Abstract
Background:
Previous studies proposed that both inflammation, oxidative stress, and impaired endothelial dysfunction have a significant role in occurrence of slow coronary flow (SCF). monocyte-to-high density lipoprotein cholesterol ratio (MHR) is a recently emerged indicator of inflammation and oxidative stress, which have been studied only in patients with chronic kidney disease.
Hypothesis:
We aimed to assess the relationship between MHR and SCF.
Methods:
Patients who had angiographically normal coronary arteries were enrolled in this retrospective study (n = 253 as SCF group and n = 176 as control group). Patients who had corrected thrombolysis in myocardial infarction frame counts (cTFCs) above the normal cutoffs were defined as with SCF.
Results:
The MHR and high-sensitivity C-reactive protein (hsCRP) were significantly higher in the SCF group. In correlation analysis, MHR has a significantly positive correlation with cTFC and serum hsCRP levels (P < .001). In multivariate logistic regression analysis, MHR was found as independently associated with the presence of SCF (odds ratio: 1.24, P < .001).
Conclusion:
Higher MHR which indicates an enhanced inflammation and oxidative stress was significantly and independently associated with the presence of SCF. Besides, MHR was positively correlated with serum hsCRP level as a conventional marker for systemic inflammation.
Introduction
Slow coronary flow (SCF) is known as an angiographic definition and recognized by delayed opacification of the epicardial coronary arteries in the absence of significant coronary artery stenosis. 1 The prevalence of SCF is at about 1% among patients undergoing coronary angiogram (CAG) for stable angina pectoris. 2 The exact etiopathogenesis of SCF is unclear, but several mechanisms have been proposed including enhanced oxidative stress and inflammation, microvascular dysfunction, small vessel disease, impaired endothelial functions, and diffuse atherosclerosis. 3 –7 However, the role of inflammation, oxidative stress, and endothelial dysfunction in the pathogenesis of SCF is well established. 5,8,9 Macrophages and monocytes are the most important cell types for secretion of proinflammatory and prooxidant cytokines at the site of inflammation. 10 In addition, high-density lipoprotein cholesterol (HDL-C) has been shown to defend endothelial cells against the unfavorable effects of low-density lipoprotein (LDL) 11,12 and to prohibit oxidation of LDL molecules. 13 Thus, it was thought that HDL-C exhibits both anti-inflammatory and antioxidant actions. Recently, monocyte count to HDL-C ratio (MHR) has emerged as a new cardiovascular prognostic marker in chronic kidney disease. 14 To the best of our knowledge, no study to date has evaluated the association of MHR with the coronary flow.
Because both inflammation and oxidative stress are the main constituents of SCF and increased monocyte count and decreased HDL-C levels were shown to be related to inflammation and oxidative stress, we hypothesized that higher MHR may be associated with the presence of SCF. Hence, we aimed to investigate the relationship between MHR and SCF.
Methods
Study Population
The clinical, laboratory and angiographic data of consecutive patients who had undergone coronary angiography for stable angina pectoris and had angiographically normal coronary arteries without any atherosclerotic lesion were retrospectively recorded and analyzed at our Cardiology Clinic. A total of 253 patients with SCF and 176 patients with normal coronary flow (NCF) were included in the final analysis. The NCF group consisted of participants who were selected in a consecutive manner from the catheterized patients during the same study period. Exclusion criteria were as follows: decompensated heart failure, significant valvular heart disease, recent acute coronary syndrome (<3 months), previous revascularization history, recent infection, malignancies, blood dyscrasias, autoimmune or inflammatory diseases, renal and/or hepatic failure, current therapy with corticosteroids and nonsteroidal anti-inflammatory drugs, and hematologic diseases including anemia.
Hypertension was defined as a systolic blood pressure of ≥140 mm Hg and/or a diastolic blood pressure of ≥90 mm Hg in at least 2 measurements or use of any antihypertensive drug. Diabetes mellitus was defined as a fasting plasma glucose level of >126 or >200 mg/dL at any measurement or use of any antidiabetic agent. Smoking was defined as current smoking. The study was in compliance with the principles outlined in the Declaration of Helsinki and approved by Institutional Ethics Committee.
Coronary Angiography
All patients underwent coronary angiography with radial or femoral approach using the Judkins technique. None of the patients experienced hypotension during the procedure, which might lead to a wrong diagnosis of SCF. Indications for coronary angiography were made according to the results of noninvasive stress tests or high clinical suspicion for atherosclerotic coronary artery disease. Selective cineangiographic images of the coronaries were recorded using a digital angiographic system (AXIOM Artis; Siemens AG, Munich, Germany). The contrast agent used was iopromide (Omnipaque; GE Healthcare, Cork, Ireland).
All coronary angiograms independently quantified the coronary flow using the thrombolysis in myocardial infarction frame count (TFC) by 2 experienced invasive cardiologists blinded to the clinical details of the study groups. 12 Briefly, the number of cine frames that was recorded at 30 frames/s required for the contrast to first reach standard distal coronary landmark in the left anterior descending (LAD) artery, left circumflex (LCX) artery, and right coronary artery (RCA) was measured. The distal ends for the coronary arteries were defined as distal bifurcation for the LAD, distal bifurcation of the longest branch for the LCX, and the first side branch of the posterolateral artery for the RCA. The LAD artery is usually longer than the other major coronary arteries, and the TFC for this vessel is often higher. To obtain corrected TFC for LAD artery, TFC was divided by 1.7. The standard mean values for normal visualization of coronary arteries are described as 36.2 ± 2.6 frames for the LAD, 22.2 ± 4.1 frames for the LCX, and 20.4 ± 3 frames for the RCA as previously described. 12 Patients with a TFC greater than 2 standard deviations from the normal published range for any 1 of the 3 coronary arteries were considered to have SCF. 12 Intra- and interobserver reliabilities were determined from a random sample of 50 patients from the study sample. The κ value for intraobserver reliability was .97 (P < .001). The interobserver reliability was a κ of .94 (P < .001) for the assessment of cTFC values.
Laboratory Measurements
Blood samples were obtained from patients in the morning, after 12 hours of fasting, for measurement of biochemistry panel including lipid parameters before the index coronary angiography. Samples for the complete blood count (CBC) analysis (with differential analysis) were collected in EDTA-anticoagulated Monovette tubes (Sarstedt, Leicester, United Kingdom). Calibration was assessed daily with the commercial calibrant (Beckman Coulter; Fullerton, California), and monitored 3 times daily with internal quality control material. Monocyte count was calculated using data obtained from the CBC differential analysis. The reference value for monocyte in our laboratory is 2% to 10%. High-sensitivity C-reactive protein (hsCRP) measurement was done using an automatized analyzer (Beckman Coulter IMMAGE) using nephelometric measurement before the index coronary angiography.
Statistical Analysis
In all statistical analysis, SPSS 20.0 Statistical Package Program for Windows (SPSS Inc, Chicago, Illinois) was used. Kolmogorov-Smirnov test was used to assess normality of distribution. Continuous variables with a normal distribution were specified as the mean ± standard deviation, and categorical variables were specified with number and percentage. To compare parametric continuous variables, Student t tests was used, and to compare nonparametric continuous variables, the Mann-Whitney U test were used. The study population was assigned into tertiles based on preprocedural MHR (T1: <86.3, T2: 86.3-105.0, and T3: >105.0). Comparisons of multiple mean values were carried out by Kruskal-Wallis tests or analysis of variance as appropriate. Chi-square (χ2) test was used to compare the categorical variables. Spearman rank test was performed to define the correlation of MHR with hsCRP and cTFC. To assess the association of clinical and laboratory parameters with the presence of SCF, univariate and multivariate logistic regression analyses were performed by including the parameters that were significantly different between the groups and possible confounding factors. To evaluate model characteristics of discrimination and calibration, we used receiver–operator characteristic (ROC) curves and Hosmer-Lemeshow goodness-of-fit tests. The ROC curve was also used to demonstrate the sensitivity and specificity of MHR and the optimal cutoff value for predicting SCF. A 2-sided P value of <.05 was considered significant.
Results
A total of 429 patients were enrolled in this study (n = 253 patients in SCF group and n = 176 patients in NCF group). Baseline clinical, laboratory, and angiographic features of the study groups were represented in Table 1. The rate of hypertension and smoking was significantly higher in SCF group (P < .05). The mean cTFC value was also significantly higher in the SCF group as compared with the NCF group (P < .001). Mean number of coronary arteries with SCF was 1.78 ± 0.65. Total white blood cell (WBC) count, monocyte count, total cholesterol, triglyceride, hsCRP levels, and MHR (Figure 1A) were significantly higher in the SCF group (P < .05), whereas HDL-C level was significantly lower in the SCF group as compared with the NCF group (P < .05; Table 1). Also, Table 2 represents the baseline clinical, laboratory, and angiographic data of the patients by tertile of MHR. Both cTFC and hsCRP levels showed a significant incremental trend from tertile 1 to tertile 3 (P < .05; Figure 1B).
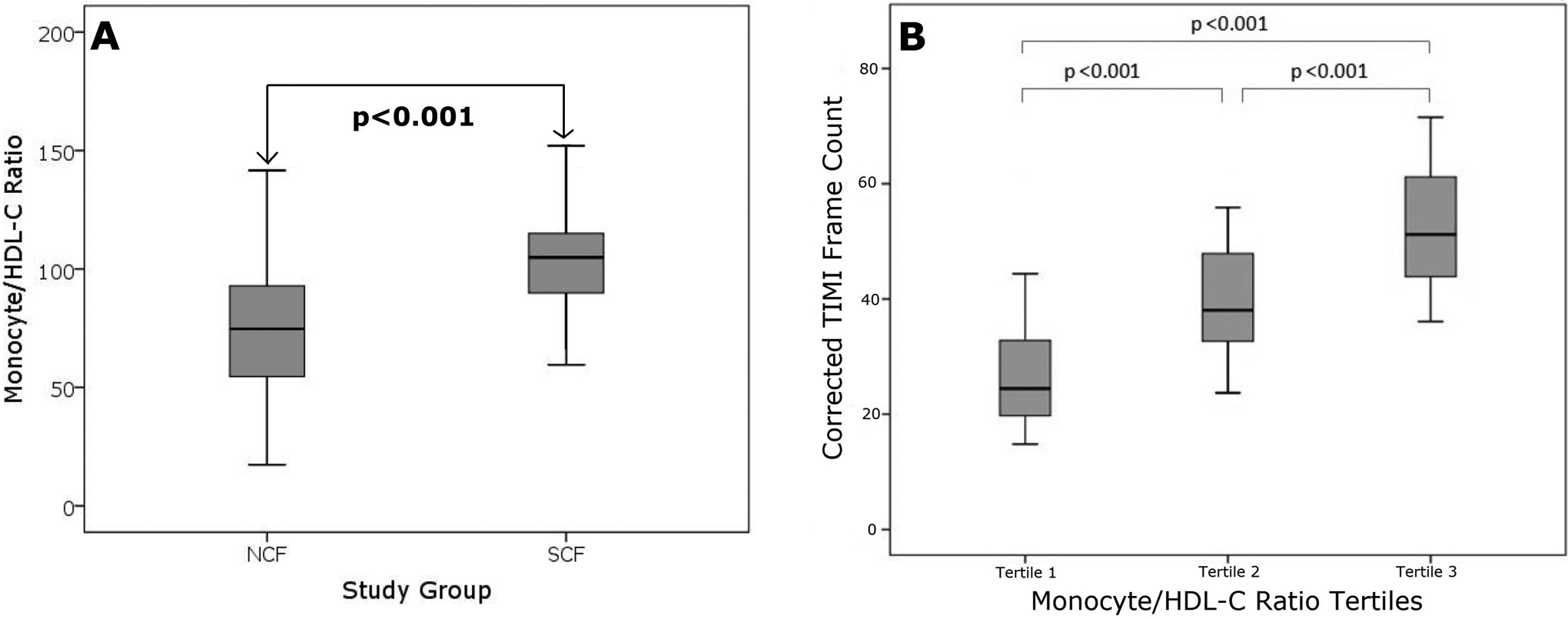
Comparison of mean circulating monocyte count to high-density lipoprotein cholesterol ratio (MHR) according to the presence of slow coronary flow (A). Corrected thrombolysis in myocardial infarction (TIMI) frame counts according to the MHR tertiles (B).
Baseline Characteristics and Laboratory Parameters of the Study Groups.a,b
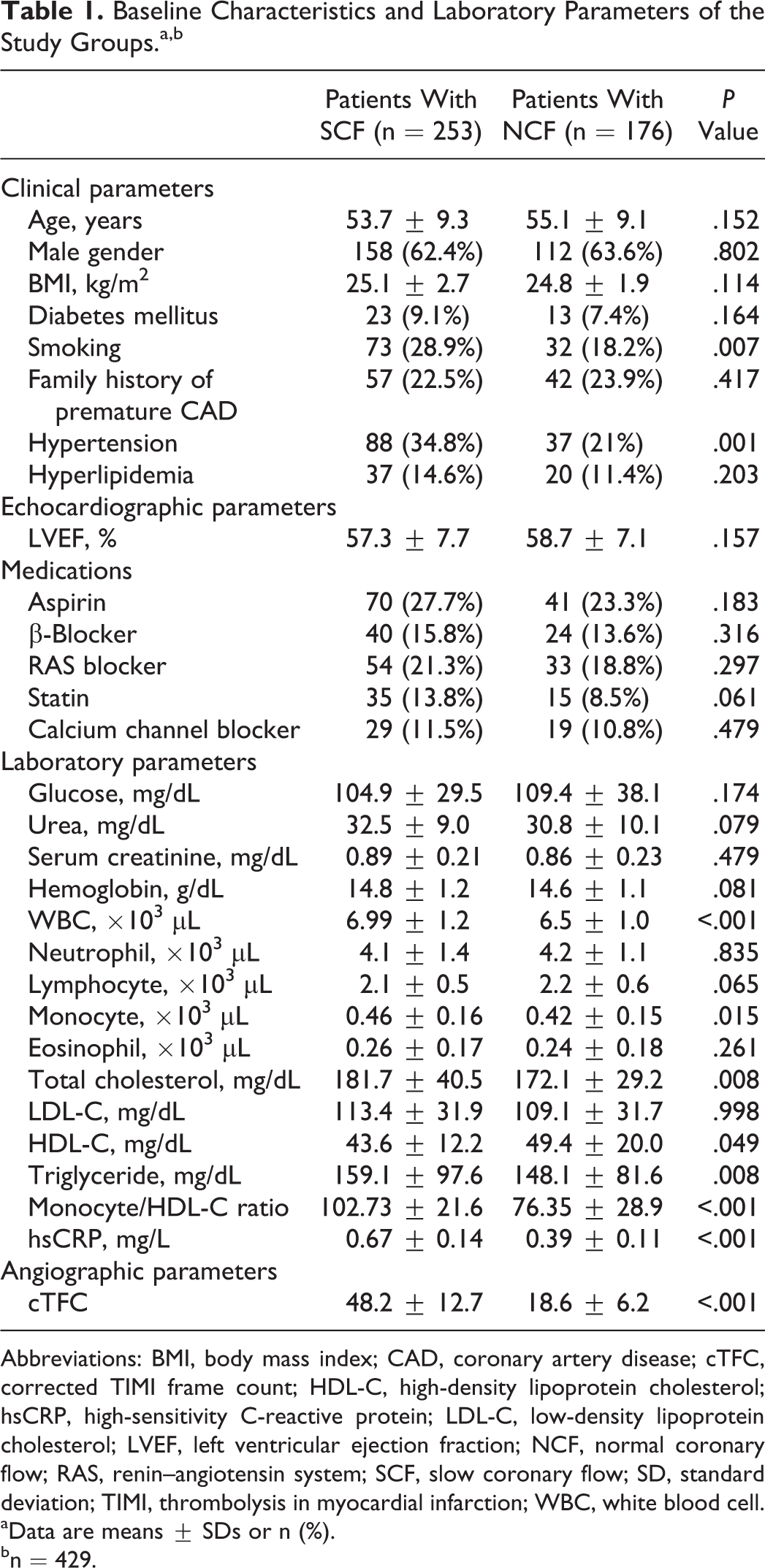
Abbreviations: BMI, body mass index; CAD, coronary artery disease; cTFC, corrected TIMI frame count; HDL-C, high-density lipoprotein cholesterol; hsCRP, high-sensitivity C-reactive protein; LDL-C, low-density lipoprotein cholesterol; LVEF, left ventricular ejection fraction; NCF, normal coronary flow; RAS, renin–angiotensin system; SCF, slow coronary flow; SD, standard deviation; TIMI, thrombolysis in myocardial infarction; WBC, white blood cell.
aData are means ± SDs or n (%).
bn = 429.
Comparison of Clinical and Laboratory Parameters of the Patients According to the Monocyte Count/HDL-C Ratio Tertiles.a,b
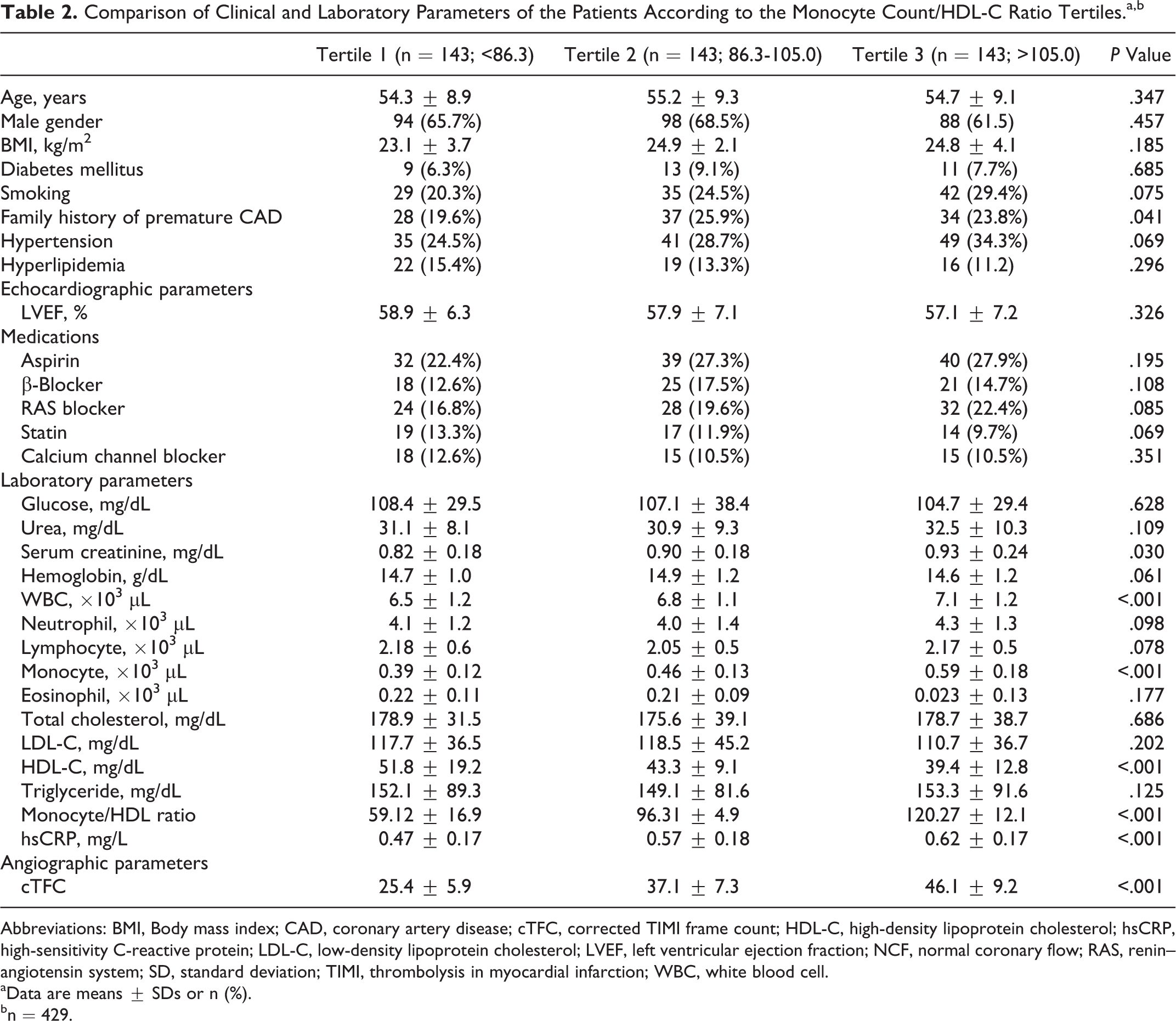
Abbreviations: BMI, Body mass index; CAD, coronary artery disease; cTFC, corrected TIMI frame count; HDL-C, high-density lipoprotein cholesterol; hsCRP, high-sensitivity C-reactive protein; LDL-C, low-density lipoprotein cholesterol; LVEF, left ventricular ejection fraction; NCF, normal coronary flow; RAS, renin–angiotensin system; SD, standard deviation; TIMI, thrombolysis in myocardial infarction; WBC, white blood cell.
aData are means ± SDs or n (%).
bn = 429.
In correlation analysis, MHR revealed a significantly positive correlation with hsCRP (r = .352, P < .001) and cTFC (r = .462, P < .001; Figure 2A and B). In multivariate logistic regression analysis, the MHR was found as independently associated with the presence of SCF (odds ratio: 1.240, 95% confidence interval [CI]: 1.230-1.451, P < .001). Moreover, hypertension, smoking, WBC count, total cholesterol, and hsCRP were also independently associated with SCF (Table 3). The ROC curve explored the discriminatory capability of MHR for the presence of SCF. Area under the curve was 0.755 (95% CI: 0.707-0.802; P < .001). Using a cutoff level of 92.5, preprocedural MHR predicted the presence of SCF with a sensitivity of 75.9% and specificity of 68.2% (Figure 2C). Moreover, MHR indicated good calibrations for the presence of SCF (Hosmer-Lemeshow; χ2 = 8.514; P value = .322).
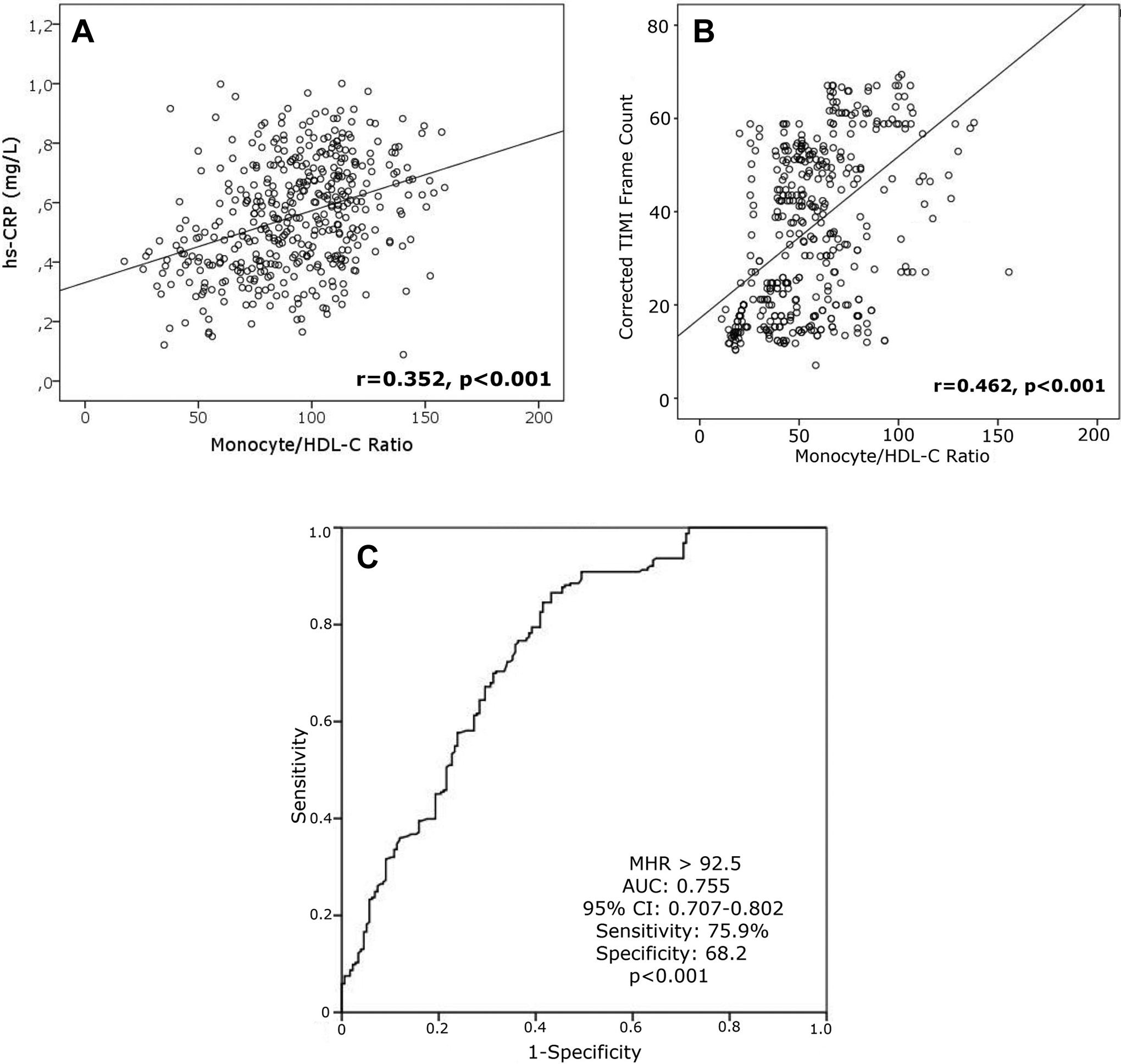
Correlation analysis representing the association of the MHR with high-sensitivity C-reactive protein (hsCRP; A) and corrected thrombolysis in myocardial infarction (TIMI) frame counts (B). Also receiver–operator characteristic (ROC) curve exploring the discriminatory capability of MHR for the presence of slow coronary flow (SCF; C). MHR indicates monocyte-to-high-density lipoprotein ratio.
Multivariate Logistic Regression Analysis Showing the Independent Predictors for the Presence of Coronary Slow Flow.
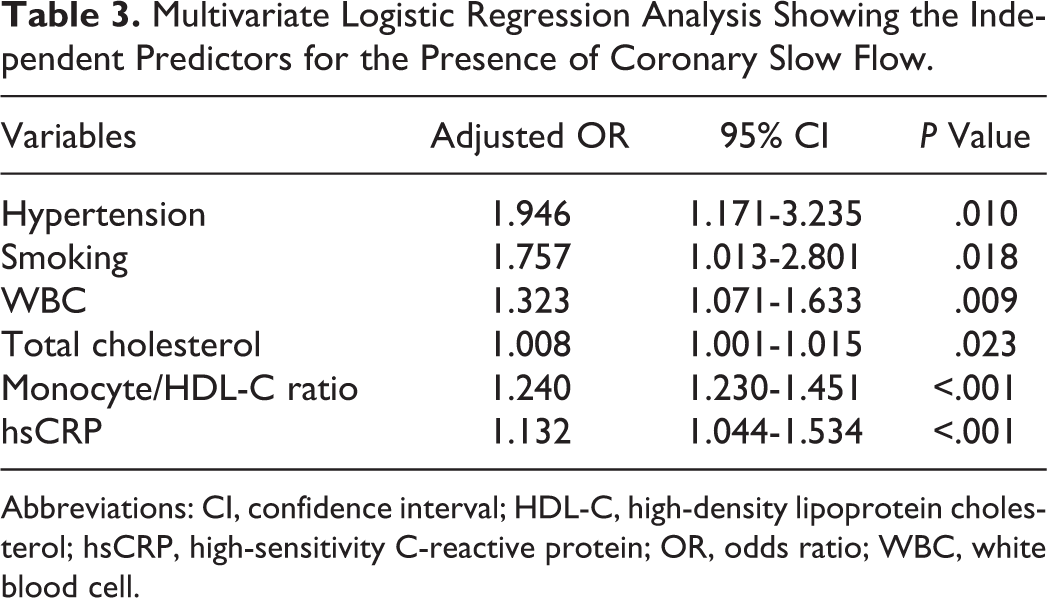
Abbreviations: CI, confidence interval; HDL-C, high-density lipoprotein cholesterol; hsCRP, high-sensitivity C-reactive protein; OR, odds ratio; WBC, white blood cell.
Discussion
Main findings of our study included (1) higher MHR before index coronary angiography significantly and independently associated with the presence of SCF and (2) MHR was also positively correlated with serum hsCRP levels showing its strong association with systemic inflammation. To the best of our knowledge, our study is the first in the literature showing the association of MHR with SCF in a relatively large number of SCF population.
Slow coronary flow is a well-known terminology by the interventional cardiologists in which opacification of major epicardial coronary arteries has been delayed at the distal segments without any atherosclerotic stenosis. 1 Besides its simple definition, the exact etiopathogenesis is unclear. However, various mechanisms have been suggested in the development of SCF-like early form of atherosclerosis, diffuse atherosclerosis, coronary vasomotor dysfunction, and impaired endothelial functions. 3,7,8 Also, both oxidative stress and systemic inflammation may be involved in the pathophysiology of SCF. 4 –6,9 Li et al 15 reported that the levels of CRP and interleukin 6 were significantly higher in patients with SCF. Barutcu et al 4 also demonstrated that hsCRP as a well-known marker of systemic inflammation might be associated with occurrence of SCF or contributes to its pathogenesis. Furthermore, it has been hypothesized that SCF phenomenon was a systemic vascular disturbance rather than a localized epicardial coronary artery pathology, which develops as an interaction between local and systemic factors. 16 As in line with previous studies, we also confirmed that total WBC count and hsCRP as a biomarker for systemic inflammation was significantly higher in patients with SCF as compared to NCF group. Therefore, the findings of all those studies as well as our study empower the knowledge that systemic inflammation plays a significant role in the etiopathogenesis of SCF.
As shown in various pathological conditions, inflammation and oxidative stress are well-known mechanisms during development and progression of atherosclerosis. Monocytes as a distinct type of leukocytes are the key player during this process. Activated monocytes interact with damaged or activated endothelium, which result in overexpression of proinflammatory cytokines/adhesion molecules including monocyte chemotactic protein 1 ligand and vascular cell adhesion molecule 1 and intercellular adhesion molecule 1. Thereafter, monocytes differentiate into the macrophages that ingest oxidized LDL cholesterol and form the dangerous foamy cells. 17 Contrarily, HDL molecules counteract the migration of macrophages and promote efflux of oxidized cholesterol from these cells. Recent studies also indicate to a role of HDL in controlling of monocyte activation, adhesiveness, and inflammation 18,19 and in controlling the proliferation of progenitor cells that give rise to monocytes. 20 Besides its anti-inflammatory and antioxidative effects, HDL molecules also enhances vasorelaxation and increased endothelial nitric oxide synthase expression. 12,21,22 Therefore, monocytes exert a proinflammatory and prooxidant effects, but HDL-C functions as a reversal factor during those processes. However, the role of both monocytes and HDL-C was less known during development of SCF. In a study, Turhan et al 23 showed that the levels of intercellular adhesion molecule 1, vascular cellular adhesion molecule, and E-selectin were significantly elevated in patients with SCF, which implies the activation of endothelium. In our study, we also found that both monocytes and WBC were higher, and HDL-C was lower in patients with SCF.
In a recent study, Kanbay et al 14 combined the circulating monocyte count and serum HDL-C in a proportion, as “MHR,” which was found as cross-sectionally associated with poor cardiovascular prognosis and arose as independent predictor of major cardiovascular events in patients with chronic kidney disease. To the best of our knowledge, no study to date has investigated the association of the MHR with SCF. We hypothesized that MHR in association with systemic inflammation and oxidative stress might play a role in SCF. Our study findings confirmed that MHR was significantly higher in the SCF group as compared with the NCF group. Besides its close association with TFCs, MHR was also has a strong positive correlation with serum hsCRP level, which supports its role in systemic inflammation in our study. From a clinical point of view, as a novel indicator combining both inflammation and oxidative stress, MHR may gain a role in prediction of SCF during daily clinical practice.
Our study should be interpreted with some limitations. First, because of the retrospective design, it lacks prognostic value of MHR on adverse cardiovascular outcomes. Second, we used a single MHR value for our analysis rather than a temporal trend. Third, we had no data regarding the coronary vasomotion and peripheral endothelial functions and intravascular ultrasound data, which clearly allows tomographic assessment of lumen area, plaque size, distribution, and composition. Also, it was unclear from our study whether the MHR and systemic inflammation have a cause and effect relationship.
In conclusion, our findings revealed that higher MHR levels were significantly and independently associated with the presence of SCF. In addition, MHR was positively correlated with serum hsCRP level as an evidenced marker of systemic inflammation. These results suggest that a higher MHR level may represent a proinflammatory and prooxidant effect on coronary blood flow. As a widely available and inexpensive parameters of the CBC and lipid panel, the MHR can be simply calculated in clinic practice for prediction of SCF. However, our findings should be confirmed in large-scale prospective studies to explain the exact mechanistic role of MHR in SCF.
Footnotes
Authors’ Note
Ugur Canpolat contributed to acquisition of data, analysis, and interpretation of data; revising it critically for important intellectual content; and final approval of the version to be published. Elif Hande Çetin contributed to acquisition of data, analysis, and interpretation of data; drafting the article; revising it critically for important intellectual content; and final approval of the version to be published. Serkan Cetin acquisition of data, analysis, and interpretation of data; drafting the article; and final approval of the version to be published. Selahattin Aydin, Osman Turak, Dursun Aras, and Sinan Aydoğdu substantial contributions to conception and design, drafting the article, and final approval of the version to be published. Mehmet Kadri Akboga contributed to acquisition of data, drafting the article, and final approval of the version to be published. Cagri Yayla substantial contributions to conception and design, revising it critically for important intellectual content, and final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
