Abstract
Postpartum hemorrhage (PPH) remains a leading cause of maternal mortality and morbidity worldwide. This retrospective observational study describes patient characteristics and hemostatic therapies administered to 352 parturients experiencing PPH and analyzes risk factors for developing severe PPH. During the study period, bleeding was controlled in all cases and 99.4% survived. The majority (98%) of patients received packed red blood cells. The most frequent hemostatic therapies administered were fibrinogen concentrate (56%), fresh frozen plasma (49%), and platelets (30%). A total of 124 (35%) women experienced severe PPH. Significant independent predictors for evolution to severe PPH were age, obstetric comorbidity, and plasma fibrinogen concentration. The latter was based on records from 267 (76%) patients. Plasma fibrinogen concentration before labor was the only modifiable prepartum risk factor independently associated with severe PPH, indicating that fibrinogen monitoring is warranted in these patients.
Introduction
Postpartum hemorrhage (PPH) is a major contributor to maternal death and severe maternal morbidity, leading to increased therapeutic intervention, mechanical ventilation, hysterectomy, and hospital stay. 1 –4 An increase in the prevalence of PPH has been reported in several developed countries between 1991 and 2006. 5 –7 Despite the potentially severe outcomes of PPH, clinical management is not evidence based and varies considerably between centers. Protocols have been proposed for PPH management 8 –11 but as yet there is little data reporting on their impact.
Postpartum hemorrhage can rapidly evolve to severe PPH, variously defined as estimated total blood loss >1500 mL, 12 >2000 mL, 11 or >2500 mL 13 within 24 hours of birth; transfusion of ≥4 units 12 or ≥5 units 13 of blood; or a decrease in hemoglobin levels ≥4 g/dL. 12 A number of risk factors for PPH have been identified, 14 but few have been specifically linked to the evolution of severe PPH. However, plasma fibrinogen levels have been linked to both the incidence and the severity of PPH. 15 –17 The identification of prepartum characteristics that highlight patients at risk of severe PPH before it occurs could be clinically useful, although there is still some uncertainty whether prepartum fibrinogen levels or any other prepartum factors can predict evolution of PPH to severe PPH. For example, one study found no association between elevated prepartum fibrinogen levels and a reduced risk of PPH. 18
Here, we present our experience of managing PPH in a Spanish tertiary hospital with the main aim of describing patients’ baseline characteristics and comorbidities, delivery and hemorrhage characteristics, and hemostatic therapies administered in line with our standardized treatment protocol, which was carried out in cases of severe or massive hemorrhage. In addition, we use our data to explore clinically important outcomes in patients where PPH evolved to severe PPH and patients where it did not. We also investigate prepartum characteristics associated with the evolution of PPH to severe PPH.
Materials and Methods
Study Design and Patient Population
This was a retrospective, observational analysis of information obtained from the general statistical data at La Paz University Hospital, Madrid, between July 1, 2005, and December 31, 2011. La Paz University Hospital is a tertiary referral hospital with 9000 deliveries yearly, more than 25% of which are high-risk parturients. All parturients experiencing PPH were eligible for inclusion. The analysis was not designed as a case–control study; data from parturients experiencing PPH were compared with the general parturient population.
Critical Management of PPH
Patients were treated according to the multi-faceted, standardized hospital protocol developed by us and described in Guasch et al 19 for use in severe or massive hemorrhage only, as determined by the anesthesiologist. Following diagnosis, surgical or medical control of the hemorrhage was initiated by the consultant obstetricians. Blood samples were taken, and 4 units of packed red blood cells (pRBCs) were administered immediately, followed by 4 g of fibrinogen concentrate, and 4 units of fresh frozen plasma (FFP). The decision to administer pRBC and FFP was taken when bleeding appeared uncontrollable, despite surgical attempts and when coagulopathy was evidenced by analytical values or was suspected during the hemorrhagic episode. If hemorrhage control was still insufficient, 4-factor prothrombin complex concentrate was administered following consultation with a hematologist; with ongoing life-threatening bleeding, activated recombinant factor VII could also be administered. The decision was also taken as to whether further steps to control the hemorrhage were required, for example, embolization, laparotomy, hysterectomy, or other surgical resource. Patients were divided into nonsevere (defined as transfusion with <5 units pRBC) and severe PPH (requiring ≥5 units pRBC). It was decided to base the definition on transfusion, rather than blood loss, as bleeding measures are frequently inaccurate.
Data Collection and Statistical Analyses
A broad range of demographic and pre-, peri-, and postpartum data were collected from all parturients experiencing PPH until discharge from the hospital. Prepartum coagulation measurements were made using blood drawn directly before start of labor, and the distributions were analyzed. Coagulation parameters were also recorded after hemorrhage and following hemostatic therapy. Postoperative complications were identified as follows: acute kidney failure was defined as serum creatinine levels greater than 1.2 mg/dL; acute pulmonary edema was assessed by radiologic criteria and clinical features; acute myocardial ischemia or infarction was identified by transient or permanent electrocardiogram changes plus laboratory-measured troponin I increase; urinary tract injury was defined as damage to the ureter and/or bladder secondary to surgery; and coagulopathy was measured by an activated partial thromboplastin time and/or international normalized ratio greater than 1.5.
Categorical variables are summarized as the number observed in each category (percentage of total population). Continuous variables are presented as mean values (± standard deviation) if normally distributed and median values (interquartile range) otherwise. Fisher exact test was used to compare outcomes between the nonsevere and the severe PPH groups. Univariate and multivariate analyses were performed to identify prepartum characteristics associated with evolution to severe PPH. Two multivariate analyses were carried out: Model 1 used only variables for which data were available for ≥85% of patients (and so did not include ethnicity, where the patient was referred from, fibrinogen levels, or abnormal coagulation), while Model 2 included variables with larger proportion of missing data (data included as for Model 1, with the addition of fibrinogen levels and abnormal coagulation).
Results
Patient Characteristics
A total of 56 984 women gave birth in our center during the study period, of which 352 (0.62%) women were treated for PPH; bleeding was controlled in all cases, and 99.4% of the patients with PPH survived. Two fatalities were reported; the first died after hepatic failure, while the second died following acute respiratory distress syndrome arising due to an influenza A H1N1 infection. The majority of patients with PPH were referred from the delivery room (45%), other maternal hospital ward (21%), the patient’s home (17%), the intensive care unit (ICU; 9%), other hospitals (4%), or postdelivery wards (3%).
The baseline characteristics and comorbidities for patients with PPH are summarized in Table 1. The number of multiple births was relatively high in the PPH population (11% vs 3% in the general parturients population), with a 4.3 times increase in relative risk of multiple births in the PPH population (P < .001).
Patient Baseline Characteristics and Comorbidities.
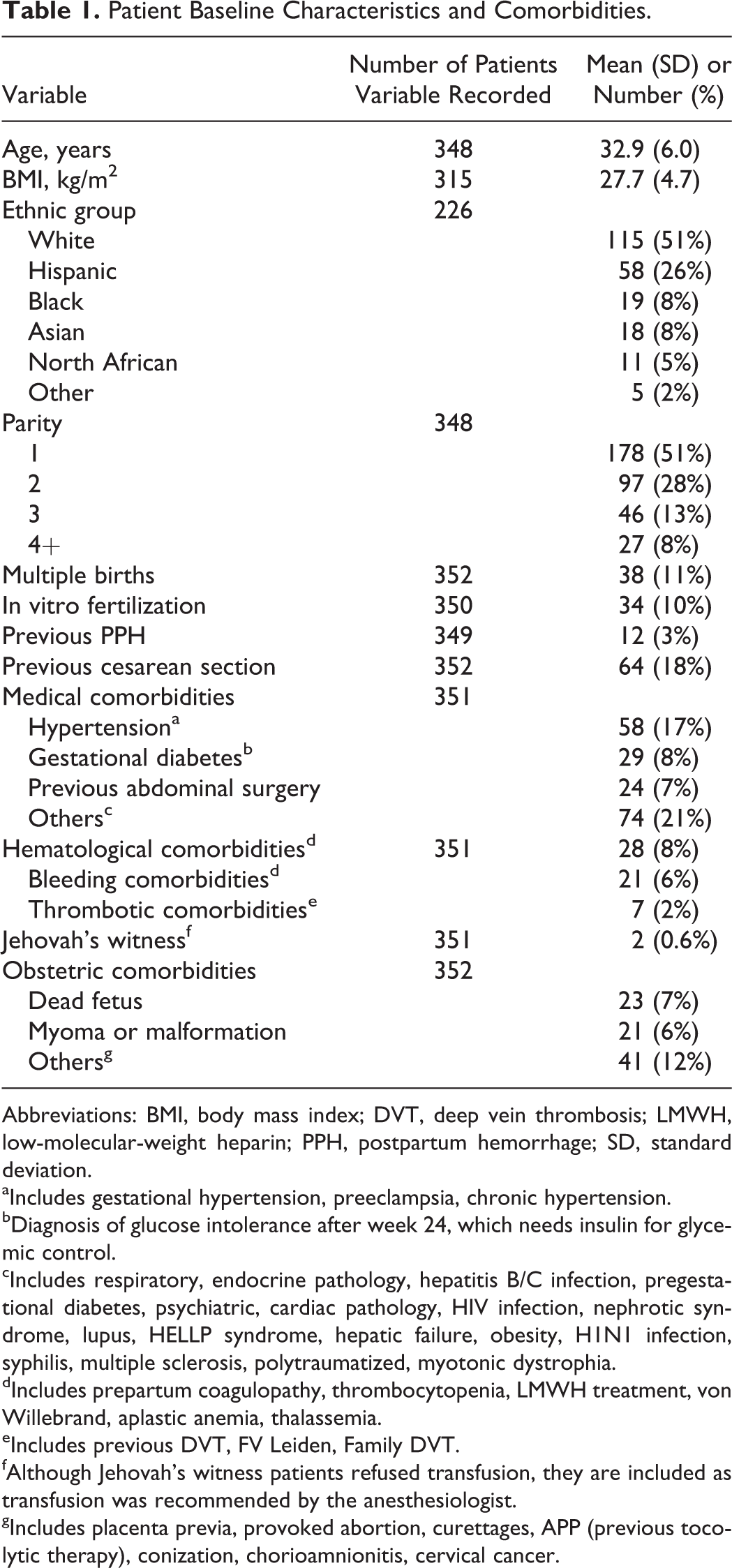
Abbreviations: BMI, body mass index; DVT, deep vein thrombosis; LMWH, low-molecular-weight heparin; PPH, postpartum hemorrhage; SD, standard deviation.
aIncludes gestational hypertension, preeclampsia, chronic hypertension.
bDiagnosis of glucose intolerance after week 24, which needs insulin for glycemic control.
cIncludes respiratory, endocrine pathology, hepatitis B/C infection, pregestational diabetes, psychiatric, cardiac pathology, HIV infection, nephrotic syndrome, lupus, HELLP syndrome, hepatic failure, obesity, H1N1 infection, syphilis, multiple sclerosis, polytraumatized, myotonic dystrophia.
dIncludes prepartum coagulopathy, thrombocytopenia, LMWH treatment, von Willebrand, aplastic anemia, thalassemia.
eIncludes previous DVT, FV Leiden, Family DVT.
fAlthough Jehovah’s witness patients refused transfusion, they are included as transfusion was recommended by the anesthesiologist.
gIncludes placenta previa, provoked abortion, curettages, APP (previous tocolytic therapy), conization, chorioamnionitis, cervical cancer.
Delivery and hemorrhage characteristics are described in Table 2. Almost 50% of patients with PPH required an emergency cesarean delivery, with an increased relative risk of 2.0 (P < .001) in the PPH population compared to the general parturients population. The most common reasons for emergency cesarean section were fetal distress, placenta previa, or repeat cesarean delivery. In comparison, 23% of patients with PPH required instrumental delivery, with the most common reasons being a long second stage or fetal distress. The gestational age was recorded for 336 patients and was found to be full term in the majority of cases (236 deliveries, 70%), but preterm (24-36 weeks) in 68 (20%) patients, puerperal in 13 (4%) patients, and not viable (<24 weeks) in 19 (6%) patients. Prematurely ruptured membranes (>24 hours) were observed in 11 (3%) of 352 patients.
Delivery and Hemorrhage Characteristics.
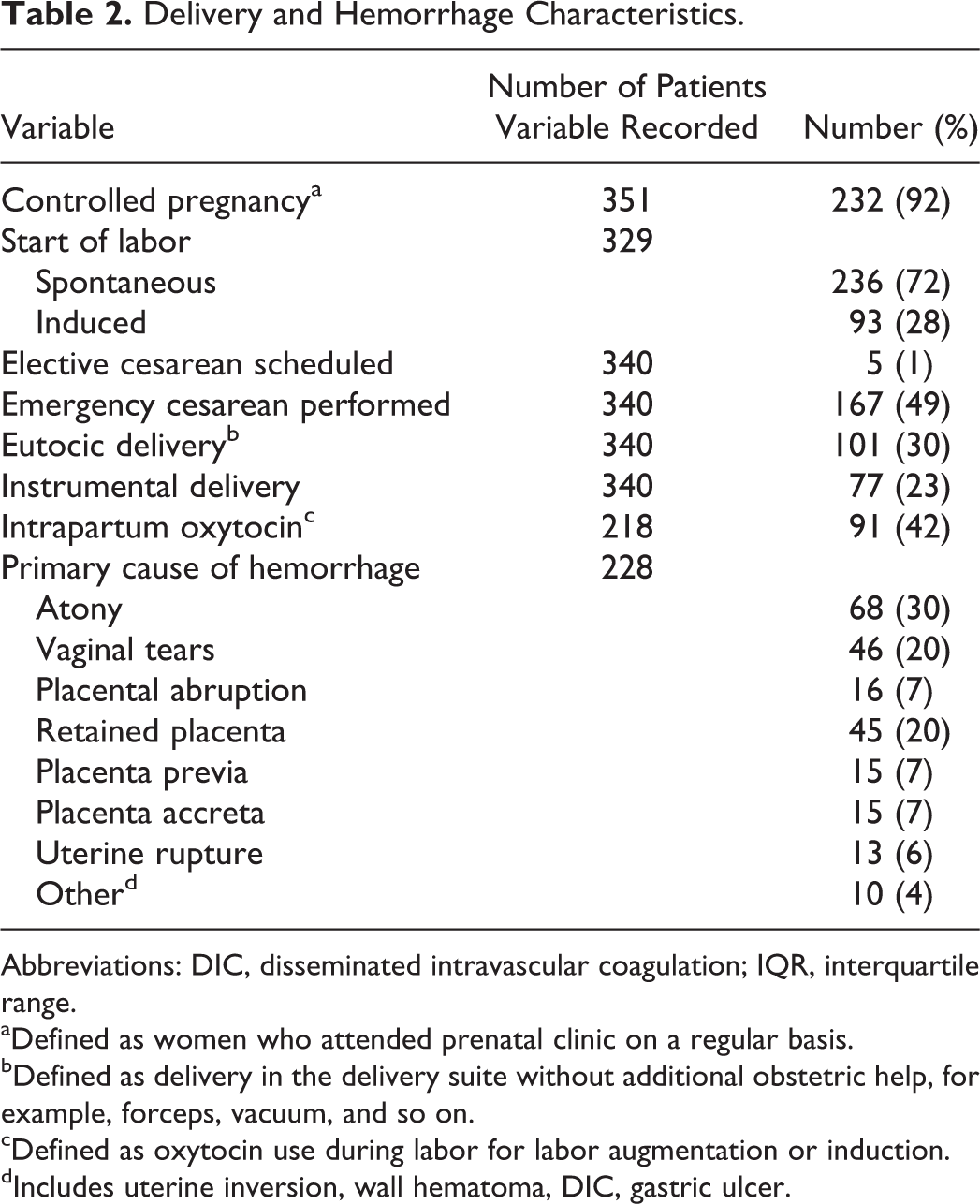
Abbreviations: DIC, disseminated intravascular coagulation; IQR, interquartile range.
aDefined as women who attended prenatal clinic on a regular basis.
bDefined as delivery in the delivery suite without additional obstetric help, for example, forceps, vacuum, and so on.
cDefined as oxytocin use during labor for labor augmentation or induction.
dIncludes uterine inversion, wall hematoma, DIC, gastric ulcer.
In the majority (92%) of patients, hemorrhage occurred following delivery but was also observed prenatally (10%), and during the first (6%), second (6%), and third (10%) stages of labor.
Coagulation Tests and Hemostatic Management
Patients were treated according to a standardized protocol; the therapies administered for hemostatic management are summarized in Table 3. The association between the amount of colloid and/or crystalloids administered and the occurrence of renal failure was examined, and no relationship was found. Coagulation parameters were recorded just before labor, after hemorrhage (lowest values pre-transfusion), and after hemostatic therapy (Figure 1). The mean (standard deviation, range) values observed were as follows: hemoglobin levels were 11.7 (1.7, 5.9-16.2) g/dL before labor, 6.7 (1.3, 2.9-11.1) g/dL posthemorrhage, and 9.4 (1.1, 7.1-13.1) g/dL following therapy; hematocrit was 35.1 (4.8, 3.6-49.3) % before labor, 19.8 (3.7, 8.2–35.2) % posthemorrhage, and 27.9 (3.3, 20.7-39.2) % following therapy; platelet levels were 225 (80, 15-484) c/mL before labor, 153 (73, 5-489) c/mL posthemorrhage, and 154 (70, 41-766) c/mL following therapy; fibrinogen levels were 555 (146, 126-1000) mg/dL before labor, 327 (127, 80-756) mg/dL posthemorrhage, and 440 (140, 151-945) mg/dL following therapy.
Volume Replacement, Transfusions, and Hemostatic Therapies Administered.
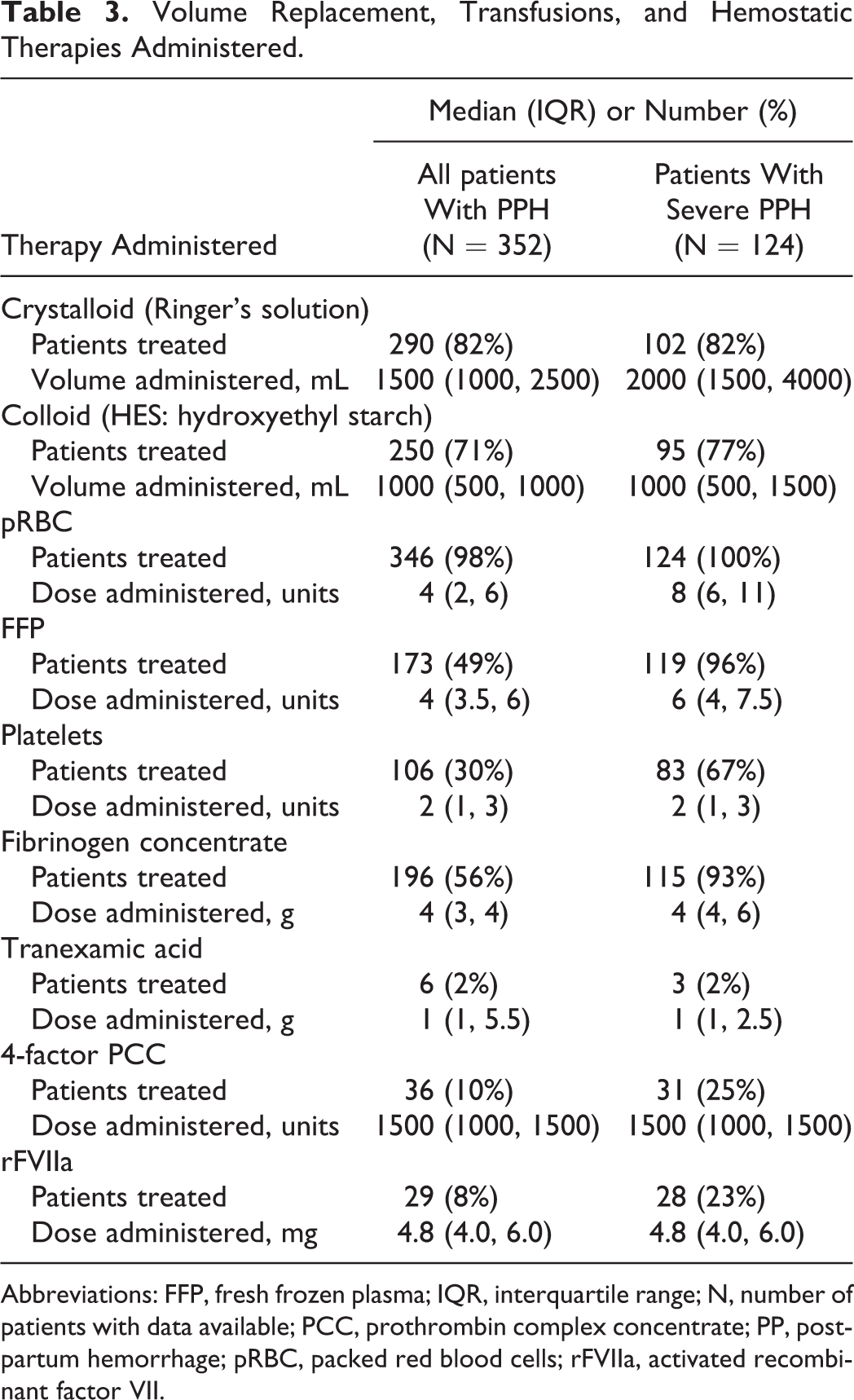
Abbreviations: FFP, fresh frozen plasma; IQR, interquartile range; N, number of patients with data available; PCC, prothrombin complex concentrate; PP, postpartum hemorrhage; pRBC, packed red blood cells; rFVIIa, activated recombinant factor VII.
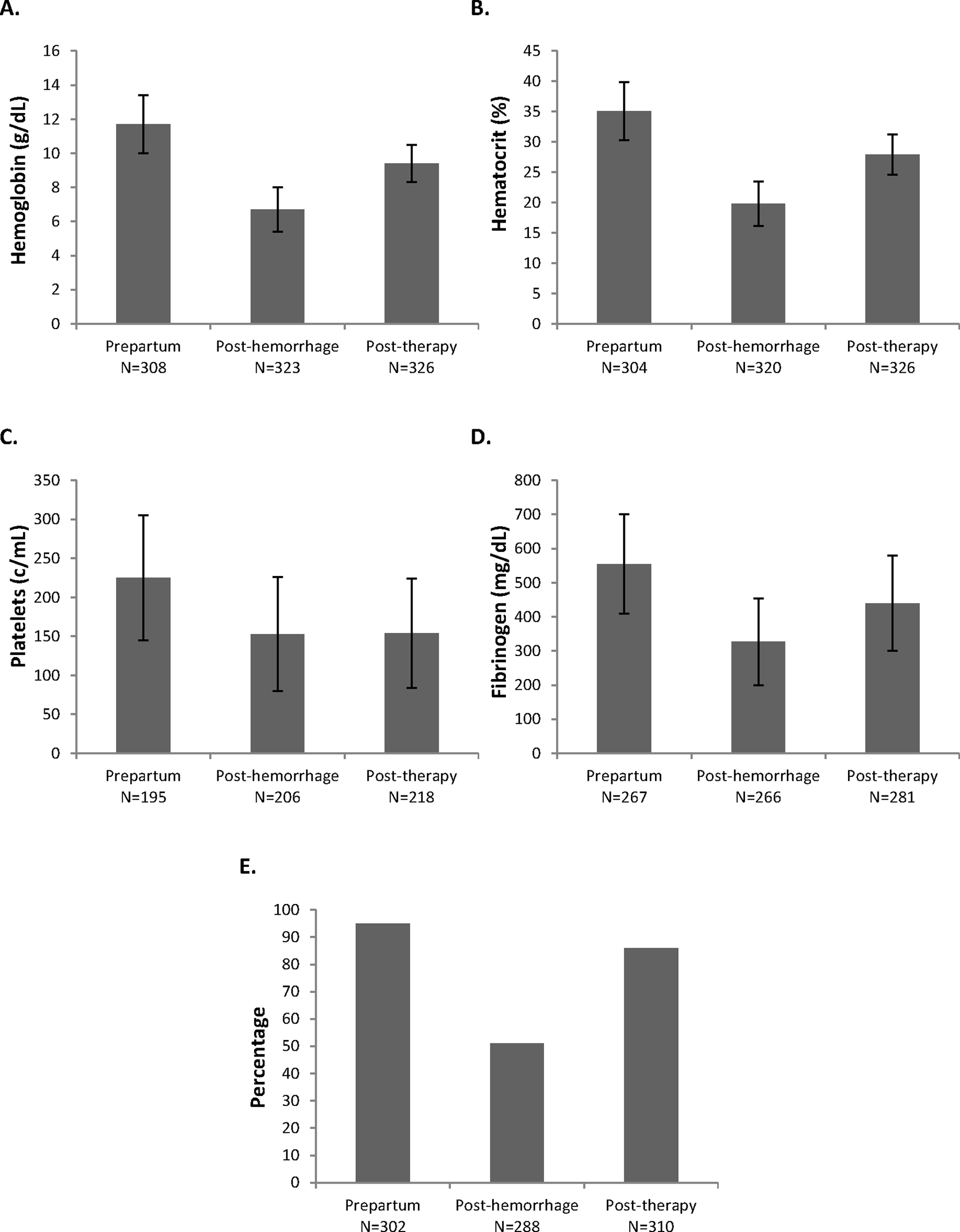
Hematology and coagulation variables: Mean levels of (A) hemoglobin, (B) hematocrit, (C) platelets, and (D) fibrinogen and (E) percentage of patients with normal coagulationa. aNormal coagulation defined as INR ≤ 1.5 and aPTT <1.5 times normal; N, number of patients with data available. aPTT indicates activated partial thromboplastin time; INR, international normalized ratio; N, number of patients with data available.
Evolution of PPH to Severe PPH
A total of 124 (35%) women experienced severe PPH (requiring ≥5 units pRBC). The key clinical outcomes for patients with severe versus nonsevere PPH are listed in Table 4. The results indicate that severe PPH is more likely to require a change from regional to general anesthesia and is associated with a more than 8-fold increase in patients requiring embolization as well as a more than 5-fold increase in the incidence of hysterectomy. Patients with severe PPH also had a 50% longer median stay in the ICU (36 [24-58] hours vs 24 [12-30] hours in the nonsevere PPH group). Before labor, mean (median, range) values for fibrinogen levels in all parturients were 555 (554, 126-1000) mg/dL and 568 (561, 204-1000) mg/dL and 527 (547, 126-952) mg/dL in patients with nonsevere and severe PPH, respectively.
Outcomes Observed in Patients With Nonsevere Versus Severe PPH.
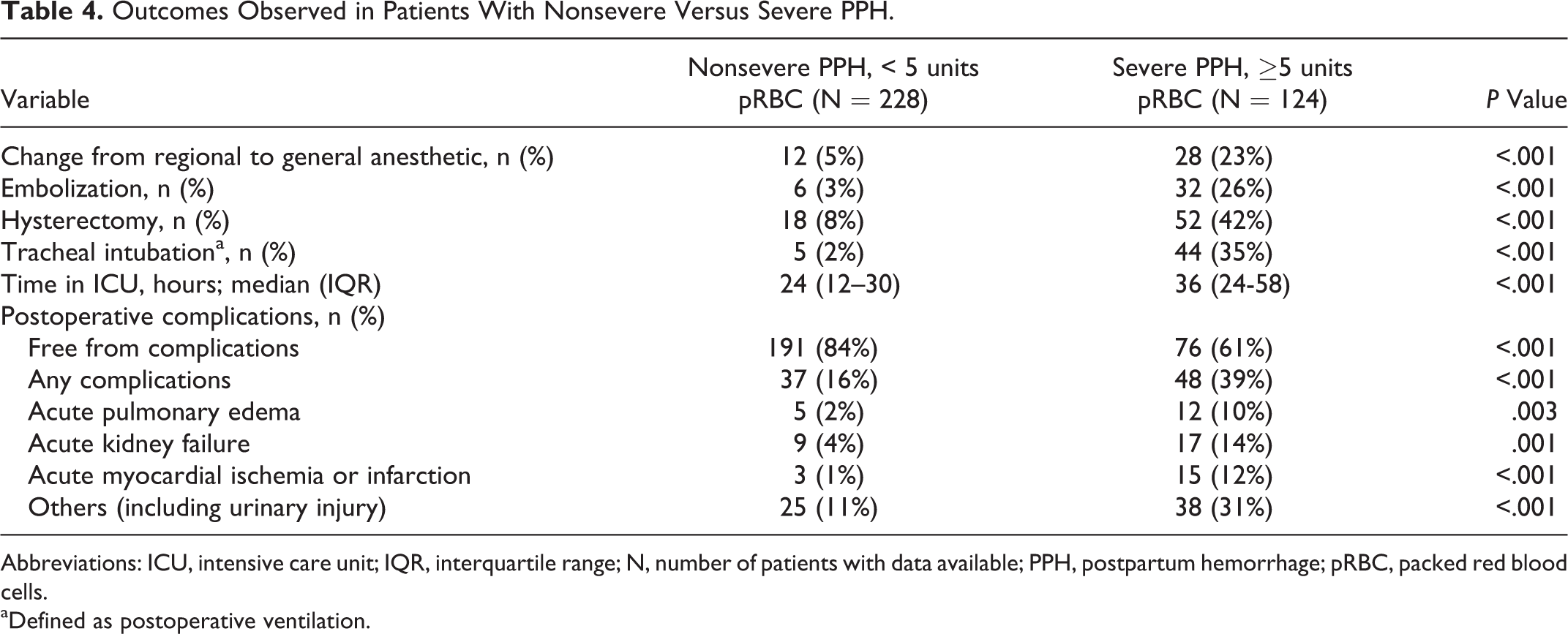
Abbreviations: ICU, intensive care unit; IQR, interquartile range; N, number of patients with data available; PPH, postpartum hemorrhage; pRBC, packed red blood cells.
aDefined as postoperative ventilation.
Analyses were carried out to identify any prepartum risk factors linked to the evolution of severe PPH. A univariate analysis was performed to identify prepartum variables associated with increased odds of severe PPH (Table 5). A 5-year increase in age was associated with a 29% increased odds ratio for severe PPH, obstetric comorbidity was associated with a 2.4 times greater odds ratio of severe PPH, and coagulation diagnosed as “abnormal” increased the odds ratio of severe PPH by 2.8-fold. Conversely, a 50 mg/dL increase in fibrinogen level resulted in a 10% reduction in the odds ratio of severe PPH.
Univariate Analysis of Effects of Baseline Characteristics on Evolution of Severe PPH (≥ 5units pRBC).a
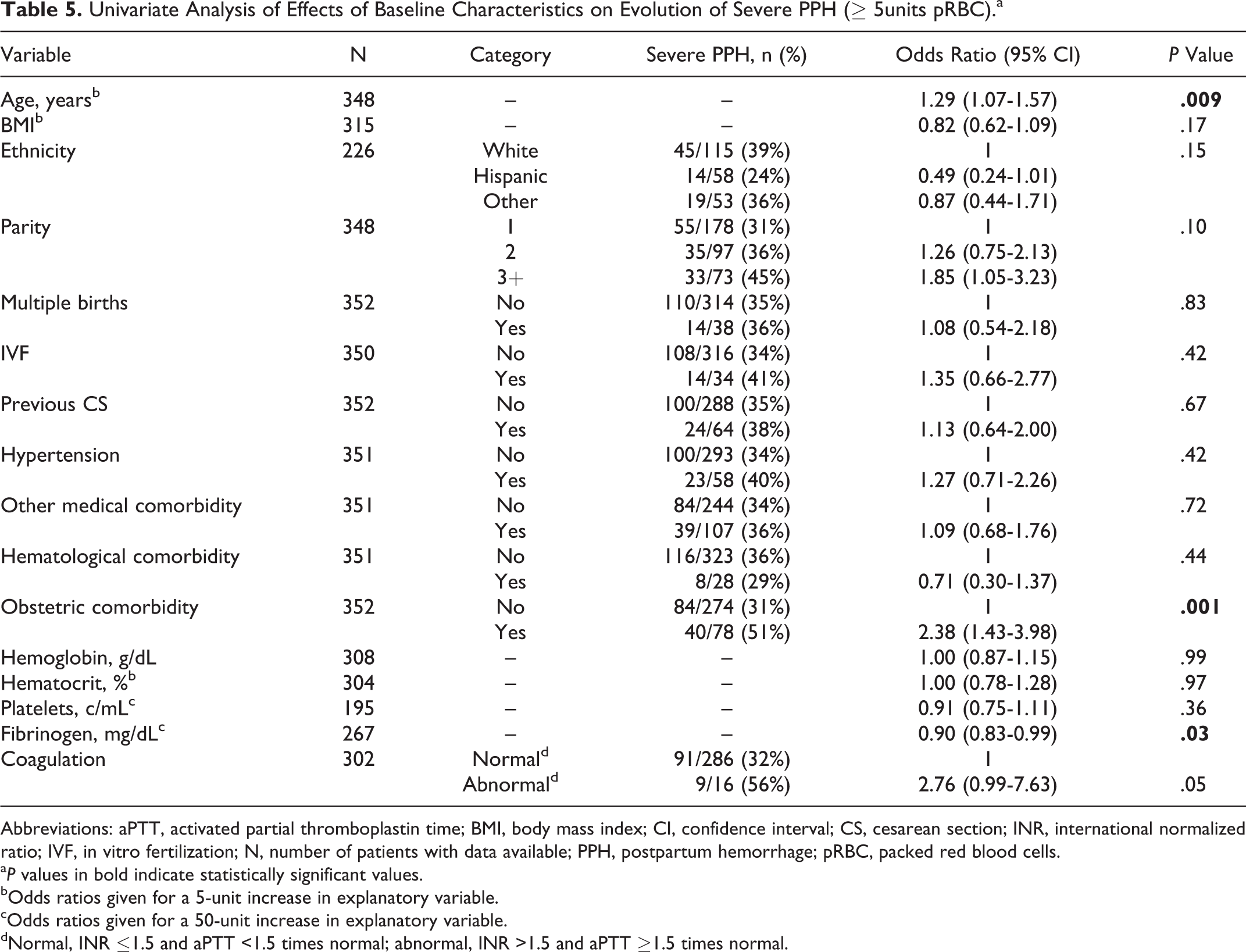
Abbreviations: aPTT, activated partial thromboplastin time; BMI, body mass index; CI, confidence interval; CS, cesarean section; INR, international normalized ratio; IVF, in vitro fertilization; N, number of patients with data available; PPH, postpartum hemorrhage; pRBC, packed red blood cells.
a P values in bold indicate statistically significant values.
bOdds ratios given for a 5-unit increase in explanatory variable.
cOdds ratios given for a 50-unit increase in explanatory variable.
dNormal, INR ≤1.5 and aPTT <1.5 times normal; abnormal, INR >1.5 and aPTT ≥1.5 times normal.
Multivariate analyses were also performed (Table 6). In each case, age and obstetric comorbidity were still significant independent predictors for evolution of PPH to severe PPH. Plasma fibrinogen concentration directly before labor also remained associated with severe PPH. However, abnormal coagulation was not independently associated with severe PPH.
Multivariable Analyses of Effects of Baseline Characteristics on Evolution of Severe PPH (≥ 5 units pRBC).
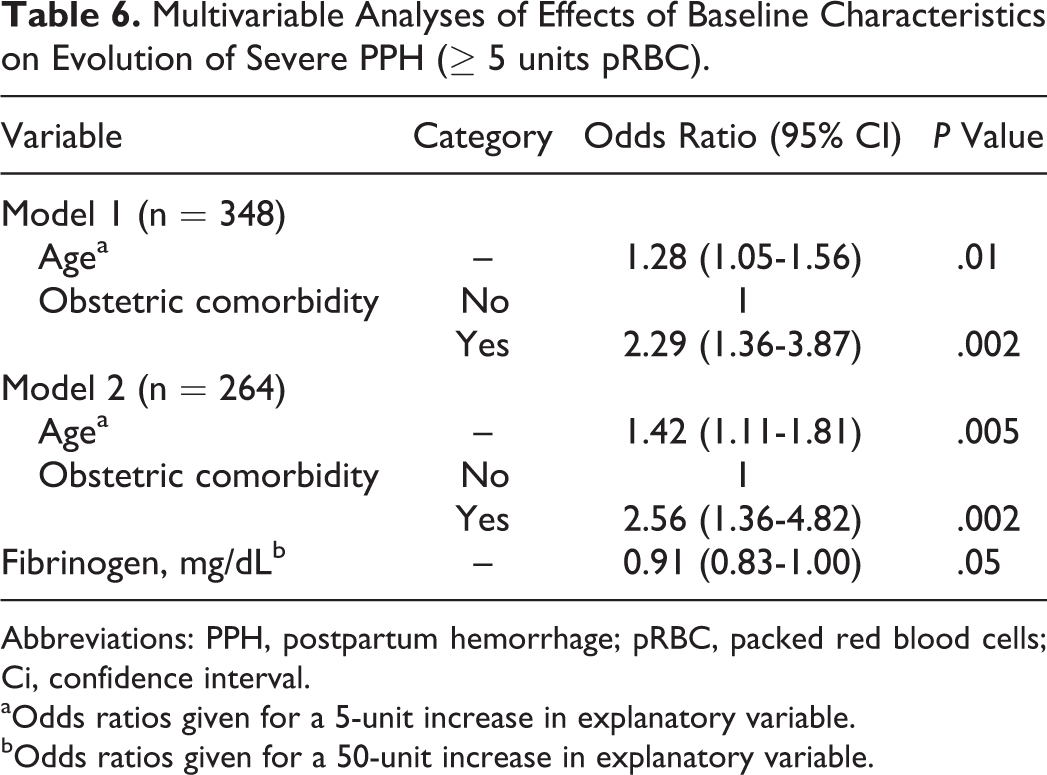
Abbreviations: PPH, postpartum hemorrhage; pRBC, packed red blood cells; Ci, confidence interval.
aOdds ratios given for a 5-unit increase in explanatory variable.
bOdds ratios given for a 50-unit increase in explanatory variable.
Discussion
In this study, we have presented our experience with 352 cases of PPH, including 124 cases of severe PPH. Hemostasis was restored in all cases and 99.4% of patients survived. Our therapeutic approach combined standard transfusion practice and coagulation factor concentrate-based therapy. This may have improved the appropriateness of therapy received by each patient and possibly explains the correction of hemostatic parameters observed posttherapy. However, new algorithms for the effective management of PPH are under development, and it is critical that these algorithms are widely tested, reported, disseminated, and applied in order to ensure the most appropriate standardized practice. 14
In our study, a small number of patients received tranexamic acid (TXA); however, TXA was only introduced to our protocol in 2011, although it was occasionally administered prior to this date at the hematologist’s discretion. Mounting evidence suggests that TXA is useful for prevention and treatment of PPH, 20 –22 and a global, multicenter, randomized, controlled trial (the WOMAN trial) is underway to test the effects of TXA on mortality and incidence of hysterectomy due to PPH. 23
One-third of all cases evolved to severe PPH, which was associated with a significantly higher rate of complications. Several of these complications (eg, use of general anesthesia) carry additional risks for the patient as well as incurring significant hospital costs. Additionally, hysterectomy means the absolute loss of reproductive potential as well as being a major surgical intervention. As such, the early identification and treatment of women at risk of severe PPH may help minimize the economic and psychological/socioeconomic costs associated with severe PPH.
The results of this retrospective analysis helped us identify risk factors that may be used in the prepartum period to recognize patients at risk of evolution of PPH to severe PPH. Interestingly, some well-established risk factors for PPH, including high parity, multiple births, previous cesarean section, and anemia, were not significantly associated with the evolution of PPH to severe PPH. The major risk factor for severe PPH was the presence of an obstetric comorbidity; the most common prepartum obstetric comorbidities in our population were dead fetus, myoma, placenta previa, medically induced abortion, and history of curettages in prior pregnancy. Physicians managing patients with these conditions should prepare for rapid deterioration of PPH to severe PPH and the need for intervention to prevent hysterectomy. Age was the next factor most strongly associated with severe PPH. As the average age of parturients is increasing, the proportion of PPH episodes that evolve to severe PPH may also increase.
Our findings build upon previous studies showing that low prepartum fibrinogen is associated with increased incidence of PPH, and low fibrinogen during PPH is associated with evolution to severe PPH. The association of reduced levels of fibrinogen with PPH severity indicates that fibrinogen replacement therapy may be a suitable addition to PPH management. Data are limited, but fibrinogen concentrate (in conjunction with other therapies) has been used as an effective therapy for controlling PPH associated with low fibrinogen levels. 24,25 A trial is ongoing to evaluate whether early treatment with fibrinogen concentrate reduces the need for blood transfusion in women with severe PPH, 26 and fibrinogen replacement has been recommended as an early intervention for the control of PPH associated with amniotic fluid embolism. 27
Low fibrinogen concentration directly before labor is the only independent risk factor that we identified for severe PPH that is readily modifiable indicating that early, targeted fibrinogen replacement therapy may be warranted during severe PPH. Consistent with this, our data show a transfusion pattern in which fibrinogen concentrate is frequently administered during severe PPH. Fibrinogen concentrate was the only available option that could elevate mean fibrinogen levels from 3.3 g/L to 4.4 g/L as observed in our population. As the fibrinogen content of FFP is only ∼2 g/L, it would not be suitable to bring about this increase. Cryoprecipitate could be considered as an alternative to increase fibrinogen concentration in this setting, but it was withdrawn from most European countries due to safety considerations. 28
There are some limitations to this study: the findings are subject to the constraints typically associated with retrospective data analyses, in particular the fact that full data were not available for all patients. For example, we only had fibrinogen data for 267 patients, representing 76% of the total study population. In addition, none of the analyses were prespecified, so a sample size calculation was not performed to assess the statistical power of the study. Moreover, many of the comparative analyses showed striking between-group differences with highly significant P values. Generalizability of the findings is also uncertain, given that our hospital is a tertiary referral center, so may receive more serious cases of PPH compared with other hospitals. Additionally, it should be noted that we did not do a precise quantification of the amount of bleeding in our patients with PPH.
In conclusion, our data describe successful management of PPH in a tertiary hospital. Over 99% of the patients with PPH survived. In our setting, the application of a multidisciplinary protocol with early administration of fibrinogen and FFP was useful in the absence of point-of-care viscoelastic measurements such as thromboelastometry or thrombelastography. Protocols or guidelines for high-risk parturients are important in this field, and plasma fibrinogen concentration directly before labor was the only modifiable prepartum risk factor independently associated with severe PPH. However, further study of peripartum plasma fibrinogen levels is needed to examine the role of fibrinogen. Close peripartum monitoring of fibrinogen levels (or fibrin-based clotting) is warranted in line with the 2013 European Society of Anaesthesiology guidelines, 8 especially in older parturients with obstetric comorbidities, as the results indicate that these patients are at risk of severe PPH.
Footnotes
Authors’ Note
All authors participated in the conception and design of the study, drafted the article, and revised it critically for important intellectual content and approved the final version of the article to be published.
Acknowledgments
Editorial assistance with manuscript preparation was provided by Meridian HealthComms and funded by CSL Behring. The authors would like to give special thanks to the obstetricians at Hospital Universitario La Paz.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Emilia Guasch has given lectures sponsored by CSL Behring. Fernando Gilsanz has no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
