Abstract
Objective:
To investigate the efficacy of recombinant human soluble thrombomodulin (rTM) in disseminated intravascular coagulation (DIC) associated with severe postpartum hemorrhage (PPH).
Patients and Methods:
We conducted a retrospective review of 36 patients with severe PPH complicated by DIC admitted to a single tertiary center. The first 26 patients were treated without rTM (control group), and the next 10 consecutive patients were treated with rTM. Clinical parameters including bleeding symptoms and coagulation indices were evaluated.
Results:
Baseline characteristics, total blood loss, and transfusion requirements were similar between the 2 groups. On day 2, there was a significant difference between the 2 groups in the decrease in
Conclusion:
Recombinant human thrombomodulin may be an effective adjunctive therapy in the management of DIC related to PPH.
Introduction
Severe postpartum hemorrhage (PPH), complicated by disseminated intravascular coagulation (DIC), is one of the leading causes of maternal morbidity and mortality. 1 The DIC related to preeclampsia, uterine atony, or placental abnormalities accounts for 31.1% of maternal deaths in Japan. 2 The standard treatment for obstetric-related DIC includes correcting the coagulopathy and consumptive processes with fresh frozen plasma (FFP) and platelet transfusion, as well as administration of a synthetic protease inhibitor, heparin, and antithrombin III concentrate. 3
Recombinant human soluble thrombomodulin (rTM) is an endothelial anticoagulant cofactor, composed of the active extracellular domain of thrombomodulin, that inactivates the coagulation cascade by binding to thrombin. 4
The rTM was recently shown to be superior to low-dose heparin in a phase III, randomized, double-blinded clinical trial for the treatment of DIC associated with hematologic malignancies and infections. 5 This has also been demonstrated in other nonrandomized trials. 6,7
At present, there are few case reports describing the use of rTM for obstetric-related DIC, 8 and the efficacy and safety of rTM have not been established in this setting. The present study compared the clinical parameters of obstetric patients admitted with obstetric-related DIC to a single tertiary center and treated with rTM, with those of similar obstetric-related DIC patients not treated with rTM.
Materials and Methods
This retrospective observational study reviewed the clinical profiles of 36 patients with PPH complicated by DIC who were admitted to the Tohoku University Hospital, Sendai, Japan, from April 2006 to May 2011. The first 26 patients were treated without rTM (control group), and the next 10 consecutive patients were treated with rTM (380 U/kg per d, rTM group). Parameters including blood loss, coagulation parameters, and laboratory indices were evaluated.
This study was carried out in accordance with the principles of the Declaration of Helsinki. The ethics committee of Tohoku University permitted this retrospective study and did not require informed consent.
The PPH was defined as blood loss over 500 mL after a vaginal delivery and over 1000 mL after a cesarean section. 9 All patients fulfilled the criteria of the International Society on Thrombosis and Haemostasis classification for overt DIC, 10 and the improvement in DIC was evaluated by the scoring system established by the Japanese Ministry of Health and Welfare (JMHW DIC criteria; Table 1 ). 11
The Diagnostic Criteria for DIC by the Japanese Ministry of Health and Welfare (JMHW) FDP, Fibrin, and Fibrinogen Degradation Products.

Abbreviations: PT ratio, prothrombin time of patients to controls; DIC, disseminated intravascular coagulation; FDP, fibrinogen degradation product.
Recombinant human soluble thrombomodulin (Recomodulin; Asahi Kasei Pharma, Tokyo, Japan), 380 U/kg per d, was drip infused for 30 minutes once daily. The patients were also treated with fresh frozen plasma (FFP), platelet concentrate (PC), red cell concentrate (RCC), or antithrombin (AT)-III concentrate by the physician’s assessment. In the control group, all the patients were treated with gabexate mesilate (GAB) as an adjunctive therapy. Patients in the rTM group were not treated with GAB.
Laboratory studies consisting of a platelet count (Plt), fibrinogen (Fib), fibrinogen degradation products (FDPs),
The posttreatment improvement in DIC was assessed by the JMHW DIC criteria, 12 and the incidence of bleeding symptoms associated with DIC was clinically evaluated on days 1 and 2 by 3 experienced obstetricians. Adverse events related to bleeding were recorded.
Data were expressed as the mean ± standard error of the mean. Continuous variables were compared between groups with Student t test or a nonparametric test where appropriate. The assessments of coagulation markers including the
Results
The baseline clinical profiles and characteristics of the patients are shown in Table 2 . There was no statistical difference in clinical baseline characteristics, total blood loss, and transfusion requirements between the 2 groups (Table 2). There was no statistical difference in the underlying obstetrical complication including placental abnormalities, atonic bleeding, and placental abruption. Hematological findings at baseline were similar between the 2 groups, as shown in Table 2. Cesarean hysterectomies were performed in 5 control cases (19.2%) and 4 rTM cases (40%). The shock index (systolic blood pressure divided by heart rate: shock index) at administration, which corresponded to the severity of PPH, was significantly higher in the rTM group than in the control group (control: 1.1 ± 0.1 vs rTM: 1.5 ± 0.2: P < .05). In the rTM group, recombinant TM (380 U/kg per d) was administered for 3.0 ± 0.6 days. All patients were treated with AT-III concentrate and sufficient blood products including FFP and RCC. Platelet concentrate was required in 13 control cases (50%) and 4 rTM cases (40%; Table 2).
Baseline Characteristics of Patients.
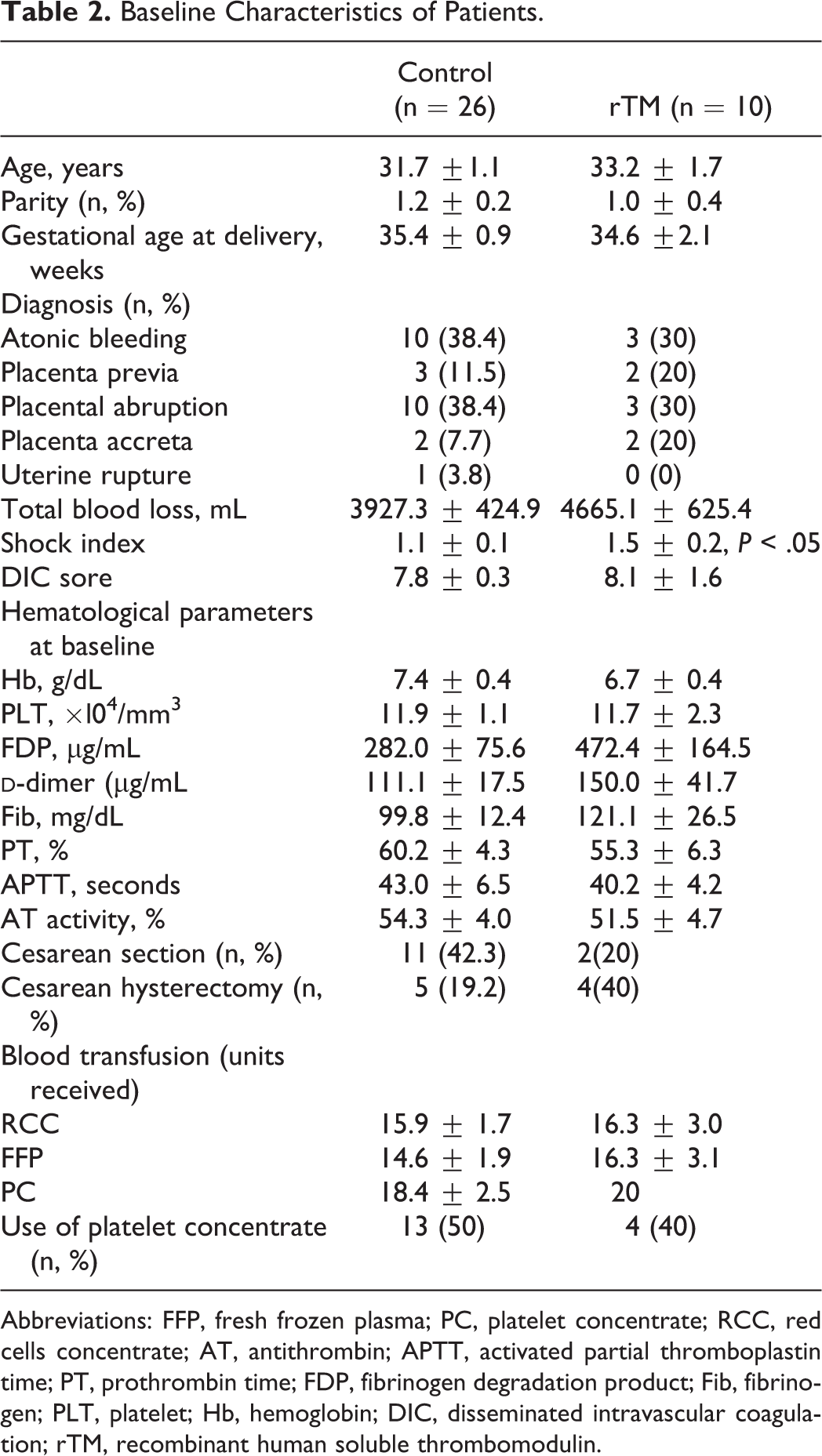
Abbreviations: FFP, fresh frozen plasma; PC, platelet concentrate; RCC, red cells concentrate; AT, antithrombin; APTT, activated partial thromboplastin time; PT, prothrombin time; FDP, fibrinogen degradation product; Fib, fibrinogen; PLT, platelet; Hb, hemoglobin; DIC, disseminated intravascular coagulation; rTM, recombinant human soluble thrombomodulin.
Figure 1
shows the serial changes from the baseline
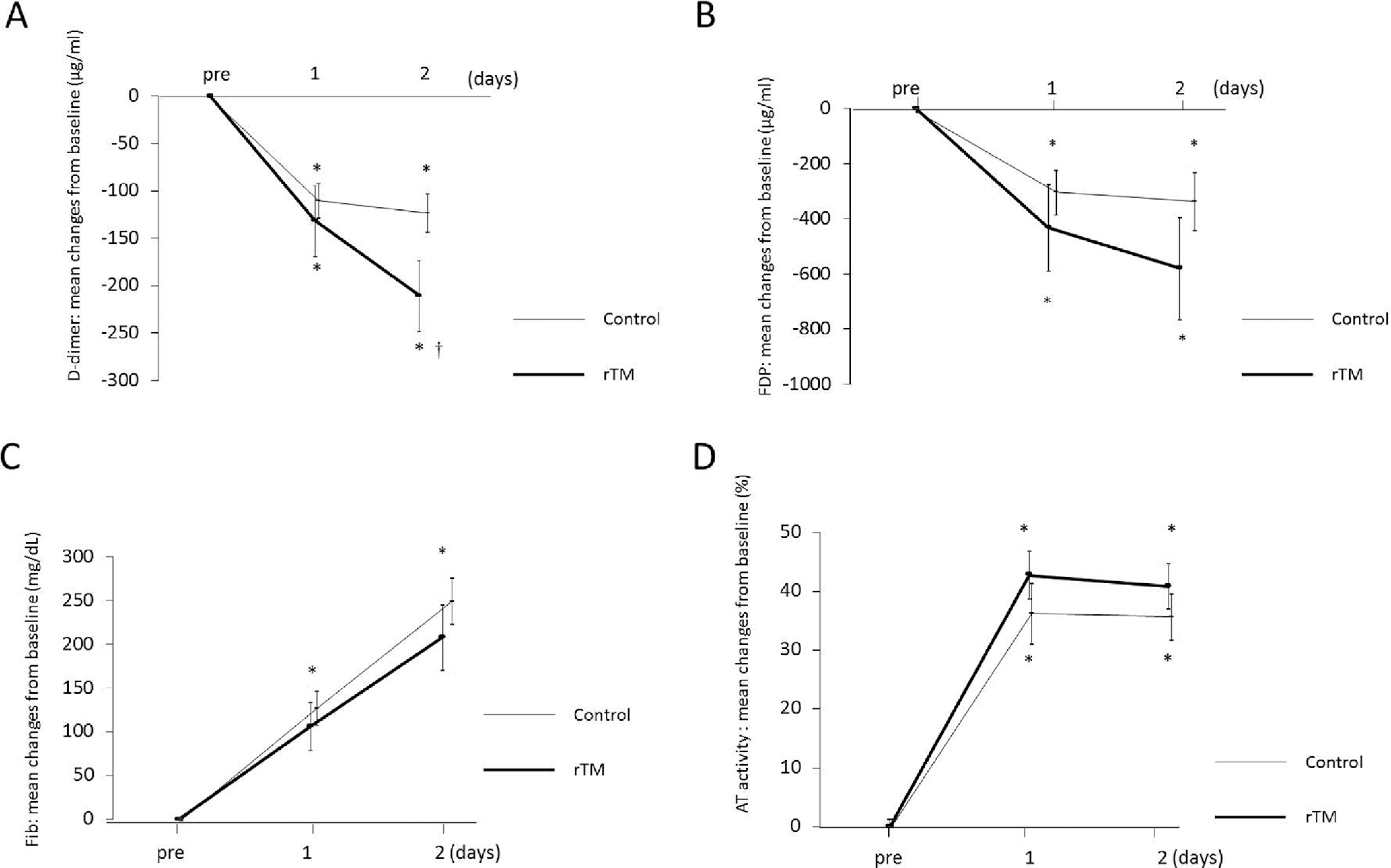
Serial changes in the posttreatment coagulation indices. Data are expressed as group mean ± standard error of the mean. *P < .05 versus baseline †
P < .05 versus control. A,
The DIC score on day 2, assessed by the JMHW scoring system, decreased significantly from baseline for each group; however, the magnitude of the decrease did not differ significantly between the groups (Figure 2 ). The incidence of bleeding symptoms in the rTM group was less than that of the control group; however, the difference was not significant (day 1: 22.2 vs 42.3%, P = .14; day 2: 11.1 vs 19.2%, P = .28; Figure 3 ). No treatment-related adverse events were observed in either group.
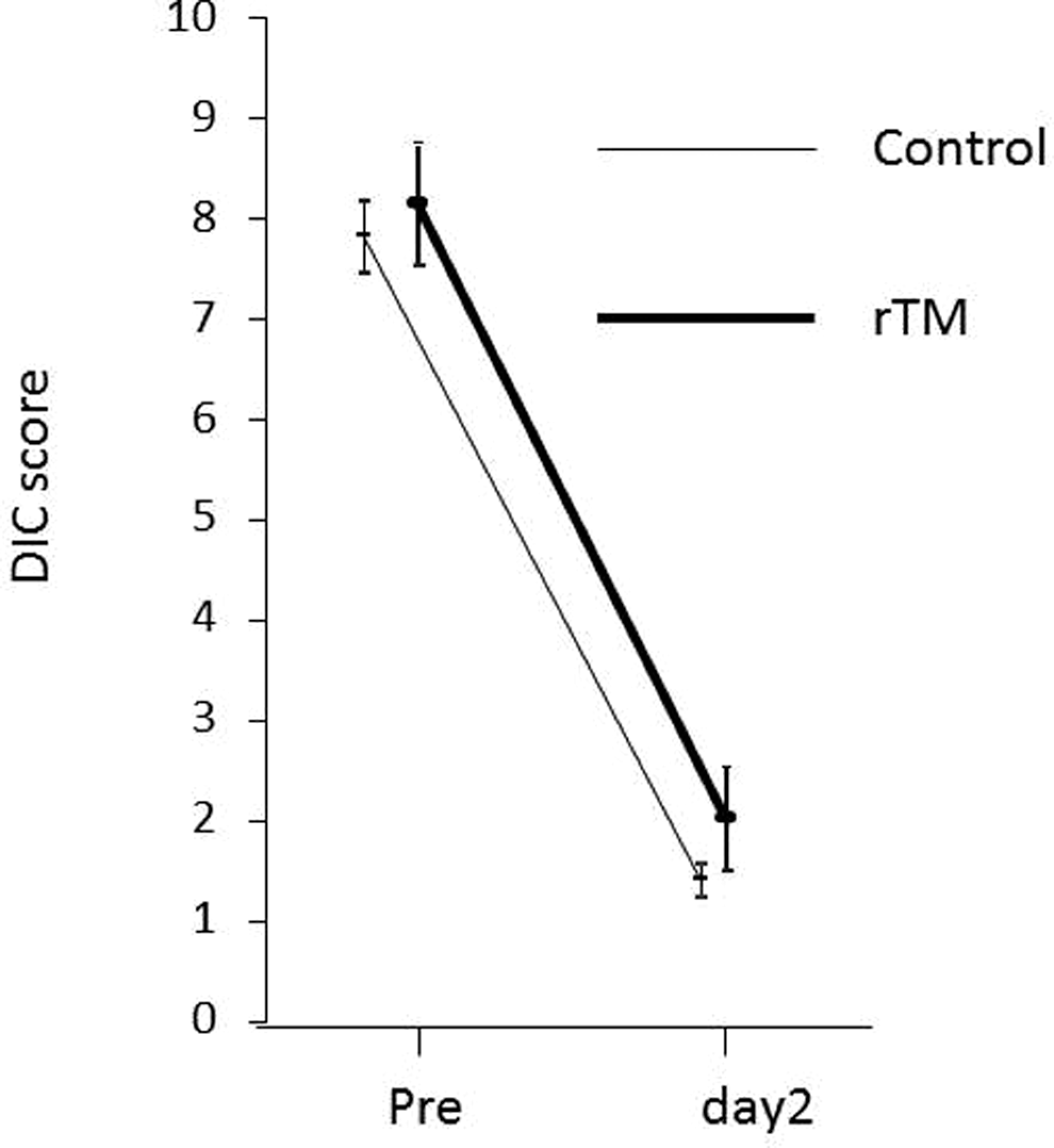
Posttreatment DIC scores. Data are expressed as group mean ± standard error of the mean. The scores decreased rapidly in both groups, although the difference between the 2 groups was not statistically significant. DIC indicates disseminated intravascular coagulation.
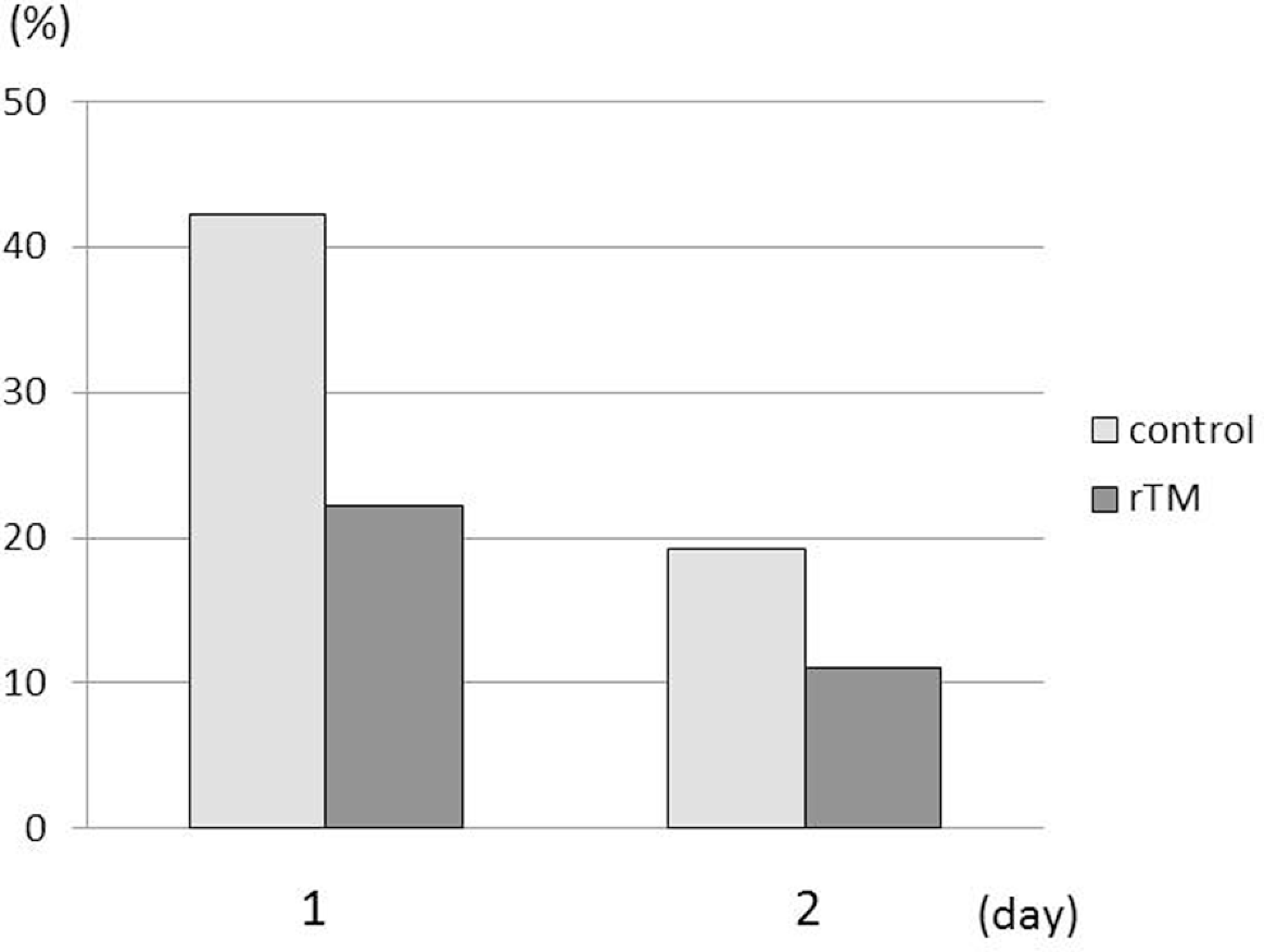
The incidence of bleeding symptoms. The incidence was decreased in the rTM group in comparison with the control, although the difference between the 2 groups was not statistically significant (day 1: P = .14, day 2: P = .28).
Discussion
The present study suggests a potential benefit of rTM in the treatment of DIC related to PPH. In the series of 10 patients, the coagulation studies rapidly normalized following the administration of rTM with no adverse events.
To date, only 1 case report exists describing the use of rTM for the treatment of DIC related to PPH.
8
This study suggested that rTM may also be useful in the setting of Hemolysis, elevated liver enzymes, low platelet count (HELLP) syndrome; however, no reports of its use in the setting of specific complications exists. The present study evaluating the potential benefit of rTM in obstetric-related DIC suggests a benefit with trends toward improvement in bleeding symptoms, DIC scores, and coagulation indices, but there was a statistically significant change only in
We found a nonsignificant increase in AT activity following the administration of rTM. In patients with DIC, AT activity is decreased predominantly by heightened vascular permeability rather than its consumption. 11 Therefore, the improvement in inflammatory changes and a decrease in vascular permeability are critical for the reversal of DIC. The rTM has been shown to reduce capillary leakage through activated protein C-dependent pathways and to stabilize vascular walls in mice. 13 Furthermore, rTM increases AT levels and decreases plasminogen activator inhibitor 1 in patients with DIC, indicating that rTM can counteract vascular injury. 5 With regard to the anti-inflammatory effect of rTM, the N-terminal domain of rTM binds to and inactivates high-mobility group box 1 protein. High-mobility group box 1 protein is a proinflammatory cytokine that stimulates the production of inflammatory cytokines through toll-like receptor 4 and receptors for advanced glycation end products. 14 These results indicate that rTM corrects some aspects of DIC through anti-inflammatory effects.
In the present study, the decrease in
We assessed the clinical efficacy of rTM by investigating the incidence of bleeding symptoms and the DIC scoring system. The incidence of bleeding symptoms was lower in the rTM group than in the control group, and the DIC score rapidly improved to a similar level in both groups. These results indicate that rTM is least comparable to the standard of care.
In the current study, GAB, a synthetic serine protease inhibitor, was not used in the rTM group; however, all control patients received GAB. Therefore, our observations further suggested that rTM showed, at least in part, similar beneficial effects with GAB, which has been widely used as an anti-DIC agents. 15 It is likely that rTM has more potent anti-DIC effects possibly through anti-inflammatory mechanisms.
There were no complications associated with the administration of rTM in the present series. The most significant adverse effect of rTM is bleeding. Unlike heparin, rTM has a long in vivo half-life and there are no blocking agents available at present. Fatal intracranial hemorrhage after the use of rTM has been repeated in a nonobstetric patient with sinusoidal obstruction syndrome. 16 Furthermore, there are no reliable biomarkers with which to measure rTM activity in patients with DIC. Therefore, patients treated with rTM must be carefully monitored for hemorrhage.
In conclusion, we demonstrated that rTM treatment may have benefit over GAB in patients with DIC related to PPH. A prospective randomized, controlled trial is necessary to confirm our findings.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
