Abstract
Background:
The outcomes of thrombolytic therapy (TT) in elderly patients with prosthetic valve thrombosis (PVT) have not been evaluated previously. We investigated the outcomes of low-dose and slow infusion TT strategies in elderly patients with PVT.
Methods:
Twenty-seven (19 female) patients aged ≥65 years (median: 70 years, range: 65-82 years) were treated with repeated TT agents for PVT. The TT regimens included 24-hour infusion of 1.5 million units of streptokinase in 2 patients, 6-hour infusion of 25 mg recombinant tissue plasminogen activator (t-PA) in 12 patients, and 25-hour infusion of 25 mg t-PA in 13 patients. Treatment success and adverse event rates were assessed.
Results:
The initial and cumulative success rates were 40.7% and 85.2%, respectively. Adverse events occurred in 6 (22.2%) patients including 4 (14.8%) major (1 death, 1 rethrombosis, and 2 failed TT) and 2 (7.4%) minor (1 transient ischemic attack and 1 access site hematoma) events. Higher thrombus burden (thrombus area ≥1.1 cm2 by receiver operating characteristics analysis, sensitivity: 83.3%, specificity: 85%, area under the curve: 0.86, P = .008) and New York Heart Association class (0% vs 15.4% vs 25% vs 100% for classes I-IV, respectively, P = .02) predicted adverse events. By multiple variable analysis, thrombus area was the only independent predictor of adverse events (odds ratio: 13.8, 95% confidence interval: 1.02-185, P = .04).
Conclusion:
Slow infusion of low doses of TT agents (mostly t-PA) with repetition is successful and safe in elderly patients with PVT. However, excessive thrombus burden may predict adverse events.
Introduction
Prosthetic valve thrombosis (PVT) is a potentially lethal complication of valve replacement surgery and may cause embolism or valve obstruction. 1 In developing countries, inadequate patient adherence and compliance to oral vitamin K antagonists play a major role in the genesis of PVT, and the prevalence is as high as 6.1% per patient-years. 2 Elderly individuals are the rapidly growing segment of the population. 3 A decision to utilize thrombolytic agents in old people with multiple comorbidities is controversial in general due to safety concerns. Older age poses an increased risk of hemorrhage in patients undergoing thrombolytic therapy (TT). 4 However, current TT protocols are based on diverse clinical syndromes such as ST-segment elevation myocardial infarction, stroke, or venous thromboembolism in which thrombolytic agents are rapidly administered after a bolus dose. Rapid thrombolysis and/or using a bolus dose has also been used in PVT treatment for several decades with satisfactory lysis rates but relatively high complications such as embolism or hemorrhage. 5 –15 We have previously and recently reported that slow infusion and low doses of thrombolytic agents have provided considerably high success rates without the cost of increased complications. 16 –19 However, there are no previous data to date that have specifically focused on the outcome of TT strategies in elderly patients with PVT. Thus, we analyzed patients with PVT aged ≥65 years who were treated with slow and/or low-dose infusion of thrombolytic agents in our institution in a 21-year period.
Rationale of TT for PVT
Current guidelines restrict TT for left-sided PVT to high-risk surgical candidates or to patients in New York Heart Association (NYHA) I and II functional capacity with recent onset symptoms, low thrombus burden, or failure of initial intravenous heparin therapy. 1,2 However our group, 16 –19 and many others, 5 –14 have reported TT as an effective and safe treatment option for patients with PVT as a first-line strategy. In addition, reoperation for PVT carries a high risk compared with TT. 20 Hence, for the last 2 decades, we have preferred thrombolysis as a first-line treatment strategy in our institution for patients with PVT without contraindications to TT.
Materials and Methods
Study Population
Overall, 27 (19 female and 8 male) elderly (≥65 years) patients with PVT who underwent TT between 1993 and 2015 with low dose and/or slow infusion of streptokinase or tissue plasminogen activator (t-PA) were included. The study consisted of 13 patients aged ≥65 years who participated in the previously published TROIA trial 17 (from 1993 to 2009) and 14 patients aged ≥65 years who underwent TT for PVT between 2009 and 2015 in our institution. Between 1993 and 2004, slow infusion of streptokinase was administered in 2 elderly patients. From 2005 to 2009, 11 elderly patients received slow infusion (6 hours) of low-dose (25 mg) t-PA. Between 2010 and 2015, 13 elderly patients were treated with ultraslow (25 hours) infusion of low-dose (25 mg) t-PA, and 1 patient was treated with slow (6 hours) infusion of low-dose (25 mg) t-PA
The study was approved by the local ethics board, and the patients were enrolled after informed consent was obtained. Inclusion and exclusion criteria of the study and the contraindications to TT were described previously. 17,19
Thrombolytic Therapy Protocols and Dosing Regimens
Slow infusion of streptokinase: 36-hour infusion of 1.5 million units of streptokinase (total 2 patients), repeat once after 36 hours if needed (maximum total dose of 3 million units).
Slow infusion of t-PA: 6-hour infusion of 25 mg t-PA (total 12 patients) without a bolus (repeat once after 24 hours, up to 6 times if needed, with a maximum total dose of 150 mg).
Ultraslow infusion of t-PA: 25-hour infusion of 25 mg t-PA (total 13 patients) without a bolus (repeat once after 6 hours, up to 8 times if needed, with a maximum total dose of 200 mg).
Intravenous heparin was not used during TT sessions to minimize the bleeding risk. Heparin infusion of 70 IU/kg bolus and 16 IU/kg per hour (up to 1000 IU/hour) with a target activated partial thromboplastin time (aPTT) of 1.5 to 2.0 times the mean of the reference range was started immediately after every TT sessions. If repeat thrombolytic infusion was needed, heparin was withhold again until aPTT was less than 50 seconds, and then repeat TT infusion was started. If the TT was successful, anticoagulation was reinitiated with heparin and warfarin.
Echocardiography
All patients underwent transthoracic echocardiography and transesophageal echocardiography (TEE) examinations at baseline to assess the burden and location of thrombus and to rule out left atrial thrombi. The indications for TEE study were described previously. 17 Real-time 3-dimensional (3D) TEE was also performed in all patients who were enrolled after 2008, since this method allows a more comprehensive delineation of PVT. 21 –24 The presence of obstruction was defined on the basis of Doppler echocardiographic measurements (peak velocity, mean gradient, effective orifice area, velocity ratio, and acceleration time as appropriate). 25 The cutoff values for these Doppler parameters were defined based on the latest recommendations. 26 The thrombus area was measured both from the atrial and ventricular sides in patients with mitral prostheses and from the aortic and ventricular sides in patients with aortic prostheses with 2D TEE examination. The 2D TEE study was performed between 0° and 180°, where there was less interference with acoustic shadowing and the largest size of thrombus could be measured. In the presence of a single mass, the thrombus was traced and measured from the atrial to the ventricular side or the ventricular to the aortic side, or otherwise each thrombus was measured separately and finally summed up.
Fluoroscopy
Fluoroscopy was performed whenever impaired leaflet mobility was suspected and TEE assessment of leaflet excursion was ill defined. The leaflet mobility was assessed in the characteristic “side view-tilting disc” projection.
Definition of Adverse Events
Major adverse events were defined as all-cause in-hospital mortality, ischemic stroke, any embolism requiring percutaneous or surgical intervention, intracranial hemorrhage, bleeding requiring transfusion or surgery, and reoperation due to TT failure. Minor adverse events were defined as bleeding without need for transfusion, transient ischemic attack (TIA), and other embolism that resolved spontaneously.
Criteria for Thrombolytic Success
In the absence of major adverse events, (1) Doppler documentation of the complete improvement in valve hemodynamics and complete normalization of leaflet mobility, (2) reduction in thrombus burden by ≥75%, and (3) symptomatic improvement were considered as the major criteria for TT success in patients with obstructive PVT. For patients with nonobstructive PVT, the only criterion was the reduction in thrombus burden by ≥50%, with a final thrombus length being less than 10 mm.
Statistical Analysis
Statistical analysis was performed using SPSS 19.0. The variables were investigated using analytical methods (Kolmogorov–Smirnov/Shapiro–Wilk test) to determine whether or not they were approximately normally distributed. Descriptive statistics were reported as mean with standard deviation for continuous variables with normal distribution, median with minimum and maximum values for continuous variables without normal distribution, and frequencies with percentages for the categorical variables. Group comparisons for continuous variables were tested using Student t test when data distribution was normal and using Mann-Whitney U test when data distributions were not normal. Comparisons for categorical variables were evaluated by χ2 test. Significance level was accepted as P < .05 in all statistical analyses. Receiver–operating characteristic (ROC) curve analysis was done to assess cutoff values. Multiple variable analysis was conducted to assess the predictors of adverse events by including the significant univariate predictors in the logistic regression analysis model.
Results
Baseline Characteristics
Over a 21-year period, 27 elderly patients (19 female and 8 male) aged ≥65 years who received TT for PVT were included in the study. The median age was 70 years (range, 65-82 years). The elapsed time since valve surgery was 26 months (range, 2-300 months). Subtherapeutic international normalized ratio (INR) was present in 77.8% of patients on admission, and 33.3% were on current aspirin use. The documented duration of suboptimal anticoagulation on the preceding days of PVT diagnosis was 4.8 ± 1.8 months. The most frequent symptom was dyspnea (55.6%). Poor functional capacity (NYHA class III and IV) was present (37%), and the median NYHA was class II. The prevalence of atrial fibrillation was 40.7%, and a history of stroke or TIA was present in 25.9% of the patients. The prevalence of coronary artery disease, hypertension, and diabetes were 29.6%, 48.1%, and 25.9%, respectively. The most common valve was the St Jude (St Jude Medical Inc, St Paul, Minnesota) bileaflet valve (68.2%). All patients had bileaflet prostheses. The median values of estimated mortality and morbidity rates as calculated by the Society for Thoracic Surgeons (STS) risk scoring system were 6.9% (range, 2.1%-14%) and 30% (range, 16%-47%), respectively. The baseline characteristics of the study population are presented in Table 1.
Baseline Characteristics of Study Patients.a
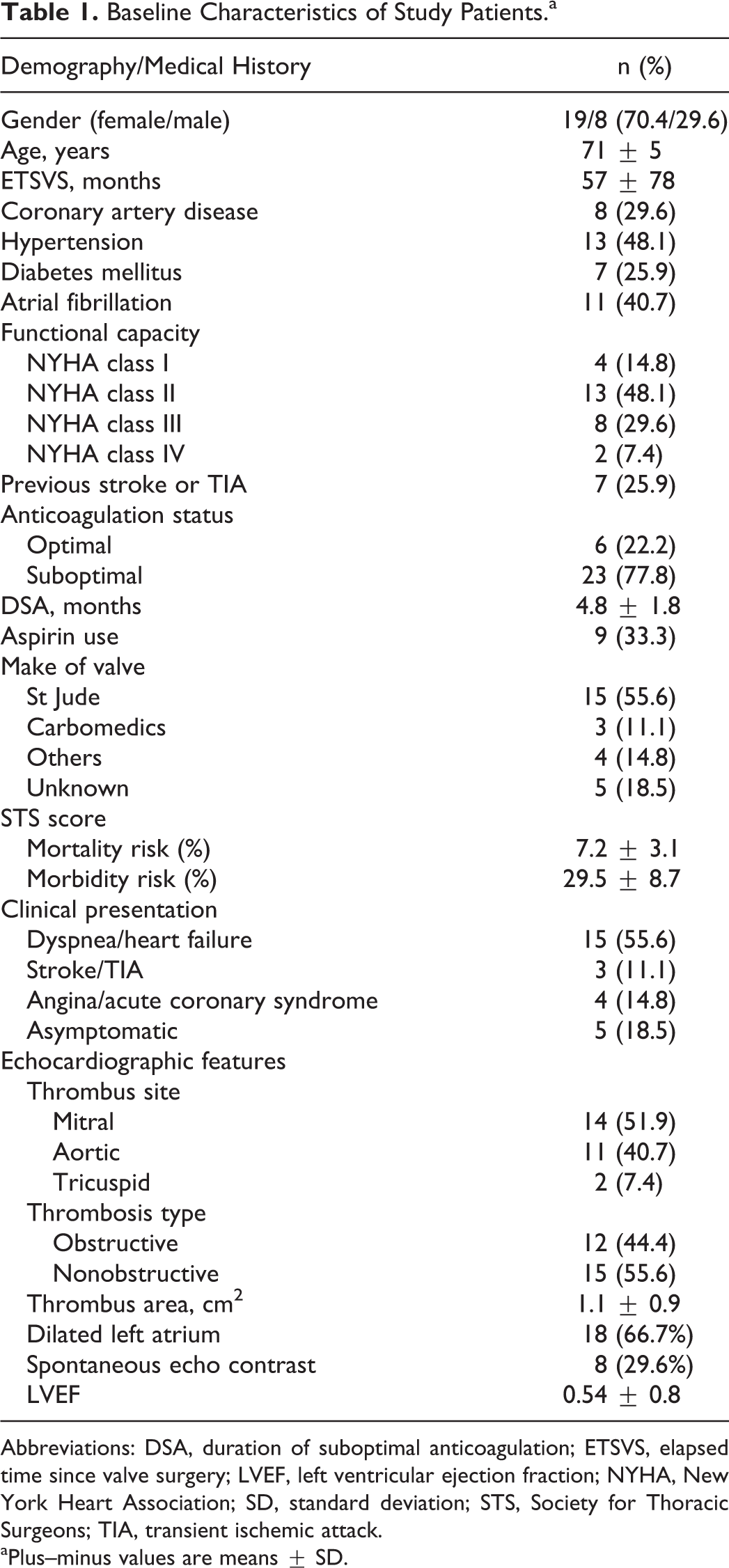
Abbreviations: DSA, duration of suboptimal anticoagulation; ETSVS, elapsed time since valve surgery; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association; SD, standard deviation; STS, Society for Thoracic Surgeons; TIA, transient ischemic attack.
aPlus–minus values are means ± SD.
Echocardiographic and Fluoroscopic Results
The median left ventricular ejection fraction was 0.54 (range, 0.30-0.65). Left atrium was dilated in 18 (66.7%) patients, and spontaneous echo contrast was present in 8 (29.6%) patients. The sites of thrombi were as follows: mitral in 14 (51.9%), aortic in 11 (40.7%), and tricuspid prostheses in 2 (7.4%) patients. Nearly half of the patients (44.4%) had obstructive PVT. The thrombus area could be measured in 96.3% of the patients (median, 0.9 cm2; range, 0.25-3.7 cm2).
In patients with obstructive PVT, the median effective orifice areas of aortic, mitral, and tricuspid prostheses were 0.68 cm2 (range, 0.40-0.85 cm2), 0.9 cm2 (range, 0.8-1.2) cm2, and 0.9 cm2 (range 0.6-1.2) cm2, respectively. The median value of mean gradients were 50 mm Hg (range, 40-80 mm Hg), 14 mm Hg (range, 10-22 mm Hg), and 12 mm Hg (range 10-14 mm Hg), respectively.
In patients with nonobstructive PVT, the median effective orifice areas for aortic and mitral prostheses were 1.5 cm2 (range 1.3-1.9 cm2) and 2.5 cm2 (range 1.85-3.1 cm2), respectively, and the median value of mean gradients were 18 mm Hg (range, 17-27 mm Hg) and 5 mm Hg (range, 3-8 mm Hg). All were obstructive in patients with tricuspid PVT.
Fluoroscopy revealed complete leaflet blockade in 5 (41.7%) of the 12 patients with obstructive PVT. Two (16.7%) patients had partial leaflet blockade. None of the patients with nonobstructive PVT underwent fluoroscopic examination.
Clinical Outcome
Adverse events occurred in 6 (22.2%) patients including 2 (7.4%) minor and 4 (14.8%) major events. The minor events included 1 TIA and 1 access site hematoma not requiring blood transfusion. The major events included 1 death, 1 rethrombosis, and 2 TT failure.
Death occurred secondary to intractable pulmonary edema during the first session of ultraslow TT in a patient who had severe obstructive mitral PVT (valve area, 0.8 cm2; mean gradient, 22 mm Hg) with a massive thrombus burden (3.7 cm2). One patient had rethrombosis after initial lytic success but underwent surgery, since repeated TT was unsuccessful. Furthermore, 2 patients who also did not respond to TT were referred for surgery. Ultimately, major adverse events occurred in 4 patients (1 death and 3 reoperation due to TT failure or rethrombosis), and hence, TT was considered as unsuccessful. Thus, the success of TT in the overall series was 85.2%. The rates of mortality (3.7%), embolism (3.7%), major (0%) and minor (3.7%) bleeding, and rethrombosis (3.7%) were considerably low.
The median dose of t-PA used was 50 mg (range, 12.5-194 mg). The median of repeated TT sessions per patient was 2 (range, 1-8). The success rate was relatively low after the first TT session (40.7%) and increased to 70.3% after the second session and to 81.5% after the third session. More than 3 TT sessions were required in only 3 patients which provided a cumulative success rate of 85.2% in the whole series. Four adverse events occurred during the first 2 TT sessions, 1 event occurred at the fourth session, and only 1 event occurred at the eighth TT session.
Age, sex, elapsed time since valve surgery, previous history of coronary artery disease, hypertension, diabetes, stroke/TIA, current aspirin use, heart rhythm, leading symptom on admission, thrombosed valve position, valve brand, presence of valve obstruction, baseline valve area, mean gradient, left ventricular ejection fraction, total TT dose per patient, and baseline STS mortality or morbidity did not seem to predict adverse events (Table 2). The adverse event rates significantly increased with increasing NYHA class (NYHA classes I-IV, 0% vs 15.4% vs 25% vs 100%, respectively, P = .018). The median NYHA class (III vs II, respectively, P = .03), median number of repeated TT sessions (1 vs 3, respectively, P = .03), and the mean thrombus area (1.9 ± 1 vs 0.9 ± 0.7 cm2, respectively, P = .008) were higher in those with adverse events compared to those without (Table 2). By multiple variable analysis (the regression model included the significant univariate predictors: the thrombus area, NYHA class, and the number of repeated TT sessions), the only independent predictor of adverse events was the thrombus area (odds ratio: 13.7, 95% confidence interval [CI]: 1.02-185, P = .048). By ROC analysis (Figure 1), a thrombus area ≥1.1 cm2 predicted the adverse events related to TT (sensitivity: 83.3%, specificity: 85%, area under the curve: 0.86, 95% CI: 0.71-0.99, P = .008).
Potential Confounders of Adverse Events.a

Abbreviations: ETSVS, elapsed time since valve surgery; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association; TIA, transient ischemic attack; TT, thrombolytic therapy; SD, standard deviation; STS, Society for Thoracic Surgeons.
aPlus–minus values are means ± SD. The continuous variables are compared between those with adverse event positive versus negative subjects.
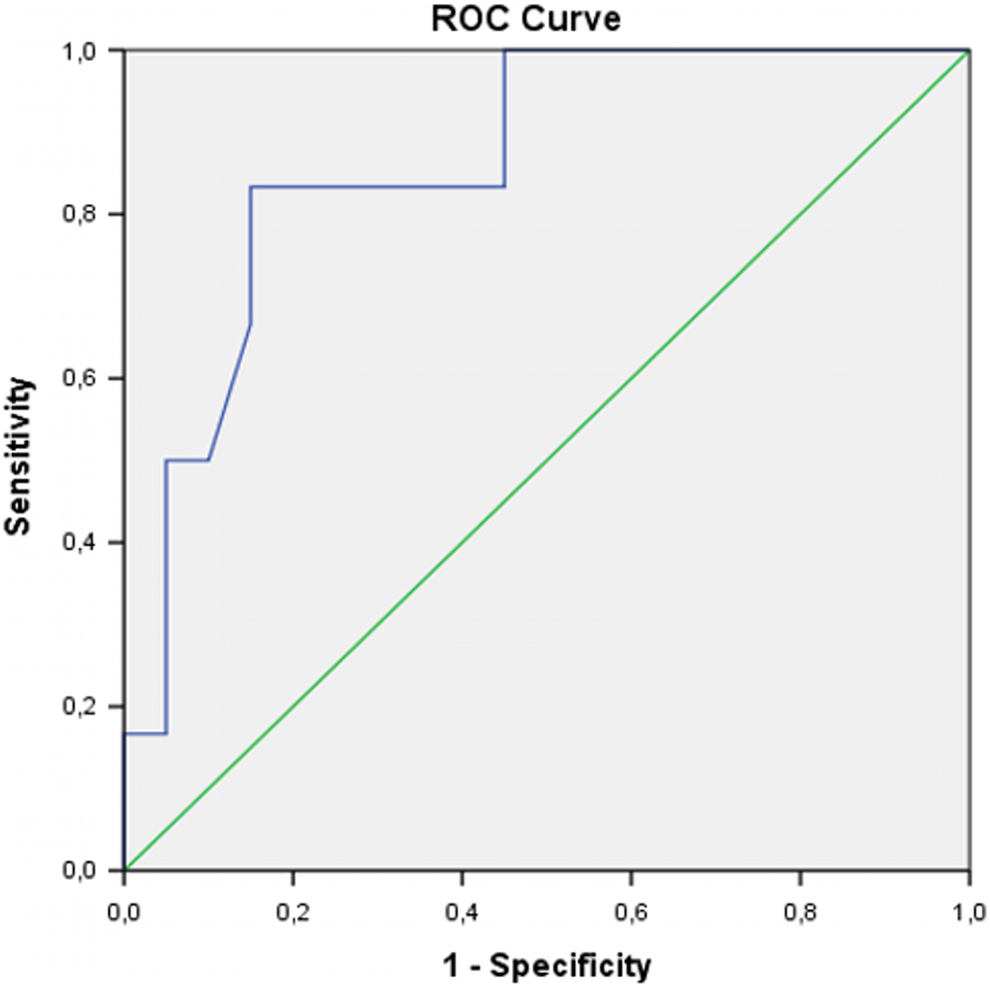
The receiver operating characteristic (ROC) curve for thrombus area in predicting adverse events related to thrombolysis.
We considered to categorize the participants into 2 age categories including those aged <70 years (10 patients) and ≥70 years (17 patients). Neither the success rate (88.2% vs 80%, respectively, P = .6) nor the incidence of adverse events (40%, 11.8%, respectively, P = .15) differed between groups 1 and 2. When the age cutoff was 75 years, again the success rates (85.7% vs 83.3%, respectively, P = 1) and the adverse event rates (19% vs 33%, respectively, P = .59) were similar between those aged <75 years (6 patients) and ≥75 years (21 patients). The STS risk of mortality and morbidity in those aged ≥70 years were 9.8% ± 2% and 32.9% ± 7%, respectively.
Discussion
The results of this single-center study demonstrate that slow or ultraslow infusions of low-dose thrombolytic agents (mostly t-PA) provide considerably high lysis rates with low major adverse event rates in elderly patients with PVT. The elderly patients included in this study would have been considered to be at moderate to high risk (calculated mean STS mortality 7.2% ± 3.1%, morbidity 29.5% ± 8.7%) if they had been candidates for valve rereplacement surgery. Furthermore, the surgical mortality rate for PVT was reported to be 18.1% (4%-69%) on average in a recent meta-analysis. 20 The mortality and major adverse event rates in the current study were only 3.7% and 14.8%, respectively. When only those aged ≥75 years are taken into account, the calculated mean STS mortality risk increases to 9.5% ± 1.7%, which renders this subgroup high-risk surgical candidates. In the current study, there was no mortality related to TT in this oldest subgroup . The major adverse event rate in this study (14.8%) is comparable to the major adverse event rate (17.7%, including major complications or failed TT) occurred in younger (<65 years) patients who underwent slow infusion of low-dose t-PA in the largest PVT series published to date. 17 In addition, the overall success rate of TT was considerably high (85.2%) in the current study which is in line with our previously published data. 16 –18 Hence, in contrast to general guideline recommendations for the treatment of patients with PVT, 1,2 most of the elderly patients may require a less aggressive initial treatment approach—that is, thrombolysis—over surgery. To date, there is no study that has previously evaluated the safety or efficacy of TT for PVT in this specific population. Hence, we believe that these data may provide a guide for clinicians and encourage using low-dose and slow or ultraslow infusion of TT protocols as an initial treatment approach in elderly patients with PVT.
The incidence of nonfatal adverse events was also relatively low (18.5%), including 3.7% embolism, 3.7% rethrombosis, 3.7% minor bleeding, and 7.4% TT failure rates. The only one patient who had cerebral embolism demonstrated transient neurological symptom that was resolved in 2 hours. This was in accordance with our previous observation that cerebral embolism during TT with slow infusion of low-dose t-PA causes only transient and/or mild neurological sequela. 15,17
The main concern in patients undergoing TT is internal bleeding, especially intracranial hemorrhage. Furthermore, contraindications to TT are also prevalent in the elderly patients. 4 The most powerful predictor of intracranial bleeding was the increasing age in one study investigating the utility of t-PA in patients with myocardial infarction. 27 Thrombolysis for pulmonary embolism was also associated with 12.9% major bleeding in patients older than 65 years. 28 However, the TT regimens used in those trials included rapid administration of higher doses of TT agents with bolus. In addition, concomitant heparin infusion is frequently used in patients undergoing TT for venous thromboembolism or myocardial infarction. Moreover, the incidence of total hemorrhage was reported to be as high as 17.3% in patients with PVT undergoing rapid infusions of streptokinase with bolus dose. 12 Major bleeding was also relatively high. By contrast, it was reported that prolonged infusion of low-dose t-PA without bolus provided successful lysis without complications in an elderly patient with pulmonary embolism. 29 Similarly, the TT regimens used in the current study included slow infusions of low doses of t-PA and streptokinase without bolus. Furthermore, heparin was interrupted during the TT sessions in all patients. Thus, only 1 (3.7%) minor bleeding occurred, and no intracranial or other major bleeding was observed in the current study.
Increased thrombus burden is associated with higher rate of complications during TT for PVT. A thrombus area ≥0.8 cm2 was shown to predict embolism in the Prosthetic Valve Thrombolysis-Role of Transesophageal Echocardiography (PRO-TEE) registry. 30 Similar results were also reported in the recently published TROIA trial. 17 In the current study, a thrombus area ≥1.1 cm2 predicted adverse events, which supports the hypothesis that excessive thrombus burden is associated with complications.
Gradual increase in TT success by repeated infusions of thrombolytic agents under the guidance of serial TEE has been noted in our previous publications, 17 –19 which was also observed in elderly patients. Furthermore, 1 embolism occurred during the first TT session and 1 minor bleeding occurred during the second TT session. No other hemorrhage or embolism occurred with repeated sessions. These data suggest that repeated TT sessions up to the protocol-specified maximum dose increase the chance of lysis without compromising safety.
Although age was not a significant univariate predictor of adverse events in the current study, most trials evaluating the safety and efficacy of TT in the elderly patients traditionally have categorized patients by a specific age cutoff (70 or 75 years) and have compared the outcome results between the groups. Accordingly, we considered to divide patients by 2 different age cutoffs. However, neither those aged ≥70 years nor the patients aged ≥75 years did worse than the remaining patients in terms of adverse events. Since the oldest patients in our cohort did not have higher mortality or complications, slow or ultraslow TT may be considered as a safe option also in this subgroup. However, since controlling INR is more difficult in the oldest patients, 31 they may be at risk of excessive thrombosis and potentially poorer outcomes.
Limitations
Our study was a single-center observational cohort that included relatively low number of elderly patients with PVT. In particular, for elderly patients with obstructive PVT, further studies with higher patient numbers are needed to support our findings. However, it should be kept in mind that elderly patients with PVT is a rare group. The subgroup of obstructive cases is much more rare. Moreover, the number of cases in the current study could be reached in a 22-year time period, despite a high-volume tertiary referral center in which special attention is paid to prosthetic valvular diseases. Hence, 12 elderly cases of obstructive PVT seem a relatively satisfactory number to draw preliminary conclusions regarding the safety and efficacy of TT in these patients. Furthermore, it should be kept in mind that the current study included a particular patient population as most of the patients were in NYHA class I (14.8%) or II (48.1%), and more than half of the patients had nonobstructive PVT (55.6%). These patients can be considered to be at low risk since their thrombi are smaller and cause less symptoms and may have a better prognosis with either surgery or other TT strategies. Thus, practicing physicians should be cautious when extrapolating our findings to all elderly patients with PVT. In addition, there was not a surgical arm to compare the outcomes of TT and surgery for the treatment of PVT in the elderly patients.
Conclusion
Slow infusion of low doses of TT agents (mostly t-PA) with repetition is successful and safe in elderly patients with PVT. However, patients with excessive thrombus burden are still at higher risk of adverse events.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
