Abstract
Venous thromboembolism (VTE) is a common complication of cancer and chemotherapy. We evaluated the baseline clinical characteristics, thromboprophylaxis patterns, frequency and timing of VTE, and clinical outcomes in 1000 adult hospitalized patients with active cancer. Overall, symptomatic VTE occurred in 5.4% of hospitalized patients with cancer. The VTE occurred in 2.3% of patients with cancer during hospitalization and in 3.4% between hospital discharge and day 90. Few (13.9%) hospitalized patients with cancer received extended duration pharmacological prophylaxis after hospital discharge. Cancer was the most frequent known cause of death in both the groups. In conclusion, VTE was common in hospitalized patients with cancer, especially after discharge. Inhospital death and death between discharge and day 90 were frequent in hospitalized patients with cancer who developed VTE.
Venous thromboembolism (VTE) is a common complication of cancer and chemotherapy. 1 –6 The frequency of symptomatic VTE in patients with malignancy during hospitalization ranges from 2% to 4% and remains high, especially in the first month after cancer diagnosis. 2,5,7 Although the risk of VTE is considered highest among patients with solid tumors, hematologic malignancies also carry substantial risk. 7 Validated risk stratification tools are available to identify patients with cancer at particularly high risk of VTE. 8
Patients with active cancer who subsequently develop VTE have a higher risk of recurrent deep vein thrombosis or pulmonary embolism despite therapeutic anticoagulation than those without malignancy. 9 Major bleeding complicates antithrombotic therapy more frequently in patients with VTE and active cancer. 9,10 Fatal pulmonary embolism contributes, in part, to a high 30-day mortality in patients with active cancer who subsequently had VTE. 11
We created a registry of 1000 patients with active cancer hospitalized in 2009, and we examined baseline clinical characteristics, thromboprophylaxis patterns, frequency and timing of VTE, and clinical outcomes. We also compared the baseline clinical characteristics, thromboprophylaxis patterns, and clinical outcomes of patients with active cancer who developed VTE versus those who did not.
Methods
Patient Population
Brigham and Women’s Hospital is a 793-bed acute tertiary care facility providing medical and surgical care for adult patients with general medical, cardiothoracic, orthopedic, oncologic, neurologic, obstetric and gynecologic, neonatal, urologic, pulmonary, and gastrointestinal conditions. Brigham and Women’s Hospital provides inpatient services for patients receiving oncologic care through the Dana Farber Cancer Institute. Both institutions utilize a medical informatics system that integrates a longitudinal electronic health record, a computerized provider order entry, and an electronic medication administration record.
Identification of Study Subjects
Utilizing the Brigham and Women’s Hospital medical informatics system, we generated a list of all patients who were hospitalized during fiscal year 2009 and carried certain diagnosis-related group (DRG) codes for cancer or cancer-related treatments. The DRG codes used in the identification of patients with cancer and cancer treatments were bone marrow transplant 9; craniotomy 2; nervous system 54, 55; ear, nose, mouth, and throat 146, 147, 148; respiratory 180, 181, 182; digestive 374, 375, 376; hepatobiliary system or pancreas 435, 436, 437; pathological fractures, musculoskeletal, and connective tissue 542, 543, 544; breast 582, 583, 597, 598, 599; kidney and urinary tract 656, 657, 658, 686, 687, 688; male reproductive system 715, 716, 722, 723, 724; female reproductive system 736, 737, 738, 739, 740, 741, 754, 755, 756; major hematologic or immunologic diagnosis 808, 809, 810; lymphoma and nonacute leukemia 820, 821, 822, 823, 824, 825, 840, 841, 842; myeloproliferative disorder or poorly differentiated neoplasm 826, 827, 828, 829, 830; acute leukemia 834, 835, 836, 837, 838, 839, 843, 844, 845; chemotherapy 846, 847, 848; and radiotherapy 849.
Potential study patients were required to be at least 18 years of age and have active cancer with admission for a cancer-related condition. Patients with active cancer were required to have had cancer-directed therapy within the prior 5 years, to be receiving palliative care, or to be refusing further cancer treatment. Patients who did not have available 90-day follow-up data were excluded. From a generated list of 4178 admissions, 1000 hospitalizations representing 1000 unique patients who met study inclusion criteria were selected at random for inclusion in the registry. For patients with active cancer with multiple hospital admissions during fiscal year 2009, only the first hospitalization was entered into the registry.
This study was approved by the Partners Human Research Committee.
Data Collection
Data were collected using REDCap, a secure, Web-based data management application (http://project-redcap.org). Patient demographics and clinical baseline characteristics were recorded, including age, gender, race, length of stay, body mass index (BMI), and comorbid conditions. A VTE risk score was calculated for each patient using a previously established risk scoring system. 12,13 Ischemic heart disease was defined as having one or more of the following: stable or unstable angina, coronary artery disease, acute coronary syndrome, myocardial infarction, coronary artery bypass surgery, or percutaneous coronary intervention. Immobilization was defined as an active order for bed rest that was not related to surgery. Cancer type was classified as breast, brain, melanoma, nonmelanoma skin cancer, osteosarcoma, lung, mesothelioma, gastrointestinal, gynecologic/urologic, hematologic, or other. Cancer treatment history and presence and location of metastatic disease were also recorded.
Data detailing VTE prophylaxis and treatment were obtained from the longitudinal medical record, computerized provider order entry system, and electronic medication administration record. Clinical outcomes of symptomatic VTE, all-cause mortality, and major bleeding were obtained from all patients using our computerized longitudinal medical record that captures any patient encounter across 6 affiliated hospitals, 17 ambulatory clinical, and numerous private practices. Patient encounters were recorded in the form of discharge summaries, office notes, diagnostic testing reports, medical treatment summaries, and procedure reports, regardless of the facility or office location. The social security death index was used to identify patients who died during the 90-day follow-up period. Mortality outcomes were confirmed using the Center for Disease Control National Death Index. Cause of death was reported according to a single International Classification of Diseases and Related Health Problems 10th Revision code. Ninety-day follow-up was completed for 100% of study patients.
Symptomatic deep vein thrombosis was confirmed by loss of compressibility on venous ultrasonography or evidence of a filling defect on invasive contrast venography. Symptomatic pulmonary embolism was confirmed using contrast-enhanced chest computed tomography, ventilation/perfusion lung scanning, or invasive pulmonary angiography. Asymptomatic VTE was excluded. Causes of death were categorized as due to cardiovascular disease, cancer, VTE, or other disease process. Bleeding events were classified according to the Global Use of Strategies to Open Occluded Coronary Arteries criteria as severe or life threatening, moderate, or mild. 14 Major bleeding was defined as intracranial, intraocular, retroperitoneal, or pericardial bleeding or bleeding that required surgical intervention or clinically overt bleeding that resulted in a hemoglobin decrease in excess of 3 g/dL.
Statistical Methods
Descriptive statistics including baseline characteristics, variables related to thromboprophylaxis and VTE treatment, and those pertaining to outcomes were stratified as continuous or binary. Continuous variables were assessed for normality and presented as means with standard deviations or medians with interquartile ranges. Binary variables were presented as numbers and proportions. Continuous variables were compared using a 2-sample t test or Wilcoxon rank-sum test. Binary variables were compared using chi-square test or Fisher exact test. All reported P values were 2 sided, and a P value of <.05 was considered statistically significant. All statistical analyses were performed using STATA version 9.2 (STATA Corp, College Station, Texas).
Results
Baseline Characteristics and Comorbid Conditions
The median age of hospitalized patients with cancer was 61 years (Table 1). The mean BMI was 27.6 kg/m2. The median length of hospital stay was 4 days. Hospitalized patients with cancer had a high frequency of prior hospitalization within 30 days (17.3%) and comorbid conditions that increased the risk of VTE, such as acute infectious illness (16.9%), ischemic heart disease (12.2%), and chronic kidney disease (11.5%). Common VTE risk factors included prior VTE (11.8%), surgery or trauma within the 30 days prior to admission (36.3%), age greater than 70 years (24.3%), and obesity (28.7%).
Baseline Characteristics and Comorbid Conditions.

Abbreviations: SD, standard deviation; IQR, interquartile range.
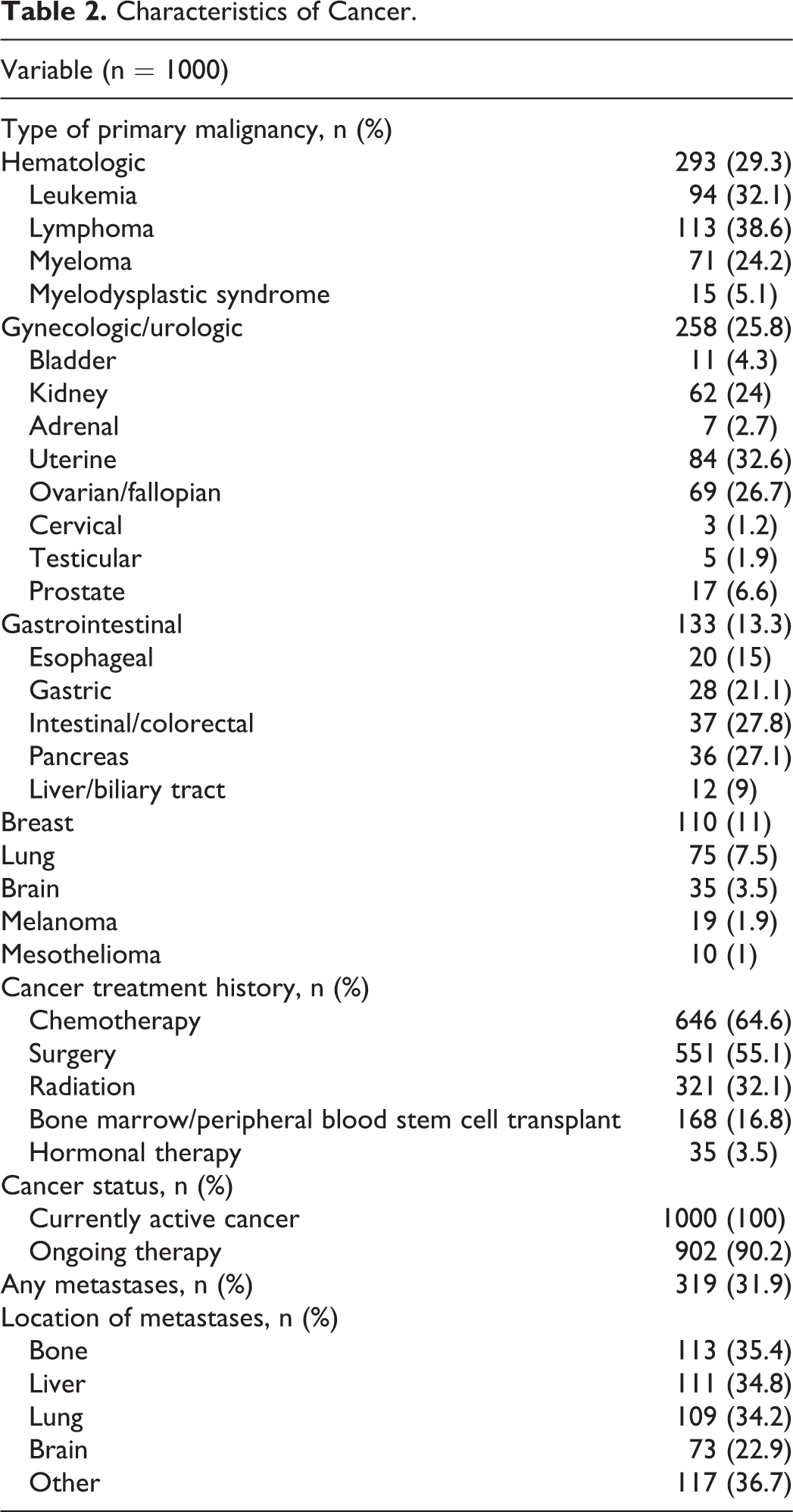
Characteristics of Cancer
The most common malignancies were hematologic (29.3%), gynecologic/urologic (25.8%), gastrointestinal (13.3%), and breast (11%; Table 2). Metastases were present in 32% of the patients. Chemotherapy (64.6%) was the most common treatment for cancer, followed by surgery (55.1%) and radiation therapy (32.1%). Ninety percent of patients in the study were actively receiving some form of cancer treatment.
Characteristics of Cancer.
The VTE Prophylaxis
Inhospital VTE prophylaxis was prescribed in 86.8% (Table 3). Mechanical and pharmacological prophylactic measures were provided in 78.8% and 69.8%, respectively. Combination mechanical and pharmacological prophylaxis was provided to 48.6% of hospitalized patients with cancer. Low-molecular-weight heparin (49.5%) and subcutaneously administered unfractionated heparin (48.7%) were the most frequently prescribed forms of prophylactic anticoagulation. A small proportion (13.9%) of hospitalized patients with cancer received extended duration pharmacological prophylaxis after hospital discharge.
Venous Thromboembolism Prophylaxis History.a

Abbreviations: GUSTO, Global Use of Strategies to Open Occluded Coronary Arteries; IV, intravenous; IQR, interquartile range; SC, subcutaneous.
aPatients could receive more than one type of thromboprophylaxis.
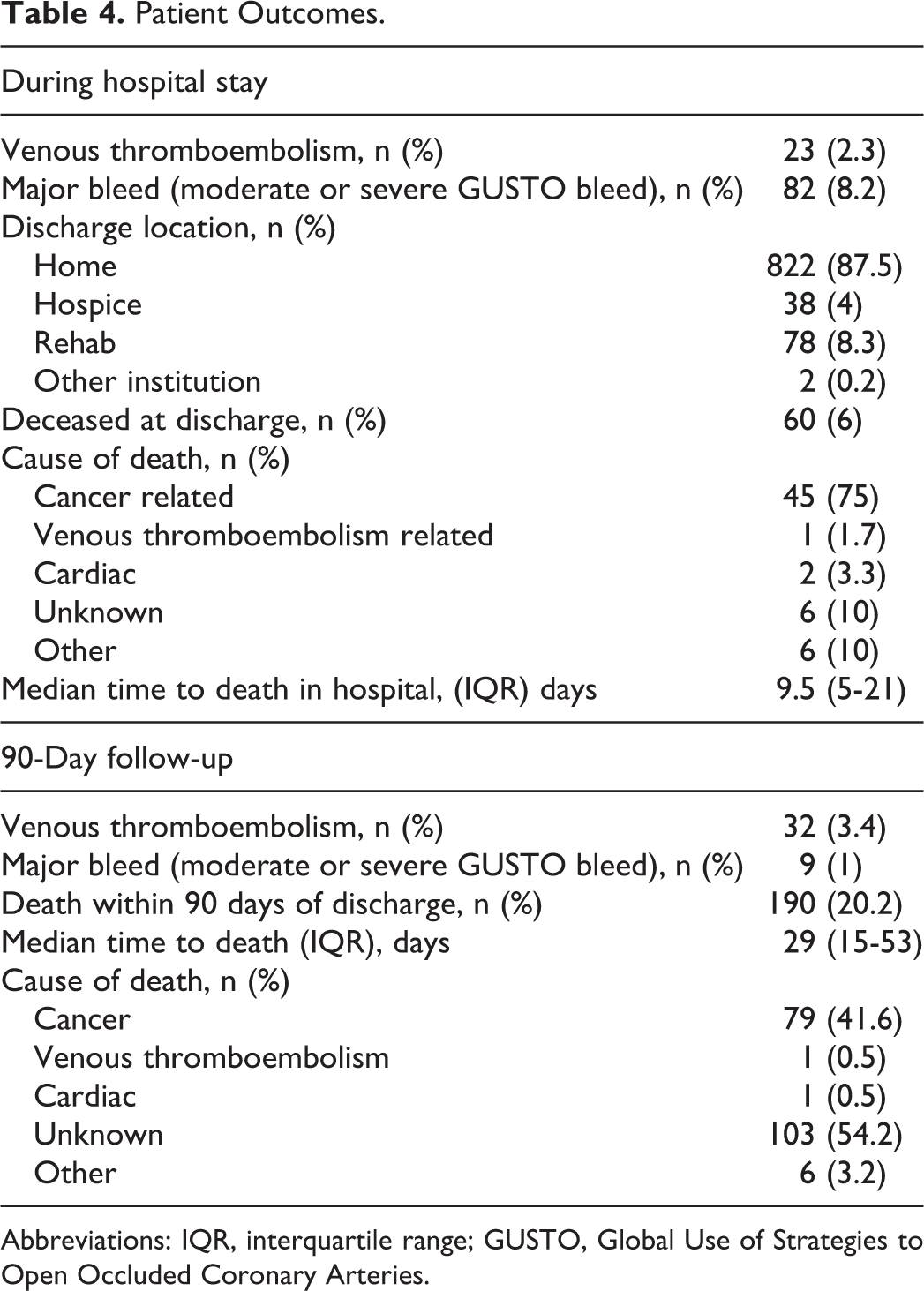
Patient Outcomes
Venous thromboembolism occurred in 2.3% of patients with cancer during hospitalization and in 3.4% between hospital discharge and day 90 (Table 4). One patient who developed VTE as an inpatient had a recurrent venous thromboembolic event between hospital discharge and day 90. The frequency of VTE during hospitalization did not differ according to the type of cancer. The VTE at day 90 was more common in patients with cancer of the brain, lung, gastrointestinal tract, gynecological organs or genitourinary tract, and those with hematological malignancy. The frequency of VTE neither during hospitalization nor at 90 days was different in patients receiving ongoing cancer therapy compared with those receiving palliative care or refusing further cancer treatments.
Patient Outcomes.
Abbreviations: IQR, interquartile range; GUSTO, Global Use of Strategies to Open Occluded Coronary Arteries.
Major bleeding was more common during hospitalization (8%) than between hospital discharge and day 90 (1%). In all, 6% of the patients with cancer died during hospitalization, while 20% of the patients died during the interval from hospital discharge to day 90. Cancer was the most frequent known cause of death during hospitalization and after discharge.
The VTE Characteristics and Treatment
Overall, symptomatic VTE occurred in 5.4% of hospitalized patients with cancer (Table 5). Deep vein thrombosis and pulmonary embolism were diagnosed in 3.5% and 1.6% of hospitalized patients with cancer, respectively. Deep vein thrombosis with pulmonary embolism occurred in 0.3%. Both lower (57.9%) and upper extremity deep vein thrombosis (36.8%) were common among hospitalized patients with cancer. The most common strategy for the treatment of VTE was low-molecular-weight heparin monotherapy without warfarin (88.1%).
Venous Thromboembolism Characteristics and Treatment.

Abbreviations: IV, intravenous; IQR, interquartile range; GUSTO, Global Use of Strategies to Open Occluded Coronary Arteries.
Patients With Cancer having VTE
Hospitalized patients with cancer who subsequently developed VTE inhospital or after discharge were more likely to have been hospitalized within the prior 30 days (27.8% vs 16.7%, P = .04), had a higher frequency of prior VTE (22.2% vs 11.2%, P = .02), and had a longer median length of hospital stay (7 days vs 4 days, P = .006). There was no difference in the rate of inpatient thromboprophylaxis between hospitalized patients with cancer who subsequently developed VTE and those who did not (90.7% vs 86.6%, P = .53). Low-molecular-weight heparins were more frequently prescribed among hospitalized patients with cancer who subsequently developed VTE (76.3% vs 47.7%, P = .001). Patients with cancer who subsequently developed VTE were more likely to have received pharmacological prophylaxis upon hospital discharge (24.1% vs 13.3%, P = .04).
Death between hospital discharge and day 90 was more common among patients with cancer who subsequently developed VTE than among those who did not (31.5% vs 18.4%, P = .03).
Discussion
Venous thromboembolism occurred in 5.4% of hospitalized patients with active cancer over a 90-day follow-up period. The majority of hospitalized patients with cancer in our registry who developed VTE did so as outpatients. They were twice as likely to have been hospitalized within the prior 30 days and to have had a prior venous thromboembolic event compared with those who did not have VTE.
Few (13.9%) patients in our study received extended duration prophylaxis after hospital discharge. Although extended duration thromboprophylaxis has not been shown to provide a net clinical benefit in the general medical population, 15 –17 outpatient pharmacological thromboprophylaxis for patients with cancer receiving chemotherapy reduces VTE without increasing the risk of major bleeding. 18 –20 A systematic review of trials evaluating primary prophylaxis for VTE in ambulatory patients with cancer receiving chemotherapy demonstrated a 38% reduction in the risk of symptomatic VTE (number needed to prevent a symptomatic VTE = 60) without significantly increasing the risk of major bleeding. 20 However, current guidelines do not endorse routine prescription of out-of-hospital thromboprophylaxis for patients with cancer. 21,22 The most recent guidelines from the National Comprehensive Cancer Network call for the assessment of VTE risk in ambulatory patients with cancer and randomized controlled trials of thromboprophylaxis in those at high risk of VTE and low risk of bleeding. 23
Paradoxically, patients with cancer who were prescribed thromboprophylaxis after hospital discharge were more likely to develop VTE in the outpatient setting. We speculate that some may have already developed asymptomatic VTE before or during hospitalization. If this were the case, prophylactic doses of anticoagulation would not be effective. Alternatively, patients with cancer who were prescribed thromboprophylaxis to continue after discharge may have had a greater medical acuity contributing to a higher risk of VTE in the outpatient setting. These findings support those of a previous case–control study of predominantly patients with cancer in which we observed a trend toward a higher rate of VTE in patients receiving extended duration thromboprophylaxis compared with those who did not (5.0% vs 4.3%, P = .03). 24
Use of low-molecular-weight heparins may help explain the low frequency of VTE-related death. 25 –28 Nevertheless, venous thromboembolic events represent an important source of morbidity 29,30 and health care costs 31 in patients with active cancer.
Our findings must be interpreted in the context of the study design. The population in the registry was predominantly white, thereby limiting generalizability. Because of the observational nature of the study, we cannot account for reasons why clinicians elected to prescribe a certain thromboprophylactic or therapeutic regimen. As with any registry, unrecognized confounders may have been present and may account for the observed differences in thromboprophylaxis, treatment, and patient outcomes. The registry did not record information regarding the presence of indwelling central venous catheters. Some deaths due to cancer, VTE, or cardiac disease may have gone undetected. Finally, the frequency of VTE may have been underestimated, because only the initial hospitalization was included in the analysis.
We present data from a large registry analysis evaluating the clinical characteristics, prophylaxis patterns, treatment patterns, and outcomes of hospitalized patients with active cancer. Our study not only corroborates findings from prior registry analyses regarding VTE incidence in patients with cancer but also provides additional insights regarding the onset of symptomatic VTE. The methodology used in this analysis is consistent with published criteria for evaluating the scientific value of clinical data registries. 32 Our electronic health record allowed for the longitudinal follow-up of large cohort of patients with active cancer.
Conclusions
Venous thromboembolism was a common complication of hospitalization in patients with active cancer, especially after hospital discharge. However, pharmacologic thromboprophylaxis after hospital discharge was prescribed to a minority of patients with cancer.
Footnotes
Authors’ Contribution
All of the authors had access to the data and participated in the writing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Goldhaber receives clinical research grant support from Eisai. This study was funded, in part, from a clinical research grant from Eisai. Dr Goldhaber (principal investigator), Dr Piazza (co-investigator), and Thanh Nha Nguyen (research pharmacist) were partially compensated with these funds.
