Abstract
Background:
Retinal vein occlusion (RVO) is the second most common retinal vein disease and an important cause of blindness and visual morbidity. Many conditions are associated with RVO but the real role of the thrombophilic mutations is still unclear.
Aim:
To evaluate the potential role of thrombophilic mutations in RVO.
Methods:
We have evaluated 113 patients with RVO and compared with 104 volunteer controls. The controls were all healthy blood donors without previous venous thromboembolism episode or arterial thromboembolism episode. All patients were tested for 5 gene variants (here all named as mutations): factor V (FV) Leiden (G1691A), factor II (FII; G20210A), 5,1-methylenetetra-hydrofolate reductase (MTHFR; C677T), plasminogen activator inhibitor 1 (PAI-1; 4G/5G), and angiotensin-converting enzyme (ACE; Del/Ins). Statistical analysis were performed by the 2-tailed chi-square test.
Results:
Statistical test showed that TT homozygous patients of the MTHFR C677T mutation (P = .017) and heterozygous GA patients of the FII G20210A mutation (P = .018) were significantly higher than that in controls. For FV Leiden, even if the values were higher in patients than in controls, P value was not statistically significant. Conversely, for the ACE (Ins/Del) and PAI-1 (4G/5G) mutations, no difference was observed among genotypes of patients with RVO and control participants.
Conclusions:
In our study, the FII G20210A and the MTHFR C677T mutations resulted significantly higher in patients than in controls; in contrast, thrombophilic mutation of FV, ACE, and PAI-1 genes was not statistically correlated with RVO. In spite of having found an association between some thrombophilic mutations and RVO, more studies with a major number of patients are necessary to determine the final role of these gene variants.
Introduction
Retinal vein occlusion (RVO) is the second most common retinal vein disease after diabetic retinopathy and an important cause of blindness and visual morbidity 1 and has been associated with genetic mutations of coagulation factors. 2,3 A recent study showed that about 16.4 million adults worldwide are affected by RVO. 4 Thrombophilic gene variants or mutations more associated with RVO are the factor V (FV) Leiden gene mutation (G1691A), the factor II (FII) gene mutation (G20210A), and the 5,1-methylenetetra-hydrofolate reductase (MTHFR) gene mutation (C677T) but the results from several studies are still conflicting. 5 In recent years, in fact, several studies have failed to demonstrate a correlation between RVO and FV Leiden, 6 –8 on contrary to a meta-analysis published by Rehak et al 9 which showed a relationship between this common mutation and retinal vein thrombosis. In the healthy people, FV Leiden is present in the 5% of the cases; regional differences are also important: in northern Europe it is more frequent than in the south, in the Chinese population, in the Japanese as well as in the native Australian and African it is not present. The prevalence of prothrombin G20210A in healthy European caucasian population is about 1% to 8% while in patients with confirmed venous thromboembolism episode (VTE) is 3% to 17%. 11 A study 12 published in 2011 showed that the angiotensin-converting enzyme (ACE) gene variant was higher in native Americans than in caucasians or Africans; conversely, the prevalence of plasminogen activator inhibitor 1 (PAI-1) polymorphism was similar among different peoples. 12 The distribution of C677T allele showed geographical and ethnic variations. Low prevalence of this mutation in shown in black Americans, Asians, and Africans. 13 The 4G/4G polymorphism of PAI-1, associated with high plasma level of PAI-1 activity, is commonly considered a risk factor for coronary heart diseases and venous thrombosis, particularly in patients with other genetic thrombophilic mutations. 14 The prevalence of hereditary thrombophilia is known in patients with lower limb thrombosis or pulmonary embolism but not clearly confirmed in patients with thrombosis in other sites. In a study published by Bombeli et al, 39% of the patients with lower extremity thrombosis presented thrombophilia, but these genetic mutations were found only in the 5.9% of patients with RVO. 15 In the literature, the role of thrombophilia in patients with RVO resulted more important in patient without acquired risk factors such as hypertension, hyperlipidemia, or diabetes. 5 Other studies have suggested that thrombophilia can play more important role in younger people with RVO than in older people. 16 The role of thrombophilia in patients with RVO resulted very controversial; hence, the aim of our study is to evaluate the potential role of thrombophilic mutations in RVO compared with healthy population.
Methods
Patients
A population of 113 consecutive male and female patients, aged between 18 and 77 years, with RVO who were referred to our hospital were selected for this study and compared with 104 anonymous control males and females aged between 18 and 65 years.
All patients were subjected to an anamnestic investigation to exclude patients with previous VTE or arterial thromboembolism episode (ATE)from the study. The controls were all healthy and regular blood donors, without concomitant or previous serious diseases and previous ATE or VTE.
For the 2 groups, we have evaluated the following genetic parameters: FV Leiden (G1691A), FII (G20210A), PAI-1 (4G/5G), MTHFR (C677T), and ACE (Del/Ins). Patients with RVO and glaucoma were excluded from the study.
Genotyping
Genomic DNA was extracted from peripheral blood samples collected in EDTA using MagNA Pure LC (Roche Diagnostics, Roche S.p.A, Milan - Italy).
Genotyping of patients with RVO and controls was performed by the CVD-StripAssay (ViennaLab, Vienna, Austria) which is based on the reverse hybridization of biotinylated polymerase chain reaction (PCR) products principle. The strips combine probes for variants and controls in a parallel array of allele-specific oligonucleotides and generate test results by enzymatic color reaction. 17 Validation of genotyping was performed by testing several patients and control participants with different procedures. In particular, variants of FV Leiden, FII, and MTHFR were genotyped by real-time PCR-based procedures by kits from Roche Diagnostics using a Light Cycler 2.0 thermal cycler (Roche Diagnostics). 18 Variants of PAI-1 gene were genotyped by an allele-specific PCR procedure using the following oligonucleotides: PAI-1 4G, 5′-GTCTGGACACGTGGGGA-3′; PAI-1 5G, 5′-GTCTGGACACGTGGGGG-3′; and PAI-1 reverse, 5′-TTTTCCCCAGGGCTGTCCA-3′. Detection of amplification products was performed by agarose gel electrophoresis. 19 Variants of ACE gene were genotyped by PCR using the following oligonucleotides: forward, 5′-CTGGAGACCACTCCCATCCTTTCT-3′; reverse, 5′-GATGTGGCCATCACATTCGTCAGAT-3′. Detection of the length of amplification product was performed by agarose gel electrophoresis (I allele, 477 bp; D allele, 190 bp). 20
Statistical analysis was performed using the GraphPad (GraphPad InStat, GraphPad Softwares, San Diego - California, USA) and MedCalc (MedCalc Softwares bvba, Ostend - Belgium) softwares.
Results
A total of 113 consecutive patients with RVO, 50.4% males, aged between 18 and 77 years were compared with 104 healthy controls, 72.1% males, aged between 18 and 65 years. Genotype results are summarized in Table 1 and indicate that FV Leiden (G1691A mutation) was present in 5 of the 113 patients and in 2 of the 104 controls. All, patients and controls were heterozygous for this mutation. FII G20210A mutation was found in 8 of the 113 patients and in 3 of the 104 controls (7.0% in RVO vs 2.9% in controls). None was homozygous for this mutation. Homozygosity of MTHFR (C677T mutation) was observed in 18 of the 109 patients and in 10 of the 104 controls. In the case of PAI-1 (5G/4G mutation), the homozygous 4G/4G genotype was found in 26 of the 113 patients and in 31 of the 101 controls. The Del/Del genotype of the ACE gene was found in 38 of the 108 patients and in 24 of the 104 controls.
Genotype Frequencies of Polymorphisms for FV, Prothrombin, MTHFR, PAI-1, and ACE Genes in Patients With Retinal Vein Occlusion (RVO) and Normal Controls (CON).
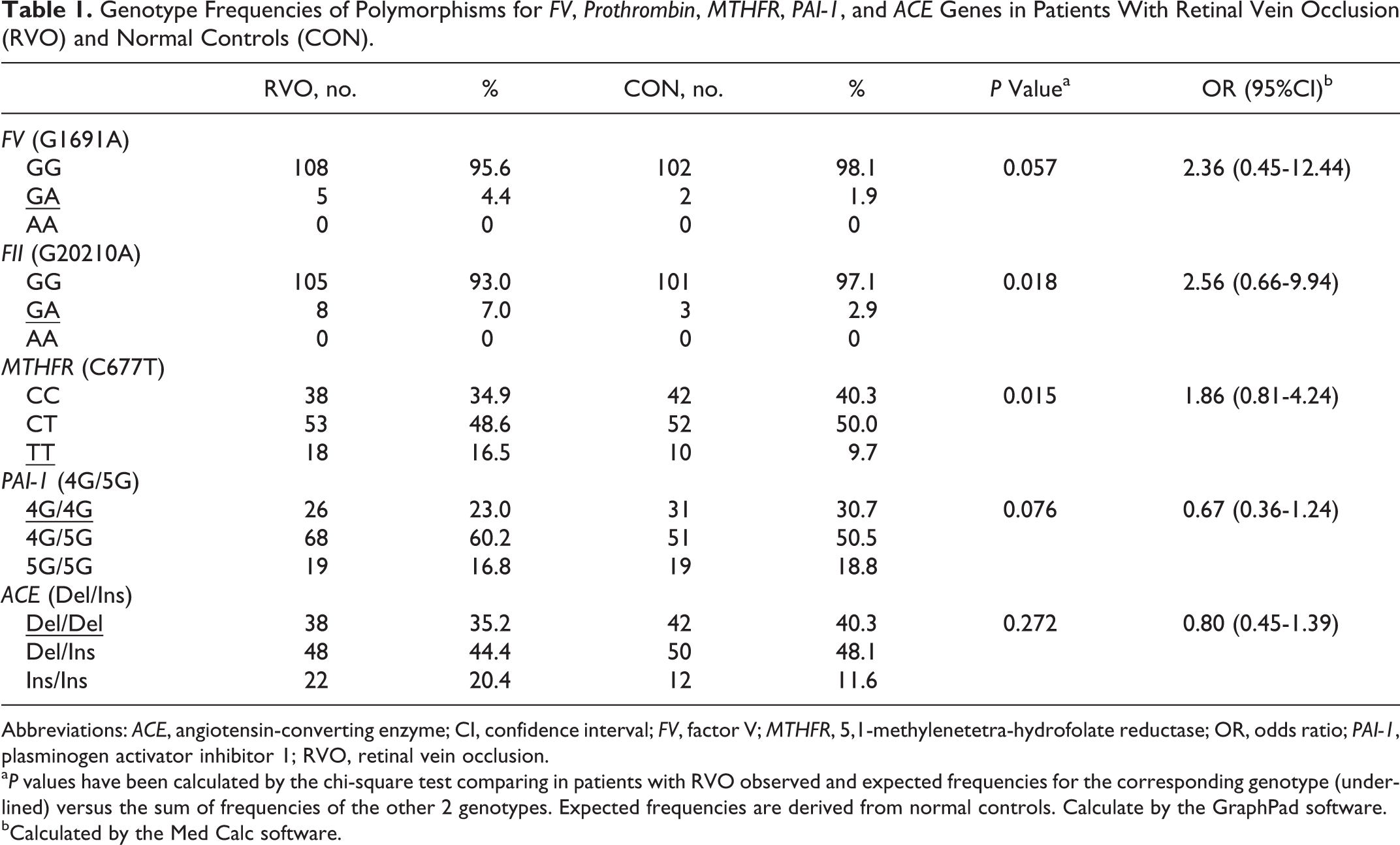
Abbreviations: ACE, angiotensin-converting enzyme; CI, confidence interval; FV, factor V; MTHFR, 5,1-methylenetetra-hydrofolate reductase; OR, odds ratio; PAI-1, plasminogen activator inhibitor 1; RVO, retinal vein occlusion.
a P values have been calculated by the chi-square test comparing in patients with RVO observed and expected frequencies for the corresponding genotype (underlined) versus the sum of frequencies of the other 2 genotypes. Expected frequencies are derived from normal controls. Calculate by the GraphPad software.
bCalculated by the Med Calc software.
The statistical analysis, performed by comparing with the chi-square test observed and expected frequencies, showed that the 2 hereditary thrombophilic mutations are significantly more present in patients with RVO than in controls. In particular, the TT genotype of the MTHFR C677T mutation (P = .017) and the heterozygous genotype of the FII G20210A mutation (P = .018) were significantly higher in patients. Even if FV Leiden mutation was less present in controls, these values were not statistically significant (P = .056).
When allelic frequencies are compared (Table 2), only the thrombophilic A allele of the FII G20210A mutation appears significantly overrepresented in RVO than in control participants. In the case of the Del/Ins mutation of the ACE gene, the allelic frequency of the thrombophilic allele was significantly lower in RVO than in control participants.
Allelic Frequencies of FV, Prothrombin, MTHFR, PAI-1, and ACE Genes in Patients With Retinal Vein Occlusion (RVO) and Normal Controls (CON).
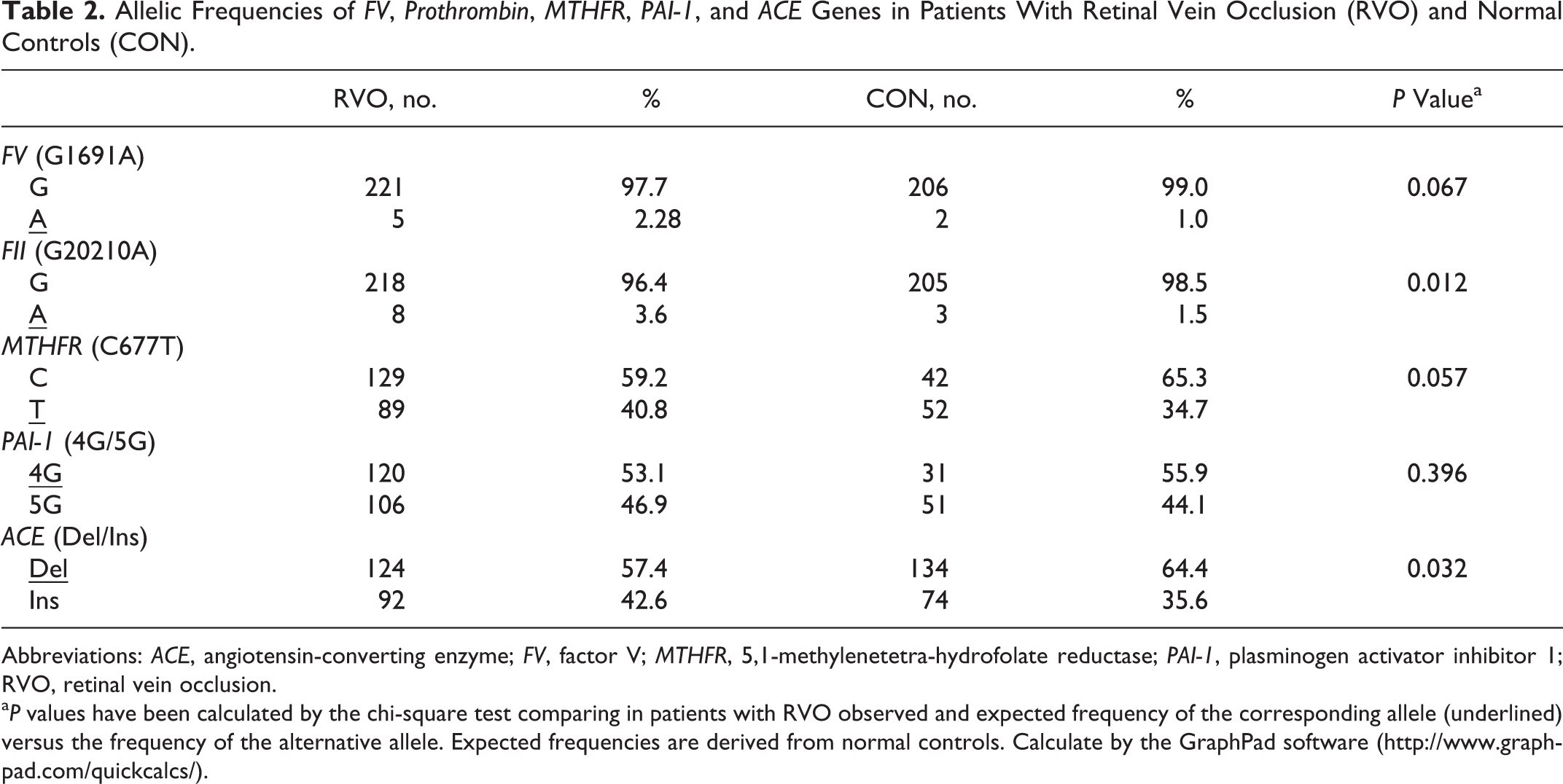
Abbreviations: ACE, angiotensin-converting enzyme; FV, factor V; MTHFR, 5,1-methylenetetra-hydrofolate reductase; PAI-1, plasminogen activator inhibitor 1; RVO, retinal vein occlusion.
a P values have been calculated by the chi-square test comparing in patients with RVO observed and expected frequency of the corresponding allele (underlined) versus the frequency of the alternative allele. Expected frequencies are derived from normal controls. Calculate by the GraphPad software (http://www.graphpad.com/quickcalcs/).
None of the observed genotypes was significantly different to the expected genotypes based on the allelic frequencies. Thus, for all analyzed gene variants, both cases and controls are in Hardy-Weinberg equilibrium.
Discussion
Retinal vein occlusion is the second most common retinal vein disease. Several trials have reported the association with classic risk factors, such as hypertension, hyperlipidemia, and atherosclerosis, and the onset of this disease. 2,9,21 Conversely, the role of thrombophilic mutations is still controversial. A recent Greek study showed that there may be an association between the presence of several polymorphisms such as ACE Ins/Del, MTHFR C677T, PAI-1 4G/5G, FV Leiden and an increased risk of RVO, but the small number of samples needed another confirmative studies. 22 This study contradicts the other Greek study performed by Giannaki et al in which the prevalence of thrombophilic risk factors was not significantly different in the patients with RVO compared to controls. 23 Two recent meta-analysis 23,24 reported a statistically significant difference in the FV Leiden prevalence between patients with RVO and controls, but these data were not supported by Stem et al in an analysis just published. 6 In our patients, without previous ATE or VTE, the prevalence of FV Leiden was found higher than controls, but this difference was not significant. Rehak et al have found that the FV Leiden was higher in patients with RVO and without acquired risk factors, suggesting that thrombophilia may play a significant role in the pathogenesis of thrombosis. 5 The A allele of the FII (G20210A) mutation increases prothrombin concentration in plasma and can cause a hypercoagulable state. In the healthy population, this polymorphism is present about 2% to 5% and it is a risk factor for VTE. An Italian trial performed in the 1990s 25 found G20210A mutation in 8.3% of patients. According to the published studies, we have found a correlation between FII mutation and RVO. Of the 113 patients and 104 controls, 8 patients and 3 controls, respectively, presented the polymorphism and the difference resulted as statistically significant (P = .018). Genotype TT of the MTHFR (C677T) mutation is very common in the healthy population with an estimated frequency about 35%. 26 The role of this mutation in increasing the risk of venous thromboembolism is reported in several publications. 27,28 Still, in our case we have found a significant difference (P = .017) between the prevalence of the TT genotype in the 2 groups. A study performed by Sottilotta et al has shown that only the hyperhomocysteinemia in plasma may play a role in the thrombosis development while the MTHFR (C677T) mutation was not associated with this disease. 29 The role of PAI-1 in the RVO is not clear. Glueck et al 3 established that the polymorphism 4G/4G was strictly related to RVO but other authors did not confirmed that hypothesis. 30 Several studies showed that the levels of PAI-1 are 25% higher in homozygotic patients for the allele 4G than in homozygotic patient for allele 5G, index for a probable relationship between polymorphism 4G, and the risk of venous thrombosis. Some authors hypothesized that the presence of the homozygosis 4G becomes a risk factor when joined to other genetic or environmental risk factors. 31 Conversely to what reported by cited authors, in our case, the polymorphism 4G/4G resulted higher in healthy controls but this difference was not statistically significant (P =.076). The ACE is present in high concentrations on the surface of vascular endothelial cells and plays an important role in the modulation of vascular tone. The ACE is also involved in the processes of fibrinolysis, platelet activation, and blood coagulation. The role of the variants of the ACE gene in the onset of venous thromboembolism is still controversial. 32,33 In a large meta-analysis performed by Gohil et al, VTE resulted statistically significant to polymorphism Ins/Del 34 but not confirmed in any other study published by Ay et al. 32 In our case, the role of polymorphism Ins/Del in RVO was not proved, in fact it resulted more present in controls than in patients.
In conclusion, we found that thrombophilic mutations of FII and MTHFR genes result significantly more frequent in patients than in controls (P < .05). In contrast, thrombophilic mutation of FV, ACE, and PAI-1 genes appears not statistically correlated with RVO. In spite of having found the association between some thrombophilic mutations and RVO, more studies with a major number of patients are necessary to unequivocally determine the role of these mutations.
Footnotes
Authors’ Note
The authors GD, GB, SP, MT, and PDR contributed equally to conception of the study, interpretation of data, and drafting the article. GD and PDR contributed to perform genetic tests. All authors had full access to all the data and take responsibility for the accuracy of the data and of the analysis. This is a retrospective, spontaneous study, not supported by public or private companies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
