Abstract
Many genetic risk factors have been identified for causing venous thromboembolism (VTE). Most of them affect the function of natural anticoagulant pathways, particularly the protein C system, although recent studies suggest a role of components of the hematopoietic pathway in the etiology of venous thrombosis. In this case–control study, we aimed to determine the frequency of prothrombin G20210A and factor V Leiden (FVL) G1691A polymorphisms and protein C, protein S, and antithrombin III deficiencies in the East Algerian population and to investigate whether these genetic factors are associated with VTE. On the other hand, our study tends to evaluate the status of JAK2V617F and calreticulin (CALR) mutations among these cases. The participants consisted of 121 cases with VTE and 146 healthy controls. Polymorphisms of FVL G1691A and prothrombin G20210A were genotyped by polymerase chain reaction (PCR) restriction fragment length polymorphism. JAK2-V617F and calreticulin mutations were analyzed by quantitative PCR and PCR followed by capillary electrophoresis sequencing, respectively. Protein C, protein S, and antithrombin levels were determined and then hereditary deficiencies were identified. Of all cases and controls, none was a carrier of the antithrombin III deficiency, prothrombin gene G20210A, and CALR mutations. Only 1 case reported having a positive JAK2 mutation (mutant allele burden was 15%). The FVL mutation (GA/AA) was found in 14 (11.6%) cases and 2 (1.4%) controls and it was significantly different between both the groups (P = .001). Deficiencies of protein S and protein C were detected in 17 (18.8%) cases. The univariate analysis resulted in a significant impact of FVL (odds ratio [OR] = 9.4, 95% confidence interval [CI] = 2.1-42.3; P = .003) and of protein S deficiency (OR = 16.9, 95% CI =2.1-132.8, P = .007) on the VTE status. Both factors stayed significant after adjustment for sex and age. The OR of the protein C deficiency was slightly elevated (OR = 6.4, 95% CI = 0.7-55.5), but it did not reach the level of statistical significance (P = .091), and it was therefore not considered as a risk factor. In conclusion, coagulant factor V gene G1691A mutation and protein S deficiency constitute important genetic risk factors in patients with VTE in Eastern Algeria. The somatic mutation of JAK2 V617F and CALR mutations are less frequent causes of VTE, thus routine testing for these mutations is not recommended.
Keywords
Introduction
Venous thromboembolism (VTE), including venous thrombosis and pulmonary thromboembolism, represents a major health problem. 1 It is a multifactorial disease, resulting from the interaction of acquired and inherited factors. 2 Among the inherited factors, we detail on the G1691A transition in exon 10 of the coagulant factor V (FV) gene known as Leiden mutation. 3 This genetic variant disorder is characterized by poor anticoagulant response to activated protein C (PC) and is considered as the most common hereditable risk factor for thromboembolic disease. 4 Heterozygosis for FV Leiden (FVL) is associated with a 5- to 10-fold increased risk of thrombosis, whereas homozygous cases have 50- to 100-fold increased risk of thrombosis. 5 –8 Another common genetic variation, which involves a guanine to adenine transition at position 20210 in the 3′-untranslated region of the prothrombin gene, has also been reported to be associated with an increased plasma prothrombin concentration. Carrier ships of this mutation are associated with an estimated 3-fold increased risk of venous thrombosis. 9 Protein C, protein S (PS), and antithrombin III (ATIII) are components of physiologically important anticoagulant systems. 10 Deficiencies of these naturally occurring anticoagulants also represent a strong genetic risk factor for VTE. Its prevalence in patients with venous thrombosis has been reported to be approximately 5% to 10%. 11,12
In European and American caucasian patients, FVL is considered as the major risk factor for venous thrombosis. 3,13 However, 2 studies from Taiwan have reported that reduced activities of the PC and PS anticoagulation system are the most important risk factors for thrombophilia in the Taiwanese–Chinese population. 14,15 Furthermore, racial differences in the prevalence of these genetic factors and their correlation with deep venous thrombosis (DVT) were found among Arab populations, 16 –19 although the constitutional background of Algerian patients with thrombosis has not been well examined, and published data are lacking.
In contrast to the most reports focused on genes involved in the clotting cascade, 20 recent studies suggest a role for the components of the hematopoietic pathway in the etiology of venous thrombosis. Philadelphia-negative myeloproliferative disorders (MPDs), especially polycythemia vera (PV) and essential thrombocythemia (ET), are found to be associated with an increased risk of venous thrombosis. 21 The identification of a dominant, gain-of-function, somatic mutation in exon 14 (1849G>T) of the Janus kinase (JAK) 2 gene, JAK2-V617F, in chronic myeloproliferative disorders has driven further research for evaluating the relevance of the mutational status in patients with thrombotic complications. 22,23 This acquired mutation has also been associated with thrombosis in special areas such as portal and mesenteric venous thrombosis, 24 splanchnic venous thrombosis, and suprahepatic veins (Bud-Chiari syndrome) in patients with or without overt signs of MPDs. 24,25 This evidence clearly suggests that further investigation is necessary in order to clarify the real implication of JAK2-V617F as a new independent thrombotic risk factor and its relationship with inherited thrombotic risk factors such as FVL and FII G20210A. For all these reasons, the detection of this mutation has become an important new acquired element in order to recognize new independent risk factor to be analyzed in patients who have developed a thrombotic event.
On the other hand, recently somatic mutations in the exon 9 of the gene encoding for calreticulin (CALR) have been identified in 88% of patients with primary myelofibrosis and in 67% of those with ET who lack the JAK2-V617F mutation and mutations in the thrombopoietin receptor gene (MPL). Of note, JAK2 and CALR mutations were found to be mutually exclusive. 26,27 Thus, the unexpected discovery of CALR mutations in MPNs has provided an important clue to the novel biological role of CALR in hematopoiesis. It has also prompted interest in its association with thrombotic risk in patients with VTE.
The purpose of the present case–control study is to determine the frequency of FVL G1691A and prothrombin G20210A polymorphisms and PC, PS, and antithrombin III deficiencies in the East Algerian populations and to investigate whether these genetic factors are associated with VTE. On the other hand, our study tends to evaluate the status of JAK2-V617F and CALR mutations among the cases.
Materials and Methods
Participants
A total of 121 cases with VTE and 146 sex- and age-matching controls were recruited from September 2012 to January 2014 within Internal Medicine and Cardiology Departments of Constantine University Hospital Center in the east of Algeria. Deep venous thrombosis was diagnosed by Doppler ultrasound, and cases were excluded if thromboembolism was due to surgery or secondary to cancer. None had clinical features of an overt MPD, 71 (58.7%) presented with deep vein thrombosis (DVT) in 1 leg, 6 (5%) with venous thrombosis of the upper limbs, 19 (15.7%) with pulmonary thromboembolism, 13 (10.7%) with cerebral vein thrombosis, and 11 (9%) with portal vein thrombosis.
The mean ages for cases and controls were 38.7 and 37.8 years, respectively. Age ranged from 12 to 85 years for cases and 16 to 85 years for controls. Traditional risk factors for VTE were recorded, including immobilization for more than 7 days, use of oral contraceptive, personal history of VTE, pregnancy, postpartum, cigarette smoking, and family history of VTE.
Normal controls were healthy laboratory workers, students, and other employees and healthy participants of the same age and with no personal or familial history of VTE. None of the women investigated was pregnant or taking oral contraceptives at the time of the blood sampling. All cases and controls participated in the study after they had given their full informed consent.
Blood Sampling and Plasma and DNA Preparation
EDTA-anticoagulated blood (5-10 mL sample) was obtained from each participant, and DNA was extracted from leukocytes by the NaCl method.
For the determination of PC, PS, and antithrombin III activity, blood was collected from fasting participants and anticoagulated with sodium citrate (0.109 mol/L; 1 volume of citrate to 9 volumes of blood). Plasma was obtained by double centrifugation at 3000g for 15 minutes at 4°C, according to the manufacturer’s recommendation, and stored in small aliquots at −80° C within 2 hours until ready for analysis.
Laboratory Methods
Genotype analysis
Detection of FV G1691A and prothrombin G20210A mutations was performed by polymerase chain reaction (PCR)-restriction fragment length polymorphism. The amplification of DNA was carried out using the primers, forward and backward sets, for each polymorphism, and the PCR products were then digested using the specific enzyme for each region (Table 1).
Sequences of Primers, Length of PCR Product, and Restriction Digestion (bp).
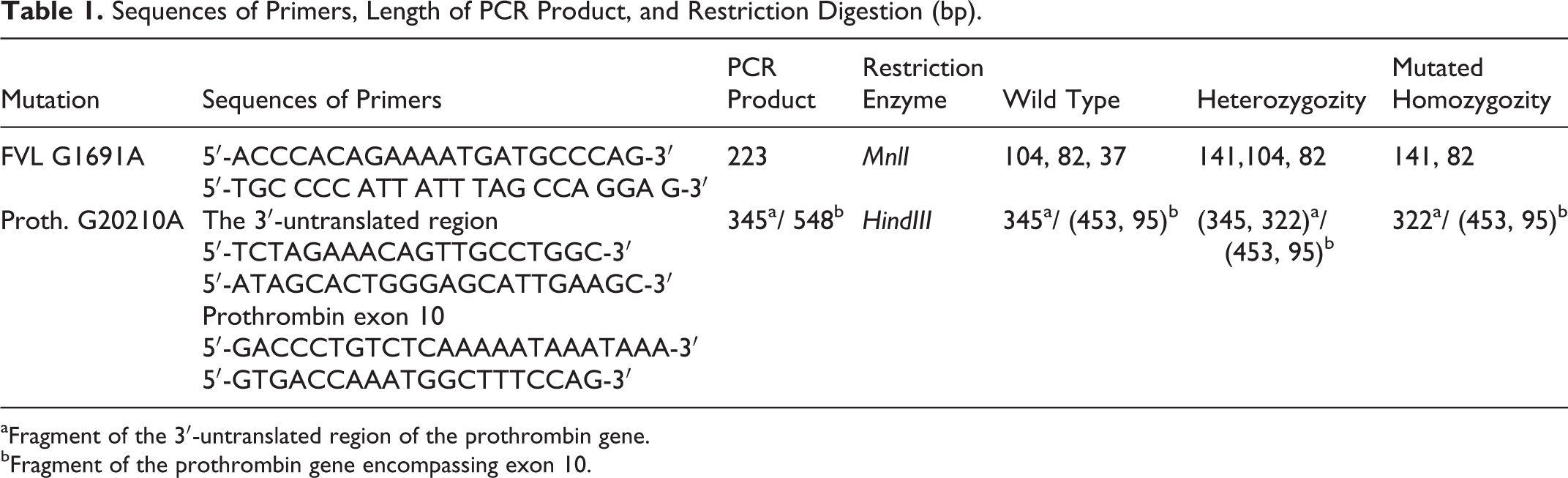
aFragment of the 3′-untranslated region of the prothrombin gene.
bFragment of the prothrombin gene encompassing exon 10.
For the detection of the G1691A mutation of FV, the mixture comprised genomic DNA (50-200 ng), 0.5 mmol/L MgCl2, 1× buffer, 0.16 µmol/L forward and reverse oligonucleotide primers (Biomatiq, Germany), 0.05 mmol/L per each deoxynucleotide (Biomatiq, Germany), and 0.04U/µL TaqDNA polymerase (Biomatiq, Germany). The PCR products were digested by MnlI (Fermentas, USA) and separated on (3:1) agarose Nusieve gel electrophoresis.
In order to detect the G20210A mutation, we performed a coamplification of 345-bp and 548-bp fragments including the 3′-untranslated region and exon 10 of prothrombin gene; according to the method of Raoul et al. 28 The 2 amplifications were performed in 2 different reaction mixtures of 25 µL.
The first contained 2.5 mmol/L MgCl2, 0.3 mmol/L of each dNTP, 1× buffer, 0.32 µmol/L forward and reverse oligonucleotide primers, and 0.04U/µL of TaqDNA polymerase for the amplification of the 3′ untranslated region. The second contained 1.5 mmol/L MgCl2, 0.25 mmol/L of each dNTP, 1× buffer, 0.16 µmol/L sense and antisense oligonucleotide primers, and 0.02U/µL of TaqDNA polymerase in a reaction mixture of 25 µL for the amplification of exon 10. This fragment contains Hind III restriction site used as an internal control for enzymatic digestion.
Amplification was made as follows: initial denaturation of 5 minutes at 94°C; followed by 35 cycles with denaturation at 94°C for 40 seconds, annealing at 56°C (G20210A) or 53°C (G1691A) for 40 seconds, extension at 72°C for 40 seconds, and a final extension of 7 minutes at 72°C.
The PCR products of the 3′ untranslated region and exon 10 of prothrombin gene were then mixed in a single tube for each sample and digested with HindIII overnight at 37°C. The products were visualized by ethidium bromide staining of a 3% agarose gel electrophoresis.
The presence of mutation G to T positioning the V617 position in exon 12 was identified by using JAK2 MutaScreen Kit (Ipsogen, France), and the quantification was performed by Ipsogen JAK2 Muta Quant Kit. These techniques are real-time quantitative polymerase chain reaction based on the allelic discrimination. Sensitivity levels of the 2 procedures are ≅ 2.5% and ≅ 0.3% mutant allele, respectively.
The exon 9 of CALR gene was amplified using the primers forward: 5′-GGCAAGGCCCTGAGGTGT-3′ and reverse: 5′-GGCCTCAGTCCAGCCCTG-3′. The PCR products are prepared by adding Hi-Deionized Formamide with ROX Size Standards mixture for the separation on electrophoresis capillary (Genetic Analyser Applied Biosystems 3730XL, Applied Biosystems, CA, USA). Then analysis of sequencing products are made by Gene Mapper (Applied Biosystems, CA, USA).
Measurement
Antithrombin III and PC functional activities were determined by an amidolytic assay using the Stachrom protein C kit and the Stachrom antithrombin III kit (Diagnostica Stago, Asnieres, France), respectively. Anticoagulant activity of the PS was tested by a clotting assay using Staclot protein S kit (Diagnostica Stago, Asnieres, France). The assays were performed at the hematology laboratory of Constantine University Hospital. Due to a lack of reagents, we have not proceeded to the classification of PC and PS deficiencies.
Values of antithrombin, PC, and PS below the 2.5th percentile of the control group were considered as pathological results (antithrombin <72%, PC <68%, and PS <58%). To eliminate the effect of sintrom on PS/PC activities, it would be ideal to measure them during a period when the cases have ceased taking drugs for at least 30 days. As it was impractical to achieve this goal for some patients, the activity of the inhibitors was not estimated among these cases.
Statistical analysis
Traditional risk factors for VTE were compared between cases and controls using chi-square test. Genotypic associations with VTE status were tested using logistic regression analysis. Univariate odds ratios (ORs; 95% CIs) were computed from logistic regression coefficients. P values less than .05 were considered statistically significant.
Multivariable logistic regression models were built to compute the adjusted OR for FVL, PC, and PS. In a first step, full models including 6 potential confounding covariables (age, sex, personal history of VTE, familial history of VTE, obesity, cigarette smoking, use of oral contraceptives, and immobilization) were built. Then, a backward elimination was performed to select important confounding factors. However, age and sex variables were systematically included in the final model to guarantee the model’s face validity. All statistical analysis were performed using R 3.1.2 (www.r-project.org)
Results
The clinical characteristics of cases and controls are shown in Table 2. No significant differences were observed between cases and controls in terms of age, sex, obesity, and cigarette smoking. It is worth noting that some cases presented specific clinical characteristics that are considered acquired risk factors of VTE. Among them we cited a personal history of VTE found in 21 (17.3%) cases, use of oral contraceptives found in 12 (9.9%) cases, a family history of VTE and pregnancy/postpartum found in 10 (8.3%) cases, and the immobilization of the lower limbs limited to 4 (3.3%) cases. One or more risk factors for VTE were present in 45% of cases; however, 55% of them presented idiopathic venous thrombosis.
Clinical Characteristics of Cases With VTE and Controls.
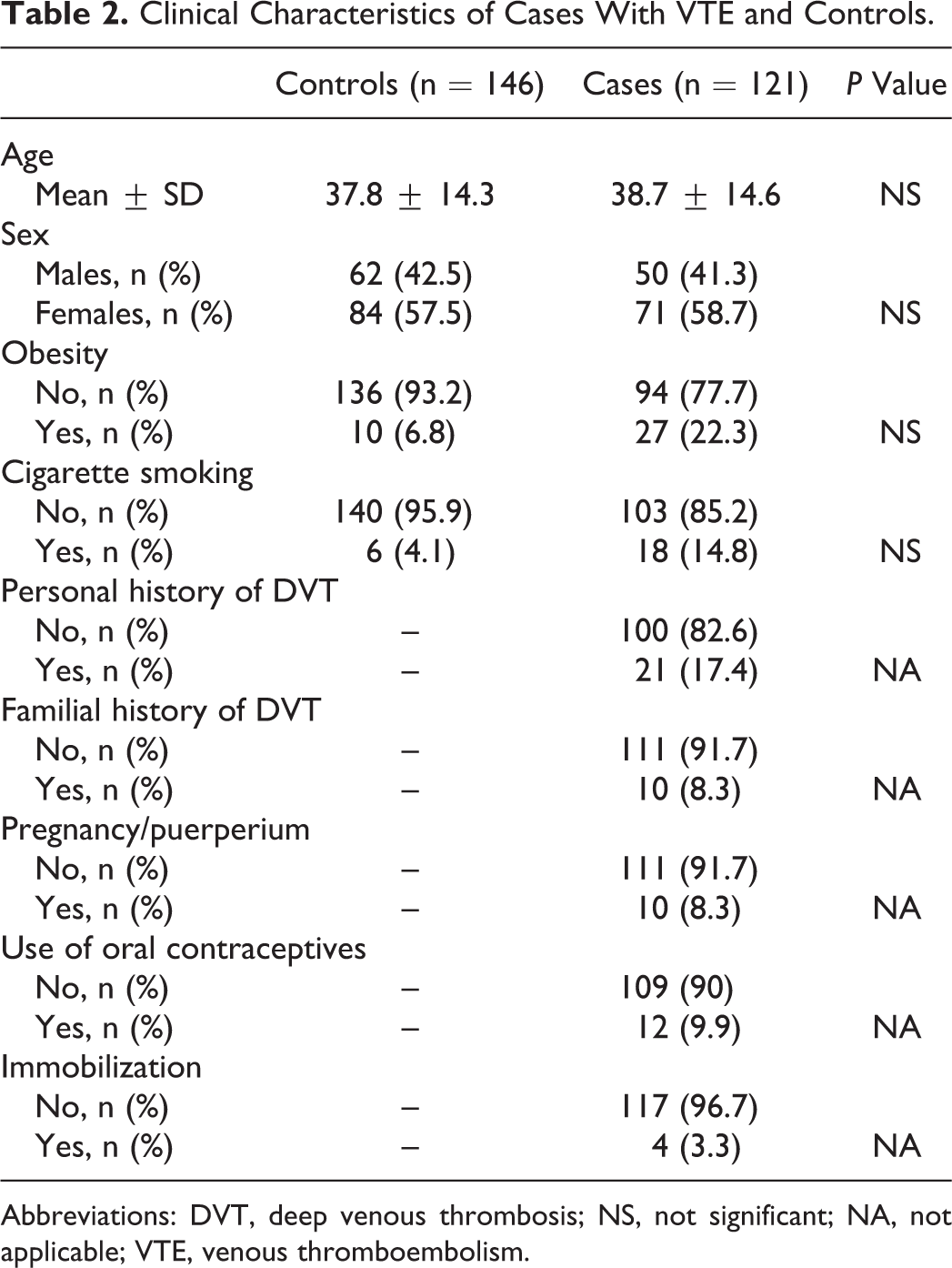
Abbreviations: DVT, deep venous thrombosis; NS, not significant; NA, not applicable; VTE, venous thromboembolism.
The genotypic frequencies of prothrombin G20210A and FVL G1691A are listed in Table 3. Among all cases (n = 121) and controls (n = 146), none was a carrier of prothrombin gene 20210G/A substitution. Regarding FVL, the observed genotype distribution was in Hardy-Weinberg equilibrium. Only 1 case presented homozygous mutation in FVL among the cases (n = 121), and none was found among the controls (n = 146). Consequently, the homozygote and heterozygote genotypes of FVL were unified in a single category (FVL mutation: GA/AA) to compute the univariate and multivariate ORs. The FVL mutation (GA/AA) was found in 14 (11.6%) cases and in 2 (1.4%) controls. The univariate analysis resulted in a significant association with VTE (OR = 9.4, 95% CI = 2.1; 42.3, P = .003).
Factor V Leiden and Prothrombin Genotypes Analysis and Thrombotic Risk Associated With Genetic Factors of VTE.
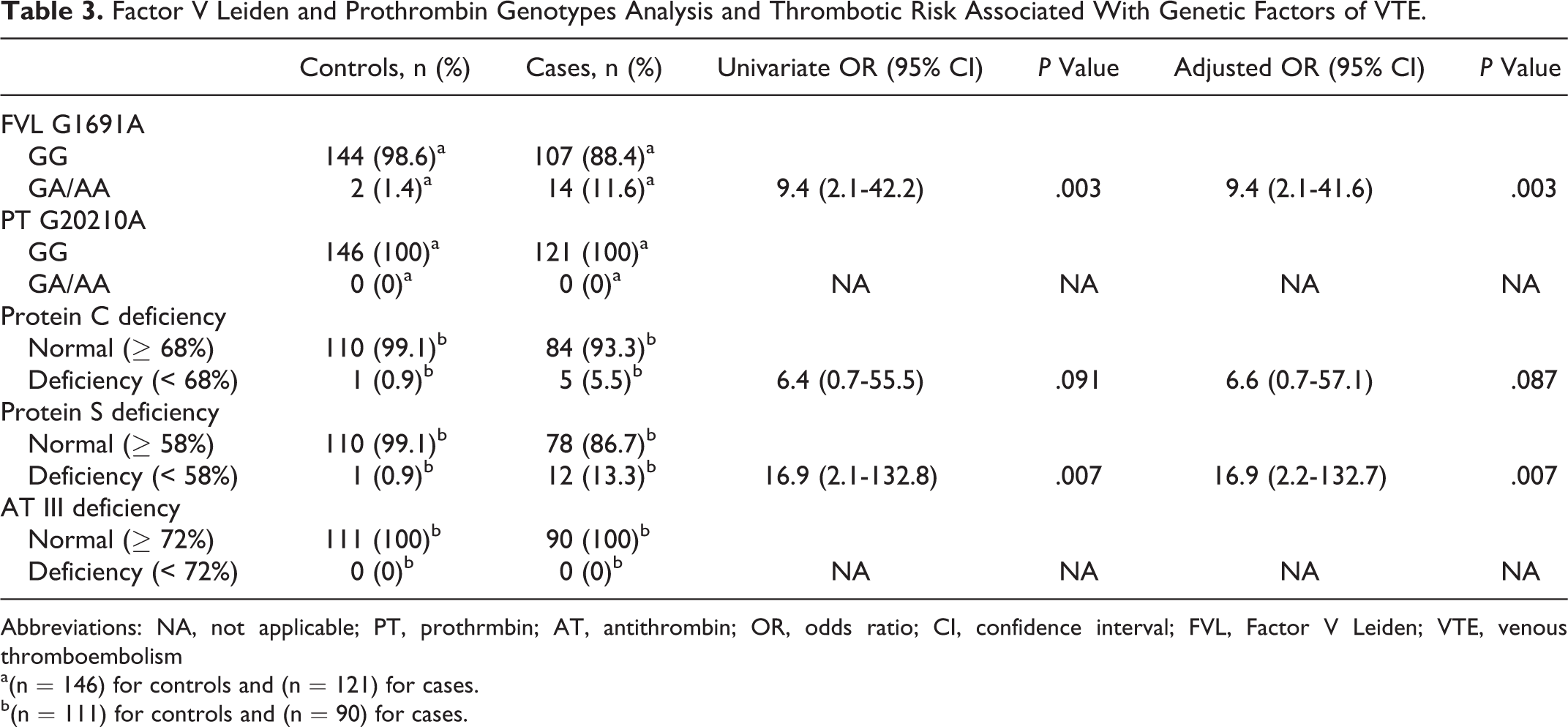
Abbreviations: NA, not applicable; PT, prothrmbin; AT, antithrombin; OR, odds ratio; CI, confidence interval; FVL, Factor V Leiden; VTE, venous thromboembolism
a(n = 146) for controls and (n = 121) for cases.
b(n = 111) for controls and (n = 90) for cases.
Family studies were carried out in 21 patients with lower levels of the PC and PS, and the hereditary deficiencies of the 2 proteins were established in 17 (18.8%) cases, while the activity determination of antithrombin was without defects for cases and controls. The coexistence of PC deficiency and FVL was found in 2 cases and deficiency of PC and PS in 1 case. In controls, only single defects were observed.
The frequency and thrombotic risk of PC and PS deficiencies are mentioned in Table 3. Univariate analysis showed a significant association of VTE status with PS deficiency and with FVL; the corresponding odds ratios were 16.9 and 9.4, respectively. The OR of PC deficiency was 6.4, but it did not reach the level of statistical significance (P = .091).
In multivariate analysis using stepwise logistic regression, the OR of the 3 variables was adjusted for age and sex. At the final step, this method confirmed that PS deficiency and FVL are significant risk factors for the development of VTE (Table 3) with the same ORs 16.9 and 9.4, respectively. Protein C deficiency remains nonsignificant in this multivariate analysis with an odds ratio of 6.6 (P = .087).
Only 1 case was reported having JAK2 positive (mutant allele burden was 15%) in a blood sample taken at the time of portal vein thrombosis. He did not present an increase in blood cell count and he had no signs of MPD. However, CALR exon 9 mutations were not detected in any of the 121 cases.
Discussion
We are well aware today that factors such as genetic predisposition, age, immobilization, recent surgery, oral contraceptives, and other hypercoagulable conditions such as pregnancy and puerperium contribute to raise the incidence of thrombosis 29,30 and synergize in enhancing VTE risk. 31 In our study, we focused on the genetic risk factors for the development of this disease and their frequency in the East Algerian population.
This study showed that the frequency of FVL (GA/AA) is estimated at 1.4% in healthy controls. This result was very similar to previous Algerian studies 32,33 that reported FVL mutation in 1.33% and 2%, respectively. There is a significant geographic variation in the prevalence of this genetic factor. In healthy controls from the Arab populations, high prevalence of FVL was observed among Jordanians 10.5% to 27.5%, 34,35,36 Lebanese 13.6% to 18.7%, 18,37,38,39 Tunisians 6.0% to 13.6%, 40,41 and Bahrainis 3.1% to 14.7%. 37,42 However, a lower frequency was reported in Saudi Arabians 0% to 2%, 37 and the mutation was not found in Morocco. 43,44 Factor V Leiden is relatively frequent in normal individuals, in populations of European origin. 45 The first reports concentrated on populations of caucasian origin in whom the mutation was found to be present in a quite high percentage of patients with VTE (15%-65%) and healthy participants (1%-15%). 46 The low frequency of FVL in our population is not comparable both with Eastern Mediterranean Arab countries: Egypt, 47 Lebanon, 48 Syria (13.6%), and Jordan (12.3%) 49 and Southern European countries, including Spain (3.3%), 50 France, and Italy (2.7%-7.5%). 51 The low prevalence rate of FVL in our region interestingly matched similar low rates established for Saudi Arabia, probably because Algerians and Arabians are related, which was brought about by the admixture of ancient Algerians (Berbers) with Arabians during the Islamic expansion era.
Prothrombin 20210A represents the second most frequent prothrombotic polymorphism in humans. 52 Its prevalence differs over a variety of countries and ethnic groups, being higher in populations living on the coasts of the Mediterranean Sea. 53 The mutation was found at 2.9% to 6.5% among Spanish healthy controls, 54,55 1.0% to 3.1% among French, 56,57 2.3% to 5.7% among Italians, 58,59 2.6% to 3.9% among Tunisians, 19 2.4% to 5.5% among Moroccans, 43,60 and 1.8% to 3.4% among Algerians. 33,61 Conversely, compared to the rates observed in neighboring communities of the Mediterranean Sea and unlike the results of published data in Algerian populations, we did not find prothrombin gene G20210A mutation in this study. Our results are similar with those of African populations 62,63 and native populations of America (Amerindians) and Australia 64 –66 evidenced by the absence of the mutation among these populations. Indeed, the heterogeneity of frequency within the same country was also reported elsewhere such as in the Italian population, characterized by the increase in the mutation in the South (3%-7%) than in the North (2%-5%). 67 This highlights the importance of establishing the frequency of prothrombin G20210A mutation in common, ethnically derived population.
The results of the study showed that FVL stands as an independent risk factor for the development of the disease with an OR of 9.4 (95% CI: 2.1-42.2), thus confirming the results of previous studies. 18,68,69 However, the mutation G20210A of the prothrombin could not be considered as a risk factor for VTE in our population. Our results are consistent with other studies including Lu et al, which did not find an association between the mutation and the risk of developing a pulmonary thromboembolism. 1 Also Salem- Berrabah et al have shown that G20210A prothrombin did not contribute to the risk of cerebral venous thrombosis. 70 Other contradictory studies have shown that this mutation contributes to a significant increase in the risk of thrombosis 71 ; Bedenčič et al showed that the G20210A mutation increased the risk of recurrence of venous thrombosis in their study population. 69 On the other hand, a high frequency of G20210A mutation was found in patients with thromboembolism in 2 studies with an allelic frequency of 6.9% and a genotype frequency of 10.5%. 72,73 This heterogeneity of the results is mostly due to the differential demographic distribution of prothrombin G20210A 74,75 and hence its association with VTE. These results are consistent with earlier reports that documented a selective association between FVL and VTE but not between FII G20210A and VTE. 75,76,77
Previous reports have indicated that abnormalities of the anticoagulation system and fibrinolytic factors contribute to thrombosis. 78,79 The results of this study show that only PS deficiency was a risk factor for venous thrombosis. This finding is in agreement with previous reports from Chinese patients in Taiwan. 15 The relative risk for thrombosis expressed as an OR stayed significant after controlling for age and sex for the PS deficiency and remains statistically nonsignificant for the PC deficiency. The frequency of PS deficiency reported in the present study was high compared to results of previous studies in Arab countries where the prevalence was estimated at 6.1% among Arabs of Kuwait, 17 2.3% in Jordan, 80 and 2.8% in Israel. 81 In caucasian unselected patients having VTE, the aggregate prevalence of these 3 conditions is approximately 5% to 7%, 20,82,83 while it was consistently reported to be significantly higher in Chinese, Thai, and Japanese patients with VTE. 84,85 Our study showed significantly lower levels of PS and slightly of PC, with an estimated prevalence of 18.8%. The latter was higher among European studies and comparable with previous studies carried out on an Algerian population. 86 At variance with the Algerian study, we did not find ATIII deficiency; therefore, it was not associated with thrombosis. A similar finding was reported in a study of Leiden Thrombophilia. 87
A thrombophilic abnormality was diagnosed in 21% of the cases, thus the present study extends these data to the JAK2-V617F and CALR mutations and further suggests mechanistic differences in the pathogenesis of venous thrombotic events in different districts.
In the past few years, a relatively large number of studies have investigated the prevalence of the somatic JAK2-V617F mutation in retrospective cohorts of patients with unexplained venous and arterial thromboembolism. 88,89 It is uncertain whether this mutation is associated with an increased risk of thrombosis. 90 –94
In our study, only 1 case harbored the JAK2-V617F allele (mutant allele burden was 15%) in a blood sample taken at the time of portal vein thrombosis; he did not present an increase in blood cell counts and he had no signs of MPDs. The frequency of JAK2-V617F mutation estimated at 0.8%. This was consistent with studies that evaluated the JAK2-V617F mutation in patients with venous thrombosis at different sites (lower limb DVT, pulmonary embolism, and retinal vein occlusion), and they have found absence or very low prevalence (0.1%-3%) of JAK2-V617F. 88,89,95 –99 Few studies have also investigated the association between the JAK2-V617F mutation and cerebral vein thrombosis. According to those, the prevalence was quite low, ranging from 0% to 6%. 93,98,100 –103 Also evident from the current study is the uniformly low mutant allele burden in patients who harbor the JAK2-V617F mutation. Other studies have shown that JAK2-V617F positivity ranged from 12% to 74% in patients with splanchnic vein thrombosis. 95,96,104 –111 This wide range was explained by several factors, such as heterogeneity in the inclusion criteria, small number of patients, different sensitivities of the assays employed for the detection of JAK2-V617F, and different types of biological samples used in the analysis. 112
The JAK2-V617F mutation was absent among controls, and this was in accordance with previous reports. 95,105 Surprisingly, in Chinese and Indian studies, this mutation was detected in blood samples of nearly 1% and 0.5%, respectively, of healthy participants. 113,114
It could be argued that identification of the JAK2-V617F mutation in patients with unexplained venous thrombosis might help to diagnose atypical or occult MPDs. Only a follow-up of the cases who carry the mutation can confirm or rule out this hypothesis. Pardanani et al 99 who followed up their 6 JAK2-V617F-positive patients for a median period of 40 months found that none developed an overt MPD or recurrent thrombosis. In contrast, others have reported a 25.0% rate of progression to overt polycythemia vera or ET in patients with splanchnic venous thrombosis and JAK2-V617F-positive after 7 to 87 months of follow-up. 104 In our study, the case who was reported having a positive JAK2-V617F died 3 months after drawing blood. It was reported that mortality is heavily related to the clinical state of the underlying disease and not only to local complications of the portal venous system, further suggesting an interdisciplinary care of these patients. 115
Our observation confirms previous data 95 that found JAK2-V617F in patients with SVT but in none of the patients presented with a thrombotic event from different districts. Thus, this suggests that in patients with SVT, screening for the JAK2-V617F mutation may be useful in recognizing patients who should be carefully observed for the subsequent development of overt MPD. In addition, a meta-analysis that evaluated the association between JAK2-V617F and venous thrombosis 116 also found that JAK2-V617F is rare or absent in patients with venous thrombosis of common sites (DVT of the lower limbs and pulmonary embolism), suggesting that screening for this mutation is not recommended as part of the routine tests for unexplained thrombosis in these settings.
The mechanism by which the JAK2-V617F mutation contributes to a hypercoagulable state or VTE without overt MPD is not clear. Xavier et al have suggested that the mutation could affect pathways involved in the activation of the hemostatic system. 112 The JAK2-V617F mutation has been associated with increased platelet and leukocyte activation 117 and increased expression of hypercoagulation markers. 118 Enhanced neutrophil activation and increased formation of neutrophil–platelet aggregates have been found in patients with Polycythemia Vera (PV) and ET. 119 Activated leukocytes can promote thrombosis by releasing granules and forming aggregates with platelets. In our study, there was no increase in the leukocyte and platelet counts of the case reported at the time of admission. This suggests that thrombosis could occur not only due to an increase in blood cell proliferation and, therefore an increase in blood viscosity but, could also be due to activation of platelets and neutrophils even without an increase in their numbers. 114
It should be taken into consideration that the case reported in this study with portal vein thrombosis may have been simply underdiagnosed because of the difficulties in applying conventional diagnosis criteria for MPD. The patient did not undergo measurement of red cell mass at the time of thrombosis, and he hadn’t a bone marrow biopsy, which is regarded as an important source of information for diagnosis of MPD. 120,121 Thus, this constitutes a limitation of the study.
The present study is the first, which evaluated the frequency of CALR mutations in patients with VTE. Based on the recent discovery of mutations in the CALR gene in a substantial proportion of patients with JAK2-negative MPNs, we hypothesized that CALR mutations could also play a role in the risk of thrombosis. None of the patients carried the CALR exon 9 mutations. So we deduced that it is not associated with a risk of VTE. Similarly Haslam and Langabeer have reported in their study that the presence of CALR mutations is not found associated with the occurrence of thrombosis in splanchnic venous thrombosis. 122 However, in a recent study among Spanish patients with SVT, Turon et al have evaluated the role of CALR mutations in the etiological diagnosis of splanchnic venous thrombosis. They reported that the screening of underlying MPNs in patients with SVT, the JAK2-V617F mutation must be evaluated first and, if negative, CALR mutations should also be investigated. 123 Whether the absence of CALR mutations in patients with VTE, it is a reflection of the size of the study population that is unknown with verification of these results required in independent cohorts.
In conclusion, we support the notion that both FVL and PS deficiency are independent prothrombotic factors that may become clinically relevant when associated with other inherited and acquired prothrombotic defects. Screening for these genetic factors in East Algerian patients with thrombophilia is therefore highly recommended. If we failed to determine that the risk of VTE disease is associated with prothrombin G20210A mutation and antithrombin III deficiency, it is because they are inexistent in our population.
Additionally, the impact of the PC deficiency on DVT pathogenesis was nonsignificant, and as VTE risk varies depending on the risk factors examined, the presence of PC deficiency in some cases within our population ensures that screening occurs for this factor in patients with thrombophilia. The somatic mutation of JAK2-V617F and CALR mutations are less frequent causes of VTE in our population. Thus, routine testing for these mutations is not recommended. Moreover, large studies in patients with SVT are required for considering the usefulness of JAK2 in recognizing patients who should be carefully observed for the subsequent development of underlying MPD.
Footnotes
Acknowledgments
We would like to thank all the team members of hematology laboratory at Constantine University Hospital Center including Professor N. Sidi Mensour for their helpful services.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
