Abstract
Keywords
Introduction
Venous thromboembolism (VTE), 1 as deep vein thrombosis (DVT) and pulmonary embolism (PE), has been considered the most common preventable cause for in-hospital mortality. 2,3 Besides causing more deaths than the combination of breast cancer, HIV/AIDS, and traffic accidents, VTE was also associated with significant morbidity by means of postthrombotic syndrome disabling patients and by increasing health costs. 4
Indicating an international problem concerning VTE prophylaxis, only about one third of the patients at VTE risk was consistently reported to receive appropriate prophylaxis. 3 Unfortunately, the cost of such neglect seems to be high, since the incidence of DVT may reach to 40% among general surgical patients 2 and also 40% to 70% in patients undergoing major orthopedic surgery who do not receive prophylaxis. 5
Despite current data on successful reduction of VTE risk by means of VTE prophylaxis, 5 –8 thromboprophylaxis still remains underestimated in hospitalized patients. 9,10 Lack of easy-to-use and convenient national guidelines has been considered among the factors leading to and was identified as the main reason underlying this neglect. 3 Moreover, some health care providers were shown to be unaware of effective measures to identify high-risk patients and provide appropriate prophylaxis, leading to gaps between evidence-based guidelines and recommendations and practice in the hospital settings. 11
Therefore, the current study, representing the Turkey arm of multinational cross-sectional Epidemiologic International Day for the Evaluation of Patients at Risk for Venous Thromboembolism in the Acute Hospital Care Setting (ENDORSE) study conducted across 32 countries was designed to evaluate VTE risk and use of thromboprophylaxis in the acute care hospital setting. 12
Methods
Study Population
Representing Turkey arm of the international cross-sectional ENDORSE study involving 68 183 patients across 32 countries including Algeria, Australia, Bangladesh, Brazil, Bulgaria, Colombia, Czech Republic, Egypt, France, Germany, Greece, Hungary, India, Ireland, Kuwait, Mexico, Pakistan, Poland, Portugal, Romania, Russia, Saudi Arabia, Slovakia, Spain, Switzerland, Thailand, Tunisia, Turkey, United Arab Emirates, UK, USA, and Venezuela, 12 the current study was conducted with 1701 patients enrolled from 11 hospitals selected via random sampling method among hospitals with more than 50 beds across Turkey (Table 1 ).
List of Participant Hospitals Across Turkey

Hospital wards occupied by acute medical and surgical patients were included, while specialized psychiatric, pediatric, eye, ear, nose and throat, dermatology, alcohol/drug treatment, rehabilitation, accident and emergency, maternity, chronic, and palliative care wards were excluded from the study. Each selected ward was assessed on a prespecified day; and data based on the medical charts of all eligible patients who were occupying a bed in the selected wards during that prespecified day of survey were collected.
Study population consisted of patients hospitalized for acute or exacerbated chronic medical illnesses or elective major surgical operation in the eligible wards including general medical, respiratory, or cardiac wards, and general surgical and orthopedic wards at the study hospitals. Patients aged ≥40 years at the eligible medical wards or ≥18 years at the eligible surgical wards were included in the study. Exclusion criteria of the study are shown in Figure 1 . Written informed consent was obtained from each participant following a detailed explanation of the objectives and the protocol of the study, which was approved by the local ethics committee, and the study was conducted in accordance with the ethical principles stated in the “Declaration of Helsinki” and International Conference on Harmonization/Good Clinical Practice (ICH/GCP) guideline.
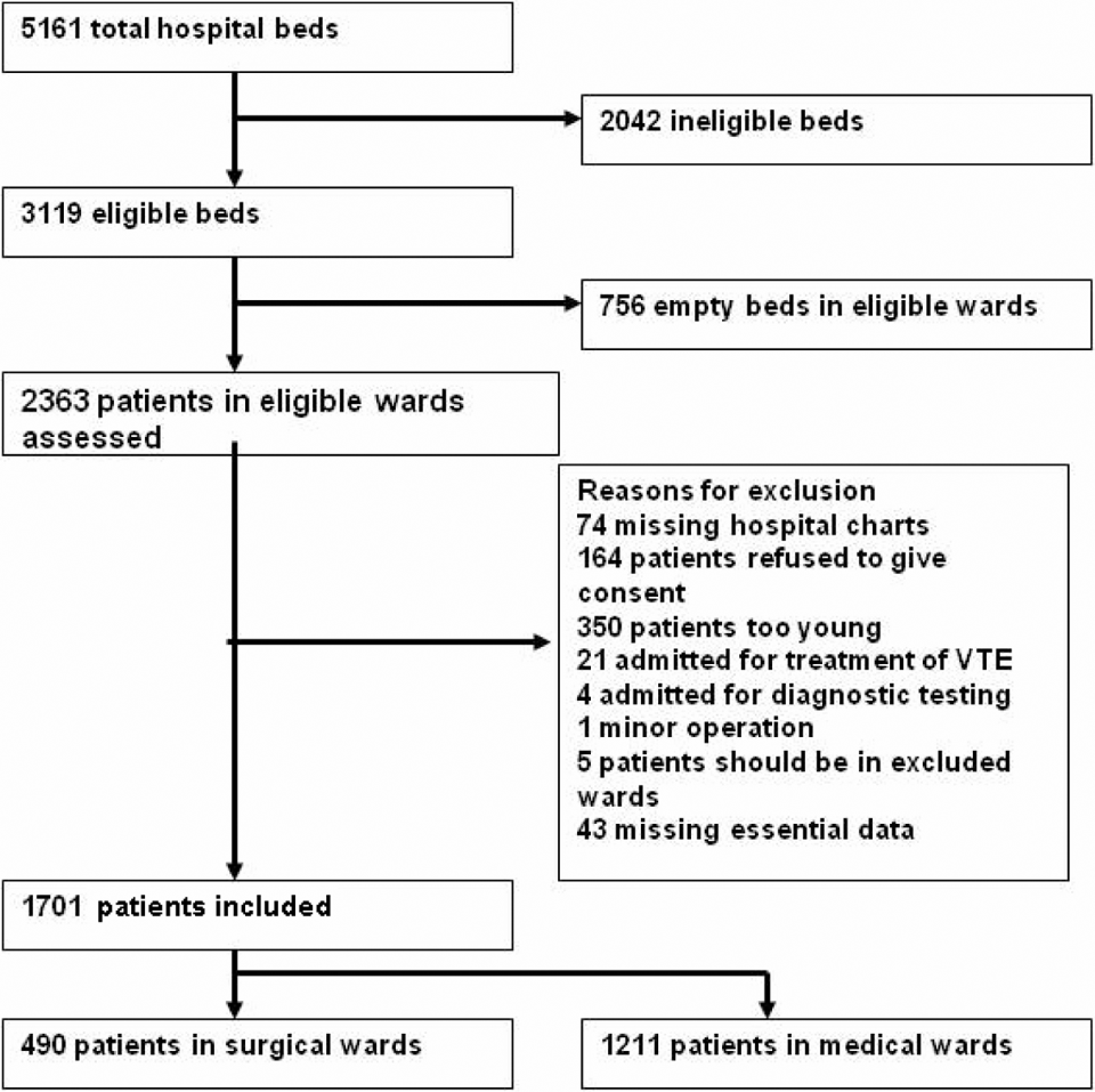
Selection of study population and reasons for exclusion.
Assessments
Data concerning patient demographics, admission and post-admission diagnoses, duration of hospital stay, risk factors associated with VTE, and type of VTE prophylaxis with respect to 2004 American College of Chest Physicians (ACCP) evidence-based consensus guideline, which was the most current guideline on the days of the survey, 9,10 were collected via retrospective evaluation of medical charts. Enrolled patients were assessed for risk of VTE in accordance with the 2004 ACCP guidelines, which included acutely ill medical patients and those admitted for major trauma or undergoing a major surgical procedure requiring general or epidural anesthesia for at least 45 minutes. 6,12 Surgical patients were first assessed for age, type of surgery, and duration of anesthesia, and then classified as being at highest, high, moderate, or low risk for VTE as per the ACCP guidelines. 6,12 Patients were considered to have a sufficient risk of bleeding indicating a contraindication to anticoagulant prophylaxis if intracranial hemorrhage, hepatic impairment, bleeding at hospital admission, active gastroduodenal ulcer, or a known bleeding disorder was apparent on admission or developed during hospitalization. 13 The use of recommended types of VTE prophylaxis received by at-risk patients was defined according to specific recommendations from the 2004 ACCP guideline that proposed implementation of standard group-specific prophylaxis in 3 to 4 defined risk-stratified groups, based on appropriate prescription and administration of pharmacological and mechanical VTE prophylaxis. 6
Statistical Analysis
Data were summarized as median and n (%) where appropriate. To assess the true occurrence of VTE risk at 25% with a margin of error of 4%, a minimum of 450 patients per analysis group were required. SAS version 9.1 was used for all statistical analyses.
Results
From a total of 5161 beds included in 11 hospitals, 2042 were recorded to be ineligible and of 3119 beds in the eligible wards, 756 were empty on the day of the survey. Therefore, 2363 patients in eligible wards were included in the study. Following exclusion of 662 patients in accordance with the exclusion criteria as presented in Figure 1, remaining 1701 patients comprised our study population, of which 490 (28.8%) were surgical patients and 1211 (71.2%) were medical patients (Figure 1).
According to the ACCP criteria, overall 35.6% (606 of 1701) of the patients were identified to be at VTE risk. Percentage of surgical patients among VTE risk group was 52.5% (n = 318 of 606); while 64.9% (n = 318 of 490) of surgical and 23.8% (n = 288 of 1211) of medical patients were at risk.
Most of the patients in the medical wards were males (70%), with a median age of 64 years and an overall median body mass index (BMI) of 23 kg/m2. However, 51.8% of the patients in the surgical wards were males, with a median age of 55 years and a median BMI of 27 kg/m2. Median length of hospital stay prior to data collection was 7.0 days in the overall study population, 7.0 days for patients in medical wards, and 8.0 days for those in the surgical wards.
The most frequent medical conditions were pulmonary infection (31.8%), chronic pulmonary disease (29.3%), chronic heart failure (9.8%), obesity (8.4%), and acute noninfectious respiratory disease (8.3%) defined before or during hospitalization in overall at-risk study population. Distribution of medical conditions in medical and surgical at-risk patients is presented in Table 2 .
Patient Demographics, Medical Conditions, and VTE Risk Factors Defined Prior to and During the Course of Hospitalization Among Surgical and Medical Risk Groups
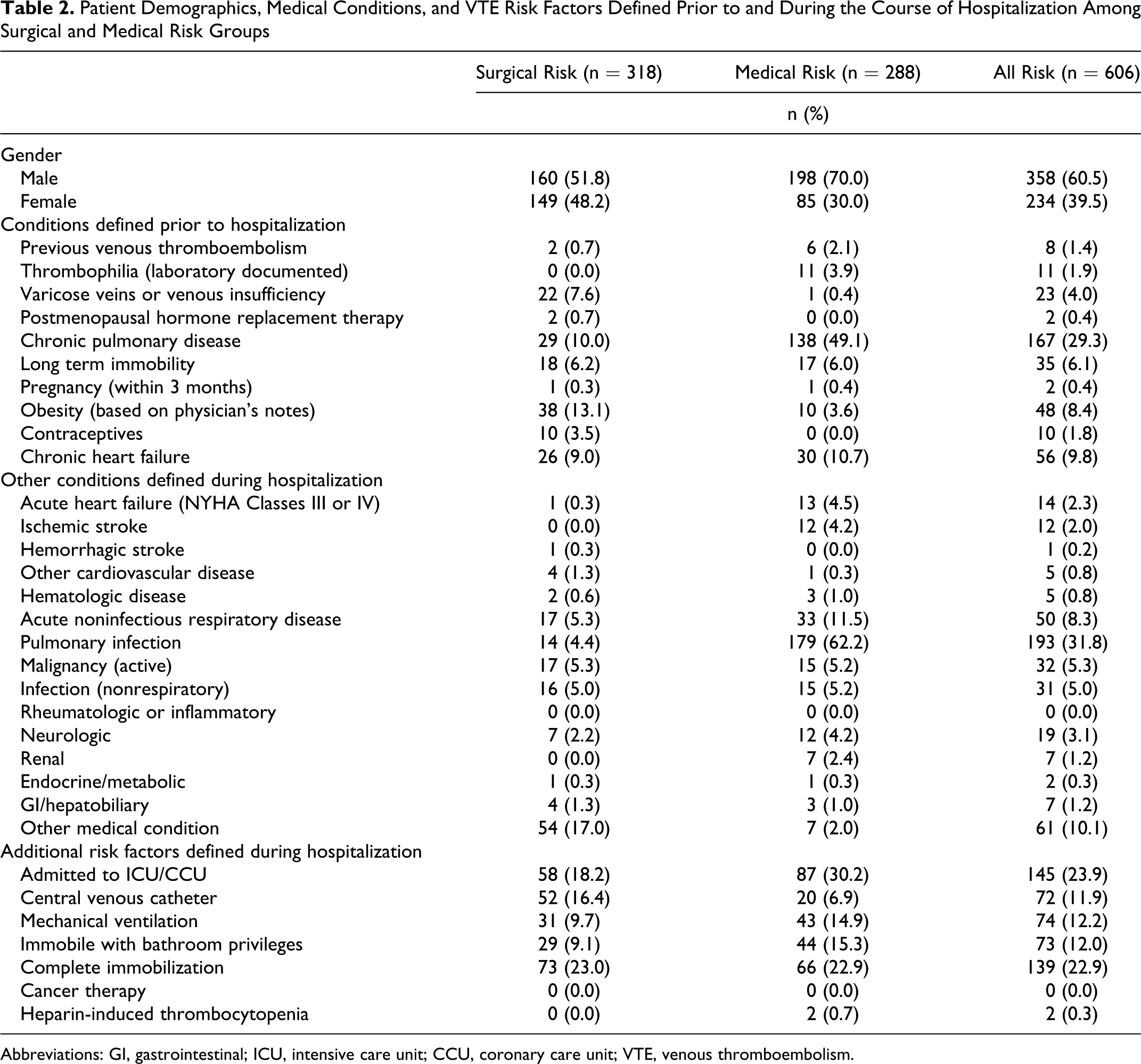
Abbreviations: GI, gastrointestinal; ICU, intensive care unit; CCU, coronary care unit; VTE, venous thromboembolism.
The most common additional risk factors were admission to intensive or NHYA classes (23.9%), complete immobilization (22.9%), and mechanical ventilation (12.2%) noted throughout the hospitalization period among all at-risk patients (Table 2). In medical at-risk group, admission to intensive care unit (ICU)/coronary care unit (CCU; 30.2%) was followed by complete immobilization (22.9%), while the reverse order was true for the surgical ward patients (23.0% for complete immobilization and 18.2% for admission to ICU/CCU; Table 2).
Although 606 (35.6%) patients were identified to be at VTE risk necessitating ACCP-recommended prophylaxis, only 235 patients among them (38.7%) were ordered prophylactic treatment according to ACCP recommendations. Among 490 surgical patients, 318 (64.9%) were found to be at risk for VTE and the ACCP-recommended prophylaxis was administered only for 124 (39.0%) of them. Among 1211 medical patients, 288 (23.8%) were found to be at risk for VTE and the ACCP-recommended prophylaxis was administered only for 111 (38.5 %) of them (Table 3 ).
Distribution of Patients Administered With ACCP-Recommended Thromboprophylaxis in Terms of Medical Diagnosis
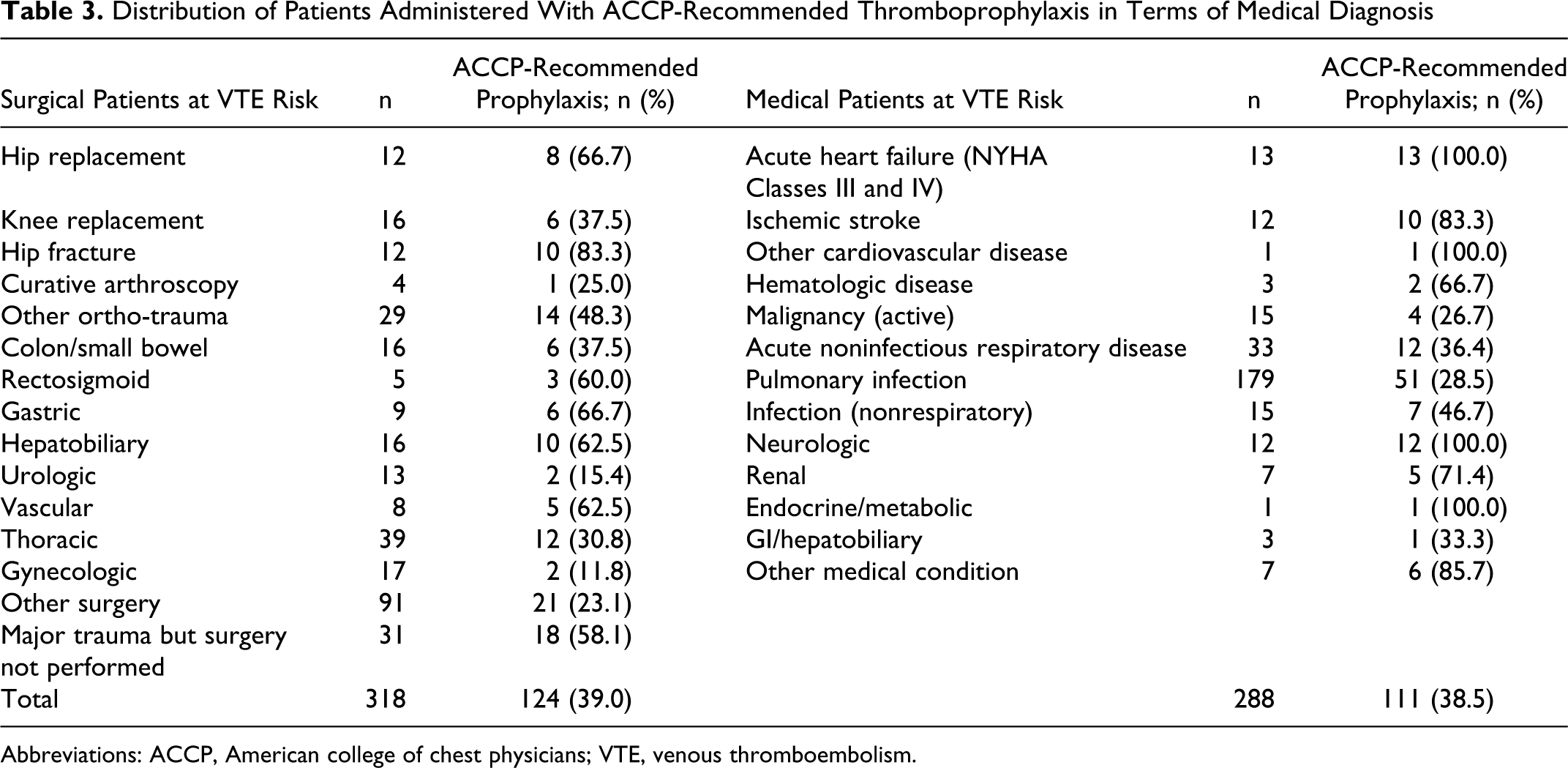
Abbreviations: ACCP, American college of chest physicians; VTE, venous thromboembolism.
American College of Chest Physicians−recommended thromboprophylaxis rate was the highest for patients undergoing hip fracture surgery (83.3%) and hip replacement (66.7%) in the surgical wards, while the highest thromboprophylaxis rates were obtained for patients with acute heart failure (NYHA classes III and IV) or neurological disease in the medical wards (100% for each; Table 3).
Low-molecular-weight heparin was the overall drug of choice for the anticoagulant prophylaxis among patients (38.1%) both in surgical (38.7%) or medical (37.5%) wards (Table 4 ).
ACCP-Recommended Thromboprophylaxis in Surgical and Medical Ward Patients at VTE risk
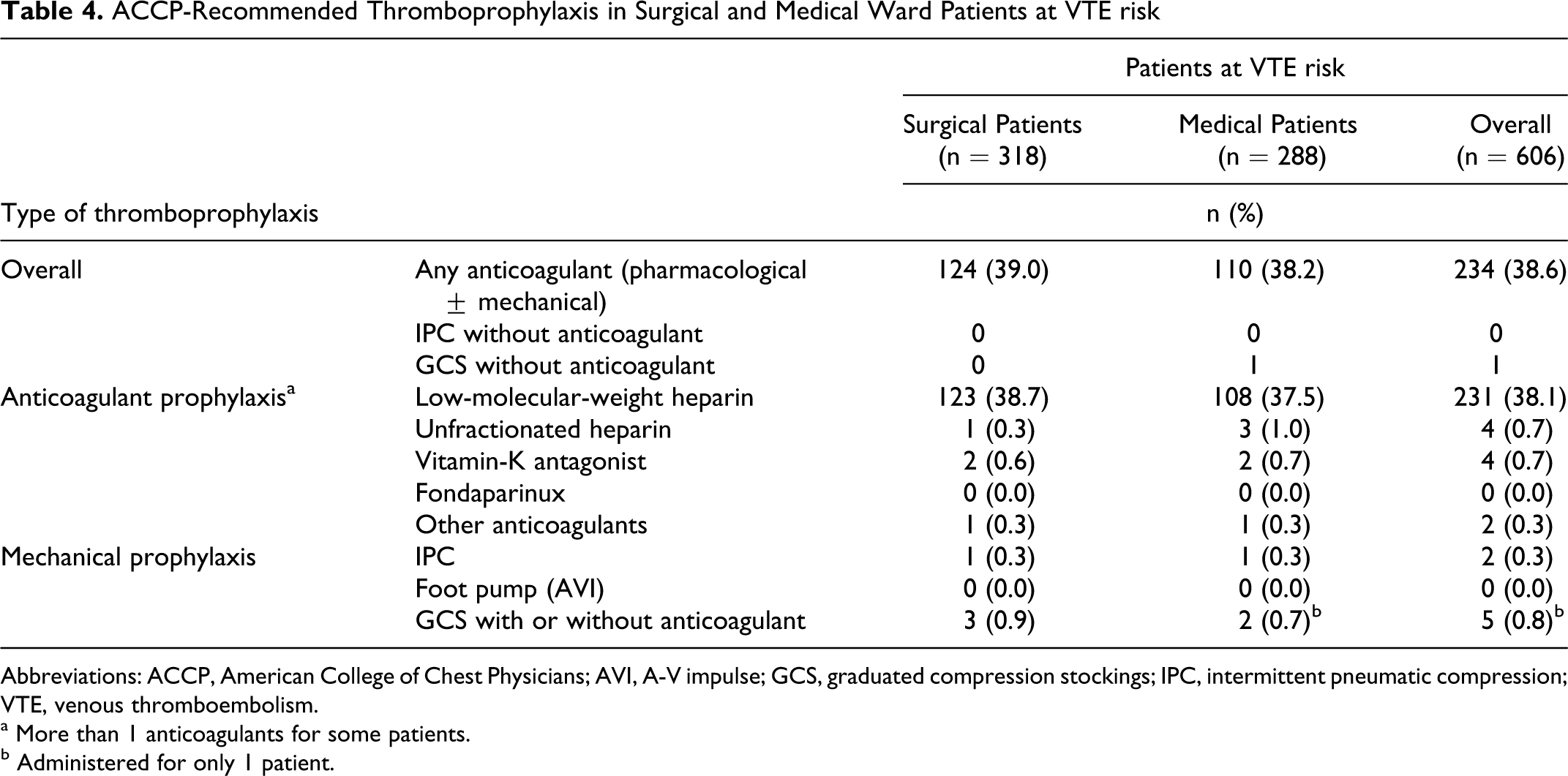
Abbreviations: ACCP, American College of Chest Physicians; AVI, A-V impulse; GCS, graduated compression stockings; IPC, intermittent pneumatic compression; VTE, venous thromboembolism.
a More than 1 anticoagulants for some patients.
b Administered for only 1 patient.
Regarding high bleeding risk, 8.7% (n = 25) of medical patients and 8.8% (n = 28) of surgical patients were considered to have a contraindication to anticoagulant prophylaxis. The most common contraindications to anticoagulant prophylaxis were bleeding on admission and thrombocytopenia in medical patients, while bleeding at hospital admission and intracranial hemorrhage were the major contraindications for the surgical ward patients (Table 5 ).
Contraindications to VTE Prophylaxis Determined in Surgical and Medical Ward Patients With VTE Risk
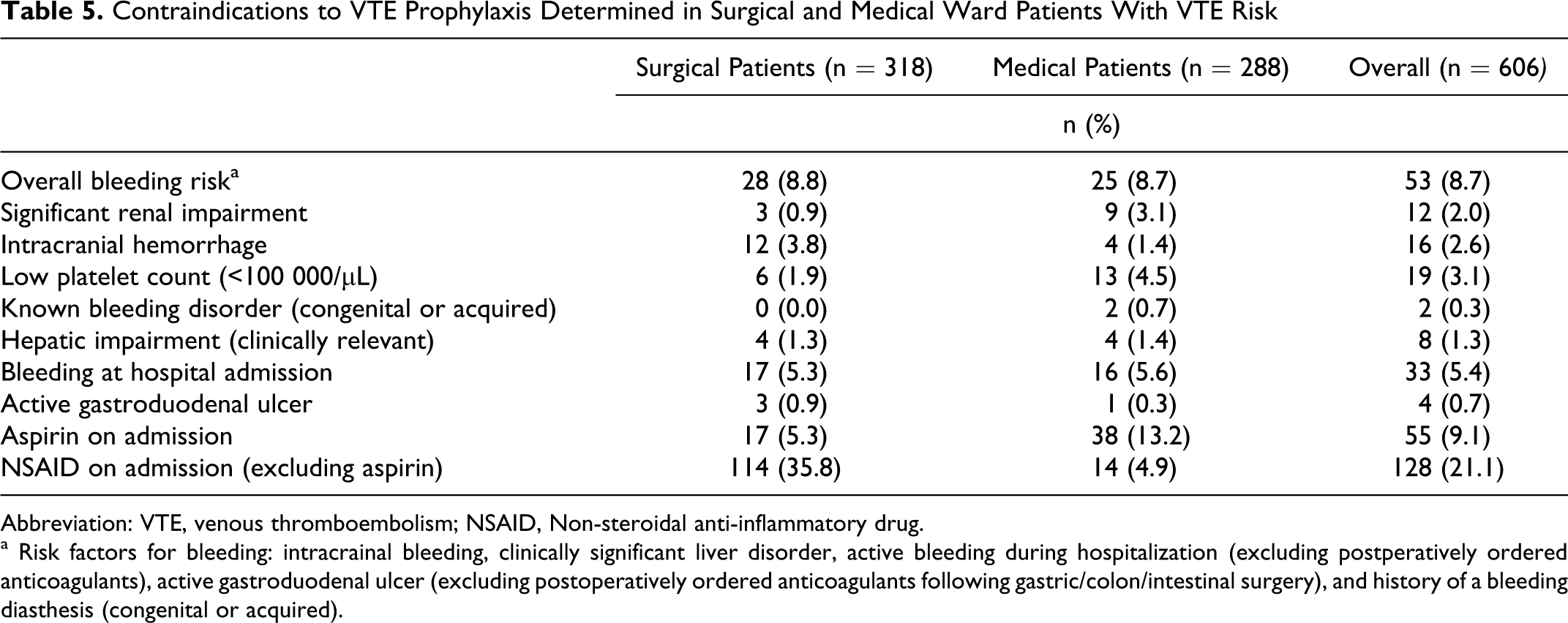
Abbreviation: VTE, venous thromboembolism; NSAID, Non-steroidal anti-inflammatory drug.
a Risk factors for bleeding: intracrainal bleeding, clinically significant liver disorder, active bleeding during hospitalization (excluding postperatively ordered anticoagulants), active gastroduodenal ulcer (excluding postoperatively ordered anticoagulants following gastric/colon/intestinal surgery), and history of a bleeding diasthesis (congenital or acquired).
Discussion
Since VTE is a leading but highly preventable cause for significant morbidity and mortality, appropriate prophylaxis of patients at high risk for VTE has been recognized as the highest ranked safety practice.
According to ENDORSE study, global risk of VTE was determined in accordance with ACCP criteria to be 51.8% in hospitalized patients (95%CI 51.4-52.2, range 35.5-72.6), with surgical patients (64.4%; 63.8-64.9; 44.1-80.2) having higher VTE risk than medical patients (41.5%; 41.0-42.0; 21.1-71.2). 12 Overall, VTE risk in our hospitalized patients (35.6%) seems to be the lowest among 32 countries included in the multinational ENDORSE study. 12 Similar low incidence of VTE risk has been related to underrecognition of risk factors. 14,15 Similarly, traditionally accepted lower VTE risk in Asian population 16 was reported to be dependent on insufficient awareness among physicians concerning VTE risk estimation rather than low thrombosis tendency specific to this population. 17,18
Venous thromboembolism risk for surgical patients in our study (64.9%) was quite similar to that obtained in ENDORSE global study (64.4%). 12 Therefore, lower incidence of overall VTE risk among Turkish patients seems to be attributable to medical rather than surgical wards. Accordingly, the risk for VTE among Turkish medical patients (23.8%) was much lower when compared to medical patients in ENDORSE study (41.5%), 12 as well as International Medical Prevention Registry on Venoous Thromboembolism (IMPROVE) 9 (60%) and Multi-Centre Chart Audit of the Utilization of Risk Assessment and of Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients in Canada (CURVE) 10 (90%) studies. In a recent study, retrospective evaluation of 49 cases with pulmonary embolism admitted to a hospital in Turkey showed that 37% of patients were determined to have VTE risk, which is most probably higher in fact, but missed. 19 One-day screening of 173 patients hospitalized in surgical and medical wards of a single hospital in Turkey has also showed that 38% of patients were determined to be at VTE risk requiring VTE prophylaxis. 20 Moreover, retrospective evaluation of 21 Turkish patients who developed PE during their hospitalization revealed that none of them was administered prophylaxis despite evidence of risk factors among 17 (81%) of them. 20 Thus, physician awareness of VTE risk assessment seems to be insufficient among overall internal medicine physicians.
Surgical patients have been shown to be more likely than medical patients to receive appropriate thromboprophylaxis and it was reported that thromboprophylaxis is underused in the medical wards. 9,10 Underestimation of overall VTE risk among medical patients may indicate the need for valid and reliable national guidelines and risk assessment tools specific to VTE screening in medical wards. Lower level of physician awareness for VTE risk assessment among medical patient population may be associated with the availability of more detailed former studies in surgical patients for much longer time than studies concerning prophylaxis of medical patients. Moreover, the assessment of VTE risk in surgical patients is easier, via simpler procedure based on determination of the type of surgery. 6,12 In accordance with previously shown 13% VTE risk in patients hospitalized for the acute episode of chronic obstructive pulmonary disease in Turkey, 21 chronic pulmonary and noninfectious respiratory diseases were the leading VTE risk factors determined during prehospitalization and in-hospitalization periods, respectively, in the present study.
However, more successful screening achieved for risk assessment among surgical patients cannot be considered as an advantage for VTE prophylaxis, as long as the VTE prophylaxis was evident only in one third of the population at risk. The neglect considering almost two thirds of the population at risk seems to be compatible with the world-wide failure in ongoing practice considering VTE prophylaxis enclosing only one third of patients at VTE risk. 3 In fact, low probability of physicians to reconsider the well-being of the patients who discharged without prophylaxis, especially at hospitals with insufficient follow-up routine, may have a role in neglecting the prophylaxis for patients at VTE risk. 20
Representing a contraindication for the thromboprophylaxis, bleeding risk among our medical and surgical patients (8.7%) was determined to be similar and also compatible with overall results of the ENDORSE study (10.0%). 12 However, anticoagulant prophylaxis was not administered in approximately 40% of our patients having no contraindication to treatment.
In conclusion, majority of adult inpatients especially in medical wards at different hospitals across Turkey seem to be underestimated in terms of determination of at-risk patients and administration of proper thromboprophylaxis. Therefore, strategies to improve national guidelines providing simple easy-to-use and reliable diagnostic criteria for the accurate estimation of patients at VTE risk and reasonable strategies making routine VTE prophylaxis a priority becomes paramount.
Footnotes
This paper was presented orally at 13th Annual Congress of the Turkish Thoracic Society which was held on May 6-9, 2010 in Istanbul, Turkey.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
Acknowledgement
We thank Cagla Isman, MD, and Prof Sule Oktay, MD, PhD, from KAPPA Consultancy Training Research Ltd, Istanbul, who provided editorial support.
ENDORSE Study Group (by the center’s name in descending order of number or patients enrolled):
Süreyyapaşa Chest Diseases & Thoracic Surgery Training & Research Hospital, Istanbul- Semih Halezeroğlu, MD; Adnan Yilmaz, MD. Dr. Suat Seren Chest Diseases & Thoracic Surgery Training & Research Hospital, Istanbul- Ali Kadri Çirak, MD; Mehmet Böncü, MD. Şişli Etfal Training and Research Hospital, İstanbul - Ali İhsan Dokucu, MD; Çiğdem Yazici Ersoy, MD. Kocaeli University Faculty of Medicine, İzmit-Faruk Erden, MD; Hüsnü Efendi, MD. Goztepe Training and Research Hospital, Istanbul- Faik Altıntaş, MD; Namık K. Özkan, MD. Gaziantep University School of Medicine, Şahinbey Hospital, Gaziantep- Celalettin Camcı, MD; Cuma Yildirim, MD. IMM Eşrefpaşa Hospital, Izmir- Hülya Güven, MD; Ali Akın, MD. Vezirköprü State Hospital, Samsun- Hikmet Tereci, MD; Ayşegül Demir, MD. Erciyes University School of Medicine Kemal Dedeman Oncology Hospital, Kayseri- Leylagül Kaynar, MD; Sedat Büyükkeklik, MD. Hatay Hospital, Hatay-Neşat Bastacı, MD; Halim Karaoğlan, MD. Anadolu Çinar Hospital, Istanbul- Nazmi Demirtaş, MD.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The study is granted by sanofi-aventis Türkiye. Subanalysis of the ENDORSE study, authorized by Center for Outcomes Research, University of Massachusetts Medical School, was sponsored by an unrestricted educational grant from sanofi aventis.
