Abstract
In-stent restenosis (ISR) is a common clinical problem in patients with coronary artery disease treated with percutaneous coronary intervention. Inflammatory process plays a pivotal role in the development of ISR. Both lymphocytes and monocytes are associated with inflammatory status. Recently, it has been shown that the lymphocyte-to-monocyte ratio (LMR) is a novel inflammatory marker. We aimed to investigate the association of serum LMR levels and ISR in patients undergoing bare-metal stent (BMS) implantation. The study included 273 patients (aged 61 ± 11 years, 66.5% men) with a history of BMS implantation and a further control coronary angiography due to stable angina pectoris. Patients were divided into 2 groups: patients with and without ISR. The LMR levels were significantly lower in patients with ISR than in those without ISR (2.50 ± 0.95 vs 3.87 ± 1.51, respectively, P < .001). On multivariate logistic regression analysis, the LMR was independently associated with ISR (odds ratio [OR]: 0.310, 95% confidence interval: 0.166-0.579, P < .001) together with high-sensitivity C-reactive protein (OR: 1.244, P = .008), reason for stent implantation (OR: 6.566, P = .003), stent diameter (OR: 0.015, P < .001), and stent length (OR: 1.137, P = .007). In conclusion, LMR levels are inversely related to ISR in patients treated with BMS implantation.
Introduction
In-stent restenosis (ISR) is one of the most widely discussed topics in interventional cardiology. Despite major advances in percutaneous coronary intervention (PCI) and antiplatelet therapy, ISR still remains a major limitation of PCI. 1 Coronary stent implantation disrupts the endothelial layer, and the cellular response to arterial wall injury includes activation of an inflammatory process, which plays a pivotal role in the development of ISR. 2 –4 Neointimal hyperplasia is the main mechanism of stent restenosis that is mostly associated with increased inflammatory status. 4,5
Many previous studies have confirmed that inflammation has been clearly associated with initiation and progression of atherosclerosis. 6,7 Immune system has a critical role in the atherosclerotic process, with lymphocytes and monocytes being the important players in this inflammatory response. 8 It has also been reported that inflammatory process is strongly related to malignancies, and these inflammatory cells have been independently associated with prognosis in different types of malignancies. 9 –11 Recently, the lymphocyte-to-monocyte ratio (LMR) has been suggested as a prognostic marker for predicting clinical outcomes of various cancers. 10,12,13
To our knowledge, relationship between this marker and ISR has not been studied previously. The aim of the present study is to investigate whether preprocedural LMR level before coronary stenting with bare-metal stent (BMS) in patients with stable angina pectoris was independently associated with ISR.
Methods
Study Population
A total of consecutive 294 patients with stable angina pectoris who underwent successful BMS implantation from September 2012 to December 2014 were enrolled in the current retrospective study. Patients were excluded from the study if they had infectious or inflammatory disease (n = 2), severe renal or liver disease (n = 4), major adverse events during follow-up (n = 2), a history of coronary artery bypass grafting (n = 6), and a history of hematologic disorder (n = 3) or malignancy (n = 4). The remaining 273 eligible patients were included.
Patients with stable angina pectoris were evaluated for inclusion when the patients underwent BMS implantation after control coronary angiography performed owing to clinical indications, including symptoms of angina and abnormal stress test results, by either exercise treadmill tests or myocardial perfusion scintigraphy. Hypercholesterolemia was described if total cholesterol >200 mg/dL and/or a low-density lipoprotein cholesterol level >130 mg/dL were revealed in biochemical tests or formerly diagnosed and treated hypercholesterolemia. Hypertension was defined as systemic blood pressure measurements exceeding 140/90 mm Hg on 2 different occasions or treatment with antihypertensive drugs for a diagnosed hypertension. Diabetes mellitus was diagnosed by fasting serum glucose levels ≥126 mg/dL, nonfasting serum glucose levels ≥200 mg/dL, or a history of diabetes mellitus. Current smokers were considered those with regular smoking in the previous 6 months. The study was approved by the local ethics committee of Ankara Education and Research Hospital, and informed consent was obtained from all participants.
Procedures
Prior coronary angiography and PCI were performed according to standard clinical practice using the femoral approach (Siemens Axiom Artis zee 2011; Siemens Healthcare, Erlangen, Germany). The diagnosis of significant coronary lesions was visually assessed in the presence of 50% stenosis in at least 1 of the 3 major coronary arteries (left anterior descending, left circumflex, or right coronary artery) or major branches by experienced interventional cardiologists. All patients received aspirin (loading dose of 300 mg) and clopidogrel (loading dose of 600 mg) before or during coronary intervention, and it was continued as a daily dose (aspirin 100 mg/d and clopidogrel 75 mg/d). Unfractionated heparin was administered in a bolus dose (100 U/kg), and further doses were used as required to maintain an activated clotting time >250 seconds. The decision to further medical therapy (glycoprotein IIb/IIIa inhibitors, β-blockers, angiotensin-converting enzyme inhibitors, and diuretics) was at the physician’s discretion. Successful PCI was defined as the reduction in a minimum stenosis diameter to <20%, with a final Thrombolysis in Myocardial Infarction grade 3 flow without any major complications. During clinical follow-up after PCI, control coronary angiography was performed as clinically indicated by recurrent symptoms or objective evidence of myocardial ischemia in patients with stable angina pectoris. Coronary angiograms were evaluated using visual estimation by 2 independent cardiologists who were unaware of the clinical and laboratory status of patients. In-stent restenosis was defined as a diameter stenosis of >50% occurring in the stented segment or 5-mm segment proximal or distal to the stent. 14 Echocardiography was performed for all patients (Vivid 3; GE Medical System, Horten, Norway), and left ventricular (LV) ejection fraction was calculated by the modified Simpson method.
Laboratory Data
Venous blood samples were obtained from catheter laboratory before coronary angiography prior to the stent implantation. Total white blood cell, neutrophil, lymphocyte, and monocyte were counted using the automated blood cell counter (XE-2100; Sysmex Inc, Japan) within 30 minutes after blood sampling. Lymphocyte-to-monocyte ratio was calculated as the ratio of lymphocytes to monocytes, and neutrophil-to-lymphocyte ratio (NLR) was calculated as the ratio of neutrophils to lymphocytes. Serum levels of high-sensitivity C-reactive protein (hs-CRP), creatinine, glucose, and fasting lipid panel, including triglyceride, total cholesterol, low-density lipoprotein, and high-density lipoprotein, were also measured via the standard laboratory techniques.
Statistical Analysis
All data analyses were performed with SPSS (version 18.0; SPSS Inc, Chicago, Illinois). Categorical variables are expressed as counts and percentages, and quantitative variables were expressed as mean ± standard deviation or median and interquartile range. Categorical variables were compared by the χ2 test or Fisher exact tests as appropriate. Comparisons of parametric values between the 2 groups were done using independent samples t test. Comparisons of nonparametric values between the 2 groups were done by Mann-Whitney U test. The goodness of fit of the model was assessed by the Hosmer-Lemeshow test. Any correlation between variables was performed by Pearson or Spearman correlation analysis. In addition, we performed multiple regression and logistic regression analyses to identify independent predictors of ISR using all clinical variables. The receiver–operating characteristic (ROC) curve was used to measure the sensitivity and specificity of LMR and optimal cutoff value for predicting ISR. A 2-sided P value of <.05 was considered statistically significant.
Results
The baseline demographic and clinical characteristics of the patients are shown in Table 1. A total of 294 patients were enrolled in the study, and 21 patients were excluded. The mean age of the study population was 61 ± 11 years, and 181 (66.5%) of the patients were male. The mean interval between the 2 coronary angiographic studies was 23 months. In-stent restenosis developed in 42.9% (n = 117) of the patients. Patients were divided into 2 groups: ISR group and no-ISR group. Left ventricular ejection fraction and body mass index were significantly lower in patients with ISR. The reason for stent implantation was more likely to be acute coronary syndrome in patients who developed ISR. However, there were no significant differences between the 2 groups in gender, age, prehospital treatments, blood pressure, heart rate, presence of hypertension, diabetes mellitus, smoking, hyperlipidemia, and family history of coronary artery disease (CAD). In addition, there was no any difference in prior medications of patients between groups.
Baseline Clinical and Demographic Characteristics of the Study Groups.
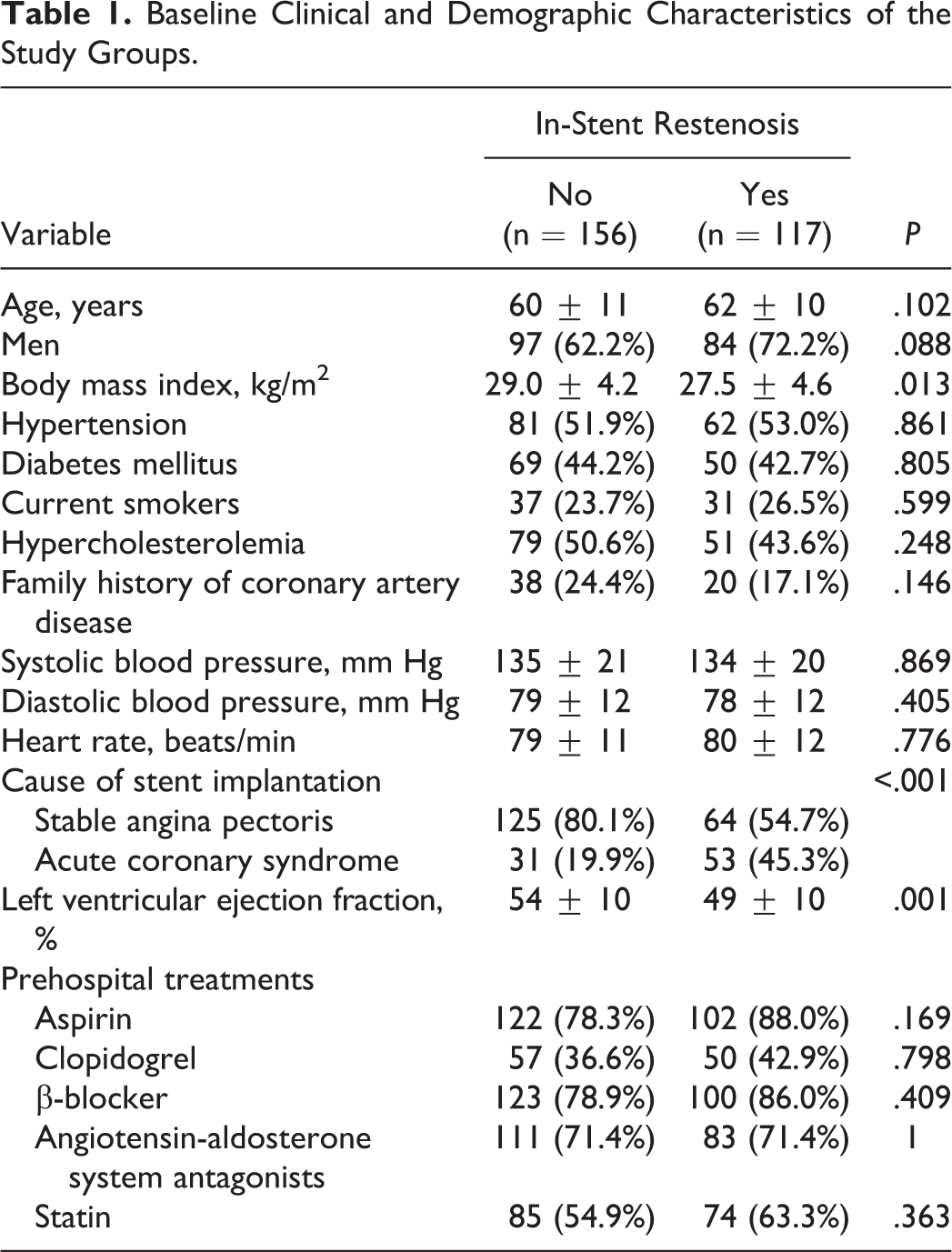
Hematological and biochemical measurements of patients with ISR and patients without ISR are listed in Table 2. Patients who developed ISR had higher serum hs-CRP levels and monocyte counts. On the contrary, these patients had lower baseline hemoglobin and lymphocyte counts. Lymphocyte-to-monocyte ratio was significantly lower in patients who developed ISR compared with those who did not (2.50 ± 0.95 vs 3.87 ± 1.51, respectively, P < .001; Table 2 and Figure 1). In addition, in Spearmen correlation analysis, there was a significant inverse relationship between LMR and hs-CRP levels (r = −.55, P < .001).
Hematological and Biochemical Measurements of the Study Population.
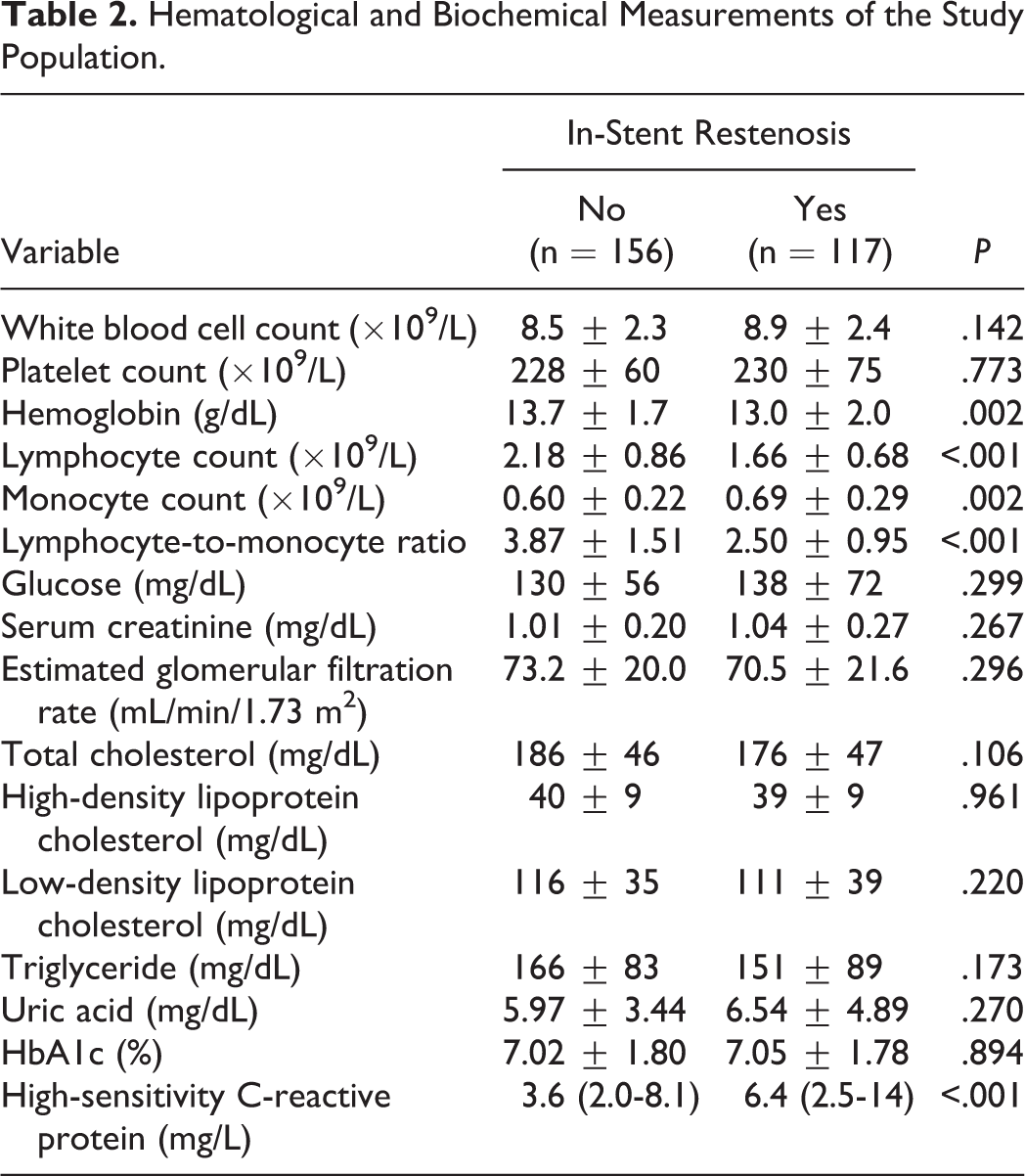
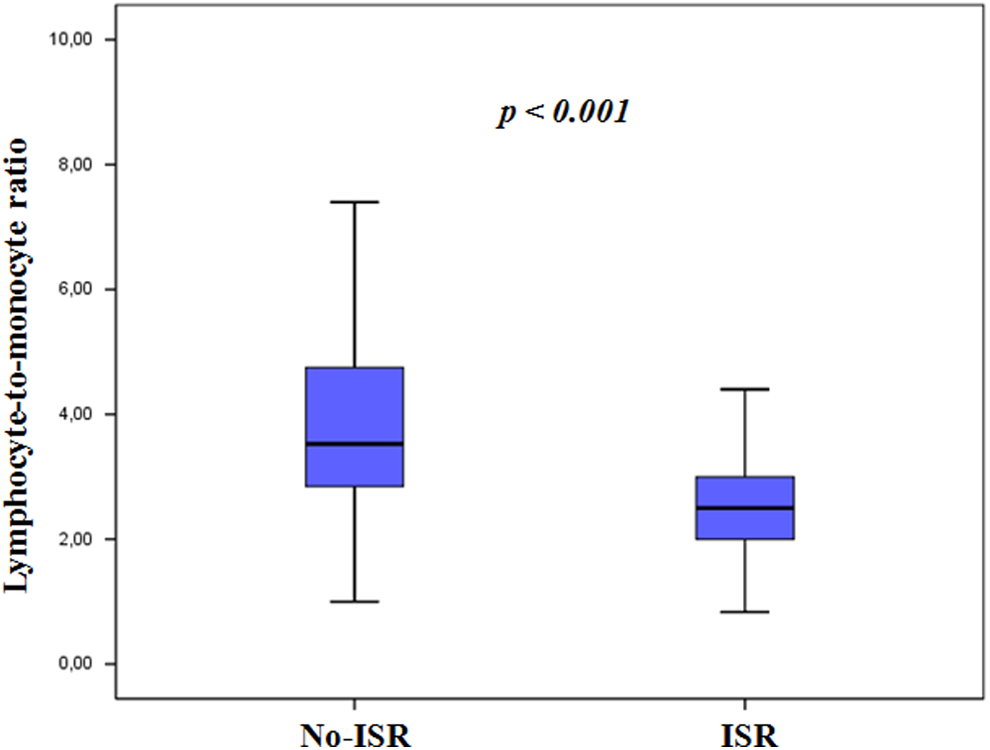
Comparison of lymphocyte-to-monocyte ratio values between patients with and without in-stent restenosis.
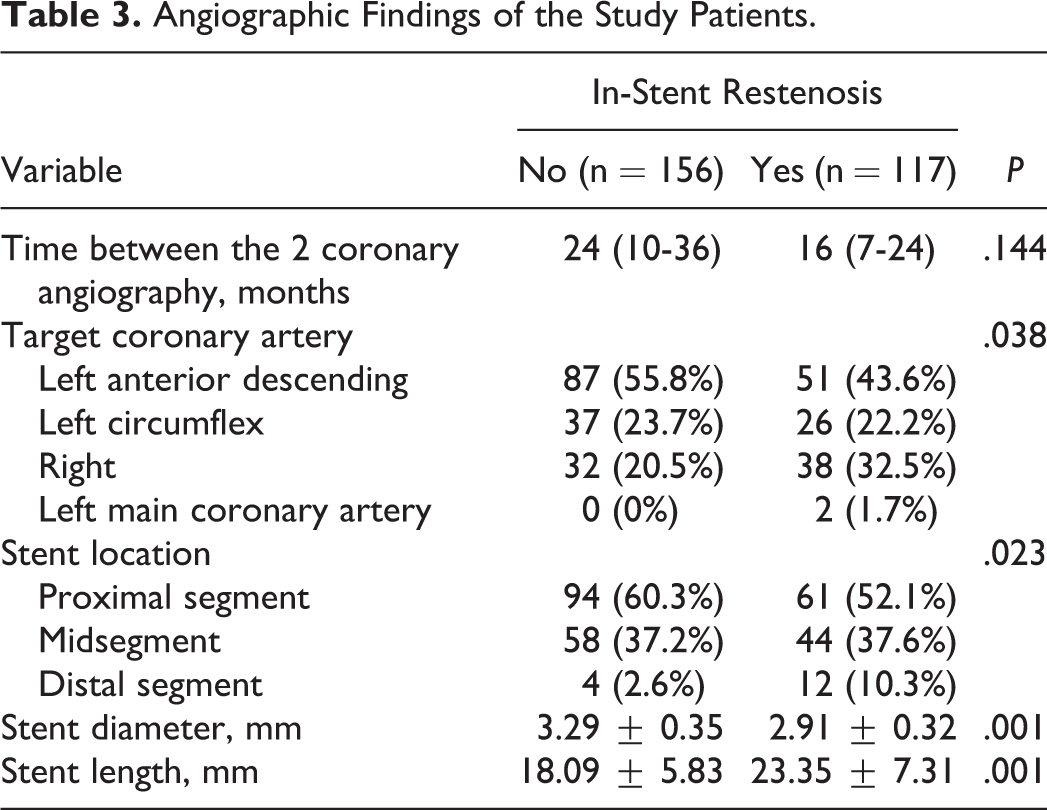
Angiographic and procedural characteristics of the study sample are summarized in Table 3. Patients with ISR had significantly longer stent length and lower stent diameter compared to those without (23.35 ± 7.31 mm vs 18.09 ± 5.83 mm, P < .001 and 2.91 ± 0.32 mm vs 3.29 ± 0.35 mm, P < .001, respectively). Angiographic restenosis rates were significantly higher in the right coronary artery. In addition, in-stenotic restenotic lesion was more common in the distal segment. However, there was no significant difference between groups in the time between the 2 coronary angiographies.
Angiographic Findings of the Study Patients.
After multivariate analysis with binary logistic regression, apart from hs-CRP level (odds ratio [OR]: 1.244, 95% confidence interval [CI]: 1.058-1.462, P = .008), cause of stent implantation (OR: 6.566, 95% CI: 1.858-23.196, P = .003), stent length (OR: 1.137, 95% CI: 1.035-1.248, P = .007), and stent diameter (OR: 0.015, 95% CI: 0.002-0.109, P < .001), LMR was an independent risk factor for ISR (OR: 0.310, 95% CI: 0.166-0.579, P < .001; Table 4).
Multivariate Logistic Regression Analysis to Identify the Independent Predictors of Bare-Metal In-Stent Restenosis.
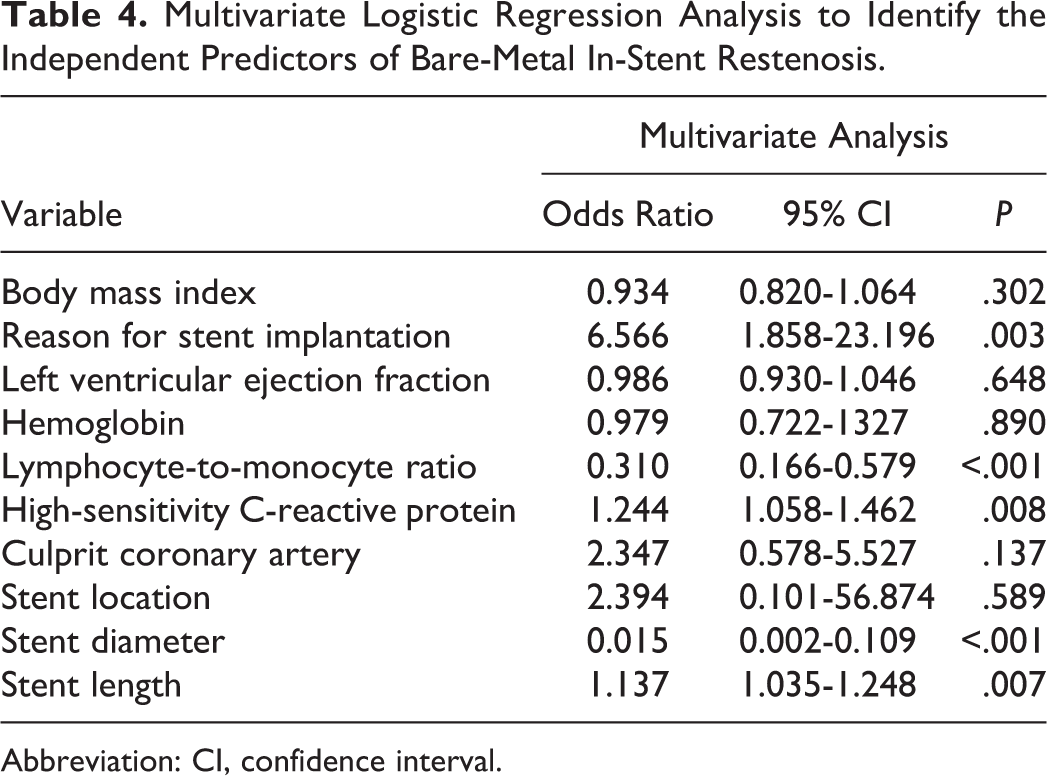
Abbreviation: CI, confidence interval.
Furthermore, the ROC curve analysis was used to determine the cutoff value of LMR to predict ISR. The cutoff value of LMR on admission to predict ISR in the entire population was 2.86, with 74.4% sensitivity and 72.6% specificity (area under the curve: 0.804, 95% CI: 0.752-0.856, P < .001; Figure 2).
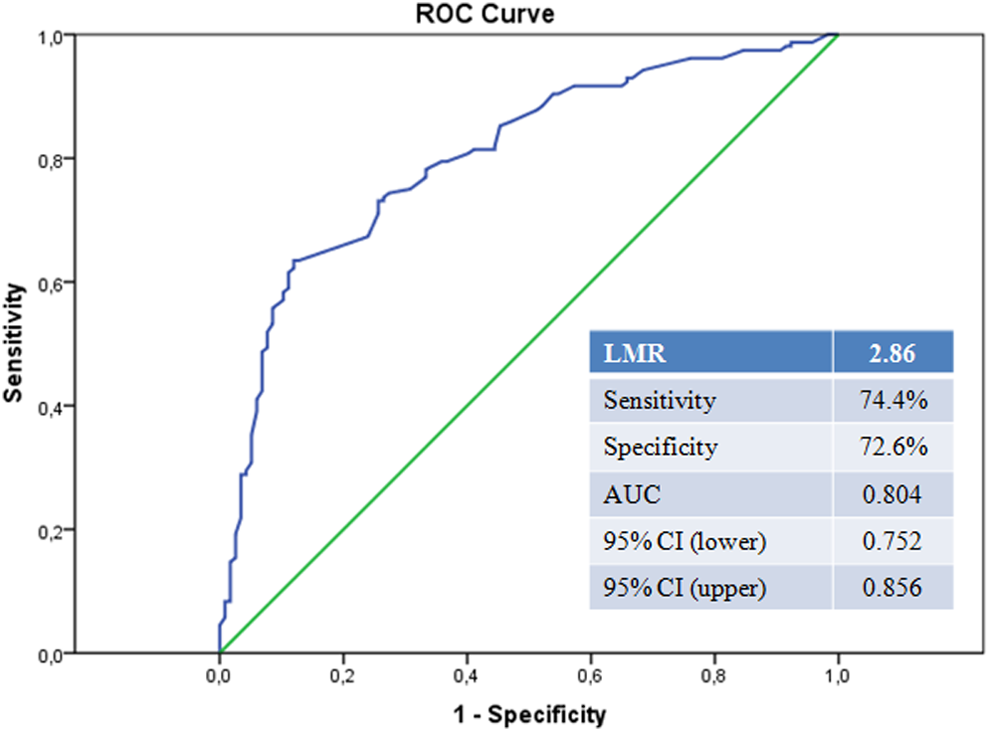
Receiver–operating characteristic curve analysis of lymphocyte-to-monocyte ratio levels for predicting in-stent restenosis.
Finally, we performed to estimate the correlation between NLR levels and LMR levels by Pearson correlation coefficient. We found a significant negative correlation between LMR and NLR levels (r = −.692, P < .001; Figure 3).
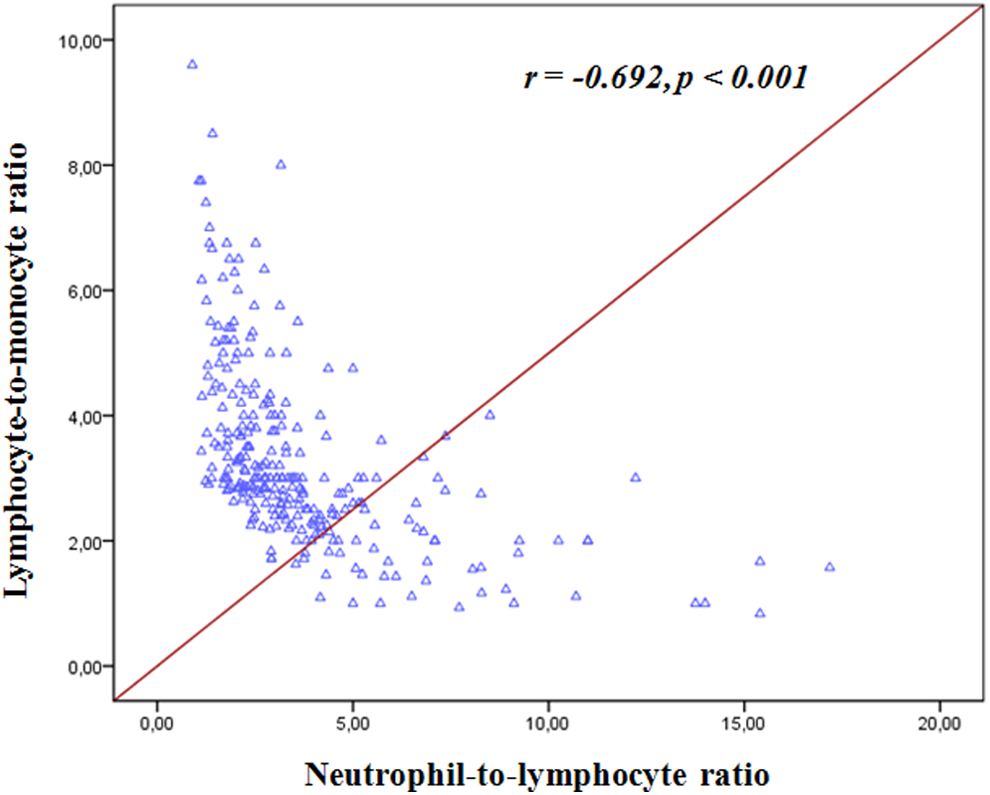
Correlation between lymphocyte-to-monocyte ratio and neutrophil-to-lymphocyte ratio.
Discussion
In our recent clinical study, we explored the impact of the preprocedural LMR for ISR in patients with stable angina pectoris who underwent successful BMS implantation and found that lower LMR on admission was strongly associated with the development of ISR. Thus, our study contributed to explain the underlying mechanisms of ISR using a new different inflammatory marker.
It is well established that coronary stent implantation has been the first-choice PCI procedure in the treatment of patients with CAD. 15 However, ISR is a common complication of coronary intervention that has been recognized as very difficult to eliminate this issue and is related to increased morbidity and costs. 1,16,17 The pathophysiology of ISR remains incompletely understood. Inflammation, granulation, extracellular matrix remodeling, and vascular smooth muscle cells proliferation and migration are thought to be major mechanisms involved in the development of ISR. 2,18,19 Percutaneous coronary intervention causes arterial wall injury that stimulates local cytokine expression leading to infiltrations of smooth muscle cells, T lymphocytes, and macrophages in that region. Thereafter, the extracellular matrix increases, and neointimal hyperplasia develops that eventually causes ISR. 20 Although the underlying causative mechanisms are still unclear, inflammatory process seems to be the main risk factor for the development of ISR. 3,21 Inflammation plays an important role in this process not only by a local vascular reaction in the vessel wall but also by a systemic inflammatory state. 21,22 Numerous previous studies have established the association of systemic inflammation with ISR that was related to various inflammatory biomarkers. For example, Ferrante et al 23 reported that higher baseline CRP levels, which has been the most commonly used inflammatory marker in several studies, are associated with increased risk of ISR after BMS implantation. In another study, Turak et al 24 found NLR, a widely accepted inflammatory marker, is a powerful and independent predictor of BMS restenosis. In this regard, we also performed the correlation between NLR and LMR levels, and we found a negative correlation between NLR and LMR levels in the study cohort. Moreover, many other proinflammatory biomarkers, such as red cell distribution width, 25 interleukin 1, 26 interleukin 6, 22 and interleukin 10, 22 were studied in several clinical trials for predicting ISR.
Inflammatory process has been implicated in various types of cancers, and it has been declared that there is a strong link between inflammation and development of different malignancies. 9 –11 In recent trials, LMR has been proposed as a prognostic marker for predicting clinical outcomes in some cancers. 10,12,13 Different inflammatory cells play a critical role in the initiation and propagation of atherosclerosis, with lymphocytes and monocytes being the important players in this inflammatory response. 8 It has also been demonstrated that elevated leukocyte count is a risk factor for ischemic heart disease in the long-term follow-up. 27 On the other hand, lymphocyte count is inversely associated with poor cardiovascular outcomes, and a lower lymphocyte count is an independent significant risk factor to have predictive value for patients with CAD. 28,29 The role of monocytes during the various stages of atherosclerosis has been identified in several studies, and monocytosis has also been shown to be involved in LV remodeling, myocardial healing, myofibroblast accumulation, and angiogenesis. 30,31 Monocytes are a critical component of atherosclerotic plaques that are activated by many growth factors and proinflammatory cytokines such as platelet-derived growth factor, interleukin 1, and interleukin 6. 30 They accumulate in atherosclerotic plaque in the dysfunctional endothelium and differentiate lipid-laden “foam” cell macrophages resulting in the formation of atherosclerotic plaque. 30 Therefore, lower LMR levels may be associated with increased inflammatory status predisposing to ISR. Furthermore, the CD14+ monocyte subset, which can be differentiated into a variety of mesenchymal cell types such as myocytes and smooth muscle cells that might potentially promote intimal hyperplasia, 30,32 may be other possible mechanism linking LMR and ISR.
This study has several limitations. First, our study was a single-center retrospective study and included patients with only BMS restenosis. Second, we could not use the intravascular ultrasound or optical coherence tomography for detecting ISR, which are more quantitative and informative compared to visual estimation. Finally, we measured LMR levels only on admission and without correction for potential variability that serial LMR measurements may provide additional information. Future large, prospective, and randomized clinical trials are required to confirm our findings.
In conclusion, we demonstrated that decreased peripheral blood LMR was an independently associated factor of BMS restenosis in patients who presented with stable angina pectoris. Also, measurement of LMR is relatively simple, inexpensive, and widely available.
Footnotes
Authors’ Note
All authors have substantial contributions to conception and design, acquisition of data, and analysis and interpretation of data; drafting the article or revising it critically for important intellectual content; and final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
