Abstract
Background
Guidelines recommend prasugrel or ticagrelor for acute coronary syndrome (ACS) patients. However, these P2Y12 inhibitors increase bleeding risk compared to clopidogrel. Although genotype-guided P2Y12-inhibitor selection has been shown to reduce bleeding risk, data on its clinical implementation is lacking.
Methods
The study included ACS patients receiving genotype-guided antiplatelet therapy, utilising either a point-of-care (POC) device or laboratory-based testing. We aimed to collect qualitative and quantitative data on genotyping, eligibility for de-escalation, physician adherence to genotype results, time to de-escalation and cost reduction.
Results
Of the 1,530 patients included in the ACS registry from 2021 to 2023, 738 ACS patients treated with ticagrelor received a CYP2C19 genotype test. The median turnover time of genotyping was 6.3 hours (interquartile range [IQR], 3.2-16.7), with 82.3% of the genotyping results known within 24 hours after admission. POC genotyping exhibited significantly shorter turnaround times compared to laboratory-based testing (with respective medians of 5.7 vs 47.8 hours; P < .001). Of the genotyped patients, 81.7% were eligible for de-escalation which was carried out within 24 hours in 70.9% and within 48 h in 93.0%. The time to de-escalation was significantly shorter using POC (25.4 hours) compared to laboratory-based testing (58.9 hours; P < .001). Implementing this strategy led to a reduction of €211,150.50 in medication costs.
Conclusions
CYP2C19 genotype-guided-de-escalation in an all-comers ACS population is feasible. POC genotyping leads to shorter turnaround times and quicker de-escalation. Time to de-escalation from ticagrelor to clopidogrel in noncarriers was short, with high physician adherence to genotype results.
Keywords
Introduction
Dual antiplatelet therapy (DAPT) with aspirin and a P2Y12 inhibitor is the cornerstone of treatment for patients with acute coronary syndrome (ACS).1,2 Guidelines recommend the use of ticagrelor or prasugrel, given their more consistent anti-platelet response, over clopidogrel for patients with ACS. This recommendation is based on the pivotal trials TRITON-TIMI 38 and PLATO demonstrating superiority over clopidogrel in reducing cardiovascular events.3–6 However, ticagrelor and prasugrel are associated with a higher bleeding risk than clopidogrel.1,4,6,7
Clopidogrel is a pro-drug and needs to be oxidised by the CYP2C19 enzyme to become effective. However, around 30% of Europeans and 60% of Asians carry at least one loss-of-function (LoF) allele for the CYP2C19 genotype (*2 or *3), leading to reduced metabolisation. 8 As a result, the United States Food and Drug Administration (FDA) warns against the use of clopidogrel in these patients and recommends the use of an alternative P2Y12 inhibitor. In line with the FDA's recommendation, the European Medicines Agency (EMA) has also acknowledged the impact of CYP2C19 LoF alleles on clopidogrel efficacy, emphasising the use of alternative antiplatelet therapies in poor metabolizers. 9 This reflects a global awareness of the clinical implications associated with CYP2C19 genetic variation in the context of antiplatelet therapy. This is based on data demonstrating that, compared to noncarriers, carriers of one (intermediate metabolizer) or two (poor metabolizer) CYP2C19 LoF alleles have a higher risk for major adverse cardiovascular events (MACE) when treated with clopidogrel.10–12
The randomised clinical trial POPular Genetics showed that applying a genotype-guided de-escalation strategy in ST-elevation myocardial infarction (STEMI) patients led to a reduction in bleeding without an increase in ischaemic events. 13 However, to date the implementation of this strategy in an all-comers ACS population has not been reported.
The objective of this study was to provide objective insights on the feasibility of implementing an early routine CYP2C19 genotype-guided de-escalation strategy in patients with ASC, using both point-of-care (POC) genotyping and laboratory-based testing (Table 1).
The Different CYP2C19 Phenotypes and Genotype, With Their Associated Expected Response to Clopidogrel and Treatment Recommendation.

Methods
Study Design and Population
The rationale and design of the FORCE-ACS registry have been described previously. 14 In brief, the FORCE-ACS registry is an ongoing prospective registry in which nine Dutch hospitals participate. All hospitals are capable of performing coronary angiography and six hospitals have on-site percutaneous coronary intervention (PCI) facilities. One hospital has on-site genotyping facilities and performs routine genotyping. On August 2021, the genotype-guided strategy was implemented. From then on, all consecutive patients who were diagnosed with an ACS and had an indication for dual antiplatelet therapy (DAPT), were eligible for a genotype-guided antiplatelet therapy. The diagnosis of ACS included STEMI, non-ST elevation myocardial infarction (NSTEMI) and unstable angina pectoris (UAP). Patients were followed up by questionnaires at 1, 12, 24 and 36 month(s) after initial admission. Clinical data abstraction from the electronic health record (EHR) was performed manually. Only patients who had complete data regarding their P2Y12 inhibitor therapy were included for this analysis. Written informed consent was obtained from each patient. The institutional review boards of all approved the protocol of the FORCE-ACS registry. The current study complies with the principles of the Declaration of Helsinki (Table 2).
Baseline Characteristics.

This table is part of the local protocol.
Abbreviations: BMI: body mass index; CABG: coronary artery bypass grafting; CAD: coronary artery disease; CVA: cerebrovascular accident; MI: myocardial infarction; PCI: percutaneous coronary intervention; TIA: transient ischemic attack.
Genotyping
Initially, genotyping was performed using the POC device of Genomadix, which requires a buccal sample to operate. This device has Conformité Européenne (CE) marking and is therefore compliant with European Union legislation for safety, health, and environmental requirements. As of March 2022, the implementation protocol was updated to also include CYP2C19 genotyping using genomic DNA isolated from venous EDTA-anticoagulated blood. These samples were analysed in the laboratory of the clinical pharmacy. The LoF alleles CYP2C19*2 (G681A, rs4244285) and CYP2C19*3 (G636A, rs4986893) were determined by real-time polymerase chain reaction using the StepOnePlus™ Real-Time PCR system (Applied Biosystems), pre-validated Drug Metabolism TaqMan Genotyping Assays (for CYP2C19*2 Assay ID C__25986767_70 and for CYP2C19*3 Assay ID C__27861809_10) and TaqMan GTXpress Master Mix (Thermo Fisher Scientific, USA), according to the manufacturer's instructions. The laboratory-based test results were entered into the EHR system by qualified laboratory personnel. The POC genotyping results were processed on a separate computer, printed, and scanned into the EHR by the nurses. Nurses also reported the CYP2C19 genotype into the EHR, which was automatically linked to the overall lab results of patients.
The CYP2C19 genotyping results were classified according to the guidelines from the Clinical Pharmacogenetics Implementation Consortium (CPIC). 8 : Normal metabolizer (NM): *1/*1;
Intermediate metabolizer (IM): *1/*2, *1/*3, *2/*17 or *3/*17; Poor metabolizer (PM): *2/*2, *3/*3 or *2/*3; Rapid metabolizer (RM): *1/*17; Ultra-rapid metabolizer (UM): *17/*17.
The POC device utilised comprehensive analysis of the CYP2C19 gene, including relevant LoF-alleles such as *2, *3, and the hyperactivity encoding allele *17. The laboratory-based testing only assessed the presence of the LoF *2 and *3 alleles. Previous studies have indicated that the *17 allele is not associated with clinically significant variations in clopidogrel metabolism, leading to its exclusion in the testing protocol of laboratory-based testing for clopidogrel users. 15 All RM and UM identified by the POC device were considered normal metabolizers. 16
Antiplatelet Therapy
All patients admitted with an ACS were initially treated with ticagrelor unless they had an indication for oral anticoagulation (mostly atrial fibrillation) or had a high bleeding risk (PRECISE-DAPT score ≥25 or at least 1 major or 2 minor criteria according to the ARC-HBR criteria) in which case clopidogrel was prescribed (Figure 1). 1 In the genotyped patients, antiplatelet therapy was adjusted based on the CYP2C19 genotyping results. Attending physicians would receive an automatic notification from the EHR to change the antiplatelet therapy from ticagrelor to clopidogrel in patients who appeared to be normal or (ultra-) rapid metabolizers, starting with a 600 mg loading dose followed by 75 mg clopidogrel once daily (Figure 2). No notification was shown in intermediate and poor metabolizers, who continued to use ticagrelor 90 mg twice daily. All clinical management was left at the discretion of the attending physician. Patients were at least followed until their first follow-up visit in order to gain information regarding the use of antiplatelet therapy.

Flowchart depicting the clinical decision-making process for the genotype-guided de-escalation strategy. This flowchart provided guidance to treating physicians, who retained full discretion in clinical management. They were free to deviate from the flowchart as needed.
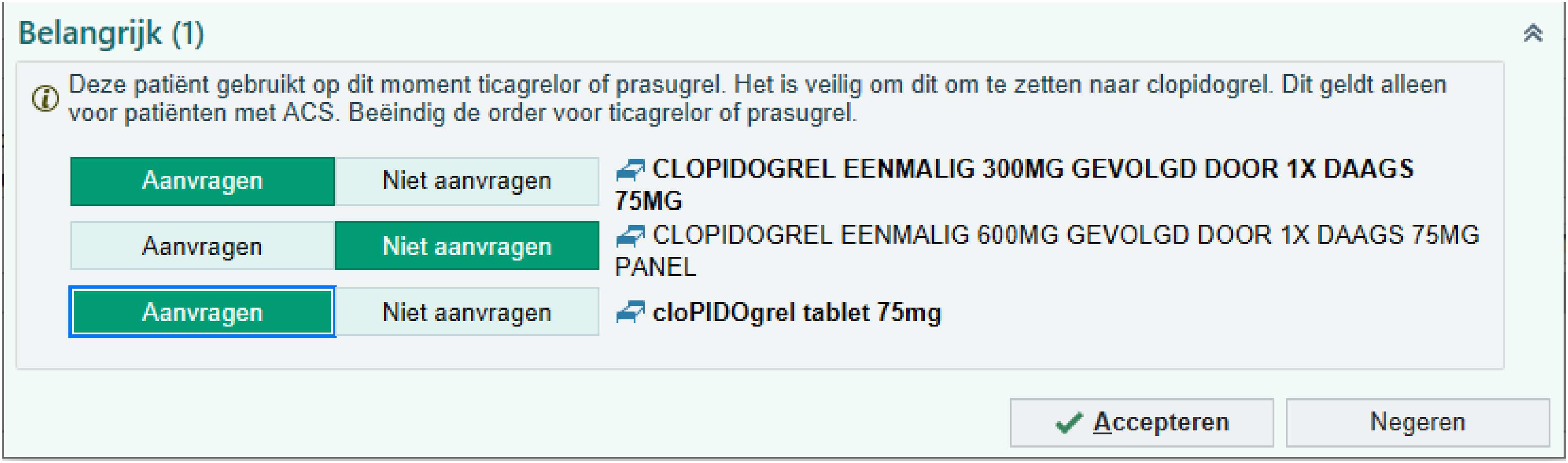
Example of the pop-up used in the medical record to propose a therapy switch to clopidogrel when an ACS patient is currently treated with ticagrelor, but is an normal metabolizer for clopidogrel. The pop-up states the following: “Important (1) This patient is currently taking ticagrelor or prasugrel. It is safe to convert this to clopidogrel. This only applies to patients with ACS. Terminate the order for ticagrelor or prasugrel” followed by a proposal for new orders for a clopidogrel loading dose and maintenance treatment, which the physician can accept or decline.
Clinical Endpoints
This registry was used to provide qualitative and quantitative data such as number of patients being genotyped, the genotyping method used, turnover times of genotyping, number of patients undergoing genotype-guided de-escalation, number of cases where physicians disregarded the genotype results and cost reduction. We separately evaluated the following time intervals: the time from admission to genetic test result, the time from test result to de-escalation and the time from admission to de-escalation.
We assessed the association between changing antiplatelet therapy and the type of genotyping method (laboratory-based testing vs POC genotyping).
Statistical Analysis Methods
Continuous variables are presented as median and 25th-75th interquartile range (IQR) or mean and standard deviation (SD). Discrete variables are presented as frequencies and percentages (%). The Mann–Whitney and Chi-square test or Fisher's exact test were used to compare quantitative and discrete variables, respectively. Differences in successful P2Y12-therapy de-escalation among the different genotyping methods were assessed using the Chi-square and Mann–Witney U test. Significance was set at a P-value of <.05. Statistical analyses were performed using SPSS version 26 (SPSS Inc., Chicago, IL).
Results
A total of 1,530 patients were included in the ACS registry from June 2021 to January 2023. In 855 patients, a CYP2C19 genotype test was performed (mean age 65 years, 28% female). From these, 269 (31.5%) patients were carriers of a CYP2C19 LoF-allele, with 28.2% of patients being intermediate and 3.3% poor metabolizers. Most patients were genotyped using the POC device (n = 752, 88.0%). The remaining 12.0% (n = 103) were genotyped using laboratory-based testing.
De-Escalation Rates
Of the 855 patients who were genotyped, 85 patients (14.5%) were already treated at admission with clopidogrel (thus not with ticagrelor or prasugrel). These patients had a prior indication for clopidogrel such as a previous cerebrovascular accident (CVA), had a high bleeding risk or were treated with an oral anticoagulant (OAC). In addition, in 22 (3.8%) patients, initial suspicion of ACS was ruled out by coronary angiography and, as such, the need for a P2Y12 inhibitor was waived. Despite being an intermediate metabolizer, nine patients (3.3%) were switched to clopidogrel for a variety of reasons, as depicted in Figure 3 (eg, new indication for OAC during admission, increased bleeding risk after coronary artery bypass grafting [CABG]). In one patient (0.1%) the test result remained inconclusive, and no additional genotype testing was performed. In total, 478 noncarriers treated with ticagrelor (64.8%) were eligible for de-escalation. Successful de-escalation from ticagrelor to clopidogrel occurred in 433 of 478 (90.4%) patients. Of the other 45 patients, 32 (71.1%) were not switched to clopidogrel due to non-compliance by the treating physician and 4 (8.9%) because of an early transfer to a referral centre before switching could take place. In three patients (6.7%) medication was not switched at the discretion of the physician based on the high thrombotic risk of the patient and three patients (6.7%) died before therapy could be switched (Figure 3).
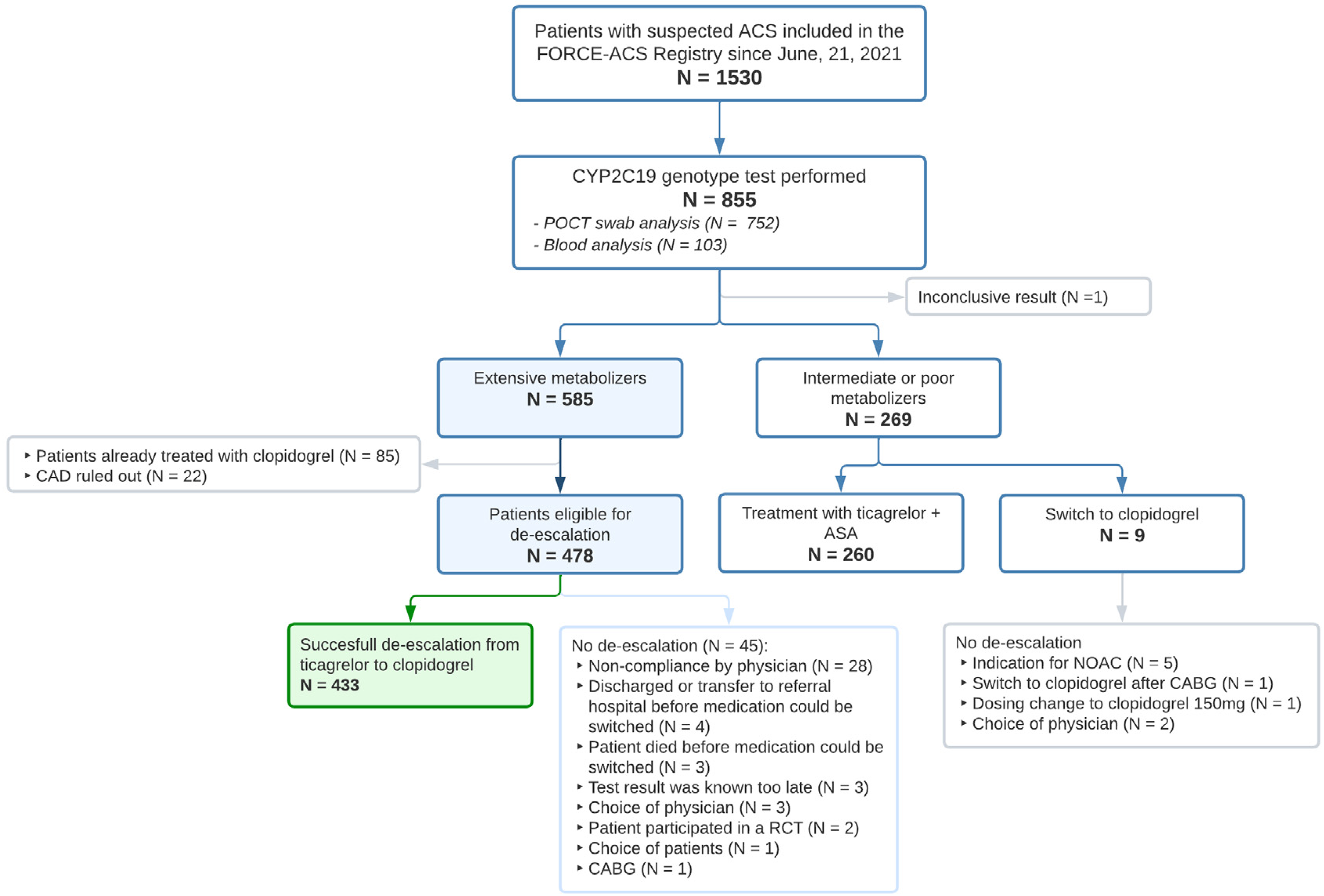
Flowchart of patients included in the FORCE-ACS registry undergoing CYP2C19 genotyping.
Turnover Time Genotyping
The median turnover time of genotyping was 6.3 h (IQR 3.2-16.7), with 82.3% of test results known within 24 h of admittance, and 91.7% within 48 h. When using POC genotyping, median turnover time was 5.7 h (IQR 3.0-12.6), with 90.5% of test results known within 24 h and 96.9% within 48 h. In patients genotyped using laboratory-based testing the median turnover time was significantly longer with 47.8 h (IQR 29.0-69.2, P < .001), with 17.0% of test results known within 24 h and 50.0% within 48 h.
Time From Test-Result to De-Escalation
After the test results were available, de-escalation to clopidogrel was carried out within 24 h in 70.9% of patients and within 48 h in 93.0%, with a median time of 20.3 h (15.3-24.9). There was no significant difference regarding time from test result to first dose of clopidogrel between POC genotyping and laboratory-based testing (20.3 vs 19.0 h).
Time From Hospital Admission to De-Escalation
The median time from hospital admission to de-escalation was significantly lower in patients analysed using POC genotyping compared to patients analysed using the laboratory-based testing (25.4 vs 58.9 h, P < .001). In patients analysed with POC genotyping significantly more patients were de-escalated within 24 h compared to the laboratory-based testing (41.4% vs 14.3%, P = .005), and 48 h (88.9% vs 32.1%, P < .001). Due to the overall longer turnover time of laboratory-based testing, de-escalation occurred less frequently compared to POC genotyping (93.0% vs 72.7%, P < .001).
Cost Reduction
A total of 855 patients underwent genotyping, of whom 752 were tested using POC genotyping and 103 using laboratory-based testing. The cost per POC genotyping analysis (sum of disposable and employee costs) was €150, while the cost per laboratory-based test was €75 (sum of machine and employee costs), resulting in a total cost of €120,525.
By implementing the genotype-guided de-escalation strategy, 433 were de-escalated from ticagrelor to clopidogrel. The daily costs of ticagrelor (still under patent protection) in the Netherlands were €2.16 per patient, resulting in an annual expenditure of €341,377.20 for the entire population. In contrast, the cost of the generic clopidogrel was €0.06 per day per patient, totalling €9482.70 annually for the overall population.17,18 This led to a net reduction of €211,369.50 (Table 3). However, no data on cost reductions related to clinical endpoints were reported in this study.
Cost Reduction by Implementing a de-escalation Strategy Whereby Genotyped Patients are Switched from Ticagrelor to Clopidogrel.

POC: point-of-care.
The hypothetical standard care group represents the projected costs if all 433 patients were treated with ticagrelor, as per standard care guidelines. The actual de-escalation group reflects the actual treatment costs after switchting these patients to clopidogrel.
Discussion
In our study, we evaluated (a) the feasibility of routine early CYP2C19 genotyping in clinical practice utilising both POC genotyping and laboratory-based testing in an all-comers ACS population and (b) the turnover times of the two genotyping strategies (POC genotyping and laboratory-based testing) as well as the rate of de-escalation to clopidogrel in ticagrelor-treated patients.
Feasibility
Our data shows that the clinical implementation and usage of CYP2C19 genotyping via POC and laboratory-based testing is feasible. In almost 90% of genotyped patients eligible for guided de-escalation, physicians prescribed clopidogrel. The median turnaround time for POC genotyping was little over 6 h, and over 90% of test results were known within 24 h. Even for laboratory-based tests, turnaround times were fair with a median of 47.8 h and over 50% of results known within 48 h. Higher turnover time of this laboratory-based test was associated with lower de-escalation rates. These findings support earlier studies showing that CYP2C19 genotyping is feasible in acute and non-acute settings.19,20
We think that several prerequisites need to be present for a successful implementation of a genotype-guided de-escalation strategy. First, staff and nurses have to be well-trained and informed about the background and clinical benefits of the treatment strategy. Second, automatic integration and interpretation of the CYP2C19 results into the EHR to guide and advise the physician to implement de-escalating from ticagrelor to clopidogrel, needs to be present. Third, the presence of genotyping facilities such as the POC device (with a turnover time less than an hour) for acute settings and laboratory-based testing facilities for non-acute settings makes it possible to get quick results and de-escalate during admission. A potential disadvantage of POC genotyping is that it is occupied for 60 min when analysing one patient. If more patients are expected to be analysed simultaneously, it is advisable to have multiple POC devices.
Routine Genotyping
Current guidelines do not encourage routine genotyping for DAPT patients, but emerging research suggests a more tailored approach is appropriate. A meta-analysis by Galli et al found that guided antiplatelet treatment selection reduced MACE and haemorrhage. 21 ACS patients undergoing PCI often receive ticagrelor or prasugrel due to their superior efficacy. Genotype-guided clopidogrel may be equally effective and even safer. 22 In a meta-analysis, Pereira et al reported that LoF carriers taking ticagrelor or prasugrel had lower MACE rates than those taking clopidogrel. 23 However, in noncarriers the efficacy of clopidogrel was comparable to that of prasugrel and ticagrelor. The test of interaction showed that CYP2C19 genotype status was a key modifier to the overall beneficial effect of ticagrelor and prasugrel compared to clopidogrel. 24 Genotype-driven treatment strategies are increasingly supported by the literature, however, our study is unique as it entails a large-scale application of a de-escalation strategy in an all-comers population. In contrast, in US and Asian centres clopidogrel remains a primary therapeutic agent and implementating a genotype-guided strategy typically results in an escalation strategy.25,26
The GIANT trial, conducted at 57 French hospitals, revealed important CYP2C19 genotyping insights in STEMI patients. 27 In this study poor metabolizers were mostly treated with prasugrel and not ticagrelor. In contrast to their findings, our research demonstrated a noteworthy improvement of the laboratory-based turnover time within 48 h (50.0% vs 18.4%), and that this can be significantly be further improved to 91.7% by using a POC device as well. These faster turnover times allow for rapid de-escalation of P2Y12 inhibition prior to patient discharge, which improves successful de-escalation and implementation.
Cost-Effectiveness
By applying a genotype-guided strategy in ACS patients treated with ticagrelor or prasugrel, 60-70% of patients can be de-escalated to clopidogrel, which results in reduced side effects and lower costs.28–30 Our findings match the POPular Genetics trial cost-effectiveness analyses. 29 This study showed that a de-escalation strategy has the potential to yield cost savings of more than €300 per patient in the first year after ACS. In a scenario analysis where there were no differences in health states at the start of the Markov model, expenditures can be reduced by €277 per patient. This cost reduction was driven by a decrease in bleeding events and medication usage. Our study showed a €247 cost savings per patient, excluding the possible cost reduction by lesser bleeding events. Our real-world data reveals that while some genetic test results were not succeeded by de-escalation, the rate of successful de-escalation was high enough to be cost saving.
Genotype-guided strategies are still uncommon in clinical practice. The cost of genetic testing appears to be a barrier to routine implementation of genotype-guided treatment. This may seem contradictory, as genotype-guided treatment has been proved to be cost-effective. Until recently, hospitals incurred the costs associated with genetic tests for genotype-guided de-escalation, while health insurers benefited from the resulting reduction in drug costs and associated healthcare savings. However, one of the Netherlands’ largest health insurers fully reimbursed our local initiative. Current evidence has however not let yet to reimbursement of this strategy on a national scale. A more widespread pre-emptive genotyping method may be reimbursed after the PREPARE (Preemptive Pharmacogenomic Testing for Preventing Adverse Drug Reactions) trial showed a 30% reduction in adverse drug reactions, in which clopidogrel was the second most common index drug. 31
POC Genotyping or Laboratory-Based Testing
The American College of Cardiology and the European Society of Cardiology recommend POC genotyping in some clinical settings for ACS management.1,32 Our data shows that ad-hoc POC genotyping has the shortest turnover times, which speeds up de-escalation and enhances the likelihood of successful de-escalation in a real-world setting. POC devices are easier to implement in hospitals without a pharmacogenetics laboratory. On the other hand, laboratory-based genotyping is cheaper than POC genotyping and has relatively fast turnaround times when a dedicated lab is available. While we showed the feasibility of ad-hoc genotyping, the most optimal scenario could involve pre-emptive testing, wherein genetic testing occurs before the initiation of actual drug therapy. This approach facilitates the immediate start of treatment with the appropriate medication. Nevertheless, implementing this approach in patients presenting with ACS is yet impractical, as it would require genetic testing in a broad population that is at-risk for ACS.
Limitations
This study has several important limitations. First, we genotyped all ACS patients, however, some were not analysed due to lack of informed consent to participate in the FORCE-ACS registry. This could have resulted in selection bias, however, only a small proportion of patients refused to participate. Second, clinical outcome data was not available for the current analysis, limiting our ability to fully interpret the feasibility of this treatment strategy. Despite this limitation, we believe our study offers valuable preliminary insights into the practical implementation of genotype-guided therapy. Third, as a single-centre Dutch registry, local practice and geographical variances in treatment may influence study outcomes and implementation. Only 855 of the 1530 patients enrolled from June 2021 to January 2023 were genotyped. Other participating hospitals participating in the FORCE-ACS registry lacked on-site genotyping capabilities at the time of data collection. Therefore, extrapolating results to other clinical settings should be done cautiously. At last, it is important to acknowledge that the present study did not record patient ethnicity, and thus, formal information on racial demographics is lacking in the FORCE-ACS registry. This limitation is particularly relevant considering the disparities highlighted in recent literature, emphasising the lack of diversity in major antiplatelet pharmacogenomic studies. 33 These studies have demonstrated that racial and ethnic diversity remains limited within the scope of investigations surrounding genotype-guided antiplatelet therapy, potentially leading to gaps in understanding the effectiveness and applicability of precision medicine strategies across underrepresented patient populations. 33 Therefore, while our study contributes valuable insights, it is essential to recognise that the broader impact of genotype-guided interventions across diverse ethnic groups warrants further investigation and consideration.
Conclusions
Early CYP2C19 genotyping as a routine procedure to guide P2Y12-inhibition in an all-comers ACS population is feasible. Time to de-escalation from ticagrelor to clopidogrel in noncarriers of CYP2C19 LoF alleles was within 24 h in the majority of patients. The shorter time to genotype-guided de-escalation with the use of POC genotyping as compared to laboratory-based testing, holds promise for implementation in sites without genotyping facilities.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Prof dr. J. M. ten Berg received research grants from AstraZeneca and ZonMw and personal fees from AstraZeneca, Accu-Metrics, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Eli Lilly, Ferrer, Idorsia, Pfizer and The Medicines Company. All other authors have nothing to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The FORCE-ACS registry is supported by grants from ZonMw, the St. Antonius Research Fund and AstraZeneca. The authors are solely responsible for the design and conduct of this study, all study analyses, the drafting and editing of the manuscript and its final contents. There was no industry involvement.
