Abstract
Cardiovascular disease (CVD) caused by atherosclerosis is the leading cause of death worldwide. The level of low-density lipoprotein cholesterol (LDL-C), considered as the initiator of atherosclerosis, is the most widely used predictor for CVD risk and LDL-C has been the primary target for lipid-lowering therapies. However, residual CVD risk remains high even with very low levels of LDL-C. This residual CVD risk may be due to remnant cholesterol, high triglyceride levels, and low high-density lipoprotein cholesterol (HDL-C). Non-high density lipoprotein cholesterol (non-HDL-C), which is calculated as total cholesterol minus HDL-C (and represents the cholesterol content of all atherogenic apolipoprotein B-containing lipoproteins), has emerged as a better risk predictor for CVD than LDL-C and an alternative target for CVD risk reduction. Major international guidelines recommend evaluating non-HDL-C as part of atherosclerotic CVD risk assessment, especially in people with high triglycerides, diabetes, obesity, or very low LDL-C. A non-HDL-C target of <130 mg/dL (3.4 mmol/L) has been recommended for patients at very high risk, which is 30 mg/dL (0.8 mmol/L) higher than the corresponding LDL-C target goal. Non-HDL-C lowering approaches include reducing LDL-C and triglyceride levels, increasing HDL-C, or targeting multiple risk factors simultaneously. However, despite the growing evidence for the role of non-HDL-C in residual CVD risk, and recommendations for its assessment in major guidelines, non-HDL-C testing is not routinely done in clinical practice. Thus, there is a need for increased awareness of the need for non-HDL-C testing for ascertaining CVD risk and concomitant prevention of CVD.
Keywords
Introduction
Low-density lipoprotein cholesterol (LDL-C) plays a key role in the pathophysiology of atherosclerosis and is a major cause of atherosclerotic cardiovascular disease (CVD)—the leading cause of morbidity and mortality globally. 1 Major international guidelines for the management of dyslipidemia recommend LDL-C as the primary target for lipid-lowering therapy (LLT).2,3 The American Association of Clinical Endocrinologists/American College of Endocrinology (AACE/ACE) recommends goals of <100 mg/dL (2.6 mmol/L) and <70 mg/dL (1.8 mmol/L) for patients at high and very high risk, respectively, for coronary heart disease (CHD), while the European Society of Cardiology/European Atherosclerosis Society (ESC/EAS) recommends goals of <70 mg/dL (1.8 mmol/L) and <55 mg/dL (1.4 mmol/L), respectively.2,3 However, patients still have residual CVD risk even when very low levels of LDL-C are attained. 4 Residual inflammatory, pro-thrombotic, and metabolic factors may drive recurrent cardiovascular events. 5 For example, metabolic factors such as remnant cholesterol, high levels of triglycerides (TG) and lipoprotein(a) [Lp(a)], and low levels of high-density lipoprotein cholesterol (HDL-C) may still exist after LDL-C lowering, 6 making LDL-C, at times, an inaccurate CVD risk predictor.
Non-high-density lipoprotein cholesterol (non-HDL-C), calculated as total cholesterol (TC) minus HDL-C, 7 has emerged as a potentially superior surrogate marker for CVD risk to LDL-C, and consequently a suitable target for CVD risk management.8–10 Despite recommendations from the Third Adult Treatment Panel (ATP III) of the National Cholesterol Education Program (NCEP) 11 for incorporating non-HDL-C as a secondary target for CVD risk lowering nearly two decades ago, a significant gap remains between current guideline recommendations and clinical practice.7,12–14
This article provides a review of the role of non-HDL-C in predicting CVD risk and the evidence for non-HDL-C as a suitable target for CVD risk lowering, provides an overview of the current guideline recommendations for non-HDL-C management, and discusses available treatments for reducing non-HDL-C levels.
Atherogenic Lipoproteins, LDL-C, and Non-HDL-C
Lipoproteins carrying apolipoprotein B (apoB) are usually atherogenic; such lipoproteins include chylomicrons, very-low-density lipoproteins (VLDL), intermediate-density lipoproteins (IDL), low-density lipoproteins (LDL), and Lp(a) 3 (Figure 1). Atherogenic lipoproteins, with a diameter of <70 nm, can cross the endothelial barrier, accumulate in the arterial wall, and initiate the formation and progression of atherosclerotic plaques. 3 LDL-C is a measure of the cholesterol mass carried within LDL particles. 15 As the most prominent cholesterol in human circulation, elevated LDL-C is indicative of atherogenesis and CVD risk. 3 Evidence based on prospective cohorts, Mendelian randomization, and lipid-lowering clinical trials have demonstrated a linear association between LDL-C levels and the development of atherosclerosis.3,16,17 Although LDL-C levels are used to estimate circulating LDL in plasma, 3 LDL-C only reflects the level of cholesterol carried by LDL particles and not the number of LDL particles. Individuals with elevated small dense LDL (which is a smaller and more atherogenic type of LDL particle than the large buoyant LDL particle) may have a high risk of CVD despite low or normal LDL-C levels. 18
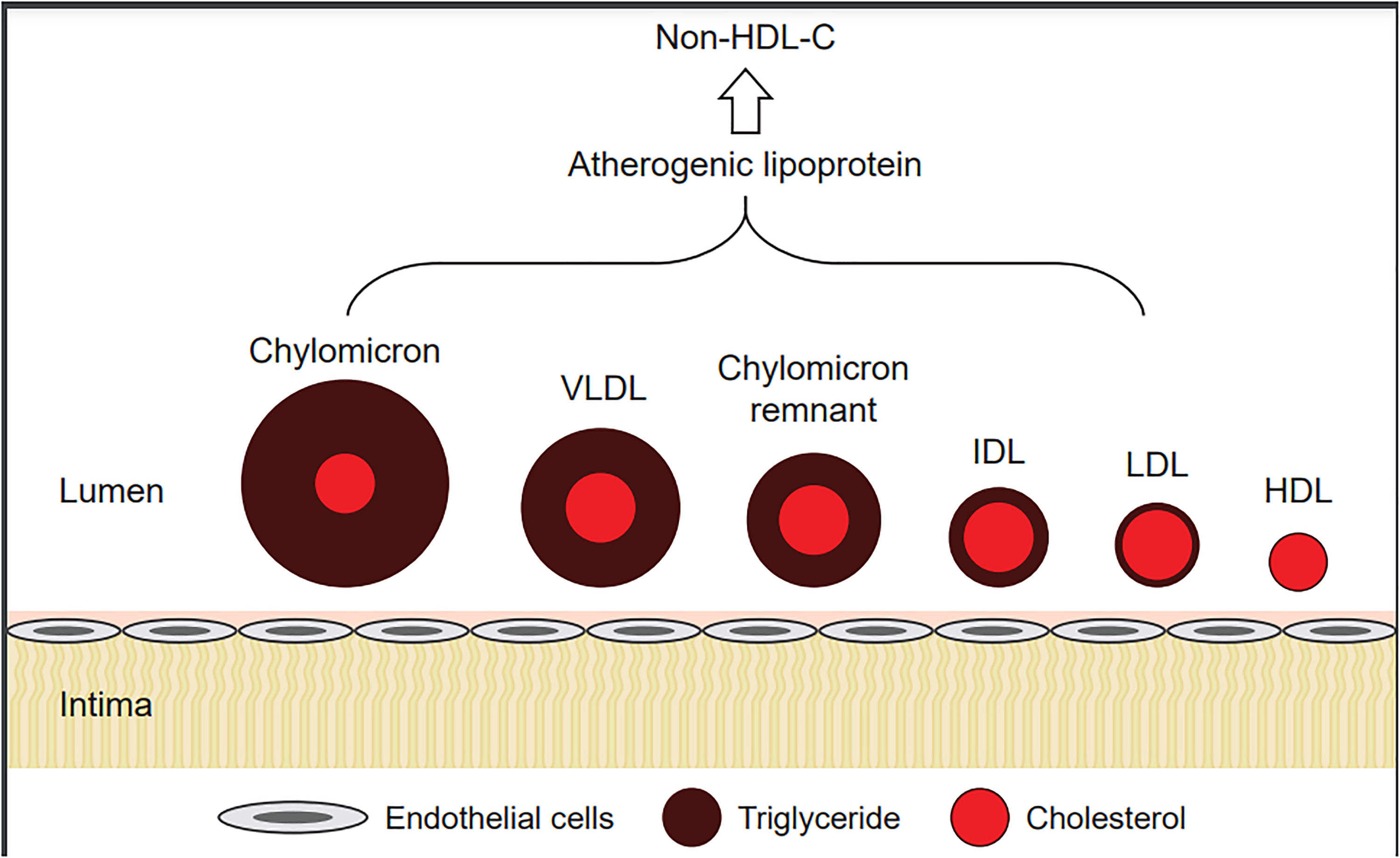
Lipid profile in human plasma. HDL, high-density lipoprotein; IDL, intermediate-density lipoprotein; LDL, low-density lipoprotein; non-HDL-C, non-high-density lipoprotein cholesterol; VLDL, very low-density lipoprotein. Figure reproduced (without changes) from Su et al. 26 under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/).
Non-HDL-C represents the overall cholesterol content from VLDL, IDL, LDL, and Lp(a) (i.e., all atherogenic lipoproteins). 12 A study in the US that used data from the National Health and Nutrition Examination Surveys from 2005 to 2010 (N = 4,986) evaluated the discordance between high non-HDL-C and high LDL-C. 19 The results revealed that, among individuals who had high non-HDL-C levels, 9.7% had normal levels of LDL-C. These individuals who had high non-HDL-C levels and normal levels of non-LDL-C were compared with individuals who had normal non-HDL-C and LDL-C; they were found to have a higher likelihood of being older, male, Hispanic, and have increased risk factors for CVD, including impaired fasting glucose and diabetes as well as metabolic syndrome. 19
To some degree, non-HDL-C is an indirect reflector of LDL particle number. Even when LDL particles are cholesterol depleted, the cholesterol is usually transferred to VLDL, and thus non-HDL-C levels still capture the overall cholesterol mass carried by LDL lipoproteins. 15 Hence, LDL particle number is more strongly correlated with non-HDL-C than with LDL-C. 15 Each atherogenic lipoprotein carries one apo B molecule, and thus, apoB concentration is a measure of atherogenic lipoprotein numbers, while non-HDL-C is a measure of atherogenic cholesterol. 20 Studies have shown that non-HDL-C correlates more strongly with apoB than LDL-C, 20 therefore, non-HDL-C is an acceptable functional surrogate for apoB. 15
Besides non-HDL-C, high levels of Lp(a) have been shown to correlate with an increased risk of atherosclerotic CVD. 21 The effect of Lp(a) on the risk of atherosclerotic CVD is proportional to the absolute plasma Lp(a) concentration, and thus large reductions (at least 40%, but up to 90%) may be needed for beneficial effects to be seen.22,23 This is much greater than the reductions required for other atherogenic lipoproteins such as LDL-C; such substantial reduction might only be achieved in patients with very high levels of Lp(a) and with the use of emerging therapies, for example, antisense oligonucleotides.22,23
Measurement of LDL-C and Non-HDL-C Levels
Preparative centrifugation of plasma samples is a direct method of measuring LDL-C and is considered the “gold standard”; however, it is expensive and time-consuming. 24 Indirect methods, such as Friedewald's equation, Martin/Hopkins equation, and Sampson's equation are more time- and cost-effective, but each of these methods has its limitations. 24 Friedewald's equation [LDL-C = (TC) − (HDL-C) − (TG/5)] requires a fasting sample, and the accuracy of the calculation is reduced with low LDL-C 24 or increasing TG levels. 25 Although Martin/Hopkins and Sampson's equations are considered more accurate than Friedewald's equation, the Martin/Hopkins method may not be a standardized method in all laboratories and Sampson's equation has not been extensively validated. 24
Non-HDL-C levels can be derived from standard lipid panels which measure TC and HDL-C levels; fasting samples are not required.7,19,26 The calculation for non-HDL-C involves subtracting HDL-C from TC 7 and the accuracy is not affected by hypertriglyceridemia or low LDL-C levels. 19 Consequently, compared with direct or calculated LDL-C, non-HDL-C shows improved accuracy for CVD risk score classification among individuals with normal and raised TG levels. 27
Non-HDL-C as a More Accurate Indicator of CVD Risk than LDL-C
As non-HDL-C is a measure of all atherogenic cholesterol, it is not surprising that non-HDL-C strongly correlates with CVD risk, and may be an even better marker and predictor of CVD risk than LDL-C. Growing evidence from genetic studies, prospective cohort studies, randomized controlled trials (RCTs), and meta-analyses of RCTs indicates the importance of measuring non-HDL-C in predicting or reflecting CVD risk, in both the general population and among treated patients with CVD receiving LLT (Table 1).8,10,28–42
Major studies that investigated the association of non-HDL-C with risk of CVD.
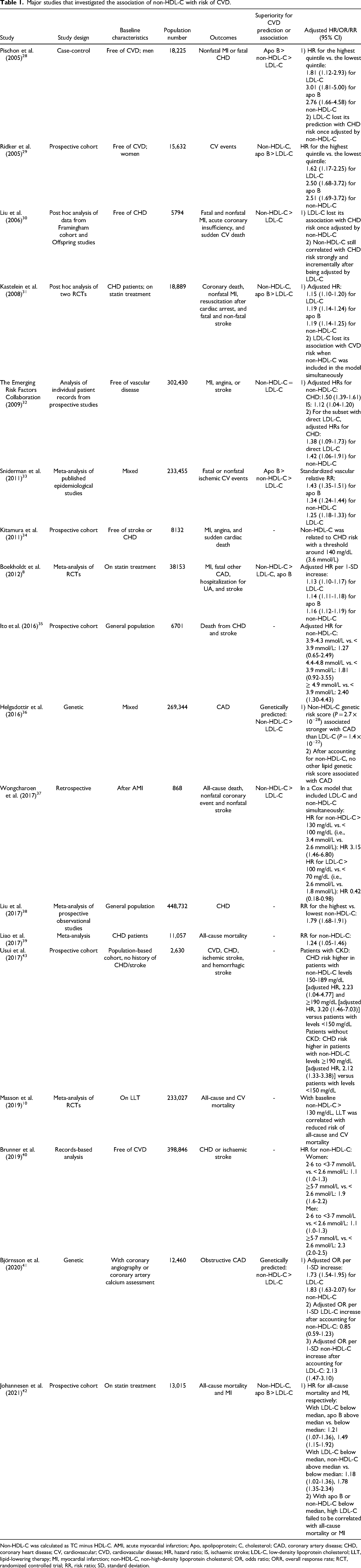
Non-HDL-C was calculated as TC minus HDL-C. AMI, acute myocardial infarction; Apo, apolipoprotein; C, cholesterol; CAD, coronary artery disease; CHD, coronary heart disease; CV, cardiovascular; CVD, cardiovascular disease; HR, hazard ratio; IS, ischaemic stroke; LDL-C, low-density lipoprotein cholesterol; LLT, lipid-lowering therapy; MI, myocardial infarction; non-HDL-C, non-high-density lipoprotein cholesterol; OR, odds ratio; ORR, overall response rate; RCT, randomized controlled trial; RR, risk ratio; SD, standard deviation.
Primary Prevention of CVD
Table 1 lists the studies that provide supporting evidence for non-HDL-C as a biomarker and a target for primary prevention of CVD. Studies conducted in patients who were CVD-free at baseline showed that higher non-HDL-C levels at baseline correlated with higher future risk for CVD.34,35,40 Some of these studies also compared the effectiveness of non-HDL-C, apoB, and LDL-C in predicting CVD risk. The Emerging Risk Factors Collaboration analyzed records of 302,430 individuals free of vascular disease at baseline and found that non-HDL-C was as effective as direct LDL-C in predicting CHD risk (adjusted hazard ratio [HR], 95% confidence interval [CI], 1.42 [1.06-1.91] vs. 1.38 [1.09-1.73]). 32 Other studies have also demonstrated a stronger association between non-HDL-C or apoB and LDL-C levels (Table 1).28–30 Although the ability of non-HDL-C and apoB to predict CVD risk varied among the studies, these lipid parameters were highly intercorrelated and were similar in predicting future CVD events.28,29 A study in Iceland that used genetic and clinical data from 269,334 individuals found that the non-HDL-C genetic risk score was more strongly associated with coronary artery disease (CAD; P = 2.7 × 10−28) than the LDL-C genetic risk score (P = 1.4 × 10−22). 36 No other lipid genetic risk scores were associated with CAD. 36 Furthermore, in the Hisayama Study, which prospectively followed 2,630 community dwellers in Hisayama for an average of 19 years, an analysis of patients with and without chronic kidney disease (CKD) showed that greater CHD risk was strongly correlated with higher serum non-HDL-C levels; this correlation was even stronger in patients who had CKD. 43 The association of non-HDL-C levels with CHD risk in patients with CKD is particularly important as patients with CKD are at high risk for developing CVD.44,45 Also, studies have shown that using LDL-C levels as a marker for CVD risk in this patient population may not be appropriate, although the existing evidence for this is limited.46,47 Moreover, dyslipidemia is common in patients with chronic renal failure and might contribute to the development of CVD, 48 thus indicating that lipid management may be important in the prevention of CHD in patients with CKD. Together, these studies suggest that non-HDL-C may be a more accurate marker for primary prevention of CVD than LDL-C.
Secondary Prevention of CVD (Lowering Residual Risk)
Patients with established CVD receiving LLT who have attained target LDL-C levels may still experience subsequent CVD events. 4 In the PROVE-IT TIMI 22 trial, the CVD event rates at 2 years were 26.3% for the standard pravastatin dose and 22.4% for the high atorvastatin dose, despite LDL-C goal attainment at the primary composite endpoint (death from any cause, MI, documented unstable angina [UA] requiring hospitalization, revascularization and stroke). 49 Moreover, even patients receiving potent LLT (e.g., statins plus proprotein convertase subtilisin/kexin type 9 inhibitors [PCSK9i]) that achieve LDL-C levels as low as 30 mg/dL may still remain at risk of CVD events. 50
Recently, a prospective cohort study in 13,015 patients receiving statin therapy reported a correlation between elevated apoB and non-HDL-C, but not LDL-C, with residual risk of myocardial infarction (MI) and all-cause mortality in patients receiving LLT. 42 Despite LDL-C levels being below the median, for patients whose apoB levels were above median, the HR (95% CI) of all-cause mortality and MI was 1.21 (1.07-1.36), while for patients whose apoB levels were below median it was 1.49 (1.15-1.92), respectively. Similarly, when LDL-C remained below median, the HR (95% CI) of all-cause mortality and MI for patients whose non-HDL-C levels were above median versus below median were 1.18 (1.02-1.36) and 1.78 (1.35-2.34), respectively. 42 Moreover, a meta-analysis comprising data from 233,455 individuals across 12 independent studies estimated that 300,000 more CVD events would be prevented by targeting non-HDL-C instead LDL-C, and 500,000 more CVD events by targeting apoB rather than non-HDL-C. 33
The Multinational Cardiovascular Risk Consortium from 19 countries in Europe, Australia, and North America analyzed data from 398,846 individuals free of CVD at baseline and established a model for evaluating CHD or ischemic stroke risk by lowering of non-HDL-C. 40 The authors found that non-HDL-C reduction was associated with a lower risk of CVD by the age of 75 years. 40 Based on this model, a 50% reduction in non-HDL-C level in people aged ≤45 years with ≥2 risk factors and with non-HDL-C of 3.7-4.8 mmol/L, could reduce the long-term CVD risk from 15.6% to 3.6% in women (12% difference) and from 28.8% to 6.4% in men (22.4% difference), with a relative risk reduction (RRR) of 77% in women and 78% in men. 40 The greatest absolute reductions in CVD risk were associated with younger age, ≥ 2 risk factors and male sex. 40
Non-HDL-C as a Treatment Target in Major Guidelines
In several international guidelines for the management of dyslipidemia and CVD2,51,52 non-HDL-C is recommended as the primary target in patients at high, very high, or extreme risk of CVD, while some other guidelines3,53–55 include non-HDL-C as the secondary target in patients who remain at high risk of CVD despite LDL-C goal attainment (Table 2). Besides the 2014 National Institute for Health and Care Excellence (NICE) 51 and the 2019 ESC/EAS 3 guidelines, most major guidelines recommend non-HDL-C levels of 30 mg/dL higher than the corresponding LDL-C target levels, with target levels of <100 mg/dL (2.6 mmoL/L) in very high-risk and <130 mg/dL (3.4 mmol/L) in high-risk patients. The 2019 ESC/EAS 3 guidelines recommend stricter non-HDL-C levels of <85 mg/dL (2.2 mmoL/L) in very high-risk patients and <100 mg/dL (2.6 mmoL/L) in high-risk patients, while the 2014 NICE 51 guidelines recommend reducing non-HDL-C levels by >40% as a treatment goal. The latest ESC Guidelines on CVD prevention 56 included estimation of non-HDL-C in the Systemic Coronary Risk Estimation 2 (SCORE2) and SCORE2-Older Persons (SCORE2-OP) risk algorithms, which help in classification of CVD risk. 56
Recommendations from major guidelines on non-HDL-C as treatment target for patients at high risk of CVD.

AACE, American Association of Clinical Endocrinologists; ACC, American College of Cardiology; ACE, American College of Endocrinology; AHA, American Heart Association; CVD, cardiovascular disease; EAS, European Atherosclerosis Society; ESC, European Society of Cardiology; IAS, International Atherosclerosis Society; LDL-C, low-density lipoprotein cholesterol; NICE, National Institute for Health and Care Excellence; NLA, National Lipid Association; non-HDL-C, non-high-density lipoprotein cholesterol.
Originally published in 2014 but regularly updated online; last updated February 2023 at the time of publication.
The 2021 recommendation does not differentiate between “high risk” and “very high risk” groups; the recommendation for the “very high risk” group applies to people at high risk of developing CVD or those who have atherosclerotic CVD.
<1.0 mmol/L (40 mg/dL) may be considered for patients with atherosclerotic CVD who experience a second vascular event within 2 years while taking maximally tolerated statin-based therapy.
In line with the international guidelines, the 2020 Chinese Guideline on the Primary Prevention of Cardiovascular Diseases 60 recommends non-HDL-C as an alternative treatment target to LDL-C. However, in patients with diabetes mellitus (DM), metabolic syndrome and obesity who have extremely low LDL-C but remain at extremely high risk of atherosclerotic CVD, the guideline recommends using non-HDL-C as the primary target for CV risk lowering, with a goal of 0.8 mmol/L (30 mg/dL) higher than the corresponding LDL-C goal. 60
Despite the accumulating evidence for non-HDL-C as a more accurate predictor of CVD risk and treatment target for lipid management, there is still a gap between recommendations from global guidelines, and routine practice in the clinic.13,61 There is a need for increased awareness of the importance of testing non-HDL-C levels, especially for managing residual CVD risk in patients who have achieved target LDL-C goals.
Effects of Lipid-Lowering Therapies on Non-HDL-C
Lipid-lowering drugs exert different effects on various lipids, including non-HDL-C. Statins have long been recognized as the mainstay of LLT, and their efficacy in reducing LDL-C and CVD risk has been established in several large RCTs.62–68 Adding remnant cholesterol-lowering treatments to statin therapy can further reduce LDL-C,50,69–71 lower non-HDL-C,50,69,71–74 and improve CVD outcomes.50,69,70,75
Lowering Remnant Blood Cholesterol
Ezetimibe and PCSK9i, combined with statin, respectively, reduce LDL-C and non-HDL-C levels and demonstrate beneficial effects on CVD outcomes.50,69,70
Ezetimibe
In the IMPROVE-IT trial, which included 18,144 patients with acute coronary syndrome, the combination of ezetimibe and simvastatin significantly reduced LDL-C, non-HDL-C, and apoB levels after 1 year of treatment (both P < .001), compared with simvastatin alone. 69 At 7 years, the simvastatin-ezetimibe group had a significant reduction in cardiovascular events, with a lower rate of the composite endpoint of death from CVD, major acute cardiovascular event (MACE) or nonfatal stroke (P = .016), compared with the simvastatin monotherapy group. 69
PCSK9i
Evolocumab and alirocumab reduce LDL-C levels on average by 60% and are most effective when combined with statin therapy. 3 PCSK9i also significantly reduce non-HDL-C levels, as shown in the FOURIER trial 50 (evolocumab use led to 52% reduction in non-HDL-C; P < .001) and in the ODYSSEY trial 70 (with alirocumab). The addition of evolocumab 50 or alirocumab 70 to maximum statin therapy resulted in an RRR of 15% (P < .001) in the composite primary endpoint of CHD death, MI, stroke, and UA requiring hospitalization.
Lowering TG
TGs are the main target for lipid-lowering therapies such as fibrates and omega-3 polyunsaturated fatty acids. These treatments are effective in reducing TG and non-HDL-C levels, but their clinical benefit in CVD outcomes remains controversial. 3
Fibrates
In the Helsinki Heart Study, 76 gemfibrozil monotherapy resulted in significant reduction in CVD events. This five-year, randomized, double-blind trial investigated the incidence of fatal and nonfatal MI and cardiac death in 4,081 middle-aged men without coronary symptoms at baseline, but who were at high risk of CVD due to abnormal blood lipid levels (non-HDL-C ≥ 5.2 mmol/L). Compared with lipid levels at baseline, gemfibrozil reduced TG levels by 43% and non-HDL-C levels by 14%; it also increased HDL-C levels by >10%. 76 At a mean follow-up of 60.4 months, gemfibrozil significantly reduced the total number of cardiac endpoints compared with placebo (56 vs. 84; P < .02; reduction of 34% (95% CI, 8.2-52.6), with the greatest reduction observed in nonfatal MI events (37%; P < .05). 76
In contrast, the BIP 77 study in 3,090 patients with previous MI or angina and without prior treatment with statin therapy, showed that bezafibrate had no benefit in terms of reducing CVD events. While treatment with bezafibrate resulted in a 21% reduction in TG levels and an 18% increase in HDL-C, no significant difference in the rate of nonfatal MI, fatal MI, and sudden death was observed between bezafibrate and placebo (13.6% vs. 15.0%; P = .26). 77
Two studies that analyzed fenofibrate in combination with statins in patients with type 2 DM and CVD (FIELD 78 and ACCORD 79 ) failed to demonstrate any benefit of treatment on CVD outcomes. 3
Similarly, the PROMINENT study, comprising 10,497 patients with type 2 DM who had a median baseline fasting TG level of 271 mg/dL and HDL-C levels of 33 mg/dL, demonstrated that the incidence of cardiovascular events was similar between those who received pemafibrate (n = 5,240) and those who received placebo (n = 5,257). 80 In this study, pemafibrate treatment led to reductions in TG and VLDL-C levels, but not non-HDL-C levels. 80
Omega-3 polyunsaturated fatty acid
In the JELIS study, 81 which included 18,645 patients with hypercholesterolemia (TC ≥6.5 mmol/L) randomized to receive eicosapentaenoic acid (EPA) 1800 mg daily in combination with statin or statin alone, EPA led to an RRR of 19% in major coronary events (HR 0.81, 95% CI, 0.69-0.95; P = .011) after a mean follow-up of 4.6 years. 81 Furthermore, in the secondary prevention subgroup, patients who received EPA with statin therapy experienced significantly fewer major coronary events compared with those who received statin alone (8.7% vs. 10.7%; HR 0.81, 95% CI, 0.657-0.998; P = .048). 81 The levels of TC and LDL-C decreased significantly in both treatment groups, but patients treated with EPA had a significantly greater reduction in TG levels compared with those who received statin monotherapy (9% vs. 4%; P < .0001). 81 In the REDUCE-IT trial, 75 addition of icosapent ethyl (2 g twice daily) to statin therapy significantly reduced the risk of a composite of CV death, nonfatal MI, nonfatal stroke, coronary revascularization, or unstable angina, compared with placebo (17.2% vs. 22.0%; HR 0.79, 95% CI, 0.68-0.83; P < .001). 75 In this study, TG levels with icosapent ethyl decreased by 18.3% compared with an increase of 2.2% with placebo, from baseline to 1 year (19.7% greater median reduction from baseline; P < .001). 75 In the same study, change from baseline to year 1 in LDL-C in the icosapent ethyl versus the placebo group was +3.1% versus +10.2% (P < .001), respectively, while change in non-HDL-C in the icosapent ethyl versus the placebo group from baseline to year 1 was −3.6% versus +10.4% (13.1% greater median reduction from baseline; P < .001), respectively. 75 In contrast, a Cochrane meta-analysis of 79 trials with 112,059 individuals found no overall effect of omega-3 fatty acids on total mortality or CV events. 82
Cholesteryl ester transfer protein inhibitors
The cholesteryl ester transfer protein inhibitor (CETPi) anacetrapib added to intensive atorvastatin therapy in patients with atherosclerotic vascular disease (n = 15,225) led to a significantly lower incidence of major coronary events than the addition of placebo (n = 15,224) over a median follow-up of 4.1 years. 83 Of note, this study was conducted in adults whose LDL-C levels were very well controlled by atorvastatin (mean LDL-C levels of 61 mg/dL or 1.58 mmol/L). 83 Moreover, at trial mid-point, mean non-HDL-C levels were lower in the anacetrapib treatment arm compared with the placebo treatment arm (relative difference of −18%). 83
The dal-OUTCOMES study, which assessed the CETPi dalcetrapib versus placebo in patients who had recent acute coronary syndrome (N = 15,871), failed to demonstrate any benefit of dalcetrapib in reducing the risk of recurrent cardiovascular events; moreover, over the course of the trial, HDL-C levels increased from baseline in both the dalcetrapib and placebo treatment arms (31% to 40% and 4% to 11%, respectively). 84
ATP citrate lyase inhibitors
In the CLEAR Harmony Phase 3 trial (N = 2,230), treatment with bempedoic acid in patients with atherosclerotic CVD and/or heterozygous familial hypercholesterolemia who had LDL-C levels of 70 mg/dL or more and were receiving statin therapy with or without additional LLT (n = 1,488), led to lower LDL-C and non-HDL-C levels from baseline to week 12 compared with placebo (n = 742). 85 No significant between-group differences were observed in the incidence of cardiovascular events or mortality. 85
Comprehensive lipid-modifying therapy
Xuezhikang (XZK) is a natural lipid-lowering “polypill.” 86 It is a purified extract of red yeast rice, which contains 13 types of natural statins and other substances including unsaturated fatty acids, ergosterol, amino acids, flavonoids, alkaloids, and trace elements. 86
Evidence shows that XZK has comprehensive lipid-modifying effects: TC levels lowered by 23.0%, LDL-C by 28.0% and TG by 36.5%; HDL-C increased by 19.6%, as compared with Jiaogulan tablets after eight weeks of treatment, in a randomized trial in 446 patients with hyperlipidemia in China. 87 It also has anti-inflammatory effects, as demonstrated by reduced levels of high-sensitivity C-reactive protein with XZK treatment. 88 A large RCT in 4,870 Chinese patients with previously documented MI showed that XZK treatment for a mean duration of 4.5 years significantly reduced the occurrence of MACE by 45%, compared with placebo, and CV and total mortality by 30% and 33%, respectively. 89
Conclusions
Dyslipidaemia is highly prevalent globally and is a major risk factor for CVD. While LDL-C is well recognized as a primary target for LLT, non-HDL-C has emerged as an alternative target and a better marker for CVD risk prediction than LDL-C. Non-HDL-C is a better marker of atherogenicity than LDL-C alone and represents the residual CVD risk in patients who have achieved target LDL-C goals. For these patients, reduction in non-HDL-C leads to further lowering of CVD risk and the incidence of MACE. This review summarizes current literature which shows non-HDL-C to be a more accurate indicator of CVD risk than LDL-C and highlights the major guidelines in which non-HDL-C has been recommended as a target for treatment. Therapeutic interventions that can help lower non-HDL-C levels are also described.
Despite increasing evidence for the importance of non-HDL-C in residual CVD risk, non-HDL-C is not routinely tested in clinical practice. We hope to improve awareness of best practices for CVD risk reduction and facilitate implementation of standardized testing for non-HDL-C, especially to prevent residual CVD risk in patients who have attained recommended LDL-C levels.
Footnotes
Acknowledgments
The authors would like to thank Alice Carruthers, PhD, and Radhika Das Chakraborty, PhD, of Nucleus Global, Asia Pacific, for providing medical writing assistance. Medical writing support was provided by AstraZeneca in accordance with Good Publication Practice guidelines.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [grant number 81870336]. Medical writing support was provided by AstraZeneca.
Institution where work was conducted
Department of Cardiovascular Medicine, The Second Xiangya Hospital of Central South University, Changsha, Hunan 410011, China.
