Abstract
Despite direct oral anticoagulants (DOACs) have overcome the most relevant limitations of vitamin K antagonists, many patients with atrial fibrillation are not receiving the appropriate anticoagulant therapy. In addition, when patients are anticoagulated with DOACs, some of them are not taking the dose recommended in the summary of product characteristics. This may be related, at least in part, to the concern about the applicability of findings from randomized clinical trials to real-life patients. In this context, performing studies in daily clinical practice to assess the efficacy and safety of DOACs in real-world setting is mandatory. The aim of this review is to update the current evidence regarding safety of rivaroxaban in clinical practice. The Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET-AF) showed that rivaroxaban was at least as effective as warfarin for preventing stroke or systemic embolism, with similar rates of major bleeding but with lesser risk of intracranial and fatal bleedings. Data from noninterventional studies and registries have confirmed the good results of ROCKET-AF. Even more, rates of bleeding may be inferior in real-life patients, since they have a lower risk profile than those included in ROCKET-AF.
Keywords
Introduction
Atrial fibrillation (AF) is the most common arrhythmia in clinical practice. 1 –5 Atrial fibrillation markedly increases the risk of stroke. In addition, strokes in which the underline cause is AF are more severe, with higher mortality, disability, and recurrence rates. 6 Therefore, reducing the risk of stroke in patients with AF with the appropriate antithrombotic therapy is mandatory. 7 However, not all patients with AF have the same risk of thromboembolic outcomes. In addition, the risk of cardioembolic complications due to AF should be balanced with the risk of hemorrhage associated with anticoagulation. 7,8
During decades, vitamin K antagonists (VKAs) have been used for the prevention of stroke in patients with AF, since they effectively reduce the risk of thromboembolic complications, with a relative low risk of bleeding. 9,10 However, VKAs have many disadvantages that together with concerns about hemorrhagic complications have led to an underuse of oral anticoagulants in patients with AF in which anticoagulation is indicated. 2,11 On the other hand, although pivotal clinical trials and meta-analyses were performed with warfarin, it has been reported that efficacy, safety, and anticoagulation control are similar between warfarin and other VKAs. 12
Direct oral anticoagulants (DOACs) have a wide therapeutic window, a predictable anticoagulant effect, low risk of interactions with other drugs, no monitoring of their anticoagulant effect is necessary, and fixed doses are taken. 13 Data from clinical trials have shown that overall, in patients with AF, DOACs are at least as effective as warfarin for preventing stroke and systemic embolism, with a similar risk of major bleeding, but a lesser risk of intracranial hemorrhage, and mortality and an increased risk of gastrointestinal bleeding. 14 –17 However, although several meta-analyses have analyzed all phase 3 clinical trials with DOACs as whether they were similar, the fact is that the clinical characteristics of patients included in these studies were markedly different, being individuals included in Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET-AF) those at higher risk (Table 1). 18 –21 As a result, particular caution should be taken when performing direct comparisons between them. 18 –21
Clinical Characteristics of Patients Included in RE-LY, ROCKET-AF, ARISTOTLE, and ENGAGE AF-TIMI 48 Trials.a
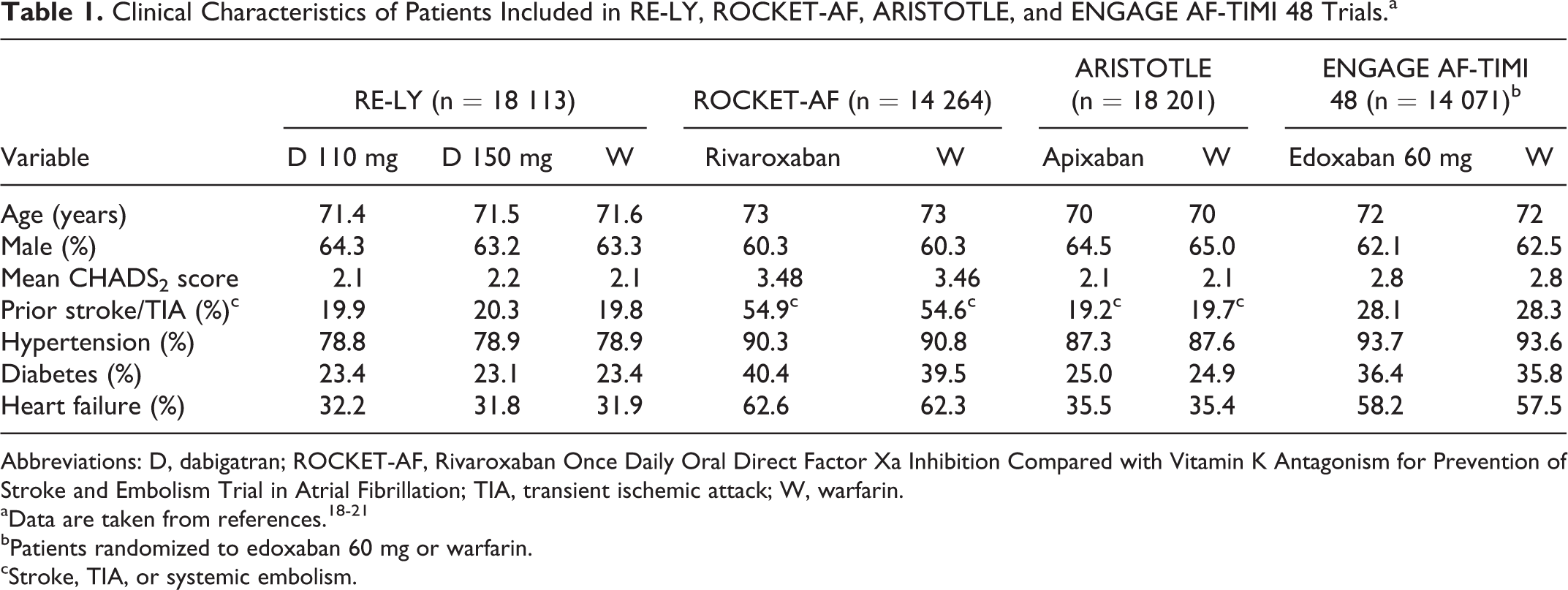
Abbreviations: D, dabigatran; ROCKET-AF, Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation; TIA, transient ischemic attack; W, warfarin.
bPatients randomized to edoxaban 60 mg or warfarin.
cStroke, TIA, or systemic embolism.
A recent study performed in the United States has shown that after DOACs have been marketed, visits with anticoagulation use have significantly increased, of which 38.2% accounted for patients taking DOACs. In 2014, rivaroxaban was the most commonly prescribed DOAC for AF. 22 However, it has been reported that low-dose DOACs usage according to the optimal dose is frequent in clinical practice, leading to an undertreatment of patients with nonvalvular atrial fibrillation (NVAF) taking DOACs. 23,24
Although evidence provided from clinical trials is well established, patients included in these studies may have lesser comorbidities and are strictly monitored. Consequently, the results of these studies regarding efficacy and safety may not be necessarily the same in real-life patients. In this context, studies performed in daily clinical practice to assess the efficacy and safety of DOACs in real-world setting are mandatory. 25 The aim of this review is to update the current evidence regarding the safety of rivaroxaban in clinical practice.
Pharmacokinetic Properties of Rivaroxaban
Rivaroxaban is an oral, direct factor Xa inhibitor, with a predictable and dose-dependent anticoagulant activity. After oral intake, rivaroxaban is quickly absorbed. In fact, the maximum concentration of rivaroxaban is achieved 2 to 4 hours after tablet intake. Despite no significant interactions between food and rivaroxaban have been described, rivaroxaban 15 and 20 mg should be taken together with food to increase oral bioavailability. Approximately two-thirds of rivaroxaban undergo metabolic degradation and are eliminated via renal and fecal routes by the same extent. The other one-third of rivaroxaban is directly excreted in the urine, largely by active renal secretion. The terminal half-life of rivaroxaban rises with age, from 5 to 9 hours in young individuals to 11 to 13 hours in elderly patients (Table 2). 26 –31
Pharmacokinetic Properties of Rivaroxaban.a

Abbreviation: P-gp, P-glycoprotein.
Pharmacokinetics (PKs) and pharmacodynamics (PDs) of rivaroxaban remain unchanged according to age, and no dose adjustment is required. Similarly, no dose adjustment is necessary in patients with extreme body weight. With regard to hepatic function, rivaroxaban is contraindicated in patients with hepatic disease associated with significant hemorrhagic risk, including patients with cirrhosis Child-Pugh B and C (Table 2). 26,32 –34
Determining renal function is important in patients taking rivaroxaban, since plasma concentrations increase as renal function declines. Thus, although no dose adjustment is required in individuals with a creatinine clearance ≥50 mL/min, rivaroxaban dose should be reduced from 20 to 15 mg once daily (OD) in patients with moderate or severe renal impairment (creatinine clearance 15 and 49 mL/min). Finally, in patients with creatinine clearance <15 mL/min, rivaroxaban is not recommended. Despite a recent study performed in hemodialysis patients without residual kidney function, a 10 mg dose of rivaroxaban resulted in drug exposure similar to that of 20 mg in healthy volunteers, rivaroxaban is not currently recommended for this population (Table 2). 26 –31,35
Rivaroxaban is metabolized via CYP3A4-, CYP2J2-, and CYP-independent mechanisms. Moreover, rivaroxaban is a substrate of the transporter proteins P-glycoprotein (P-gp) and breast cancer resistance protein. Concomitant use of rivaroxaban with strong inhibitors of both CYP3A4 and P-gp or potent CYP3A4 inducers should be avoided. However, those drugs that strongly inhibit only 1 of the 2 elimination pathways or that moderately inhibit both pathways can be concomitantly used with rivaroxaban, but particularly caution should be taken when renal impairment is present, with a more strict follow-up (Table 2). 26,27 –31,36,37 Remarkably, in a post hoc analysis of ROCKET-AF, no evidence of differential outcomes in those patients treated with ≥1 combined CYP3A4 and P-gp inhibitors was observed. 38
Dabigatran and apixaban should be taken twice daily for the prevention of stroke or systemic embolism in patients with NVAF. 18 –21 By contrast, rivaroxaban and edoxaban should be taken only OD. The OD dose of rivaroxaban chosen for ROCKET-AF trial was largely based on the data of 2 phase 2 studies together with PK/PD studies. All these studies reported that the dose of 20 mg OD presented the best benefit–risk ratio compared with twice-daily doses of rivaroxaban or other OD doses. 26,39 –41
Rivaroxaban and ROCKET-AF
The ROCKET-AF trial was a phase 3 noninferiority study that analyzed the efficacy and safety of rivaroxaban compared with warfarin in patients with NVAF who were at moderate to high risk for stroke. In this study, 14 264 patients were randomized to receive rivaroxaban 20 mg OD (15 mg if creatinine clearance is 30-49 mL/min/1.73 m2) or dose-adjusted warfarin (international normalized ratio [INR] 2.0-3.0). The clinical characteristics of patients are shown in Table 1. Remarkably, patients included in ROCKET-AF were at high risk of both thromboembolic and bleeding complications (mean CHADS2 score was 3.48, and 62.5% of patients had a HAS-BLED score ≥3). 18
After a median follow-up period of 707 days (median duration of treatment exposure of 590 days), in the per-protocol population (all patients who received at least 1 dose of a study drug, did not have a major protocol violation, and were followed for events while receiving a study drug or within 2 days after discontinuation), rates of the primary efficacy end point (stroke or systemic embolism) were 1.7% per year in the rivaroxaban group and 2.2% per year in the warfarin group (hazard ratio [HR]: 0.79; 95% confidence interval [CI]: 0.66-0.96; P < .001 for noninferiority); in the as-treated safety population (patients who received at least 1 dose of a study drug and were followed for events, regardless of adherence to the protocol, while they were receiving the assigned study drug or within 2 days after discontinuation), these numbers were 1.7% per year and 2.2% per year, respectively (HR: 0.79; 95% CI: 0.65-0.95; P = .02 for superiority); and in the intention-to-treat analysis (all patients who underwent randomization and were followed for events during treatment or after premature discontinuation), 2.1% per year and 2.4% per year, respectively (HR: 0.88; 95% CI: 0.74-1.03; P < .001 for noninferiority; P = .12 for superiority). 18
The principal safety end point (major and nonmajor clinically relevant bleeding) occurred similarly in rivaroxaban and warfarin groups (14.9% and 14.5% per year, respectively; HR: 1.03; 95% CI: 0.96-1.11; P = .44). Rates of major bleeding were also similar in both treatment groups (3.6% and 3.4% per year, respectively; HR: 1.04; 95% CI: 0.90-1.20; P = .58). However, the risk of intracranial hemorrhage (HR: 0.67; 95% CI: 0.47-0.93; P = .02), critical bleeding (HR: 0.69; 95% CI: 0.53-0.91; P = .007), and fatal bleeding (HR: 0.50; 95% CI: 0.31-0.79; P = .003) were less frequent with rivaroxaban. By contrast, major bleeding from a gastrointestinal site was more common with rivaroxaban compared with warfarin (3.15% vs 2.16%, respectively; P < .001). 18
After the publication of ROCKET-AF, several substudies and post hoc analyses determining the efficacy and safety of rivaroxaban compared with warfarin in different subgroups of patients and clinical conditions have been published (Table 3). 42 –59 Overall, the results of these substudies were consistent with the main results and emphasize the robustness of ROCKET-AF.
Efficacy and Safety of Rivaroxaban Compared With Warfarin According to Different Clinical Conditions.a,b

Abbreviations: CI, confidence interval; HR, hazard ratio; NYHA, New York Heart Association; ROCKET-AF, Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation; TIA, transient ischemic attack; VKA, vitamin K antagonists.
bData from ROCKET-AF.
Japanese guidelines recommend lower levels of anticoagulation (ie, in this population, an INR target of 1.6-2.6 is recommended, rather than 2.0-3.0). In addition, it has been reported that considering PK–PD properties, the dosage of 15 mg OD is more adequate than 20 mg OD in Japanese population. 60,61 In J-ROCKET-AF, 1280 Japanese patients were randomized to rivaroxaban 15 mg OD (10 mg if creatinine clearance is 30-49 mL/min) or dose-adjusted warfarin according to the Japanese guidelines. The risk of major and nonmajor clinically relevant bleeding similarly occurred in both groups. Intracranial bleeding rates were 0.8% and 1.6%, respectively. Importantly, there was a strong trend for a reduction in the rate of stroke/systemic embolism with rivaroxaban compared with warfarin (HR: 0.49; P = .05). 62
It has been reported that the device used in ROCKET-AF for INR monitoring may provide an INR result that is lower than an automated, plasma-based laboratory INR in patients with some specific medical conditions (ie, patients with anemia, bleeding, or situations associated with high levels of fibrinogen). However, recent post hoc analyses of the ROCKET-AF data have shown that the results of the additional sensitivity analyses were consistent with those already reported for the primary efficacy and safety end points in patients with or without recall conditions. In addition, the European Medicines Agency has ruled out that the possible malfunction of the point-of-care device used for INR measurement in the ROCKET-AF trial had any significant clinical effect on the primary efficacy and safety outcomes in the trial. 63
Rivaroxaban in Real-Life Patients
Bleeding and Anticoagulant Therapy
Although in the last years the introduction of DOACs has increased the proportion of patients receiving anticoagulants, the fact is that a significant number of individuals with NVAF do not take anticoagulants despite a clear indication, or when used, many physicians prescribe low-dose DOACs instead of the optimal dose. 23,24,64,65 Moreover, there is concern about the applicability of findings from randomized clinical trials to real-life patients. Fortunately, in the last years, several studies performed in daily clinical practice have been published or are ongoing.
Bleeding represents the most important complication of anticoagulation. However, the risk of hemorrhage differs according to the clinical characteristics of patients. In this context, it is not only important to stratify the risk of stroke (ie, CHA2DS2-Vasc score) but also the risk of bleeding (ie, HAS-BLED score). 7 Thus, in a registry that included 7243 patients with AF from 7 European countries, 70.6% of patients with bleedings had a HAS-BLED score ≥3. 66 While in ARISTOTLE trial 36% of patients had a HAS-BLED score of 2 and 23% a HAS-BLED score ≥3, these numbers were 30% and 62.5%, respectively, in ROCKET-AF. This higher risk profile was in parallel with higher rates of major bleeding observed in ROCKET-AF. Thus, the annual rates of major bleeding in the warfarin arm were 3.09% in ARISTOTLE and 3.4% in ROCKET-AF. 18,20 Unfortunately, many physicians underestimate the risk of both, stroke and bleeding. 67 This underestimation of risk may explain, at least in part, the discrepancies between current recommendations and clinical practice.
On the other hand, not all bleedings are equally relevant. Thus, when prescribing anticoagulant therapy, it is more important to consider major hemorrhages, particularly intracranial and fatal bleeding, rather than any hemorrhage. 14,68 In the meta-analysis of Agarwal et al that included 8 clinical trials, the annual rates of major bleeding with warfarin varied from 1.40% to 3.40% and the annual rates of intracranial bleeding from 0.33% to 0.8%. 69 In a meta-analysis of 11 studies (5 on AF and 6 on venous thromboembolism), the risk of fatal bleeding was significantly and markedly reduced by DOACs compared with VKA. 70 In ROCKET-AF, compared with warfarin, rivaroxaban significantly reduced the risk of intracranial and fatal bleeding by 33% and 50%, respectively. 18
In addition, it is not only important to analyze the frequency of bleeding but also the intensity. Thus, a recent study that compared patients with intracerebral hemorrhage while taking rivaroxaban or warfarin showed that rivaroxaban-associated intracerebral hemorrhage occurred in patients at high risk for major bleeding. In addition, compared with the group of patients taking warfarin, those patients in the rivaroxaban group had a relatively small hematoma, no expansion of hematoma, and favorable functional and vital outcomes. 71
Safety of Rivaroxaban in Real-Life Patients
Several studies have analyzed the safety of rivaroxaban in clinical practice. In a study that retrospectively analyzed data from the US Department of Defense, 27 467 patients with NVAF taking rivaroxaban between January 1, 2013, and March 31, 2014, were investigated. In this study, the incidence of major bleeding was 2.86 per 100 person-years. Compared with those patients without major bleeding, those patients with major bleeding were older (78.4 vs 75.7 years) and had more comorbidities, including hypertension (95.6% vs 75.8%), ischemic heart disease (64.2% vs 36.7%), heart failure (48.5% vs 23.7%), and chronic kidney disease (38.7% vs 16.7%). CHADS2 score was also significantly higher in those patients with major bleeding (3.0 vs 2.2). Major bleeding was most commonly gastrointestinal (88.5%) or intracranial (7.5%). The incidence of fatal bleeding was 0.08 per 100 person-years (Table 4, Figure 1). 72 Updated data of this study recently presented at 2015 American College of Cardiology congress showed that in the first 18 months of the study, with 31 833 patients taking rivaroxaban, the incidence of major bleeding and fatal bleeding remained 2.85 and 0.09 per 100 person-years, respectively. 75
Safety of Rivaroxaban in Real-Life Patients With AF.
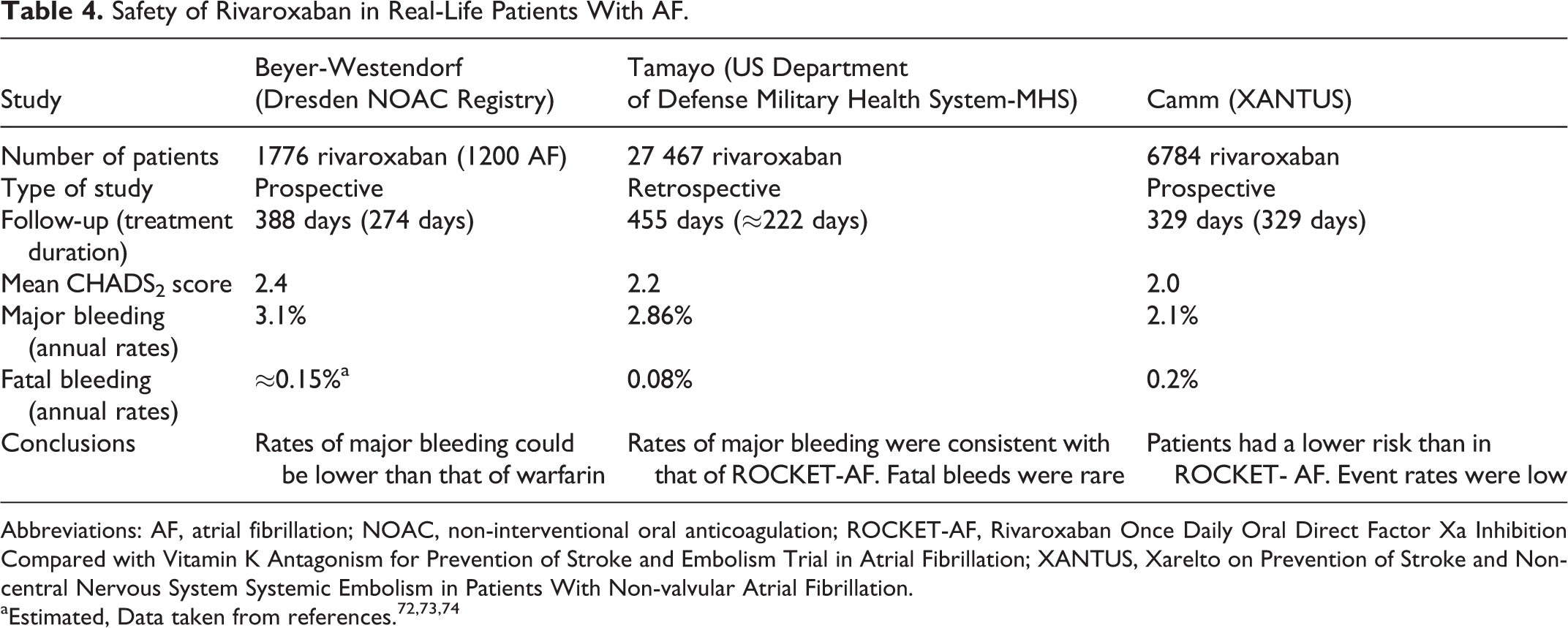
Abbreviations: AF, atrial fibrillation; NOAC, non-interventional oral anticoagulation; ROCKET-AF, Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation; XANTUS, Xarelto on Prevention of Stroke and Non-central Nervous System Systemic Embolism in Patients With Non-valvular Atrial Fibrillation.
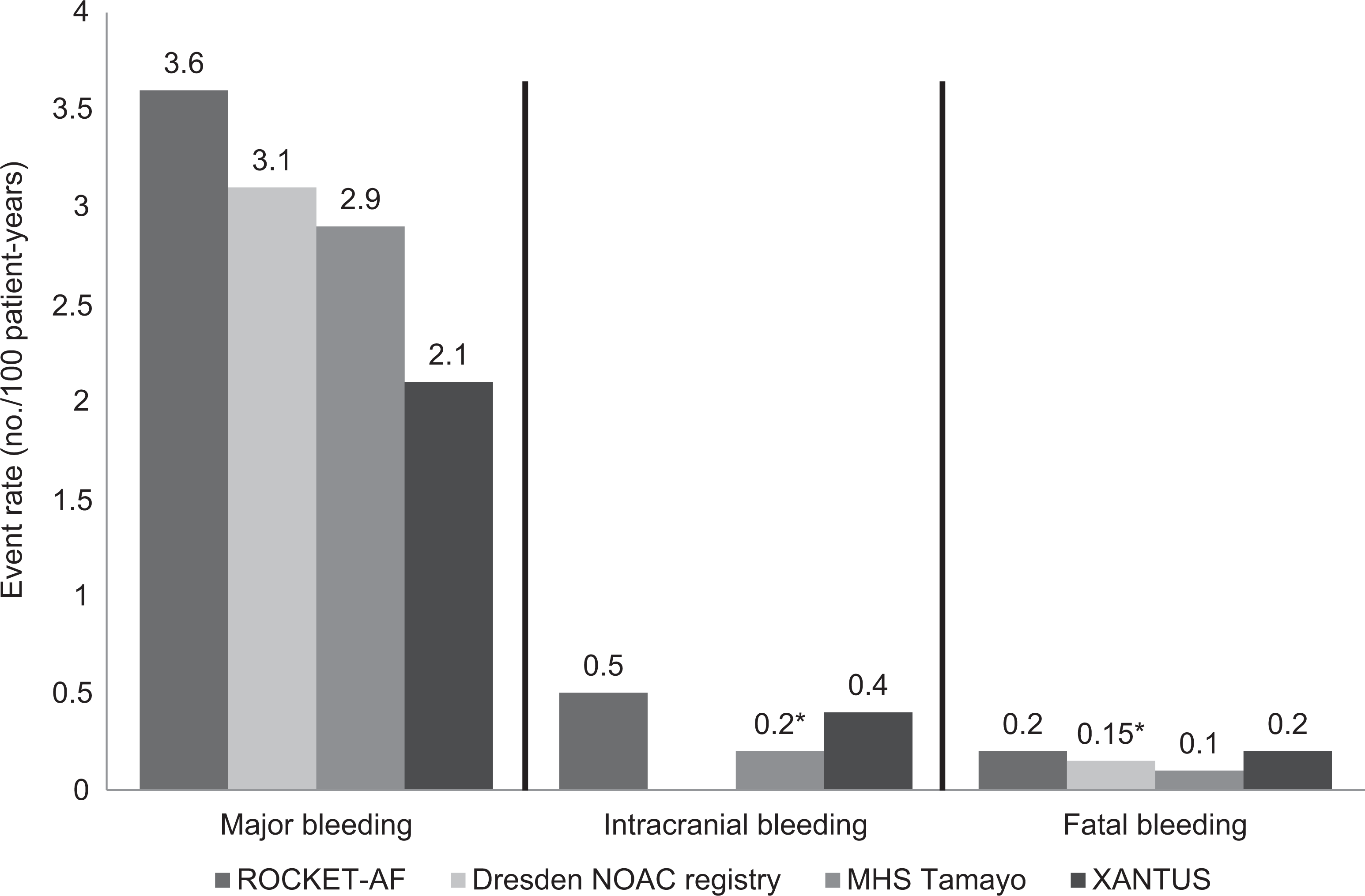
Risk of major bleeding, intracranial bleeding, and fatal bleeding in Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET-AF; rivaroxaban arm) and real-life cohorts of patients taking rivaroxaban. *Estimated, data are taken from references. 18,72,73,74
A recent study performed in primary care setting in Switzerland included 537 consecutive patients with NVAF who started treatment with rivaroxaban. Mean age of patients was 73 years, 57% were men, mean CHADS2 score was 2.2, and mean HAS-BLED score was 2.4. About 56% of patients were switched from VKA to rivaroxaban, and the remaining 44% were VKA naive. After 3 months of treatment, 2 (0.37%) major nonfatal bleeding and 11 (2.05%) minor bleeding events occurred. In those patients who changed from VKA to rivaroxaban, both patient’s satisfaction and overall physician satisfaction significantly increased. About 5.6% of patients withdrew from rivaroxaban treatment, mainly due to their side effects. 76
The Dresden Non-Interventional Oral Anticoagulation registry is an ongoing large and prospective noninterventional registry in which more than 230 physicians from Dresden (Saxony), Germany, include patients treated with a DOAC. The main inclusion criteria were age ≥18 years, planned DOAC anticoagulation for at least 3 months, and therapeutic DOAC indication including AF, deep vein thrombosis, pulmonary embolism, and other indications. In this study, efficacy and safety of rivaroxaban were analyzed. Between October 1, 2011, and December 31, 2013, 1776 patients treated with rivaroxaban were included (age 74 years; 51.4% male; 17.5% prior coronary artery disease; 11.4% previous stroke or systemic embolism; 10.8% impaired renal function). During this time, rates of major bleeding were 3.1 per 100 patient-years in patients with AF, and 4.1 per 100 patient-years in patients with venous thromboembolism. Case fatality rates of bleeding leading to hospitalizations were 5.1% at day 30 and 6.3% at day 90 after bleeding (Table 4, Figure 1). 73
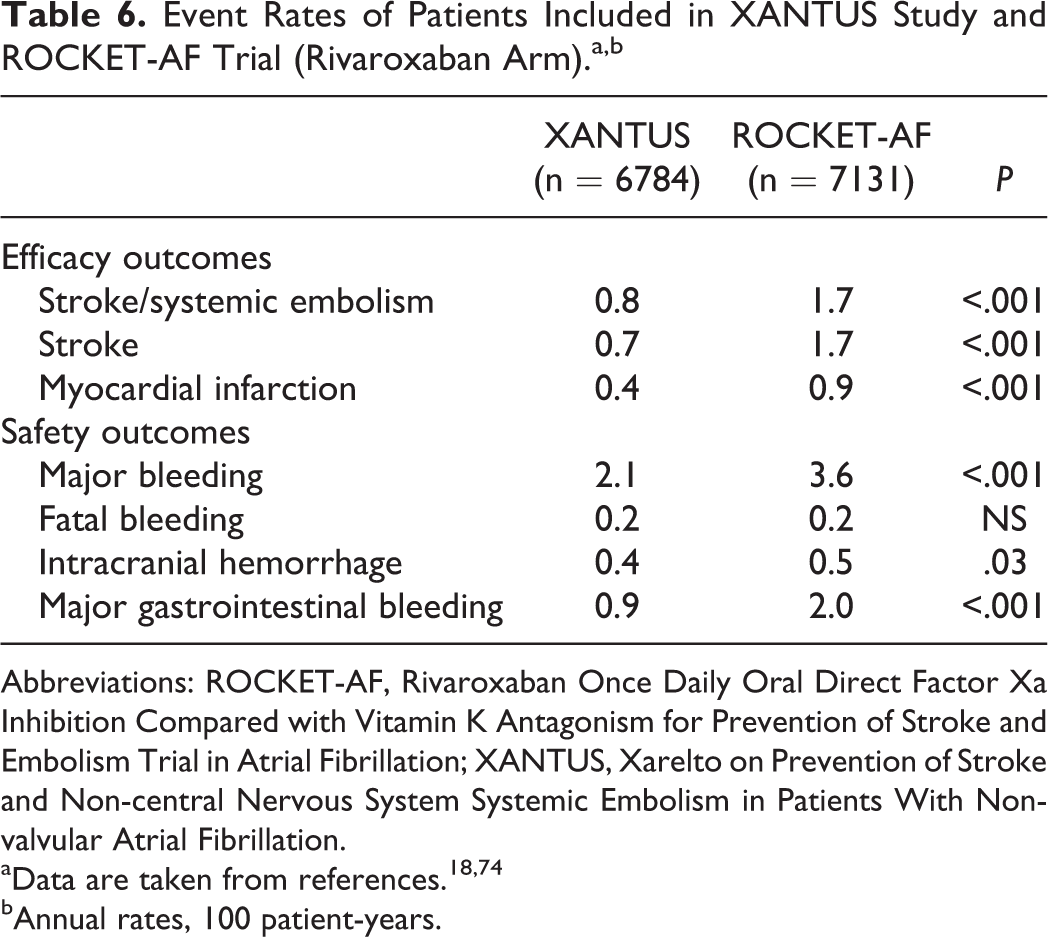
Data from Xarelto on Prevention of Stroke and Non-central Nervous System Systemic Embolism in Patients With Non-valvular Atrial Fibrillation (XANTUS) study have been recently reported in the 2015 European Society of Cardiology congress. The XANTUS was an observational, prospective, postauthorization, noninterventional, and single-arm study aimed to collect data in real-life patients with NVAF, with a special focus on safety outcomes. In fact, XANTUS was the first international, prospective, and observational study aimed to describe the use of rivaroxaban in patients with NVAF. In XANTUS, 6784 consecutive patients from 311 centers in Europe, Israel, and Canada with NVAF newly started on rivaroxaban were included. The mean observation period was 329 days. With regard to the clinical characteristics of the patients, mean age was 71.5 years, 41% were women, 19% had prior stroke/transient ischemic attack (TIA)/systemic embolism, and 9.4% had documented severe or moderate renal insufficiency. Mean CHADS2 score was 2.0 and mean CHA2DS2-VASc score was 3.4. A total of 78.7% of patients were taking rivaroxaban 20 mg OD, 20.8% 15 mg OD, and 0.5% another dose (Tables 4 and 5). Rates of major bleeding and fatal bleeding were 2.1 and 0.2 events per 100 patient-years, respectively (Tables 4 and 6, Figure 1). 74 Compared with patients included in ROCKET-AF (rivaroxaban arm), XANTUS study patients had lesser comorbidities and a lower mean CHADS2 score. This translated into a lesser risk of bleeding (Tables 5 and 6). 18,74
Clinical Characteristics of Patients Included in XANTUS Study and ROCKET-AF Trial (Rivaroxaban Arm).a
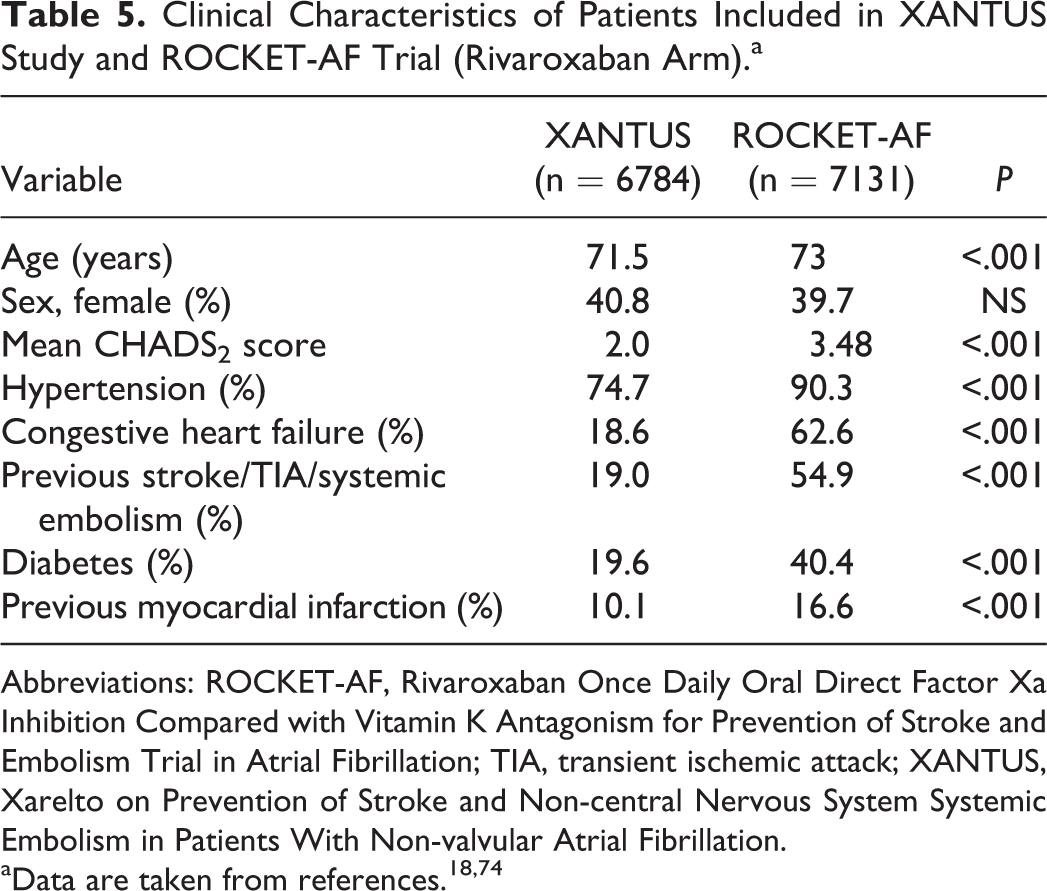
Abbreviations: ROCKET-AF, Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation; TIA, transient ischemic attack; XANTUS, Xarelto on Prevention of Stroke and Non-central Nervous System Systemic Embolism in Patients With Non-valvular Atrial Fibrillation.
Event Rates of Patients Included in XANTUS Study and ROCKET-AF Trial (Rivaroxaban Arm).a,b
Abbreviations: ROCKET-AF, Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation; XANTUS, Xarelto on Prevention of Stroke and Non-central Nervous System Systemic Embolism in Patients With Non-valvular Atrial Fibrillation.
bAnnual rates, 100 patient-years.
In other study that included patients with AF newly initiated on rivaroxaban (n = 3654) or warfarin (n = 14 616), with a CHADS2 score ≥1, bleeding events similarly occurred in both groups. 77 Another retrospective analysis of 2579 patients taking dabigatran or rivaroxaban reported similar rates of major bleeding than that of randomized clinical trials. 78
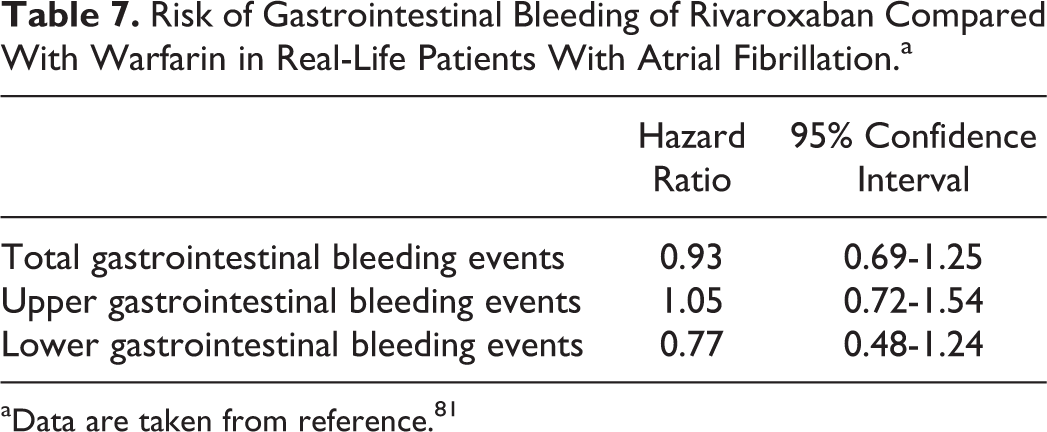
With regard to major bleeding from a gastrointestinal site, these were more common with rivaroxaban compared with warfarin in ROCKET-AF (3.15% vs 2.16%, respectively; P < .001). However, the most severe gastrointestinal bleeding, those who required transfusion of >4 U of whole blood or packed red blood cells, similarly occurred in both treatment groups (49 patients with rivaroxaban compared with 47 patients with warfarin). Only 1 patient in the rivaroxaban group and 5 patients in the warfarin group had a fatal gastrointestinal bleeding. Even more, in ROCKET-AF, there were 27 fatal bleedings, but only 1 had a gastrointestinal origin. 18 On the other hand, it has been reported that this increased risk of anticoagulant-related gastrointestinal bleeding may facilitate the unmasking of preexisting malignancies from this origin, leading to the early detection of gastrointestinal cancer. 79 Rates and severity of gastrointestinal bleeding have been specifically analyzed in real-life patients. In a retrospective analysis of 170 consecutive patients with AF from a TIA clinic, who started either dabigatran (n = 88) or rivaroxaban therapy (n = 93), with a total of 86 patient-years of exposure, gastrointestinal bleeding occurred in 5% (n = 4) of the patients treated with dabigatran (mean age 84 years, all of them with at least 2 comorbidities) and in 1% of the patients treated with rivaroxaban (this patient was 96 years old and had many comorbidities). 80 In a study that included patients with AF with a CHADS2 score ≥1, newly initiated on rivaroxaban or warfarin, the risk of gastrointestinal bleeding was similar in both groups (HR: 1.28; 95% CI: 0.99-1.63). 77 In a retrospective, propensity-matched cohort study, new users of dabigatran, rivaroxaban, and warfarin from November 1, 2010, to September 30, 2013, were analyzed. In patients with AF, the incidence of gastrointestinal bleeding associated with rivaroxaban was 2.84 events per 100 patient-years, compared with 3.06 per 100 patient-years in those treated with warfarin (Table 7). 81 In summary, although in ROCKET-AF major bleeding from a gastrointestinal site was more frequent with rivaroxaban than with warfarin, severe gastrointestinal bleeding similarly occurred in both groups. In addition, studies performed in real-life patients have not confirmed this increased risk of gastrointestinal bleeding compared with warfarin.
Risk of Gastrointestinal Bleeding of Rivaroxaban Compared With Warfarin in Real-Life Patients With Atrial Fibrillation.a
aData are taken from reference. 81
Another relevant question to be answered is whether those patients at higher risk of bleeding can be identified in order to reduce their hemorrhagic risk. In ROCKET-AF, predictors of major bleeding were increasing age, baseline diastolic blood pressure ≥90 mm Hg, history of chronic obstructive pulmonary disease or gastrointestinal bleeding, prior acetylsalicylic acid use, and anemia. By contrast, female sex and diastolic blood pressure <90 mm Hg were associated with a decreased risk of major bleeding. 82 In other study, prescriber errors, impaired renal function, concomitant treatment with antiplatelet drugs or P-gp inhibitors, old age, and low body weight were associated with increased risk of hemorrhage under DOACs. 83 As a result, a correct selection of patients, an adequate prescription of DOACs, the control of factors that increase the risk of bleeding, such as avoiding antiplatelet drugs or P-gp inhibitors and reducing blood pressure values to recommended targets, and a close follow-up of frail patients may reduce the risk of bleeding in patients with AF taking DOACs.
Efficacy of Rivaroxaban in Real-Life Patients
Different studies have analyzed the efficacy of rivaroxaban in clinical practice. REal-LIfe Evidence on stroke prevention in patients with atrial Fibrillation was a retrospective study aimed to compare the effectiveness of newly initiated rivaroxaban or VKA therapy among patients with NVAF in Germany. The primary end point was the time to composite of ischemic stroke, TIA, intracerebral hemorrhage, other nontraumatic intracranial hemorrhage including subdural hemorrhage, and myocardial infarction within 1 year of treatment initiation. Remarkably, the incidence of the primary end point was lower in rivaroxaban compared with VKA users (1.97 vs 3.68 events per 100 person-years; HR: 0.54; 95% CI: 0.31-0.92). In addition, rates of individual end points were numerically less frequent in the rivaroxaban cohort. 84 In the registry performed in Switzerland, after 3 months of treatment, only 1 (0.19%) ischemic stroke occurred. 76 In a study that included patients with AF newly initiated on rivaroxaban or warfarin, stroke or systemic embolism events similarly occurred in both groups. Importantly, rivaroxaban was associated with a lesser risk of venous thromboembolic events (HR: 0.36; 95% CI: 0.24-0.54; P < .0001) and treatment nonpersistence (HR: 0.66; 95% CI: 0.60-0.72; P < .0001). 77
In XANTUS, after 329 days of treatment, the rates of stroke were 0.7 events per 100 patient-years (Table 6). 74 Compared with patients included in ROCKET-AF (rivaroxaban arm), XANTUS study patients had a lesser risk of stroke and myocardial infarction (Table 6). 18,74
Discussion
Despite the introduction of DOACs that mitigate many of the limitations of VKA, a significant proportion of patients with NVAF remain without antithrombotic treatment, and when anticoagulated with DOACs, some of them are not taking the dose recommended in the summary of product characteristics. 2,23,24 For example, in XANTUS, despite only 9.1% of patients had a creatinine clearance 15 to <50 mL/min, 20.8% of patients were taking rivaroxaban 15 mg OD. 74 Similarly, data from the GLORIA-AF registry also showed that patients with NVAF at high risk of stroke were undertreated with oral anticoagulants, since initial therapy was often not prescribed in accordance with guideline recommendations. 85,86 Although it is uncertain whether this has an impact on a higher risk of thrombotic complications, overall, in “real-life” patients, the rates of stroke or systemic embolism seem lower than that reported in clinical trials (0.8 vs 1.7 events per 100 patient-years; Table 6). 18,74 On the other hand, since anticoagulants increase the risk of bleeding, this may be balanced with the risk of stroke that patients with NVAF have. In this context, it is mandatory to perform a correct risk stratification of this population. 7 In addition, the identification and correction when possible of those factors that increase the risk of bleeding (ie, blood pressure control) may improve the risk–benefit ratio in favor of anticoagulation.
Clinical trials have shown that in patients with NVAF, DOACs are at least as effective as warfarin for preventing stroke and systemic embolism, with similar rates of major bleeding, but importantly, with a lesser risk of intracranial hemorrhage and mortality. 14 –17 Therefore, DOACs are safer than VKA. However, the doubt that emerged was whether these results could be applied to real-life patients. In the last years, a number of noninterventional studies and registries have confirmed the safety of DOACs in clinical practice. In addition, some registries performed with rivaroxaban suggest that the rates of bleeds are similar or even lower than that of ROCKET-AF. 72–74 This could be explained by the fact that patients included in ROCKET-AF were at very high risk of stroke and bleeding 18 and that in clinical practice not all patients with AF have the same risk.
In ROCKET-AF, treatment with rivaroxaban was associated with an increased risk of gastrointestinal bleeding. However, very severe gastrointestinal bleeding occurred similarly in both groups. 18 In addition, studies performed in real-life patients have not confirmed this increased risk of gastrointestinal bleeding compared with warfarin. 77,81 As a result, patients with a high risk of gastrointestinal bleeding can be treated with rivaroxaban, but a close follow-up is recommended.
The lack of a specific antidote has been suggested as another reason for not prescribing DOACs. However, first, there is a specific antidote currently marketed for one of the DOACs. In fact, idarucizumab, an antibody fragment, has been shown to completely reverse the anticoagulant effect of dabigatran within minutes. 87 Second, the current management of patients with severe bleeding is actually very similar in case of VKA or DOACs. Moreover, since DOACs have a short half-life, reversing their anticoagulant effect may not be necessary in a great number of individuals. Nevertheless, in some patients, the use of prohemostatic agents may be necessary. In case of rivaroxaban, activated prothrombin complex concentrate and prothrombin complex concentrate are particularly recommended, and when insufficient, recombinant factor VIIa may be also useful. Importantly, specific reversal agents are currently under investigation, such as andexanet alfa or PER977. 88 –90 It is very likely that in the next future, some of these reversal agents will be available for rivaroxaban.
Since in patients taking VKA the anticoagulant activity must be monitored, no important concerns about medication adherence had been suggested. Since this is not necessary with DOACs, doubts about treatment persistence have emerged. However, different studies have actually shown the opposite, medication persistence is significantly higher with DOACs than with VKA. 91,92 It has been reported that overall persistence with rivaroxaban is high, with a discontinuation rate of approximately 15% in the first year of therapy and few additional discontinuations thereafter. Unfortunately, data regarding whether the dosage of rivaroxaban had an impact on medication adherence were not reported. 92 On the other hand, in patients with NVAF, OD dosing regimens are associated with a higher likelihood of adherence compared with patients on twice-daily regimens. 93 A recent study has reported that rivaroxaban users have a higher medication adherence when compared to dabigatran or apixaban users. 94 Therefore, rivaroxaban may assure a good adherence to treatment in this population.
Another controversial issue is the management of patients who fail rivaroxaban and present with an acute ischemic stroke. 95 First, the time in which anticoagulation should be restarted is challenging, since it depends on many factors. In many patients, if hemorrhagic transformation has not occurred, oral anticoagulation may generally be initiated 1 to 2 weeks after the stroke. 96 Second, adherence to rivaroxaban should be determined. In case of poor adherence, it is likely that the best option may be the use of VKA, since anticoagulation monitoring is mandatory for these drugs. Third, since oral bioavailability of rivaroxaban 15 and 20 mg decreases in fasting conditions, it is mandatory to assure that patients take rivaroxaban together with food. 95,26
Finally, one disadvantage of DOACs is that they are more expensive than VKA. As a result, cost-effectiveness studies are mandatory to assess which patients will benefit more from this therapy. Overall, rivaroxaban is a cost-effective therapeutic alternative in patients with NVAF, particularly in those patients at high risk of stroke or bleeding or when INR control is not optimal. 97,98
Conclusion
Data from noninterventional studies and registries confirm the good results of ROCKET-AF. Even more, the rates of bleeding are inferior in real-life patients, since they have a lower risk profile than those included in ROCKET-AF. Therefore, rivaroxaban is a good alternative for the prevention of stroke and systemic embolism in real-life patients with NVAF.
Highlights
Despite DOACs have overcome many of the limitations of VKA, a significant proportion of patients with NVAF remains without antithrombotic treatment, and when anticoagulated with DOACs, some of them are not taking the dose recommended in the summary of product characteristics.
The ROCKET-AF showed that rivaroxaban was at least as effective as warfarin for preventing stroke or systemic embolism, with similar rates of major bleeding but with lesser risk of intracranial and fatal bleedings.
A number of noninterventional studies and registries have confirmed the safety of rivaroxaban in real-life patients. Even, more, the rates of bleeding are inferior in real-life patients, since they have a lower risk profile than those patients included in ROCKET-AF.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Domingo Marzal-Martín has collaborated as a speaker, scientific advisor, and training activities with Bayer.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Marcelo Sanmartín-Fernández has received grant support from Bayer; has received honoraria as advisor from Bayer, Boehringer, and BMS; and has received honoraria as speaker from Bayer, Boehringer, and Pfizer. Editorial assistance was provided by Content Ed Net, Madrid, Spain.
