Abstract
Considering the high frequency of bleeding complications following fibrinolytic treatment in neonates, peripheral nerve blockade (PNB) has been proposed alone or in association with lower doses of tissue plasminogen activator, as a possible new therapeutic approach in the management of neonatal limb ischemia (LI) secondary to vasospasm and/or thrombosis. The present article provides a review of the current knowledge about the topic, in order to evaluate the efficacy and safety of this therapeutic approach. According to the few case reports documented in literature and to our experience, PNB could be considered as valid procedure for the treatment of LI, especially during neonatal period, when the risk of serious bleeding associated with fibrinolytic or anticoagulant therapy is higher. Peripheral nerve blockade resulted in a safe and effective procedure for the treatment of neonatal vascular spasm and thrombosis.
Introduction
Peripheral nerve blockade (PNB) is an anesthetic technique usually performed during general anesthesia and/or as an analgesic treatment in the postoperative period. 1,2 Peripheral nerve blockade has been proposed, alone or in association with other therapies, for the treatment of vascular spasm or thromboembolic events causing limb ischemia (LI). Although thrombosis is a rare event in the pediatric population, a higher incidence (1%-9%) is reported among neonates, probably due to the physiological hypofibrinolytic state of the neonate and to the common use of indwelling catheters. 3 –5 An increased risk of catheter-related thrombosis has been reported among small-for-gestational-age neonates. 6 In this population, prenatal events leading to impaired fetal growth might be associated with structural changes in the main vessels, responsible for remarkably increased cardiovascular disease risk even in adults. 7 –9
Limb Ischemia in Neonates
During neonatal period, vascular limb compromise is being increasingly registered. An incidence of 1:4500 births per year has been recently reported. 4
Both intrauterine or postnatal precipitating events may lead to LI. In utero, LI may be secondary to prenatal thrombosis, embolism, and compression of the main arteries supplying the limb. In contrast, postnatal LI is most commonly due to iatrogenic causes and mainly related to complications arising from the use of umbilical arterial catheters (UACs) and peripheral arterial catheters. Umbilical arterial catheters used for repeated blood gas analysis sampling, continuous monitoring of blood pressure, and fluid infusion represent the main risk factors for neonatal LI. 10 Cannulation of peripheral arteries, such as the radial, ulnar, dorsalis pedis, and posterior tibial artery, is used less frequently than UAC in neonatal period but could also lead to complications such as thromboembolism, vasospasm, and thrombosis. 11
Moreover, a recent case report described the development of peripheral arterial vasospasm and diffuse peripheral ischemia after umbilical vein catheterization. 12
While vascular spasm is a transient and reversible event due to arterial constriction and often triggered by intravascular catheterization or arterial blood sampling, thrombosis is a complete or partial long-lasting obstruction of the arteries. Although early clinical signs (pallor, cyanosis, weak, or absent peripheral pulses) are similar in both conditions, clinical effects of vascular spasm usually last less than 4 hours from the onset. In this case, a complete bloodstream recovery can be obtained with early diagnosis and treatment. Furthermore, a rapid management of vascular spasm is recommended since it could represent the primary event leading to thrombosis.
The salvageability of the limb depends on the interval between the initial vascular event and the onset of clinical signs. Since prolonged ischemia can lead to necrosis and loss of large amounts of tissue, early diagnosis and treatment represent the key for a successful management. 4 Treatment of LI is still a challenge for clinicians. Among neonates, the risk of serious bleeding associated with fibrinolytic or anticoagulant therapy must be balanced against the possibility of limb loss or death without an appropriate therapy. Reflex vasodilatation by warming the contralateral limb should be considered as a first-line treatment but may not induce a complete resolution of ischemia. 11 Topical administration of 2% nitroglycerin is a further therapeutic option. A successful and safe use of this technique was reported in 3 neonates with vasospasm-induced LI occurring after the positioning of UAC or radial artery line. 13
In case of persisting clinical signs and documented arterial obstruction, antithrombotic therapy is currently considered the therapy of choice. 3,14 During neonatal period, thrombosis must be always objectively confirmed before undertaking fibrinolytic or anticoagulation therapy, since the immature hemostatic system may result in a higher risk of bleeding. Both these treatments are absolutely contraindicated among neonates with preexisting or existing hemorrhage. 15 In particular, due to the risk of bleeding, fibrinolytic treatment has been recently recommended only in case of limb threatening or organ threatening (via proximal extension) when no satisfactory response to the anticoagulant treatment has been achieved. 14
Mechanism of Action of PNB in the Treatment of LI
Peripheral nerve blockade has been proposed as therapeutic strategy for the treatment of LI. Peripheral nerve blockade is able to induce vasodilatation of the peripheral vessels by means of a sympathetic blockade. Furthermore, it stops afferent impulses and inhibits peripheral vasoconstriction because of a reduction in the pain-induced release of catecholamines, cortisol, and antidiuretic hormone. 16
This procedure was also successfully used by Hargreaves and coworkers in a preterm neonate (29 weeks of gestation) with aortic thrombosis triggered by umbilical catheterization. 17 In this case, antithrombotic therapy was contraindicated because of the preexisting intraventricular hemorrhage (IVH). The authors also reported that administration of bupivacaine (1.5 mL of a solution equal to 0.17%) into the extradural space via the caudal route provided good pain relief and improvement in the discoloration of the leg. Since the beneficial effect lasted only 6 hours, an extradural catheter was positioned for the administration of continuous infusion of the drug.
Clinical Experiences With PNB in the Treatment of LI
Limb Ischemia already evident at birth
Few data exist regarding incidence, diagnosis, and treatment of early LI occurring in delivery room. 4 An ischemic limb caused by a vascular spasm can spontaneously exhibit tendency to normal circulation. 18 Warming of the contralateral limb is usually recommended in case of still viable but ischemic limb. If this approach is not sufficient and the limb is seriously compromised, PNB could represent a valid therapeutic intervention while waiting for Doppler ultrasonography.
Table 1 reports our experience with 2 neonates, presenting LI already at birth, who were successfully treated with PNB. In the first neonate (27 weeks of gestation), axillary nerve blockade of the brachial plexus was helpful to solve the persistent (>45 minutes) and severe arm ischemia secondary to external compression during labor. 19 The second neonate, born of twin pregnancy at 31 weeks of gestation, presented pale, cold, and pulseless left leg immediately after birth (Figure 1). Oxygen saturation was not detectable at the affected limb until 25 minutes of life, when PNB was performed. Doppler ultrasonography, performed 2 hours later, was normal.
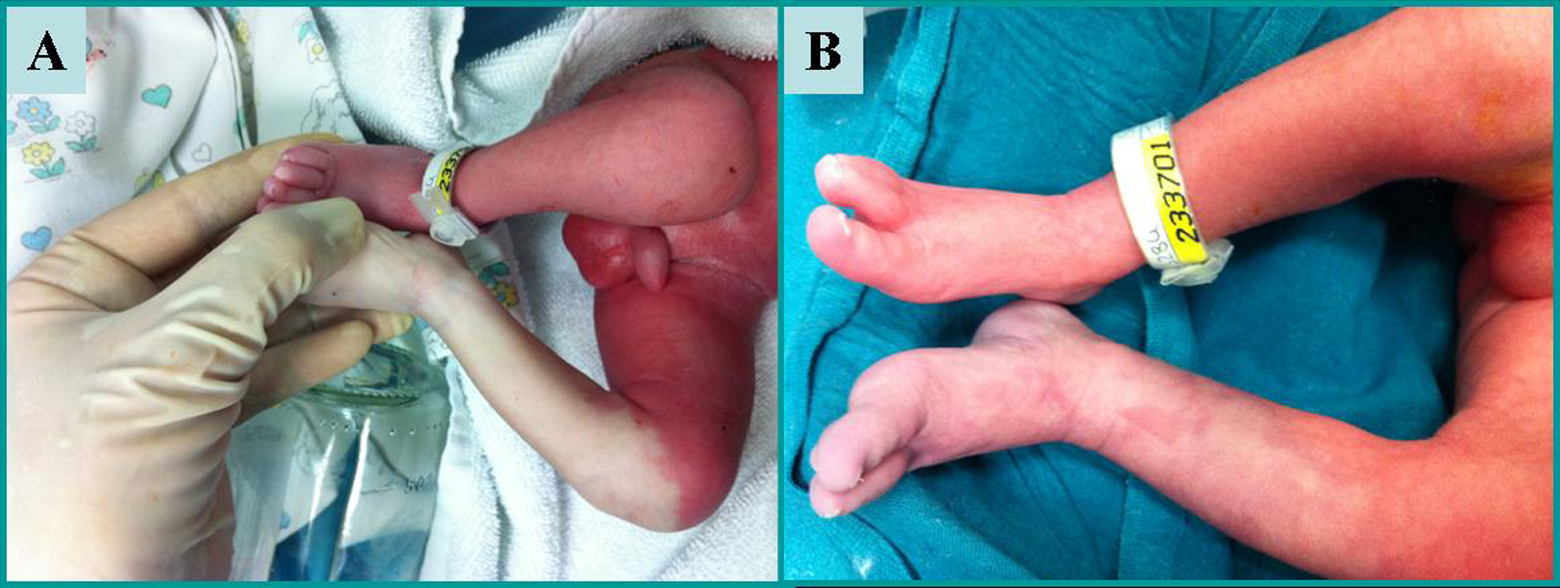
Severe ischemia of the left leg (A) and improvement after caudal nerve blockade (B).
Clinical Data of Neonates With LI Receiving PNB at Birth.
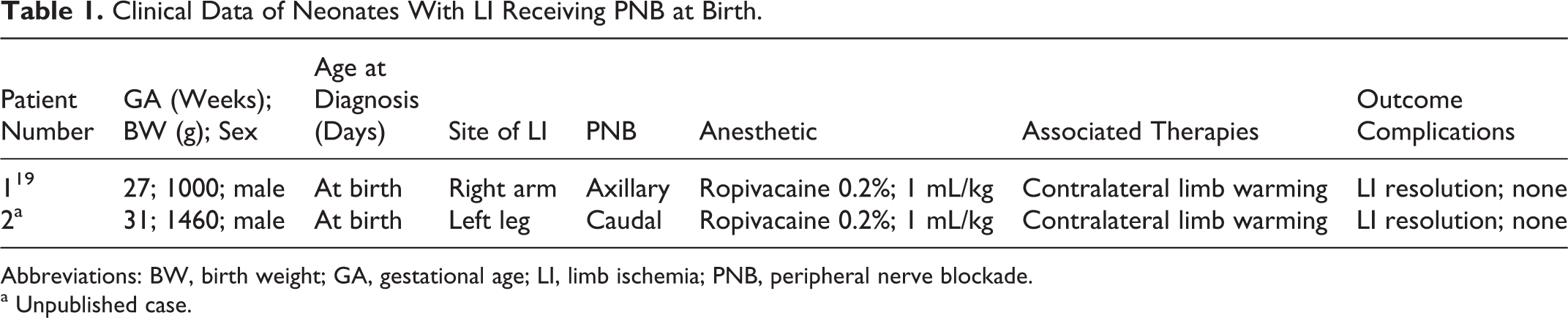
Abbreviations: BW, birth weight; GA, gestational age; LI, limb ischemia; PNB, peripheral nerve blockade.
a Unpublished case.
Since a precocious treatment is able to allow the salvaging of the limb, PNB should be considered as valid therapeutic approach in the management of persistent and/or severe LI already evident at birth.
LI in the postnatal period
According to the scientific literature, only case reports and small series addressed PNB as possible therapeutic approach in case of arterial thrombosis, documented by Doppler ultrasonography, when contraindication to anticoagulant or fibrinolytic therapy existed. 17,20 –22
Table 2 summarizes the available data concerning neonates with documented thrombosis and contraindication to the antithrombotic therapy. In all cases, except for 1 neonate with low gestational age, a preexisting IVH contraindicated the fibrinolytic treatment. In 4 of the 6 cases, thrombosis affected peripheral vessels, whereas in 2 cases (patients 1 and 5) major arteries were involved. In these 2 cases, PNB treatment was more aggressive: in 1 case after 2 blocks a continuous infusion of anesthetic through extradural catheter per 48 hours was necessary, while in the second one a high number of PNB (up to 14) was performed. In all cases, PNB was effective in reversing LI, although spontaneous amputation of one or more toes/fingers occurred in 3 neonates.
Clinical Data of Neonates With Thrombosis Receiving PNB.
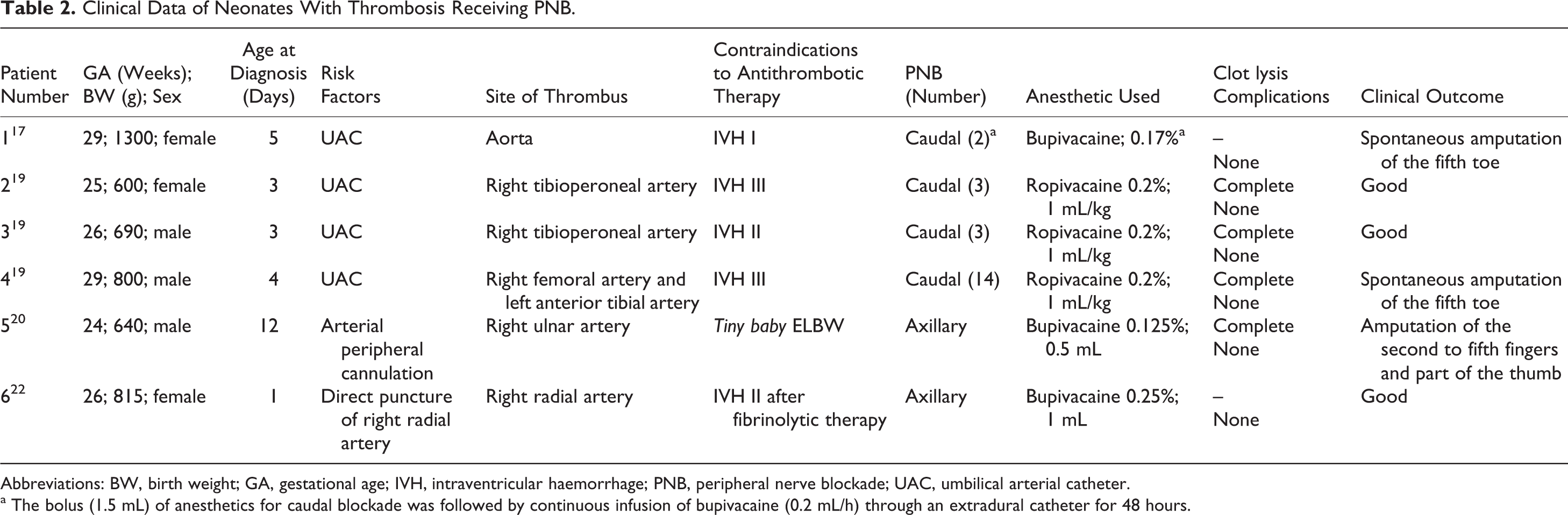
Abbreviations: BW, birth weight; GA, gestational age; IVH, intraventricular haemorrhage; PNB, peripheral nerve blockade; UAC, umbilical arterial catheter.
a The bolus (1.5 mL) of anesthetics for caudal blockade was followed by continuous infusion of bupivacaine (0.2 mL/h) through an extradural catheter for 48 hours.
In our experience, clinical improvement obtained with PNB was extremely rapid, as confirmed by Doppler ultrasonography carried out 2 hours after the procedure. 21
Based on the reported efficacy of PNB in case of documented thrombosis and/or contraindications to the fibrinolytic treatment, we recently performed the PNB in association with the fibrinolytic treatment among 4 neonates with LI due to thromboses affecting major arteries (Table 3). After this therapeutic approach, clinical signs of LI clearly improved, although a complete vascular recanalization was achieved only in 1 case. The neonate presenting with aortic thrombosis and twin–twin transfusion syndrome (patient 3) died at 3 days of life, because obstruction was not resolved and renal failure occurred. In patient 4, showing bilateral iliac occlusion, total recanalization was obtained in the right iliac artery, while only partial canalization of the left iliac artery caused leg gangrene requiring amputation. Multiple inherited prothrombotic risk factors (factor V Leiden and prothrombin 20210A) were documented in this neonate. After the infusion of fresh frozen plasma, all the patients, reported in Table 3, received thrombolytic treatment for 24 hours with tissue plasminogen activator (Actilyse, Boehringer Ingelheim, Italy; bolus 0.5 mg/kg, then continuous infusion of 0.3 mg/kg per h) followed by unfractionated heparin.
Clinical Data of Neonates With Thrombosis Receiving Both PNB and Antithrombotic Therapy.

Abbreviations: BW, birth weight; GA, gestational age; PNB, peripheral nerve blockade; t-PA, tissue plasminogen activator; TTT, twin–twin transfusion syndrome; UAC, umbilical arterial catheter; UFH, unfractionated heparin.
Side Effects of PNB
The side effects of PNB may be related to the procedure itself or to toxic reactions to the drug used. Procedure complications may be local or systemic infection, dural puncture, vascular puncture, and failure to identify the sacral hiatus. In particular, important age-related anatomy differences in the spinal cord and dural sac should be considered in case of caudal blockade. In fact, among neonates the spinal cord extends as far as L3 (and not L1 as at 1 year of age) and the dural sac extends as far as S3 (and not S1 as at 1 year of age). 23 During the neonatal period, a potentially higher toxicity of the amide local anesthetics should also be taken into consideration due to lower hepatic metabolism and decreased binding to the plasmatic proteins, such as albumin and alpha-1 acid glycoproteins. However, these conditions could be offset by either a higher blood distribution volume or the lower minimum concentration of local anesthetics required to block impulse conduction—due to decreased nerve sizes and immature myelination. 23
In all cases described in this article no side effects due to the procedure or the drug itself have been reported. Our experience also confirms the safety of this approach. In fact, a series of blood tests including serum electrolytes, transaminases, total and direct bilirubin, complete blood count, and methemoglobin levels were assessed in all our patients prior, 24 hours, and 48 hours after PNB, and they resulted normal. Moreover, normal arterial blood pressure, heart rate, and oxygen saturation were recorded and the occurrence of local or generalized infection was excluded.
Peripheral Nerve Blockade Technique
If PNB of the upper limbs is required, the axillary approach is currently used to induce brachial plexus blockade. 20,24 –26 The technique for axillary nerve blockade includes the application of an eutectic mixture of local anesthetics (EMLA cream, AstraZeneca, Rueil-Malmaison, France; a 1:1 mixture of lidocaine and prilocaine) in the axillary region. The arm should be abducted at 90° and the elbow flexed at 110°. The axillary artery should be palpated and the puncture site infiltrated subcutaneously with lidocaine. A 24-gauge needle, should be inserted high in the axilla under aseptic conditions and eventually connected to a peripheral nerve stimulator. The site should be aspirated to ensure that no vessel has been inadvertently punctured. Finally, local anesthetic drug should be slowly injected.
The technique for caudal nerve blockade involves, after the application of EMLA cream, the insertion of a 24-gauge needle through the sacral hiatus into the extradural space under aseptic conditions, with the patient in left lateral position. Initial aspiration should be performed to exclude the presence of blood or cerebrospinal fluid, followed by subsequent injection of the local anesthetic drug.
Recent findings show that the pediatric population, in particular neonates and infants, may profit from direct ultrasonographic visualization of the anatomical structures during these invasive procedures, in order to improve PNB efficacy and safety. Advantages of ultrasonography-guided regional anesthesia are higher success rate, improved block qualities, and avoidance of complications. 27,28 However, since the visualization of the plexus itself on ultrasound picture may be challenging, electrostimulation has also been evaluated as possible technique of identification of the appropriate space. 23
Both bupivacaine and ropivacaine have been used in neonatal LI to perform PNB (Tables 1, 2, and 3), although the doses varied between the reported cases. Ropivacaine is a long-acting amide local anesthetic, which is structurally similar to bupivacaine but induces less motor block (due to its lower lipid solubility), as well as less cardiac and central nervous system toxicity. In our experience, we used 1 mL/kg ropivacaine 0.2% as recommended in the neonatal period. 29
Conclusions
During neonatal period, PNB is a safe and effective procedure for the treatment of vascular spasm and thrombosis. Further investigations are warranted to validate this innovative approach and to verify whether the combination of PNB and systemic antithrombotic therapy could reduce its dosage and duration.
Furthermore, prospective studies are also recommended to identify the best dosage and injection intervals of the local anesthetics administered during PNB.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
