Abstract
Introduction:
Inappropriate use of oral anticoagulants (OACs) have not been well investigatedand, however, may be frequent in real-world practice in patients with nonvalvular atrial fibrillation (NVAF). This study was designed to evaluate the prescription patterns and appropriateness of OACs in patients with NVAF in real-world clinical settings.
Methods:
We performed a prospective, observational study (NCT02366338). A total of 148 patients with NVAF were screened for OAC prescription. Appropriateness of prescribing was evaluated using 9 criteria of the Medication Appropriateness Index (MAI): indication, choice, dosage, modalities and practicability of administration, drug–drug interactions, drug–disease interactions, duplication, and duration. For each criterion, the evaluator has to rate whether the medication is (A) appropriate, (B) inappropriate but with limited clinical importance, and (C) inappropriate.
Results:
Of 148 patients, 73 (50%) were on warfarin (group 1), 39 (26%) were on rivaroxaban (group 2), and 36 (24%) were on dabigatran therapy (group 3). The MAI showed that 83% of group 1, 28% of group 2, and 47% of group 3 patients had at least 1 inappropriate criterion. Moreover, according to the choice criterion, 37% of group 1, 8% of group 2 and 5% of group 3 were rated as inappropriate, and dosage was not appropriate in 77% of group 1, 23% of group 2, and 42% of group 3.
Conclusion:
Inappropriate drug use is frequent among patients with NVAF not only for warfarin but also for NOACs. Although there is an apparent improvement in thromboprophylaxis of NVAF, much more effort is needed for appropriate use of OACs.
Keywords
Introduction
Atrial fibrillation (AF), the most common sustained arrhythmia, is the major cause of acute ischemic stroke. 1 Vitamin K antagonists (VKAs) have been the only available oral anticoagulants (OACs) for decades in nonvalvular AF (NVAF) for prevention of stroke. The risk of stroke was reported to decline up to 68% with OAC therapy, associated with good anticoagulation control with VKAs, assessed by time in therapeutic range (TTR). 2 However, previous randomized controlled trials and real-life settings have controversies regarding TTR values. 3 In low TTR values, VKAs were found to be associated with severe complications, and a minimum TTR of 58% should be achieved to expect a net benefit from being on OAC therapy. 4 Furthermore, the recently updated guidelines of the European Society of Cardiology (ESC) for the management of AF recommended a higher percentage of TTR (>70%) for patients on VKA therapy. 5 Narrow therapeutic index, drug–drug interactions, and the need for close monitoring are the main disadvantages of VKAs, and management of patients has dramatically improved after the introduction of non-vitamin K antagonist OACs (NOACs). The NOACs have a more predictable anticoagulant affect, which allows a fixed-dose regimen. Although these new agents are user friendly, previous studies have shown an inappropriate use up to 25% with NOACs. 6 The main advantages of NOACS such as “fixed-dose regimen” and “no need for regular anticoagulant therapy monitoring” may also be the Achilles heel of the use of these agents. Fixed-dose regimen may not be appropriate for elderly individuals, chronic kidney disease, and for patients using interacting drugs. Medication appropriateness index (MAI) is a comprehensive and validated tool for evaluating prescribing quality in older adults. 7 The MAI examines the appropriateness of a medication by 10 questions which can be rated as A, B, or C. There are general instructions and specific definitions of each criterion to provide a better inter- and intrarater reliability. It has been used in various trials both randomized or prospective and recently in an NOAC trial. 6 Although there are limited number of studies demonstrating inappropriate use of NOACs, a comprehensive study evaluating inappropriate use of VKAs and NOACs has not been performed yet. Therefore, we planned a prospective study for the assessment of appropriate use of VKAs and NOACs in NVAF.
Methods
We performed a prospective, observational study in Mugla Sitki Kocman University Education and Research Hospital outpatient cardiology clinics between August and November 2014 (NCT02366338). Patients were included if they were ≥18 years of age and had NVAF. Nonvalvular atrial fibrillation was defined as AF without mitral stenosis or prosthetic heart valve. Patients were excluded if they were not receiving OAC at enrollment. We also excluded patients who were acutely ill or requiring hospitalization. Dabigatran, rivaroxaban, and warfarin were the drugs of interest. Patients who had been treated with warfarin for less than 3 months were excluded, while all dabigatran and rivaroxaban patients were included. Apixaban was not considered because this drug has recently been approved in NVAF at the time of the study in Turkey. All patients gave written informed consent, and the study protocol was approved by the institutional ethics committee.
Detailed history was obtained from all patients, including medical history, medications, and appropriate use of anticoagulants. Patients’ history regarding hypertension, diabetes mellitus, coronary artery disease, and myocardial infarction was questioned. We inquired about any history of stroke, transient ischemic attack, or noncentral nervous system embolism. Atrial fibrillation was diagnosed if the patient had a physician-confirmed diagnosis or by a 12-lead electrocardiography with irregular rhythm.
Thromboembolic risk score was calculated by CHA2DS2VASc score (1 point each for congestive heart failure/left ventricular systolic dysfunction, hypertension, diabetes, vascular disease [prior myocardial infarction, complex aortic plaque, peripheral artery disease], age 65-74 years and female gender, and 2 points for prior stroke/transient ischemic attack or age ≥75 years). The bleeding risk score was calculated by HAS-BLED (1 point each for uncontrolled hypertension, [eg, systolic blood pressure >160 mg] abnormal liver tests [eg, AST/ALT >3 ULN], abnormal renal tests [eg, hemodialysis patient], previous stroke, history of bleeding, labile international normalized ratio [INR; TTR <60%], elderly population [age >65 years], and concomitant antiplatelet or nonsteroidal anti-inflammatory drug use and alcohol excess). The TTR was calculated with the Rosendaal method for each patient. 8 This method is also known as linear interpolation method. Gaps of 56 days or more among INR values were not interpolated. We also calculated the percentage of INRs at range for each individual. SAMe-TT2R2 score (1 point each for female gender, age <60 years, medical history [2 of the following: hypertension, diabetes, myocardial infarction, peripheral artery disease, congestive heart failure, previous stroke, pulmonary disease, and hepatic or renal disease], treatment strategy [eg, rhythm control with amiodarone], and 2 points for tobacco use [within 2 years], and race [non-Caucasian]) is a clinical decision-making tool that was proposed to identify those patients who would do well on VKA (SAMe-TT2R2 score ≤2) or those who would not (SAMe-TT2R2 score >2). 9 The score was calculated to assess whether the individual patient was a candidate for warfarin therapy.
Prescribing appropriateness of OACs was assessed using the MAI. 7 The MAI is a comprehensive, validated tool to measure appropriateness of prescribing that uses 10 criteria: indication, choice, dosage, modalities and practicability of administration, drug–drug interactions (DDIs), drug–disease interactions, duplication, duration, and cost-effectiveness. The medication is rated “A” if it is appropriate, “B” if it is inappropriate with limited clinical importance, and “C” if it is inappropriate. The last criterion cost-effectiveness was not evaluated in our study due to the complexity of the cost-effectiveness calculation. We adopted MAI for NOACs (dabigatran and rivaroxaban) and for warfarin. A summated MAI score was used for the assessment of appropriateness of NOACs and warfarin. 10 Duration criterion was used to evaluate patient adherence for NOACs. The number of capsules dispensed by the pharmacy and duration of anticoagulant therapy for each patient was calculated. Adherence was obtained by dividing the number of daily doses dispensed to the number of days from the first prescription until the last refill and expressed as percentage. A cutoff value of 80% was used for appropriateness. 11 The appropriateness of warfarin therapy according to choice criterion was assessed by the time intervals between INR monitoring. At least 2 intervals >8 weeks were considered as inappropriate choice of the drug. Adherence to warfarin treatment was assessed by TTR. A TTR <60% was considered inappropriate according to dosage criterion. Detailed instructions of the MAI are listed in Appendices A and B. The primary outcome of interest was inappropriate use of OACs.
Descriptive statistics are given as mean ± SD for continuous variables and as numbers and percentages for categorical variables.The chi-square or Fisher exact test was used to compare categorical variables, and Student t test or analysis of variance was used for continuous variables. The Kruskal-Wallis test was used for the comparison of an ordinal scaled variable or a quantitative variable not normally distributed. Differences were considered statistically significant if P < .05. The statistical analysis was performed using SPSS Inc Released 2009 PASW Statistics for Windows, Version 18.0 (Chicago, Illinois: SPSS Inc).
Results
A total number of 148 patients were enrolled. Among the patients, 78 (50%) were on warfarin (group 1), 39 (26%) were on rivaroxaban (group 2), and 36 (24%) were on dabigatran therapy (group 3). The median durations of OAC therapy were 16 (3-35), 8 (1-15), and 12 (3-18) months for groups 1, 2, and 3, respectively. Eleven (15%) patients of group 1, 5 (13%) patients of group 2, and 11 (30%) patients of group 3 had a history of ischemic stroke. The mean CHA2DS2VASc score was 3.5 ± 1.5, and the mean HAS-BLED score was 2 ± 0.9 for the entire group. The baseline characteristics of patients are listed in Table 1.
Baseline Characteristics.a
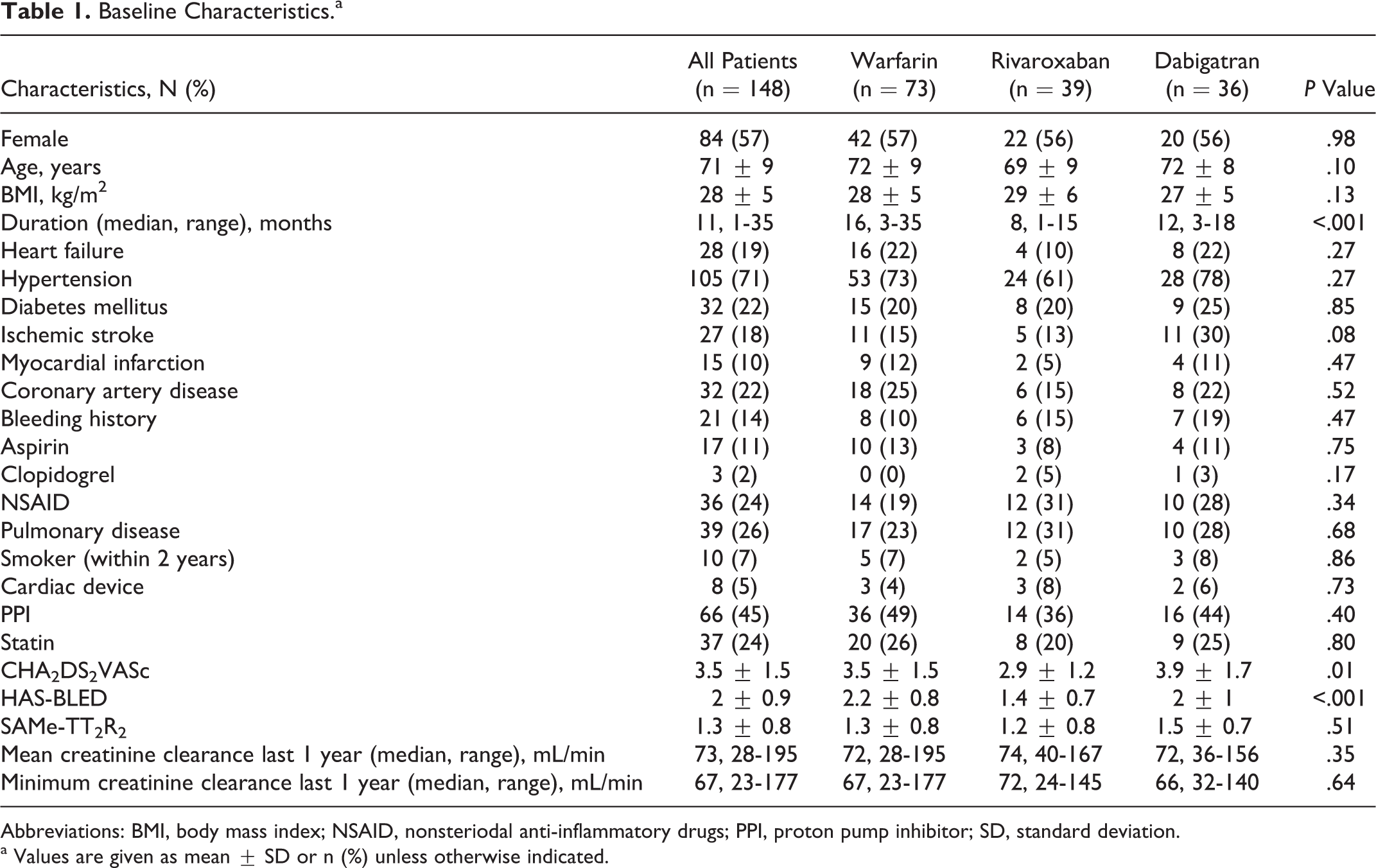
Abbreviations: BMI, body mass index; NSAID, nonsteriodal anti-inflammatory drugs; PPI, proton pump inhibitor; SD, standard deviation.
a Values are given as mean ± SD or n (%) unless otherwise indicated.
Table 2 illustrates the MAI criteria for each group. Seven patients were rated inappropriate in the “indication” criterion due to CHA2DS2VASc score: 2 patients in group 1; 3 patients in group 2; and 2 patients in group 3. If a patient is rated inappropriate for indication criterion, he or she is also rated inappropriate for choice, dosage, modalities, and practicability of administration and duration criteria according to the instructions of the MAI.
Medication Appropriateness Index Criteria According to Oral Anticoagulant Drugs.
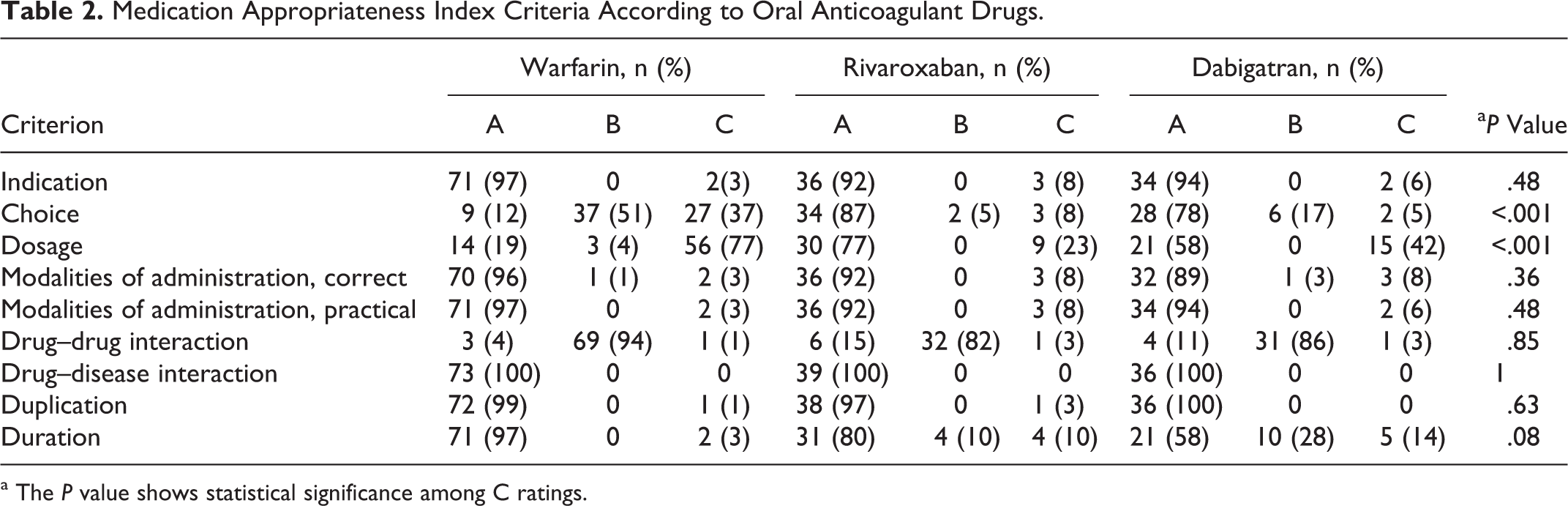
a The P value shows statistical significance among C ratings.
Choice and dosage were the most frequent inappropriate criteria for all OACs. The INR monitoring frequency and SAMe-TT2R2 score were used for the evaluation of choice criterion for group 1; however, only SAMe-TT2R2 score was used for groups 2 and 3. Thirty-seven percent of patients in group 1, 8% of patients in group 2, and 5% of patients in group 3 were inappropriate according to the choice criterion (Figure 1).
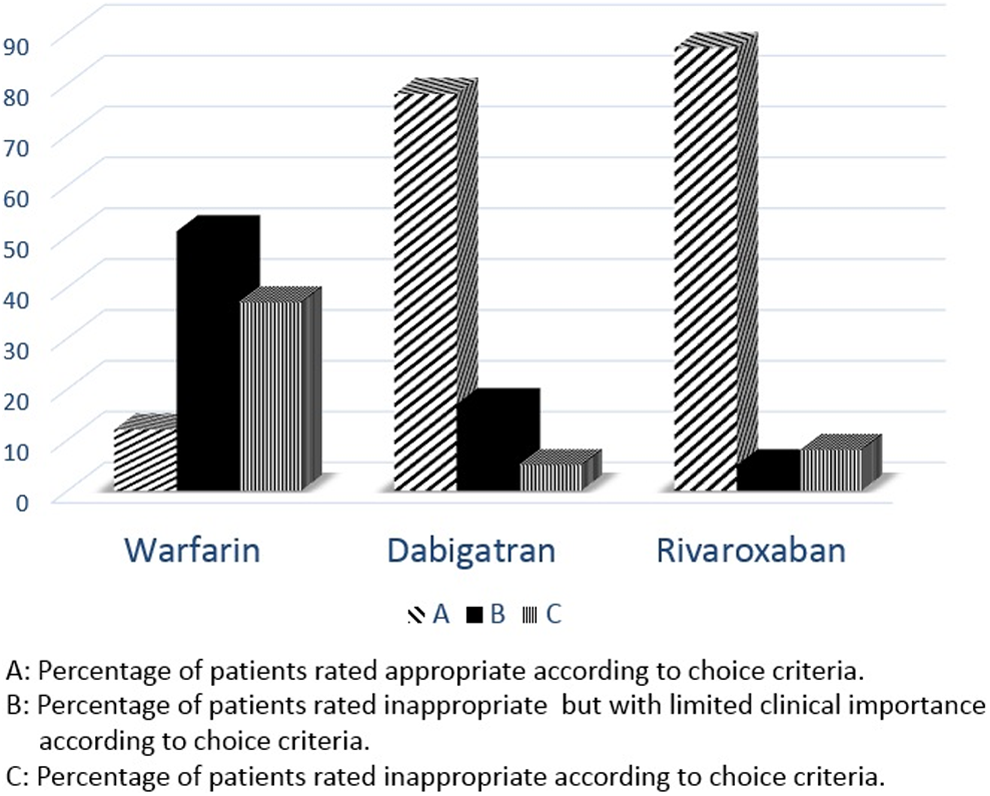
Appropriateness of oral anticoagulants (OAC) therapy according to choice criterion.
Inappropriateness of dosage criterion was evaluated by TTR in group 1, and the appropriateness of the dosage of the drug was assessed by patients’ creatinine clearance, age, hemorrhagic risk, and concomitant drug use (eg, verapamil with dabigatran) in groups 2 and 3. For dosage criterion, 56 (77%) of group 1, 9 (23%) of group 2, and 15 (42%) of group 3 patients were rated as inappropriate (Figure 2). The mean TTR was 40.5 ± 24.4, while only 18% of patients had 65% or higher TTR.
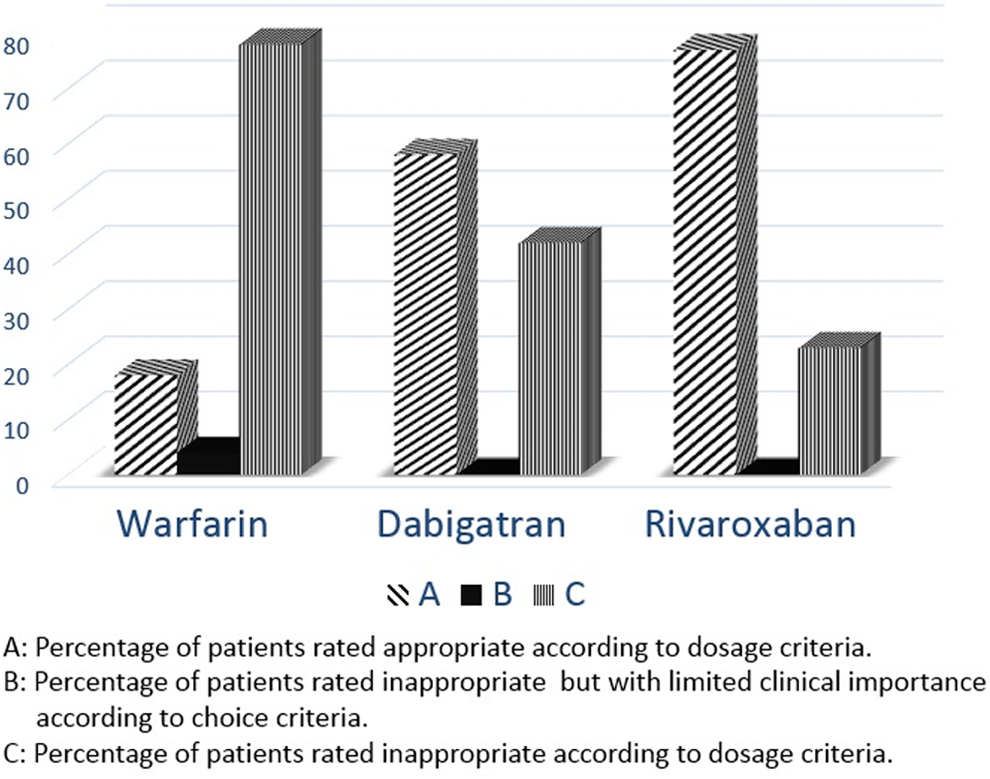
Appropriateness of oral anticoagulants (OAC) therapy according to dosage criterion.
Eighty-three percent of patients in group 1, 28% of patients in group 2, and 47% of patients in group 3 had at least 1 inappropriate criterion. If dosage criterion was removed from MAI, the inappropriateness ratios would decline to 38%, 13%, 11% for groups 1, 2, and 3, respectively. Although higher doses were indicated, 13% of group 2 and 27% of group 3 patients were receiving lower dosages of the drugs (15 mg for rivaroxaban and 110 mg for dabigatran) according to the dosage criterion.
One hundred and twenty-one (82%) patients were taking OACs for primary stroke prevention, while 27 (18%) patients were on OAC therapy for secondary stroke prevention. The MAI revealed a higher proportion of inappropriateness in secondary stroke prevention subgroup; 11 (100%) of 11 in group 1, 0/ (0%) of 5 in group 2, and 4 (36%) if 11 in group 3 had at least 1 inappropriate criterion.
The mean summated MAI score was 2.9 ± 2.5 for group 1, 1.4 ± 3.5 for group 2, and 1.5 ± 3 for group 3. Patients in the group 1 had significantly higher MAI summated score than groups 2 and 3 (P < .001). Adherence for NOACs was good, only 10% of group 2 and 14% of group 3 patients were rated inappropriate for duration criterion. The detailed ratings of MAI are listed in Appendix C.
Discussion
To our knowledge, this is the first comprehensive study evaluating the appropriateness of warfarin with MAI. This study demonstrated that inappropriate prescribing of OACs is common among patients with NVAF, and nearly four-fifths of warfarin, half of dabigatran, and a quarter of rivaroxaban patients had at least 1 inappropriate criterion according to MAI. The most frequent inappropriate criteria were wrong choice and dosage of the OACs.
The recently updated guidelines of the ESC for the management of AF recommended no antithrombotic therapy for patients with CHA2DS2VASc score of 0 who are at low risk. 5 However, OAC therapy was reported to be used in 2% to 20% of patients with CHA2DS2VASc score of 0 in recent studies. 12,13 In our study, inappropriate indication (3% to 8%) was relatively low and was comparable between VKAs and NOACs. These results may be explained by the fact that the prescription of NOACs is restricted to cardiologists in our country, and patients with NVAF at risk of stroke are primarily assessed by cardiologists in our region. Although CHA2DS2VASc score is convenient for use in daily clinical practice, serial assessment of this score is necessary to select appropriate candidates for OAC therapy.
Another problematic issue on the prevention of stroke in patients with NVAF is the choice of the drug that has come into prominence after the introduction of NOACs. In our study, choice of the drug was rated inappropriate for the patients on warfarin therapy if regular INR measurements could not be obtained. The recommended follow-up interval after obtaining an INR in range of 2.0 to 3.0 varies from 4 to 8 weeks, 14 and according to choice criterion, 37% of warfarin patients were rated inappropriate in our study. The SAMeTT2R2 score that has been developed to assist clinicians in deciding whether the individual patient is likely to benefit from VKA (SAMeTT2R2 score ≤2) or not (SAMeTT2R2 score >2) was also used for the evaluation of choice criterion. The SAMeTT2R2 score has been validated in prospective and real-world clinical trials, and for patients with a SAMeTT2R2 score >2, an NOAC should be considered instead of warfarin. 15
The main reason for inappropriate NOAC prescribing is erroneous dosage and can be avoided with a careful assessment of patients. Inappropriate NOAC dosage varies from 16% to 25% in previous studies. 6,16 Twenty-three percent of rivaroxaban and 42% of dabigatran patients had inappropriate rating due to dosage criterion in our study. Sixty-one percent of dabigatran and 15% of rivaroxaban patients were receiving the lower dosages (110 mg dabigatran and 15 mg rivaroxaban) of the drugs. However, the lower dosage of the drug was not appropriate in 13% of rivaroxaban and in 27% of dabigatran patients. Prescribing patterns of physicians may be influenced by their experience and fear of bleeding events which might have lead to the strategy of “play it safe” by giving the lower doses. This concern was noticed by Food and Drug Administration and dabigatran 150 mg but not 110 mg was approved in the prevention of stroke in patients with NVAF. 17
Warfarin dose adjustment is based on INR values, and the quality of warfarin treatment can be assessed by TTR. Although the recommended TTR values could be achieved in randomized controlled trials, observational studies in real-life settings revealed lower TTR than expected. 18 –20 A recent tertiary center study from our country revealed a TTR value of 47% among patients on warfarin therapy. 21 Patients’ nonadherence to therapy, difficulties in access to health care, and lower INR goals for patients at high risk of bleeding may lead to undertreatment of patients. In our study, overall TTR was found to be 40.5%, and warfarin dosage and adherence assessed by TTR were not appropriate in 77% of patients.
Medication nonadherence in chronic use of cardiovascular drugs may be up to 50% which is a major problem regarding the safety and effectiveness of any medical therapy and may be a topic of interest in chronic use of OACs. 22,23 Adherence to NOAC therapy seems adequate in recent studies, however, there are some concerns regarding chronic use of these drugs. 24 –26 Modalities of administration and duration criteria were used to evaluate adherence to NOACs in our study. Twice-daily dosing and gastrointestinal adverse effects of dabigatran may be the reason of poor adherence, whereas the need to take rivaroxaban with food may be a cause of undercoagulation. In our study, 10% to 14% of patients were nonadherent to NOAC therapy according to proportion of days covered >80%. As the NOACs are relatively new drugs, long-term follow-up data are needed for a precise judgment. The DDI is another issue that may make a drug less or more effective. Although DDI is common among patients on OAC therapy, the clinical significance is not clear. The NOACs have rare DDIs comparing VKAs. However, there is a clinically significant DDI for NOACs with drugs effecting cytochrome P450 3A4 and P-glycoprotein.
The MAI provides objective information regarding inappropriate use of OACs. However, inappropriateness rates may change by the description of these criteria. 27 A summated MAI score which may partly overcome this problem was previously used to compare interventions made by pharmacists, and a higher score indicated a worse outcome. 28 In our study, patients who were on warfarin had significantly higher summated MAI score compared to NOACs. In a recent study by Larock et al, MAI was used to assess appropriateness of NOACs in hospitalized patients. 6 However, they did not include patients on VKAs, and therefore they could not compare appropriateness of prescribing between NOACs and VKAs. In our study, MAI revealed NOACs do better though there is a need for improvement.
The strength of our study is the comparison of appropriateness between OACs. A limitation is that this is a single-center study with relatively small number of patients. The study included cardiology outpatient clinics that might cause a selection bias. However, prescription of NOACs is restricted to cardiologists in our country, and follow-up of patients on OAC therapy is mainly done by cardiologists. We showed a high proportion of prescriptions were inappropriate, however, these results cannot be generalized to all patients. Second, we did not follow-up patients for adverse events which might correlate with MAI. Finally, accuracy of MAI depends on the description of the criteria, however, there is no uniform definition of every MAI criterion. We also have to attract attention that there is a growing need of clinical decision support for all involved tools not just for anticoagulants. These well-documented, clinically comprehensive tools will be quite helpful in daily clinical practice.
Conclusion
Thromboprophylaxis is a challenging topic in NVAF. Patients who may benefit from OAC therapy should be identified by CHA2DS2VASc score and anticoagulated if needed. Although VKAs are effective agents in thromboprophylaxis of stroke in NVAF, a low TTR is associated with worse outcome. Thus, long awaited invention of NOACs in the field of anticoagulation brought physicians a safer option, especially in centers with low TTR. Although these new agents offer many advantages over VKAs, they require new approaches regarding good prescribing. Choosing the right dosage is one of the problems and can be overcome by a careful assessment of patients’ characteristics. Adherence to NOAC therapy is another concern as it may be as low as 50% in the chronic use of cardiovascular drugs, especially if the drug has no apparent affect to the patient. Our study showed that although inappropriate use of NOACs are lower than the VKAs, it may be more than anticipated when evaluated with MAI, and physicians should be encouraged for appropriate use of these new agents.
Footnotes
Appendix A
Medication Appropriateness Index Adapted for Non-Vitamin K Antagonists.
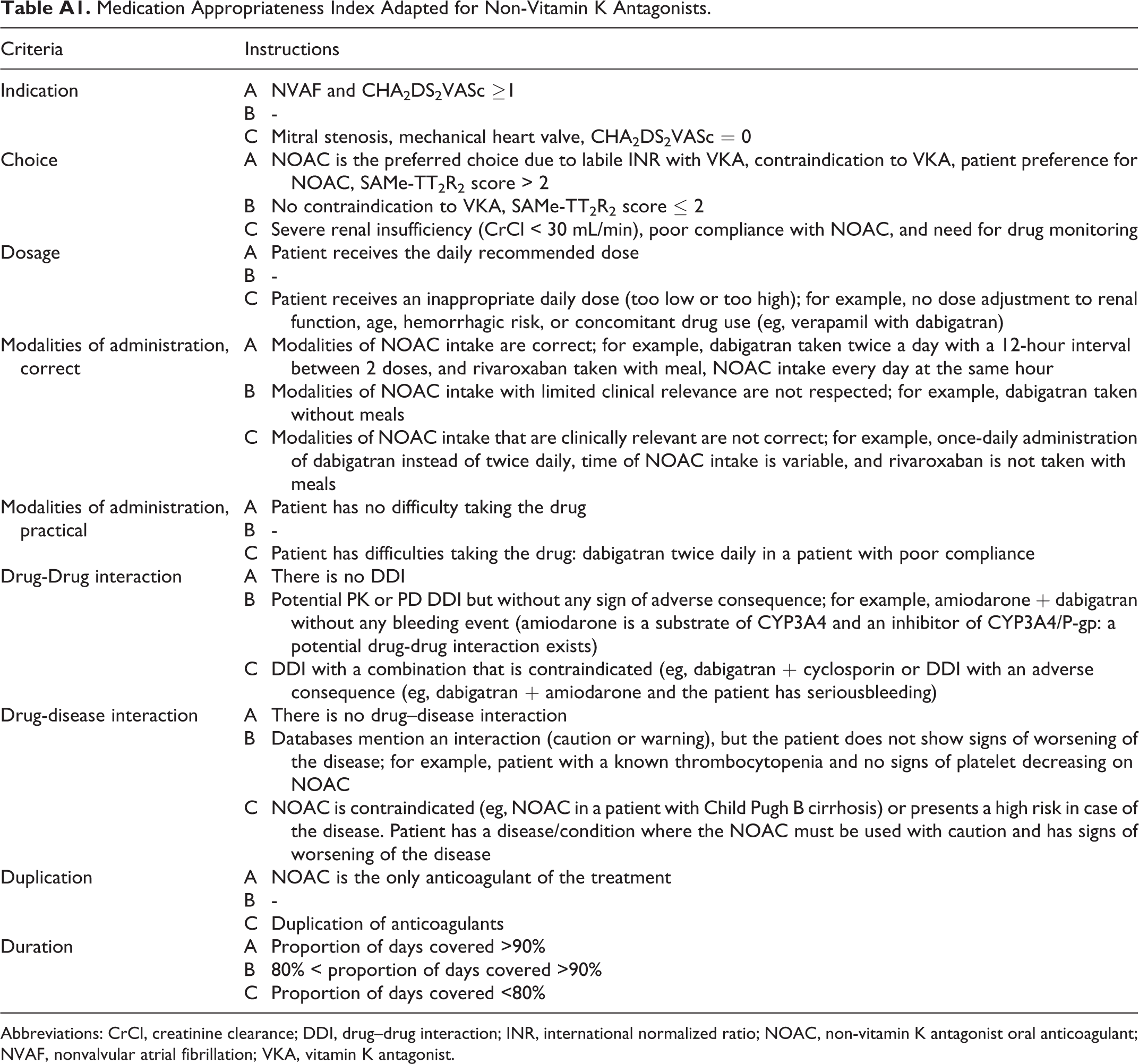
| Criteria | Instructions | |
|---|---|---|
| Indication | A | NVAF and CHA2DS2VASc ≥1 |
| B | - | |
| C | Mitral stenosis, mechanical heart valve, CHA2DS2VASc = 0 | |
| Choice | A | NOAC is the preferred choice due to labile INR with VKA, contraindication to VKA, patient preference for NOAC, SAMe-TT2R2 score > 2 |
| B | No contraindication to VKA, SAMe-TT2R2 score ≤ 2 | |
| C | Severe renal insufficiency (CrCl < 30 mL/min), poor compliance with NOAC, and need for drug monitoring | |
| Dosage | A | Patient receives the daily recommended dose |
| B | - | |
| C | Patient receives an inappropriate daily dose (too low or too high); for example, no dose adjustment to renal function, age, hemorrhagic risk, or concomitant drug use (eg, verapamil with dabigatran) | |
| Modalities of administration, correct | A | Modalities of NOAC intake are correct; for example, dabigatran taken twice a day with a 12-hour interval between 2 doses, and rivaroxaban taken with meal, NOAC intake every day at the same hour |
| B | Modalities of NOAC intake with limited clinical relevance are not respected; for example, dabigatran taken without meals | |
| C | Modalities of NOAC intake that are clinically relevant are not correct; for example, once-daily administration of dabigatran instead of twice daily, time of NOAC intake is variable, and rivaroxaban is not taken with meals | |
| Modalities of administration, practical | A | Patient has no difficulty taking the drug |
| B | - | |
| C | Patient has difficulties taking the drug: dabigatran twice daily in a patient with poor compliance | |
| Drug-Drug interaction | A | There is no DDI |
| B | Potential PK or PD DDI but without any sign of adverse consequence; for example, amiodarone + dabigatran without any bleeding event (amiodarone is a substrate of CYP3A4 and an inhibitor of CYP3A4/P-gp: a potential drug-drug interaction exists) | |
| C | DDI with a combination that is contraindicated (eg, dabigatran + cyclosporin or DDI with an adverse consequence (eg, dabigatran + amiodarone and the patient has seriousbleeding) | |
| Drug-disease interaction | A | There is no drug–disease interaction |
| B | Databases mention an interaction (caution or warning), but the patient does not show signs of worsening of the disease; for example, patient with a known thrombocytopenia and no signs of platelet decreasing on NOAC | |
| C | NOAC is contraindicated (eg, NOAC in a patient with Child Pugh B cirrhosis) or presents a high risk in case of the disease. Patient has a disease/condition where the NOAC must be used with caution and has signs of worsening of the disease | |
| Duplication | A | NOAC is the only anticoagulant of the treatment |
| B | - | |
| C | Duplication of anticoagulants | |
| Duration | A | Proportion of days covered >90% |
| B | 80% < proportion of days covered >90% | |
| C | Proportion of days covered <80% |
Abbreviations: CrCl, creatinine clearance; DDI, drug–drug interaction; INR, international normalized ratio; NOAC, non-vitamin K antagonist oral anticoagulant; NVAF, nonvalvular atrial fibrillation; VKA, vitamin K antagonist.
Appendix B
Medication Appropriateness Index Adapted for Warfarin.

| Criteria | Instructions | |
|---|---|---|
| Indication | A | AF and CHA2DS2VASc score≥1 |
| B | - | |
| C | CHA2DS2VASc score = 0 | |
| Choice | A | Patient preference, CrCL < 30 mL/min, poor compliance to NOAC, need for drug monitoring, and drug monitoring intervals <4 weeks, SAMe-TT2R2 score ≤2 |
| B | Recurrent stroke on VKA, drug monitoring intervals 4-8 weeks, and SAMe-TT2R2 score >2 | |
| C | Severe hepatic impairment, VKA contraindicated, recurrent stroke on VKA and TTR<60%, and drug monitoring intervals >8 weeks | |
| Dosage | A | TTR >65% |
| B | TTR 60%-65% | |
| C | TTR <60% | |
| Modalities of administration, correct | A | Patient receives daily dosage at the same time of the day. |
| B | Patient receives daily dosage at the different times of the day. For example, one day in the morning the other day in the evening | |
| C | Patient forgets to take daily dosage | |
| Modalities of administration, practical | A | Patient has no difficulty taking the drug |
| B | - | |
| C | Patient has difficulties taking the drug. For example, patient cannot split the drug (only 5 and 10 mg of warfarin are available in Turkey) | |
| Drug–drug interaction | A | No drug–drug interaction |
| B | Drug–drug interaction without any bleeding event or change in INR | |
| C | Drug-drug interaction with bleeding event or change in INR | |
| Drug–disease interaction | A | There is no drug-disease interaction |
| B | Drug–disease interaction without any bleeding event or change in INR | |
| C | Drug–disease interaction with bleeding event or change in INR | |
| Duplication | A | Only using VKA. |
| B | - | |
| C | VKA and other anticoagulant concomitant use | |
| Duration | A | Appropriate |
| B | - | |
| C | Inappropriate |
Abbreviations: AF, atrial fibrillation; CrCl, creatinine clearance; INR, international normalized ratio; NOAC, non-vitamin K antagonist; TTR, time in therapeutic range; VKA, vitamin K antagonist.
Appendix C
Detailed Ratings of Medication Appropriateness Index.

| Ratings, n | ||||
|---|---|---|---|---|
| Criterion | A | B | C | Additional information for C rating |
| Indication | 141 | 0 | 7 | CHA2DS2VASc score = 0 (n = 6), CHA2DS2VASc score = 1 and female sex (n = 1) |
| Choice | 71 | 45 | 32 | Drug monitoring intervals >8 weeks for warfarin (n = 25) and inappropriate indication (n = 7) |
| Dosage | 65 | 3 | 80 | TTR <60% for warfarin (n = 54), too low dose of the NOAC (n = 15), too high dose of the NOAC (n = 4), Inappropriate indication (n = 7) |
| Modalities of administration, correct | 137 | 2 | 8 | Dabigatran taken once daily (n = 1), Inappropriate indication (n = 7) |
| Modalities of administration, practical | 141 | 0 | 7 | Inappropriate indication (n = 7) |
| Drug–drug interaction | 13 | 132 | 3 | Warfarin and dabigatran duplication (n = 1), rivaroxaban and warfarin duplication (n = 1), dabigatran concomitant use with carbamazepine (n = 1) |
| Drug–disease interaction | 148 | 0 | 0 | - |
| Duplication | 146 | 0 | 2 | Concomitant use of warfarin and NOAC (n = 2) |
| Duration | 123 | 14 | 11 | Patient NOAC adherence <80% (n = 4), Inappropriate indication (n = 7) |
Abbreviation: NOAC, non-vitamin K antagonist.
