Abstract
Plasma-soluble platelet glycoprotein VI (sGPVI) levels were examined in patients undergoing living donor liver transplantation (LDLT), and the relationship between platelet activation and thrombocytopenia was evaluated to understand the mechanism of thrombocytopenia in LDLT. Platelet counts were significantly higher in the donors compared to the recipient, and the plasma sGPVI levels increased in both groups after the operation. Regarding the relationship between the platelet counts and the sGPVI levels, the slope varied on different days, and it became negative on day 3, suggesting that the plasma sGPVI levels are related to platelet activation in LDLT. The frequency of complications was high in the nonsurvivors. The platelet counts were higher in the survivors than in the nonsurvivors on days 14 and 28. Although the plasma levels of sGPVI in the survivors increased after the operation, those in the nonsurvivors were high only on day 3. Although the ADAMTS13 (a disintegrin and metalloproteinase with a thrombospondin type 1 domain, member 13) levels were markedly reduced, von Willebrand factor (VWF) and VWF propeptide (VWFpp) were markedly elevated during LDLT. The antithrombin activity was significantly lower (day 14) and VWFpp (day 28) was significantly higher in the nonsurvivors than in the survivors. These findings suggest that platelet activation first occurs after LDLT, and it is high in the nonsurvivors on day 3. Thereafter, the hemostatic abnormality and vascular endothelial cell injuries may appear on days 14 and 28.
Introduction
Living donor liver transplantation (LDLT) was first performed in Japan in 1989, 1 and unique technical, physiological, and logistical innovations in LDLT 2,3 have developed since then. Technical improvements in living donor surgery have led to the generalization of pediatric LDLT with excellent patient and graft survival outcomes. Despite the application of preoperative plasma exchange, splenectomy, and enhanced immunosuppression, the 5-year graft survival rate is less than 70% in ABO-incompatible LDLT in children. 4,5 Specific diseases and preoperative patient conditions are associated with the transplantation outcomes. 6 –8 In the registry of the Japanese Liver Transplantation Society between November 1989 and December 2010, the 1-, 5-, 10-, and 20-year patient survival rates were 88.3%, 85.4%, 82.8%, and 79.6%, respectively. 9 Nationwide surveys of acute liver failure (ALF) are conducted annually in Japan, and 20% of patients with ALF undergo liver transplantation (LT). 10 In LDLT for patients with ALF, the cumulative patient survival rate at 1 year after LT was 79%. 10
Transient thrombocytopenia is a common phenomenon after LT, and the recovery of platelet counts is clinically significant. In 1992, McCaughan et al 11 revealed the nadir platelet counts after LT to predict allograft dysfunction. Following this report, 3 additional studies 12 –14 confirmed that severe thrombocytopenia after LT was associated with graft and patient survival. However, the precise mechanisms of posttransplant thrombocytopenia and its relationship with graft dysfunction still remain unclear. The mechanisms contributing to graft dysfunction are multifactorial and include ischemic reperfusion injury, sinusoidal endothelial cells injury, platelet aggregation, immunological reactions, and inflammatory responses. 15,16
Platelet glycoprotein VI (GPVI), a type I transmembrane glycoprotein of the immunoreceptor family, is constitutively associated and expressed with the Fc receptor γ-chain, an immunoreceptor tyrosine-based activation motif-bearing receptor. 17,18 Upon platelet activation, the platelet surface GPVI is cleaved off by proteases, such as ADAM10 (a Disintegrin and metalloproteinase domain-containing protein 10), releasing the soluble form of GPVI (sGPVI). 19,20 The soluble form of GPVI has recently received much attention as a platelet activation marker, as described subsequently. Several groups have reported that sGPVI is a useful biomarker of diseases caused by platelet activation, such as acute coronary syndrome and stroke. 21,22 C-type, lectin-like receptor 2 is a transmembrane glycoprotein similar to GPVI that has been reported to be a potential thrombotic marker. 23
A disintegrin and metalloprotease with thrombospondin type I domain, member 13 (ADAMTS13) is a metalloprotease that specifically cleaves the multimeric von Willebrand factor (VWF). 24 The pre–pro-VWF is synthesized in endothelial cells and megakaryocytes, and the VWF propeptide (VWFpp) is cleaved but remains stored together with mature VWF in α-granules (megakaryocytes) and Weibel-Palade bodies (endothelial cells). After the secretion of VWFpp and VWF into the plasma from endothelial cells induced by physiological or pathological stimuli, VWFpp dissociates from VWF. 25 The behaviors of ADAMTS13, VWF, and VWFpp have been reported previously in LDLT patients with thrombotic microangiopathy (TMA), 26,27
In this study, the sGPVI levels were measured in 79 patients undergoing LDLT, and the relationship between the sGPVI levels and thrombocytopenia was examined to evaluate the mechanism of thrombocytopenia in LDLT patients with a poor outcome.
Materials and Methods
The plasma levels of sGPVI and platelet counts were determined in 79 recipients (34 females and 45 males) and 12 donors during LDLT from January 1, 2002, to March 31, 2014. The patients who provided consent were continuously included in this study, but the patients with unavailable plasma were excluded. The underlying diseases necessitating LDLT were liver cirrhosis (LC) without hepatic cell carcinoma (HCC) in 40 cases, LC with HCC in 23 cases, HCC without LC in 3 cases, and other diseases in 13 cases (ie, 6 cases of biliary atresia, 4 cases of acute hepatitis, 2 cases of primary sclerosing cholangitis, and 1 case of Alagille syndrome). The causes of LC were hepatitis C virus infection in 23 cases, hepatitis B virus infection in 12 cases, non-B and non-C hepatitis in 11 cases, primary biliary cirrhosis in 9 cases, alcoholic disturbances in 4 cases, progressive intrahepatic cholestasis in 3 cases, and secondary biliary cirrhosis in 1 case. The mortality rate was evaluated on day 90 after surgery. 13 The study protocol was approved by the Human Ethics Review Committee of Mie University School of Medicine, and written informed consent was obtained from each participant. The immunosuppression protocol consisted of tacrolimus and low-dose steroids. The target trough level for tacrolimus in whole blood was 10 to 15 ng/mL during the first 2 weeks, approximately 10 ng/mL during the next 2 weeks, and 5 to 10 ng/mL thereafter. Methylprednisolone (10 mg/kg body weight) was administered intravenously immediately before perfusion of the graft portal vein. Methylprednisolone (1 mg/kg) was given intravenously daily on postoperative days (PODs) 1 to 3, and a dose of 0.5 mg/kg was given daily on PODs 4 to 6. On POD 7, the steroid was switched to oral prednisolone (0.5 mg/kg) daily, and at 1 month, the dose was reduced to 0.1 mg/kg. Patients whose liver function was stable were weaned off the steroid approximately 3 to 6 months postoperatively.
The Child-Pugh (CP) score, prothrombin time–international normalized ratio (PT-INR), total bilirubin, albumin, graft-to-recipient weight ratio (GRWR), platelet counts, and plasma sGPVI levels were measured in the patients undergoing LDLT.
The plasma level of sGPVI was quantified using a sandwich enzyme-linked immunosorbent assay, which consisted of 2 mouse anti-GPVI monoclonal antibodies, F1232-7-1 and F1232-10-2 able to recognize the extracellular domain 1 (D1) N-terminal loop and extracellular domain D2 loop of GPVI, respectively. 28 –30
The platelet counts were measured using the fully automated hematology analyzer XE-2100 (Sysmex, Kobe, Japan). The PT-INR was measured using a Thromborel S (Sysmex) by the fully automated hematology analyzer CA-7000 (Sysmex). The ADAMTS13 was measured using FRETS-VWF73, which was chemically synthesized by the Peptide Institute, Inc (Osaka, Japan) according to the method of Kokame et al.
31
The VWF and VWFpp levels were measured with a VWF & Propeptide Assay kit (GTI Diagnostics, Waukesha, Wisconsin).
27
The AT activity was measured by a chromogenic assay, and
The behaviors of biomarkers such as sGPVI and platelet counts in the recipients were first compared with those in the donors to examine the relationship between the change in the biomarkers and the stress of the operation. Markers including VWFpp and ADAMTS13 in the nonsurvivor were subsequently compared with those in the survivors.
Statistical Analysis
The data are expressed as the median and 25th to 75th percentiles. Differences between groups were examined for significance using the Wilcoxon test, and analysis of variance (ANOVA) was also performed. A P value of less than .05 was considered to indicate a significant difference. All statistical analyses were performed using the Stat flex, version 6, software package (Artec Co Ltd, Osaka, Japan).
Results
Twelve patients died within 90 days of transplantation (nonsurvivors) and 67 patients survived over 90 days (survivors; Table 1). The causes of death were liver failure in 6 cases, sepsis in 2 cases, gastrointestinal bleeding in 1 case, cardiac failure in 2 cases, and cerebral hemorrhage in 1 case. There were no significant differences in age, sex, LC, HCC, PT-INR, total bilirubin, albumin, and GRWR between survivors and nonsurvivors. The CP score was higher in the nonsurvivors than in the survivors (P < .01). The incidence of complications, especially bleeding, was significantly higher in the nonsurvivors than in the survivors (P < .01; Table 2).
Survivors and Nonsurvivors With LDLT.
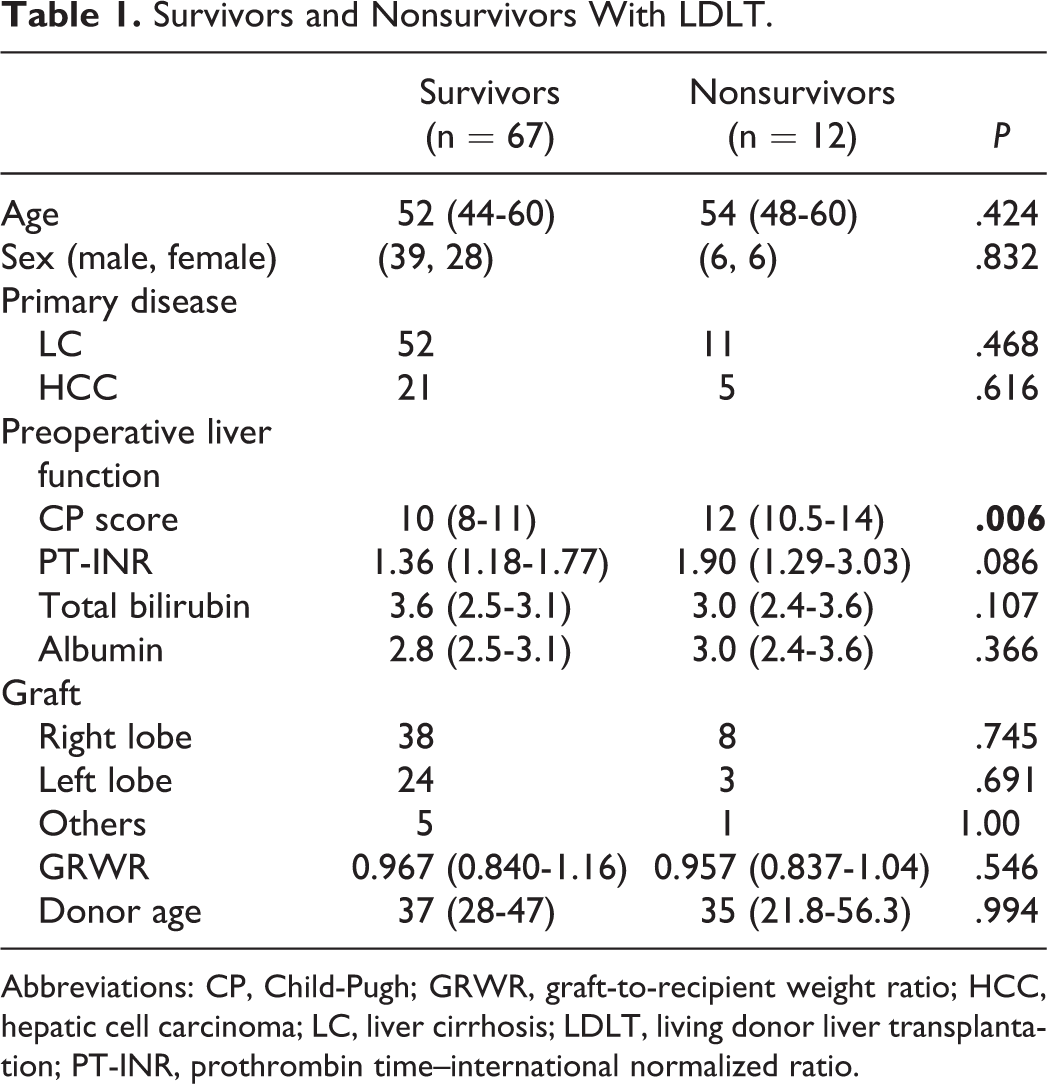
Abbreviations: CP, Child-Pugh; GRWR, graft-to-recipient weight ratio; HCC, hepatic cell carcinoma; LC, liver cirrhosis; LDLT, living donor liver transplantation; PT-INR, prothrombin time–international normalized ratio.
Postoperative Complications.

In comparison between donor with mild stress by LDLT and recipient who had marked stress by LDLT, the platelet counts in the donors were significantly decreased on day 3 (P < .05), but they were increased on day 14 (P < .001). They were significantly lower in the recipients than in the donors on days 1 (P < .001), 3 (P < .001), 7 (P < .001), and 14 (P < .05; Figure 1A). The platelet counts in the recipients were significantly decreased on day 3 (P < .05) and increased on day 14 (P < .05). The plasma levels of sGPVI in the donors were significantly higher on days 1 (P < .01), 3 (P < .001), 7 (P < .01), and 14 (P < .001) compared to the levels before the operation, but those in the recipient were significantly higher on days 1, 3, and 14 (P < .05, respectively) compared to the values before the operation (Figure 1B). The plasma levels of GPVI were significantly higher in the recipients than in the donors on day 14 (P < .05). According to the ANOVA, the F value was 8.83 (P < .001) in platelet counts and 4.08 (P < .01) in sGPVI. In terms of the relationship between the platelet counts and the sGPVI levels, the slope varied on different days and it became negative on day 3 (Figure 2 and Table 3).
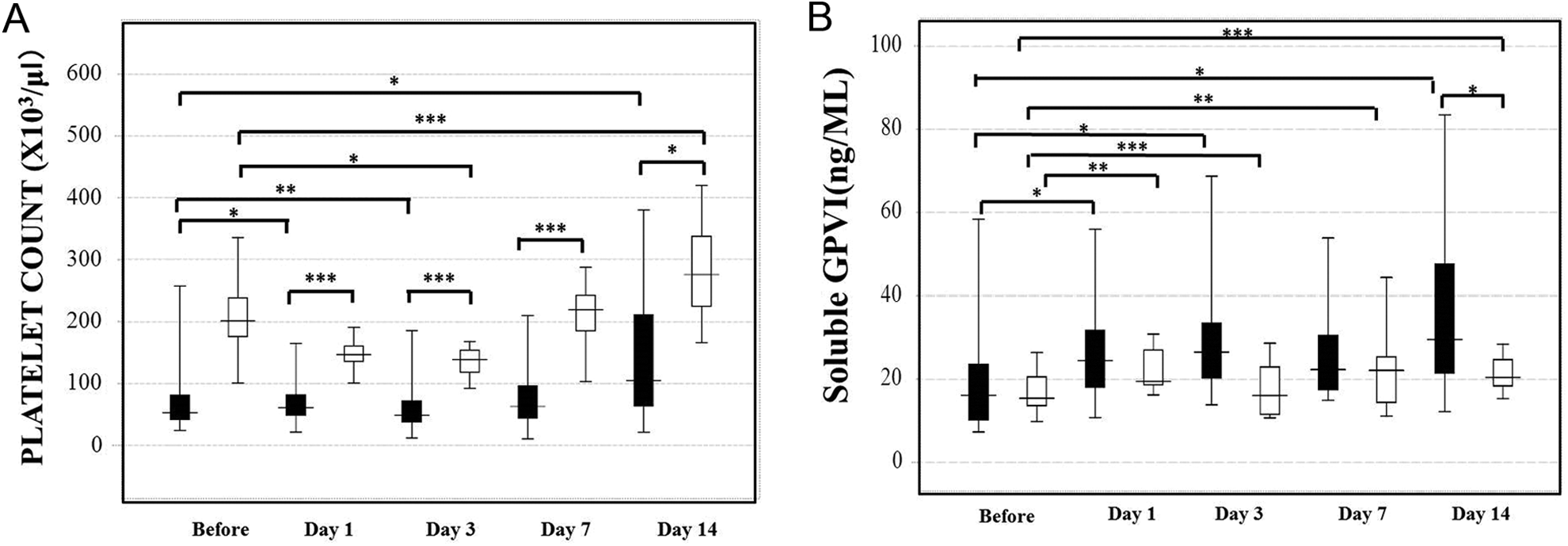
Platelet counts (A) and soluble platelet glycoprotein VI (sGPVI) levels (B) in the donors and recipients for living donor liver transplantation (LDLT). Closed bar: recipient, open bar: donor. *P < .05, **P < .01, ***P < .001.
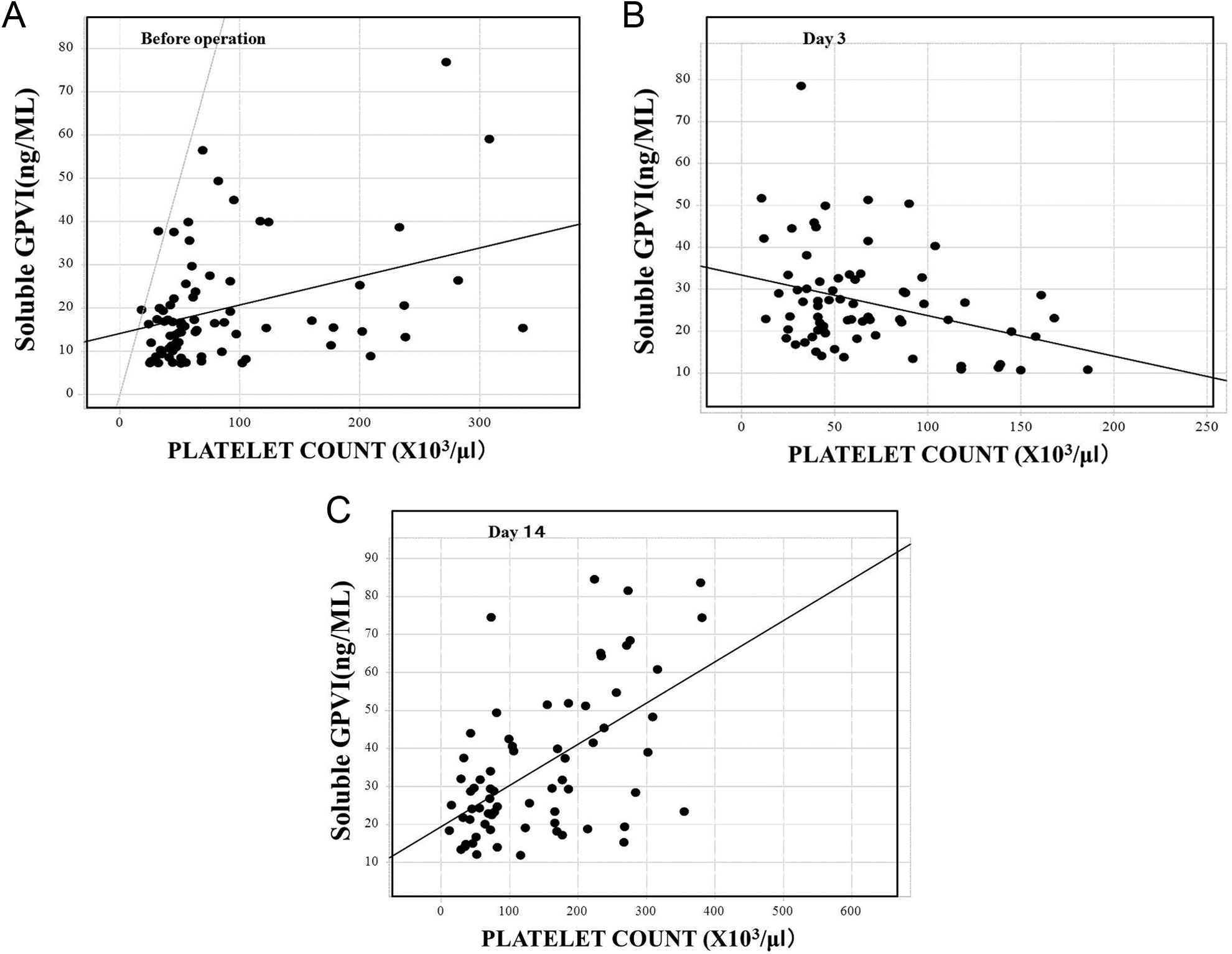
Relationship between the soluble platelet glycoprotein VI (sGPVI) levels and platelet counts in the recipients for living donor liver transplantation (LDLT). Before the operation (A), day 3 (B), and day 14 (C).
Relationship Between the Platelet Count and sGPVI Levels.
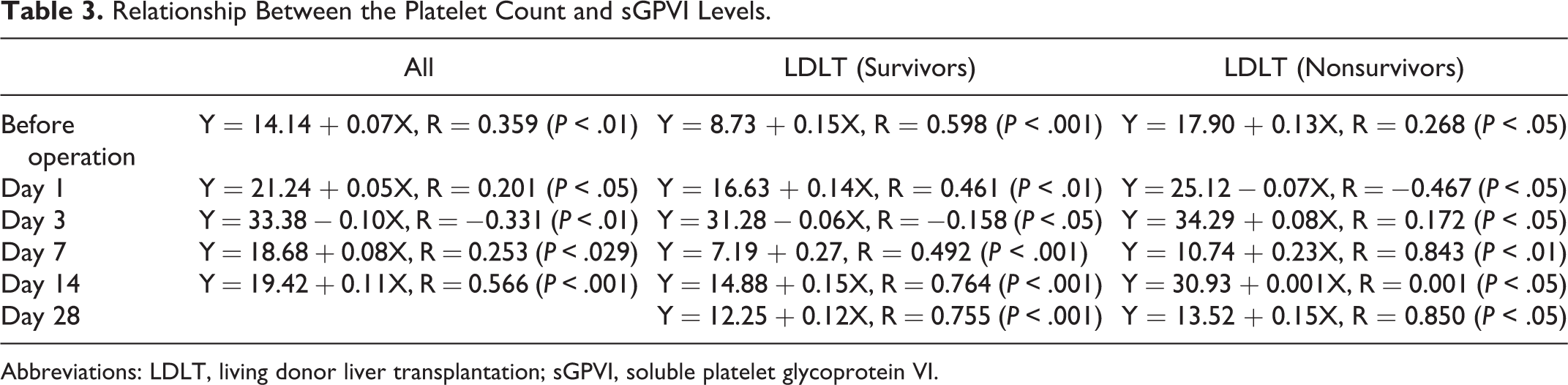
Abbreviations: LDLT, living donor liver transplantation; sGPVI, soluble platelet glycoprotein VI.
The platelet counts significantly increased in survivors on days 14 and 28 compared to values before the operation (P < .001). They were significantly higher in the survivors than in the nonsurvivors on days 14 (123 × 103/μL: 72.0-223.5 × 103/μL vs 56.0 × 103/μL: 43.5-73.8 x 103/μL; P < .001) and 28 (131.0 × 103/μL: 68.0-194.0 × 103/μL vs 60.5 × 103/μL: 41.5-118.5 × 103/μL; P < .05), although there were no differences in the platelet counts between the groups before the operation and on days 1, 3, and 7 (Figure 3A). The plasma levels of sGPVI in the survivors were significantly higher on days 1, 3, 7, 14, and 28 (P < .001) compared to values before the operation (14.7 ng/mL: 9.7-24.7 ng/mL), and those in the nonsurvivors were significantly higher (P < .05) on day 3 compared to values before the operation. The plasma levels of sGPVI on day 3 were significantly higher (P < .01) in the nonsurvivors (38.1 ng/mL: 32.3-44.1 ng/mL) than in the survivors (22.5 ng/mL: 17.3-35.8 ng/mL; Figure 3B). The ratio of sGPVI to platelets of the survivors was significantly higher on days 1 (P < .01), 3 (P < .001), and 7 (P < .01) and significantly lower on day 28 (P < .05) compared to values before the operation. The ratio for nonsurvivors was significantly higher (P < .01) on day 3 (Figure 3C), and it was significantly higher in the nonsurvivors than in the survivors before the operation (P < .05) and on days 3 (P < .01) and 28 (P < .05). In the ANOVA between the survivor and the nonsurvivors, the F values for the platelet counts, plasma levels of sGPVI, and the ratio of sGPVI to platelets were not significant.
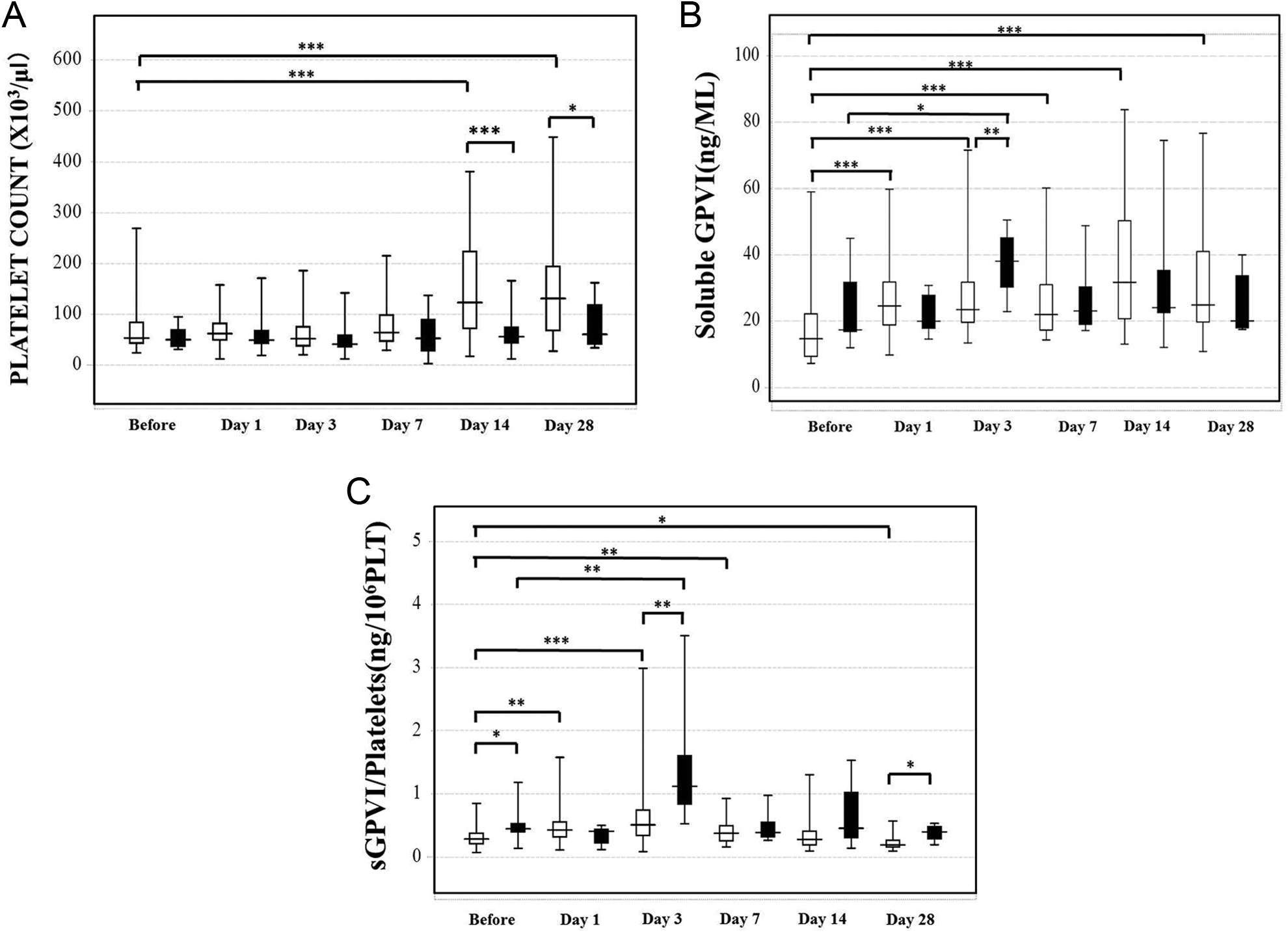
Platelet counts (A), soluble platelet glycoprotein VI (sGPVI) levels (B), and the sGPVI-platelets ratio (C) in the recipients for living donor liver transplantation (LDLT). Open bar: survivors, closed bar: nonsurvivors. *P < .05, **P < .01, ***P < .001.
The plasma levels of ADAMTS13 in patients with LDLT were significantly decreased on days 1, 7, 14, and 28 and were significantly lower in the nonsurvivors than in the survivors on days 7 and 28 (P < .05; Figure 4A). Although the plasma levels of VWF were high in the patients during LDLT, they were significantly decreased in the survivors on day 1 (P < .001; Figure 4B). The plasma levels of VWFpp were markedly higher on day 14 and decreased on day 28 in the survivors (P < .05; Figure 4C), and these levels were significantly higher in the nonsurvivors than in the survivors (P < .01). There were no significant differences in the PT-INR and
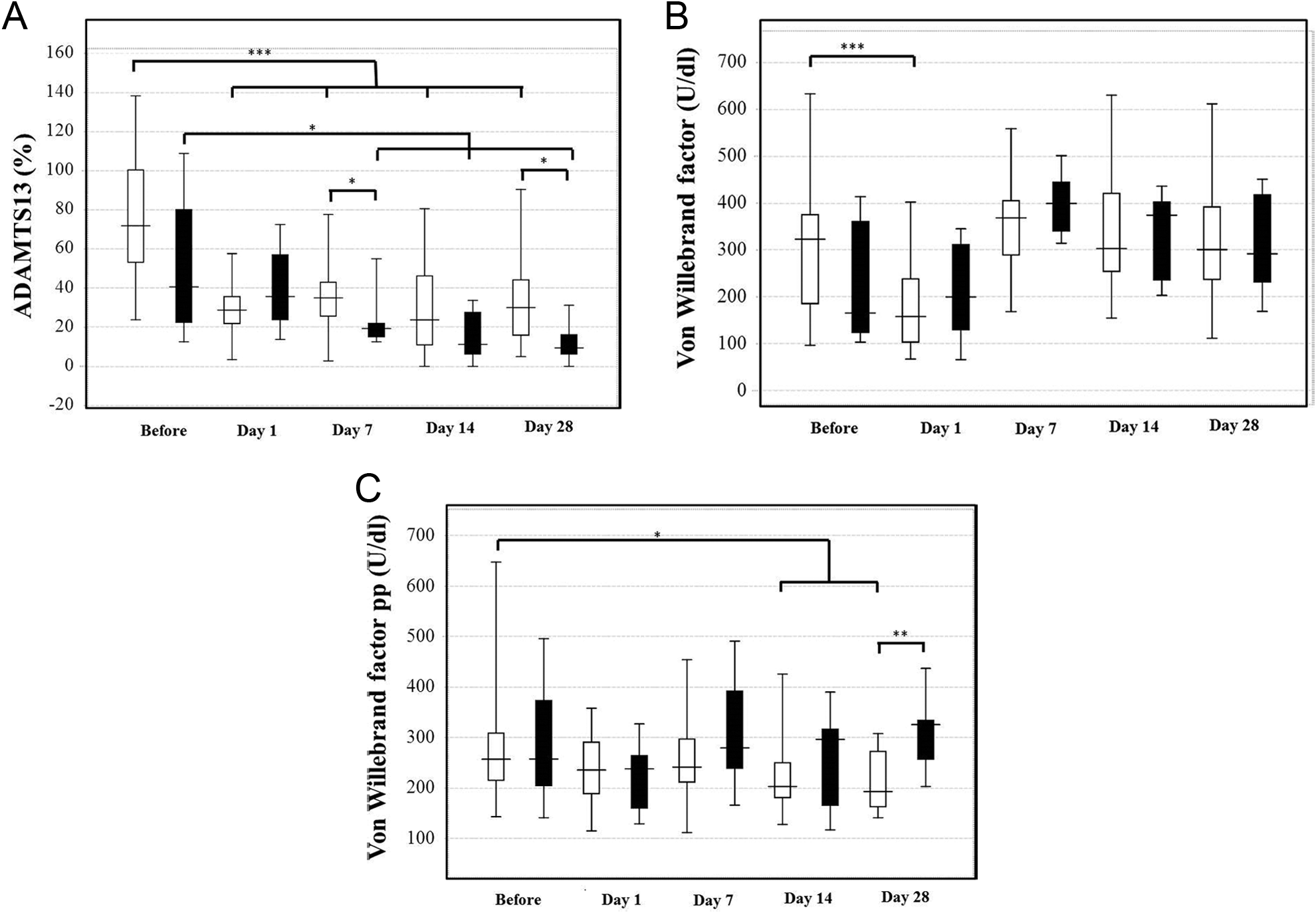
ADAMTS13 (A), von Willebrand factor (VWF) (B), and VWF propeptide (VWFpp) (C) in the recipients for living donor liver transplantation (LDLT). Open bar: survivors, closed bar: nonsurvivors. *P < .05, **P < .01, ***P < .001.
Hemostatic Data in the Survivors and Nonsurvivors With LDLT.
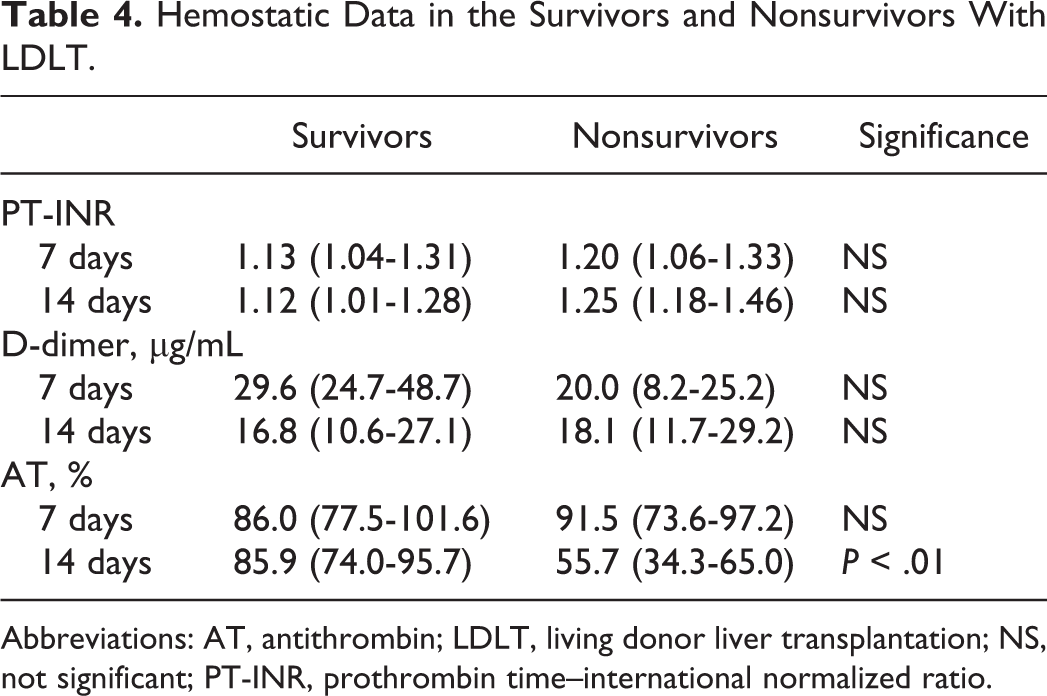
Abbreviations: AT, antithrombin; LDLT, living donor liver transplantation; NS, not significant; PT-INR, prothrombin time–international normalized ratio.
Discussion
The causes of a poor outcome for LDLT are considered to be graft dysfunction and complications such as infection, thrombosis, bleeding, graft-versus-host disease, 33 TMA, 27,34 disseminated intravascular coagulation (DIC), 35 and others. Thrombocytopenia is commonly associated with LDLT and many complications of LDLT, suggesting that it is important to know the degree of platelet activation in LDLT patients with thrombocytopenia. The plasma sGPVI levels have been reported to significantly increase in patients with thrombosis during the postoperative period 36 and those with DIC 35 or TMA, thus suggesting that the plasma sGPVI levels increase in a thrombotic state, which thus activates platelets. 30 In a cynomolgus monkey model of lipopolysaccharide-induced thrombocytopenia, the change in sGPVI was more pronounced than the existing platelet activation biomarker, soluble P-selectin. 37
An ANOVA showed that LDLT had an important effect on the platelet count and the plasma GPVI level in the present study. These levels were not significantly different among the various underlying diseases. It is therefore typically considered in LDLT that the stress and platelet activation by LDLT are higher in the recipients than in the donors. After LDLT, markedly low platelet counts and relatively high sGPVI levels were observed in the recipients, and slight changes were observed in the donors, suggesting that sGPVI levels and platelet counts will be well affected by LDLT and its complication. The low platelet counts and the high sGPVI levels were observed in the recipients on days 1, 3, and 14. In terms of the relationship between the platelet count and the sGPVI levels, the slope became negative on day 3. These findings indicate that the activation of platelets might occur the highest on day 3. In the nonsurvivors, peak platelet activation might occur earlier. These findings suggest that thrombocytopenia in LDLT may be caused by platelet activation and consumption. The ADAMTS13 levels were markedly low after day 1 and the VWF levels were markedly high during LDLT, which increased the platelet aggregations, thereby leading to thrombosis, organ failure, or TMA. There was no significant difference in the PT-INR between the survivors and the nonsurvivors until day 14, suggesting that the liver function remained stable until day 14. The decreased AT activity in the nonsurvivors on day 14 indicates that hemostatic abnormalities occurred on day 14 following elevated PT-INR on day 28. The elevated VWFpp suggested that vascular endothelial injuries occurred on day 28.
The platelet counts were significantly higher in the survivors than in the nonsurvivors on days 14 and 28, similar to those in previous reports, 12,32 indicating that thrombocytopenia on day 14 or 28 may be a good marker for a poor outcome in LDLT. ADAMTS13, 38 VWF, and VWFpp 39 were studied previously and reported to be changed on day 14 as biomarkers for complications and poor outcomes for LDLT. Thrombopoietin levels were also reported to be decreased in LDLT patients with poor outcome on day 28. 39 The incidence of complications was high in the nonsurvivors, suggesting that several complications might have contributed to the observed deaths within 90 days of surgery. The plasma VWFpp level was significantly higher in the nonsurvivors than in the survivors. The diagnosis of liver failure on day 14 or 28 may be too late to improve the outcome in the patients with LDLT. The plasma levels of sGPVI in the nonsurvivors were significantly increased on day 3, and those levels were significantly higher in the nonsurvivors than in the survivors on day 3. Although this study is small and the statistical power is low, our findings suggest that elevated plasma sGPVI levels may be an early marker for poor outcome in LDLT. An increased sGPVI to platelets ratio may therefore be a good marker for platelet activation. Decreased ADAMTS13 and elevated VWF, which are related to the platelet aggregations, were observed on day 7, suggesting that these changes may occur after marked platelet activation and aggregation.
We hypothesize that platelet activation occurs after LDLT and it is high in the nonsurvivors on day 3 and thereafter that the hemostatic abnormality and vascular endothelial cell injuries increased on days 14 and 28.
Footnotes
Authors’ Note
K.N. contributed to obtaining the measurements of sGPVI but was not involved in interpreting the results.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.N. is an employee of Mochida Pharmaceutical Co, Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Grant-in-Aid from the Ministry of Health, Labour and Welfare of Japan for Blood Coagulation Abnormalities, and the Ministry of Education, Culture, Sports, Science and Technology of Japan. The study was funded by Mochida Pharmaceutical Co, Ltd, and the sGPVI ELISA system was provided by the company.
