Abstract
Background:
The aim of this study was to investigate the relationship between endocan levels with the presence of slow coronary flow (SCF).
Methods:
In this cross-sectional study, a total of 88 patients, who admitted to our hospital, were included in this study. Of these, 53 patients with SCF and 35 patients with normal coronary flow were included in the final analysis. Coronary flow rates of all patients were determined by the Timi Frame Count (TFC) method.
Results:
In correlation analysis, endocan levels revealed a significantly positive correlation with high sensitive C-reactive protein and corrected TFC. In multivariate logistic regression analysis, the endocan levels were found as independently associated with the presence of SCF. Finally, using a cutoff level of 2.3, endocan level predicted the presence of SCF with a sensitivity of 77.2% and specificity of 75.2%.
Conclusion:
In conclusion, our study showed that higher endocan levels were significantly and independently related to the presence of SCF.
Introduction
The slow coronary flow (SCF) phenomenon is an angiographic clinical entity, described as delayed coronary opacification without obstructive coronary artery disease (CAD). The SCF is a relatively common angiographic finding, with an incidence of 1% in patients undergoing coronary angiography for the doubt of CAD. 1 Prior studies have demonstrated that SCF may cause myocardial ischemia, angina, and infarction. 2 –4 Although it is well recognized by the cardiologist, the underlying mechanisms are not fully understood. Small-vessel disease, endothelial dysfunction, inflammation, and diffuse atherosclerosis have been suggested for the etiology of SCF. 5 –7
Endocan, also known as endothelial cell-specific molecule 1, is a soluble 50 kDa proteoglycan, constituting a mature polypeptide of 165 amino acids and a single dermatan sulfate chain covalently linked to the serine residue at position 137. 8 Activated vascular endothelial cells synthesize and secrete endocan. 9 Previous studies have shown that endocan may have a key role in the pathophysiology of endothelial dysfunction by regulating major processes such as cell adhesion in inflammatory disorders. 10 –12 Since endothelial dysfunction and increased inflammatory status have a key role in SCF and increased endocan levels have been shown to be related to endothelial dysfunction and inflammation, we hypothesized that higher endocan levels may be associated with the presence of SCF.
The aim of this study was to investigate the relationship between endocan levels with SCF. To the best of our knowledge no studies up to date investigated endocan levels as a novel marker in patients with SCF and compared the results with normal coronary flow (NCF) group.
Methods
Study Population
The laboratory, clinical, and angiographic data of consecutive patients who had underwent coronary angiography for stable angina pectoris (identified by the European Society of Cardiology guidelines) 13 and had angiographic normal coronary arteries without any atherosclerotic lesion were included in the study. In this cross-sectional study, a total of 88 patients, who admitted to our hospital, were included in the study. Of these, 53 patients with SCF and 35 patients with NCF were included in the final analysis. Our hospital’s Local Ethics Committee approved the study protocol, and all participants provided their written informed consents.
Patients with an acute coronary syndrome defined as ST-segment elevation myocardial infarction (STEMI) and non-STEMI were excluded from the study. We also excluded patients with left ventricular systolic dysfunction (left ventricular ejection fraction [LVEF] <40%); malignancy and liver, kidney, or other acute or chronic inflammatory diseases; current therapy with corticosteroids and nonsteroidal anti-inflammatory drugs; and hematologic diseases including anemia or the ones that had underwent percutaneous coronary intervention and coronary artery bypass grafting before.
Arterial hypertension was considered in patients with repeated blood pressure measurements >140/90 mm Hg or current use of antihypertensive drugs. Diabetes mellitus was defined as fasting plasma glucose levels ≥126 mg/dL on multiple measurements or current use of antidiabetic medications.
Coronary Angiography
The standard Judkins technique and 6F catheters (Expo; Boston Scientific Corporation, Natick, Massachusetts) were used to perform baseline angiography through the femoral or radial artery, and Siemens Axiom Sensis XP device was used. All angiograms were recorded at 30 fram per second. We used Iopamiro-300 (612.4 mg/ml iopamidol) as the contrast agent in all the study groups. Coronary flow rates of all patients were determined by the Timi Frame Count (TFC) method as described by Gibson et al. 14 The TFC was evaluated for each coronary vessel by 2 trained cardiologist blind to the patient’s clinical information. The intra- and interobserver coefficients of variation were 3.0% and 5.0%, respectively. The first frame was considered to be that at which >70% lumen opacification with antegrade filling was noted. The final frames were determined when contrast opacification reached a certain distal landmark in each vessel. The distal bifurcation was used (“whale’s tail”) for the left anterior descending artery (LAD). The most distal bifurcation of the obtuse marginal branch furthest from the coronary ostium was used as the distal landmark for the left circumflex artery (LCx). The first branch of the posterolateral segment was used for the right coronary artery (RCA). The standard mean values for normal visualization of coronary arteries are described as 36.2 ± 2.6 frames for LAD, 22.2 ± 4.1 frames for LCx, and 20.4 ± 3 frames for RCA. As the LAD is generally longer than the other major coronary arteries, the TFC for this vessel is often higher. Therefore, the TFC for LAD is divided by 1.7 to obtain the corrected TFC. The standard corrected mean value (cTFC) for LAD coronary artery is 21.1 ± 1.5 frames. All participants with a TFC greater than the 2 standard deviations of the previously published range for the particular vessel were considered to have SCF. 14
Data Collection
Peripheral venous blood samples of the patients were obtained on their admission to the inpatient ward. An automated blood cell counter (Beckman Coulter analyzer; Beckman Coulter, Brea, California) was used for measuring complete blood count parameters. Blood biochemistry parameters that were measured were creatinine, total cholesterol, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and total bilirubin. High-sensitivity C-reactive protein (hsCRP) measurement was done using an automatized analyzer (Beckman Coulter analyzer) using nephelometric measurement during or after the coronary angiography. Blood samples for endocan from the patients were collected into plain tubes, and serum was separated after centrifugation at 1500g for 10 minutes and stored at −80°C until analysis. Blood samples from calcium–EDTA tubes were analyzed in an auto-analyzer. Endocan levels were determined using a commercially available sandwich enzyme-linked immunosorbent assay (ELISA) kit with high sensitivity and specificity for detection of human endocan (Boster, Wuhan, China). All the serum samples were routinely analyzed by ELISA in duplicate, and the results were averaged. Preliminary data obtained in our laboratory showed that the intra-assay and interassay coefficients of variation for endocan were 4.1% and 4.3, respectively. Transthoracic echocardiography was performed in all patients, and left ventricular ejection fraction (LVEF) was calculated using Simpson method.
Statistical Analyses
In all statistical analysis, SPSS 22.0 Statistical Package Program for Windows (SPSS Inc, Chicago, Illinois) was used. Kolmogorov-Smirnov test was used to assess the distribution pattern. Normally distributed numerical variables were presented as mean ± standard deviation, and the ones not normally distributed were presented as median and interquartile range (IQR). Categorical variables were presented as number and percentage. To compare parametric continuous variables, Student t test was used and to compare nonparametric continuous variables, the Mann-Whitney U test was used. Comparisons of multiple mean values were carried out by Kruskal-Wallis tests or analysis of variance as appropriate. Chi-square test was used to compare the categorical variables. Spearman rank test was performed to define the correlation of endocan levels and hsCRP and cTFC. Logistic regression analysis was performed in order to find independent predictors of presence of SCF. Variables that had an unadjusted P value of <.10 in logistic regression analysis were identified as potential risk markers and then included in the full model. We eliminated potential risk markers with likelihood ratio tests with reduced model using multivariate logistic regression analyses. A P value of <.05 was considered statistically significant with a confidence interval of 95%. The receiver–operating characteristic (ROC) curve was used to show the sensitivity and specificity of endocan levels and optimal cutoff value for predicting SCF.
Results
A total of 88 patients were enrolled in this study (n = 53 patients in SCF group and n = 35 patients in NCF group). Clinical, laboratory, and angiographic features of the study groups are presented in Table 1. The rate of hypertension and current smoker were significantly higher in the SCF group. The mean cTFC value was also significantly higher in the SCF group as compared with the NCF group. Total white blood cell (WBC) and platelet counts, hsCRP, and endocan levels were significantly higher in the SCF group (Table 1).
Clinical, Biochemical, and Angiographic Characteristics of the Study Population and Univariate Analyses.
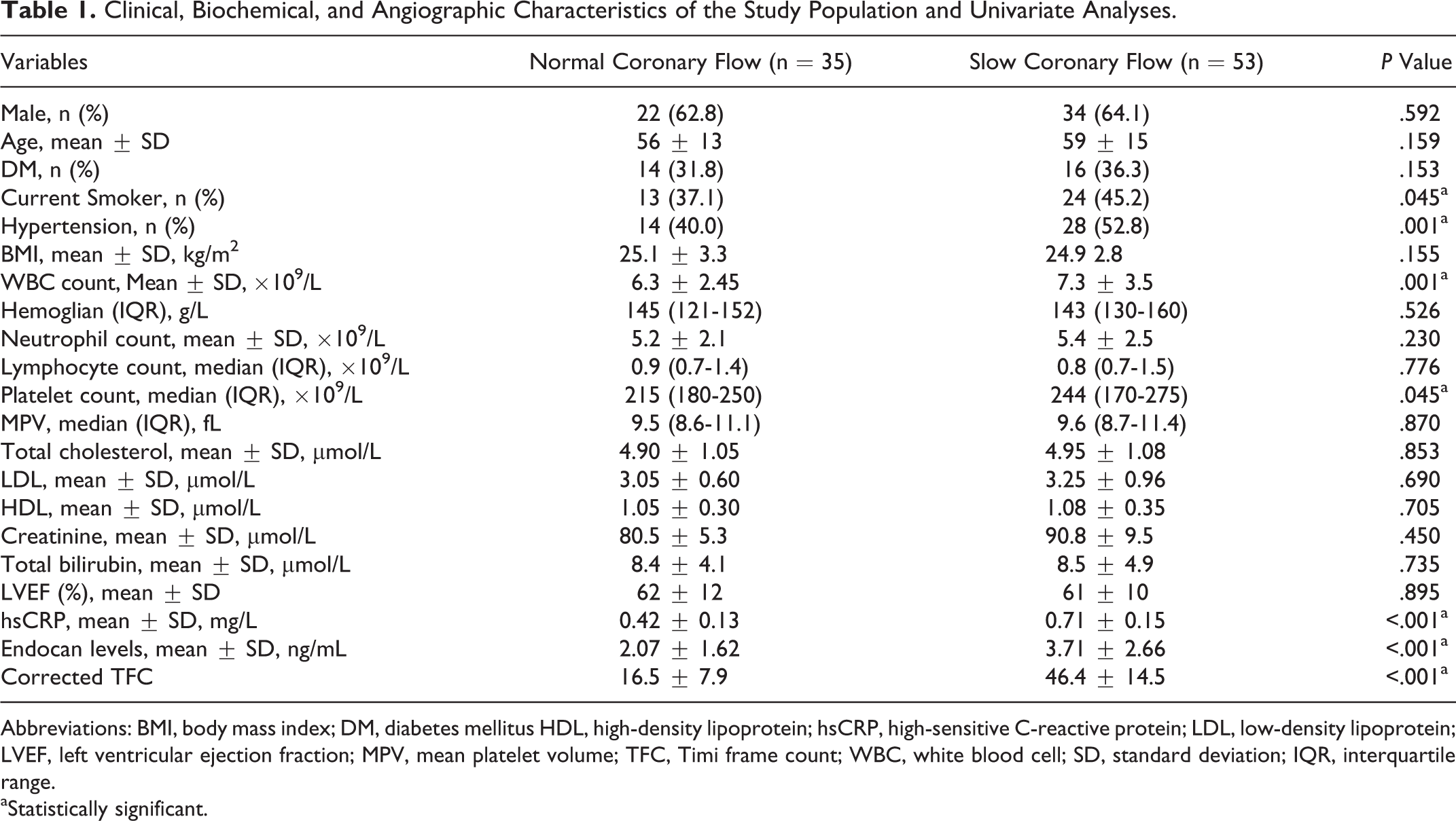
Abbreviations: BMI, body mass index; DM, diabetes mellitus HDL, high-density lipoprotein; hsCRP, high-sensitive C-reactive protein; LDL, low-density lipoprotein; LVEF, left ventricular ejection fraction; MPV, mean platelet volume; TFC, Timi frame count; WBC, white blood cell; SD, standard deviation; IQR, interquartile range.
aStatistically significant.
In correlation analysis, endocan levels revealed a significantly positive correlation with hsCRP (P < .001) and cTFC (P < .001; Figure 1 A and B). In multivariate logistic regression analysis, the endocan levels were found as independently associated with the presence of SCF (odds ratio: 1.060, 95% confidence interval [CI]: 1.032-1.085, P < .001). Moreover, hsCRP was also independently associated with SCF (Table 2). The ROC curve explored the discriminatory capability of endocan level for the presence of SCF. Area under the curve was 0.810 (95% CI: 0.712-0.902; P < .001). Using a cutoff level of 2.3, endocan level predicted the presence of SCF with a sensitivity of 77.2% and specificity of 75.2% (Figure 2). The positive and negative predictive values were 83.7% and 69.7%, respectively. Finally, the patients were divided into 2 subgroups based on an endocan cutoff level of 2.3. In addition, WBC count, neutrophil count, platelet count, hsCRP, and corrected TFC were significantly higher in the high endocan level subgroup (Table 3).
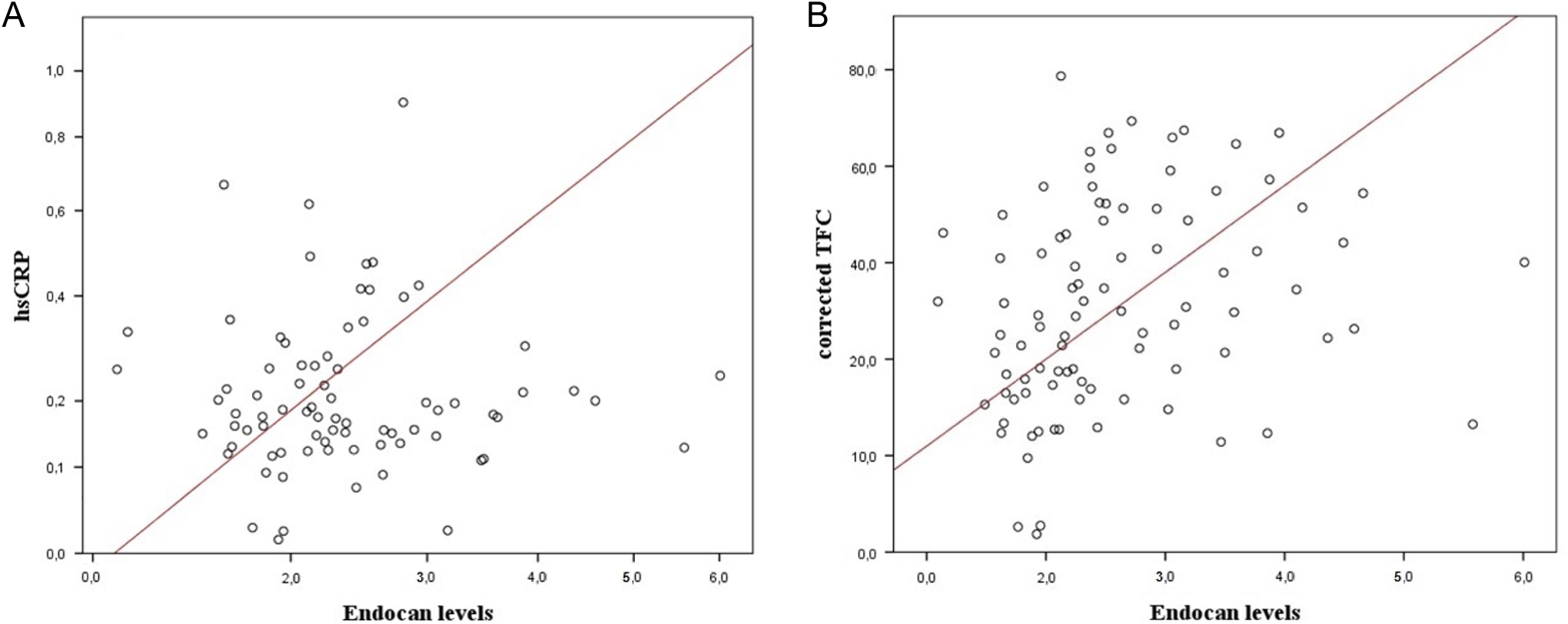
A and B, Graph shows a significantly positive correlation between endocan levels and high-sensitive C-reactive protein and corrected Timi Frame Count.
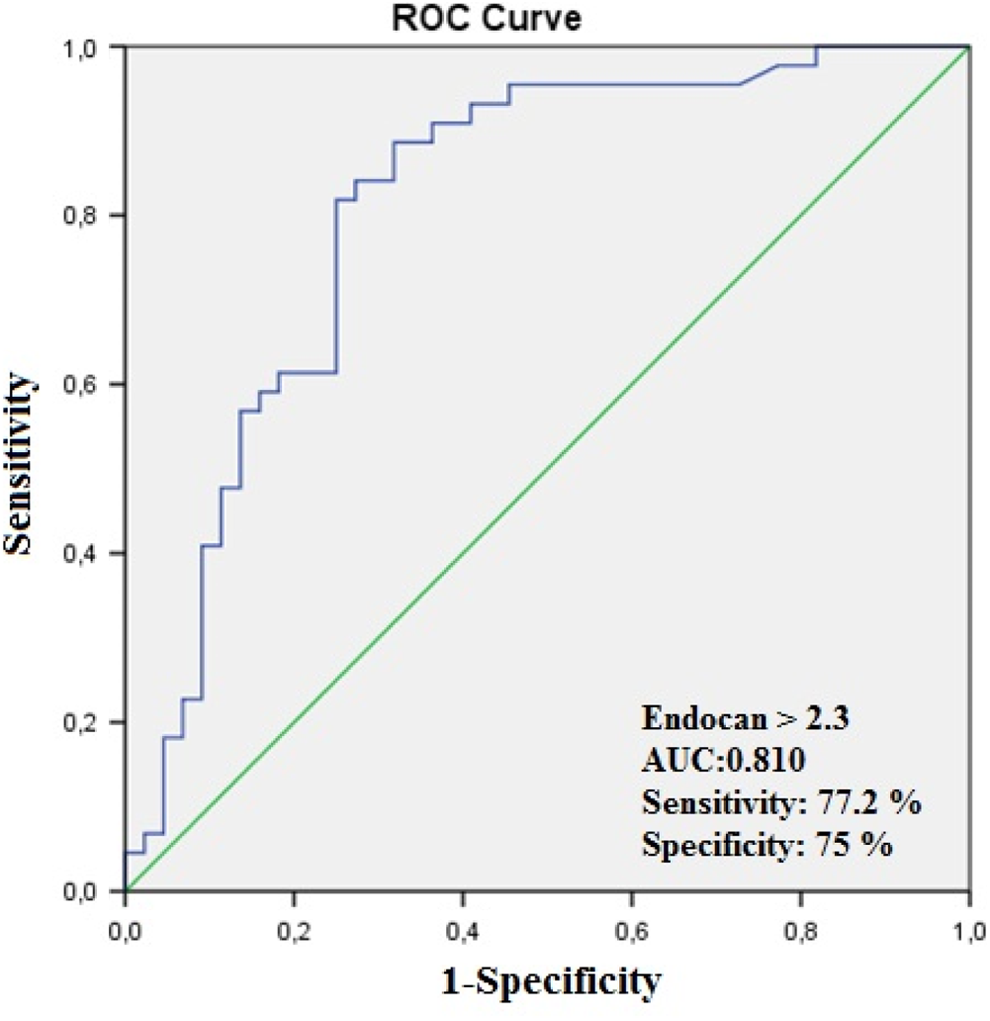
The receiver–operating characteristic (ROC) curve of endocan level for prediction of coronary slow flow.
Multivariate Logistic Regression Analysis Showing Independent Predictors of CSF.
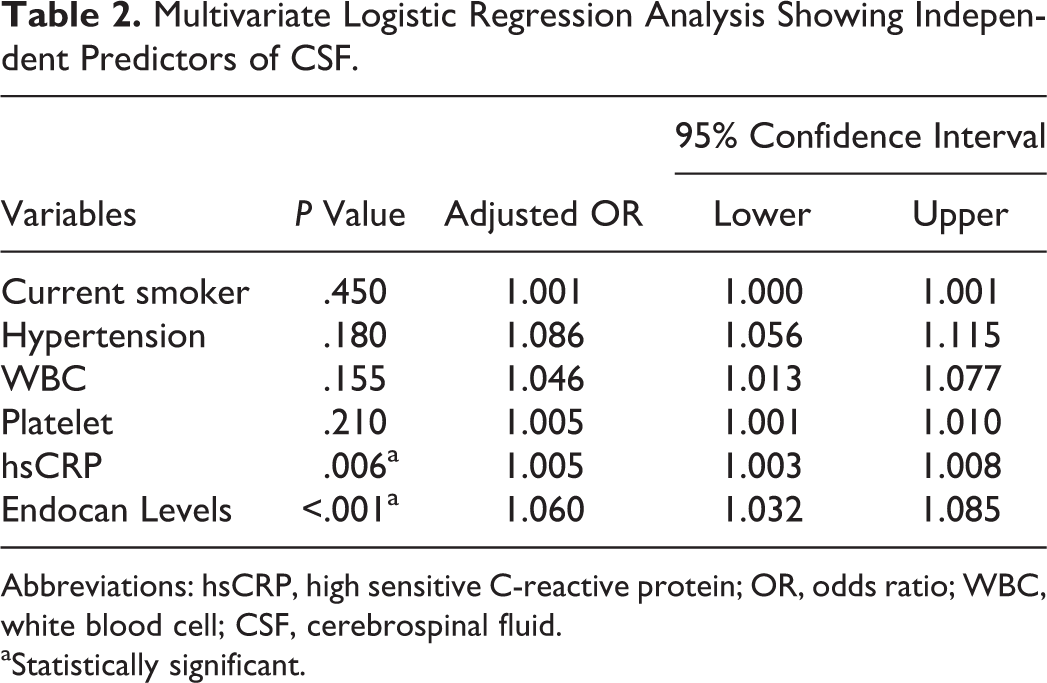
Abbreviations: hsCRP, high sensitive C-reactive protein; OR, odds ratio; WBC, white blood cell; CSF, cerebrospinal fluid.
aStatistically significant.
Clinical, Biochemical, and Angiographic Characteristics of the Study Patients According to the Endocan Level.
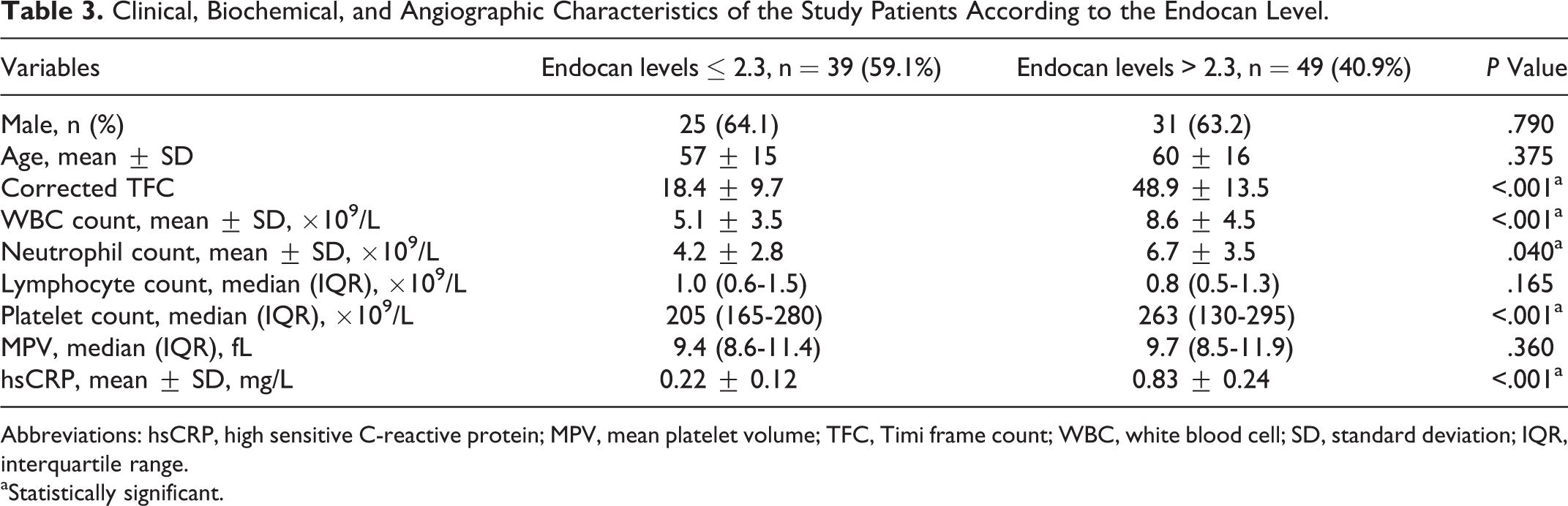
Abbreviations: hsCRP, high sensitive C-reactive protein; MPV, mean platelet volume; TFC, Timi frame count; WBC, white blood cell; SD, standard deviation; IQR, interquartile range.
aStatistically significant.
Discussion
In this study, we found significant differences between the SCF and the NCF groups for endocan levels, and the results of our study indicated that high endocan levels were independently associated with SCF. To the best of our knowledge, this is the first study that investigated endocan levels as a novel marker of inflammatory and endothelial dysfunction in patients with SCF group, and compared the results with NCF group.
Slow coronary flow, due to which opacification of major epicardial coronary arteries has been delayed at the distal segments without any atherosclerotic stenosis, is a well-known terminology by the cardiologists. 15 Besides its simple definition, the pathophysiology of SCF is still not fully understood. However, various mechanisms have been suggested in the development of SCF, endothelial dysfunction, inflammatory conditions, diffuse atherosclerosis, and coronary vasomotor dysfunction. 5,6,16 –19 On the contrary, intravascular ultrasound studies have identified epicardial CAD as a pathophysiological factor for SCF as well as microvessel disease. 20 –22 Slow flow pattern in coronary arteries has been found to be a manifestation of diffuse atherosclerotic disease due to endothelial injury without any angiographic coronary lesion. Hence, SCF may be an early manifestation of diffuse atherosclerosis involving both the microvascular system and the epicardial coronary arteries. 20,22 These observations suggest that a pathophysiological relevant interaction exists between the SCF phenomenon with endothelial dysfunction and inflammation.
Endocan is a soluble 50-kDa proteoglycan expressed by vascular endothelial cells, epithelial cells lining bronchi, renal tubules, and lung submucosal glands. 23 –26 Previous studies showed that endocan is overexpressed in several diseases such as sepsis, cancer, obesity, or inflammatory conditions. 23,27 Expression of endocan is regulated by a number of cytokines and growth factors. The lipopolysaccharide, tumor necrosis factor α (TNF-α), and interleukin (IL) 1 have been shown to induce endocan expression in vitro, while interferon γ inhibits TNF-α-induced upregulation of endocan. 28 Endocan may have a key role in the vascular contribution to endothelium-dependent pathological disorders and may represent a novel endothelial dysfunction marker. 12,29 Balta et al have previously shown that patients with Behcet disease had significantly higher levels of endocan. Also, they have shown in patients with behcet disease that endocan levels correlated positively with C-reactive protein (CRP), erythrocyte sedimentation rate, and activity of disease. Endocan levels were also higher in patients with systemic involvement. 30 Balta et al have also previously shown that patients with psoriasis vulgaris had significantly higher levels of endocan. Additionally, in patients with psoriasis, endocan levels correlated positively with cardiovascular risk and activity of disease. 10 Scherpereel et al reported that in patients with sepsis, endocan levels are related to the severity of illness and the outcome of the patient and may represent a novel endothelial dysfunction. 29 In a recent study, Altintas et al reported that endocan levels correlated with obstructive sleep apnea severity and were significantly and independently correlated with carotid intimal–media thickness. 31 Finally, in another study demonstrated that endocan levels are independently associated with the presence and severity of CAD in patients with hypertension. 32 According to these results, endocan levels, an endothelial dysfunction and inflammatory marker, may influence the pathogenesis of SCF. Therefore, we aimed to investigate a significant correlation between the presence of SCF and endocan levels.
Our findings revealed that endocan levels were significantly higher in the SCF group as compared with the NCF group. Besides its close relation with corrected TFCs, endocan also has a positive correlation with serum hsCRP level and associated with other blood inflammatory parameters such as WBC, neutrophil, and platelet counts which support its role in systemic inflammation in our study. Endocan, an indicator combining both inflammation and endothelial dysfunction, may gain a role in the prediction of SCF during daily clinical practice.
Our study has several limitations. The main limitation of our study was the relatively small sample size. Another limitation of our study is about the normal coronary arteries; even though it looks normal from the angiogram, we are not sure whether it is normal coronary arteries because we didn’t use intravascular ultrasound or optical coherence tomography. Finally, our findings were not compared with other inflammatory markers such as TNF-α, IL-1β, and IL-6.
In conclusion, our study showed that higher endocan levels were significantly and independently related to the presence of SCF. In addition, endocan levels were positively correlated with serum hsCRP level and other inflammatory markers such as WBC, neutrophil, and platelet counts. These results suggest that a higher endocan level may represent an impaired endothelial function and increased inflammatory status on coronary blood flow. The increased endocan levels may simply demonstrate an impaired function of endothelial cells, that is, endocan levels may be a surrogate of impaired endothelial function. However, it does not mean that endocan influences endothelial function, that is, endocan is the responsible factor causing the dysfunction. However, further larger studies are needed to explain the exact mechanistic role of endocan levels in SCF.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
