Abstract
This study was undertaken to evaluate the impact of progestins as part of low-estrogen (ethinyl estradiol [EE2] ≤35 μg) combined oral contraceptives (COCs) on hemostatic variables. One hundred ninety-five healthy women took oral contraceptives with following formulations: 35 EE2/norgestimate (NGM), 35 EE2/cyproterone acetate, 35 EE2/norethisterone, 30 EE2/levonorgestrel, 30 EE2/drospirenone (DRSP), 20 EE2/gestodene, and 20 EE2/DRSP, for 6 months. Hemostatic assays (prothrombin time, activated partial thromboplastin time, fibrinogen, resistance to activated protein C ratio, protein C, protein S, factor VIII [FVIII], antithrombin, plasminogen, α2-antiplasmin, inhibitor of plasminogen activator type 1 [PAI-1] and
Introduction
There are numerous combined oral contraceptives (COCs) present at the market. Past studies established that COCs with estrogen component lower than 50 μg of ethinyl estradiol (EE2) have lower risk of venous thromboembolic events (VTEs). 1,2 Consequently, new forms of COCs were developed containing 20 to 35 μg EE2 and new progestins that can provide effective contraception with less breakthrough hemorrhaging. Today, the fourth generation of progestins (drospirenone [DRSP]) is used in parallel with first (cyproterone acetate [CPA], norethisterone [NET]), second (levonorgestrel [LNG]), and third generation (gestodene [GSD], norgestimate [NGM], desogestrel [DSG]). However, the risk of VTE, although reduced, is still present because progestins alone or in combination with estrogen are independent risk factor. 3 Estimated odd ratios for the risk of VTE in COCs users differ for progestins from 3.6 for LNG to 7.3 for DSG, compared with nonusers. 4 The effects of progestins, other than pregnancy prevention, are used to treat acne vulgaris, ovarian cysts, metrorrhagias, premenstrual symptoms, regulation of menstrual cycles, and so on. 5 Nevertheless, pharmacokinetics of progestins is not clear, despite their wide use. Most studies have examined progestins in COCs. It is determined that the activity of progestins is mostly dependant on type, dosage, receptor affinity (toward estrogen, progesterone, androgenic, glucocorticoid, and mineralocorticoid receptors), distribution and storage in tissues (especially fat), manner of intake (orally or parenterally), and liver first-pass effect. Cumulative effect is intrapersonal, interpersonal, and ethnic diversity in response to progestins. 6 –9
After lowering EE2, number of studies showed controversial results for the COCs of second and third generation—some pointed the absence of significant difference in thrombogenic tests in the risk of VTE, 10 but others explained, procoagulatory impact on hemostasis and therefore increased risk of VTE, through biological activity of progestins. 11 One of the possible ways is affinity for binding sex hormone binding globulin (SHBG). Specifically, estrogens cause increase in SHBG levels, while progestins do the opposite—decrease in SHBG levels. Therefore, it was suggested that the hemostatic effect of COCs, as cause for increased VTE risk, are presented with sum of estrogenic (from EE2) and antiestrogenic (from progestins) characteristics. 12
In this work, the aim is to determine the effect of COCs with ≤35 μg EE2 and progestins: CPA (2 mg), triphasic NET (50/75/100 μg), LNG (150 μg), GSD (75 μg), NGM (25 μg), and DRSP (3 mg) on hemostasis using tests of coagulation and fibrinolytic pathway, in accordance to the consent committee conclusion that COC with minimal hemostatic changes is considered best option for women 13 and therefore the least risk for VTE development.
Materials and methods
Study Participants
The study included 227 women from the northern part of Croatia, referred by their gynecologists for blood testing on voluntary basis to Cakovec County Hospital, in the period from June 2009 to February 2011. Women should be at least 3 months free from COC use; a parental consent was required for underage participants. Dropout rate was 14%, so 195 participants were included in per protocol analysis. The mean age of the study participants was 23.51 (range 15-43) years, mean body mass index [BMI] was 21.8, and 58 (30%) of the participants were smokers.
Study Design
Study was performed in 3 time points in prospective manner—baseline (before taking of COCs), after 3 and 6 cycles. The gynecologists suggested several COCs from the market to the participants who selected one for use. The participants were asked to fill out a standardized questionnaire concerning their medical history, body height and weight were measured, and blood samples were obtained for laboratory testing. After the third and sixth cycle, participants stated which COC was used.
The following hemostatic assays were performed in all 3 time points: prothrombin time (PT), activated partial thromboplastin time (aPTT), fibrinogen, resistance to activated protein C ratio (aPCR), protein C activity, protein S activity, factor VIII (FVIII) activity, antithrombin activity, plasminogen activity, α2-antiplasmin activity, inhibitor of plasminogen activator type 1 (PAI-1), and
Study protocol was approved by the ethics committee of Faculty of Pharmacy and biochemistry, University of Zagreb and by the local hospital ethics committee. An informed consent in writing was obtained from all study participants.
Samples
Blood samples were collected between 8.00 and 10.00
Assays for Hemostatic Variables
All tests were performed on a Behring Coagulation System (BCS) automated analyzer using reagents manufactured by Siemens Healthcare Diagnostics, Germany, as follows: PT (Thromborel S, reference range 10-15 seconds), aPTT (Actin FS, reference range 23.0-31.9 seconds) and fibrinogen (Multifibren U, reference range 2.1-4.0 g/L) were determined in fresh citrated plasma; FVIII activity (FVIII chromogenic assay, reference range 50%-150%), aPCR ratio (ProC AcR, reference value ratio >1.8), protein C activity (Berichrom Protein C, reference range 70%-140%), protein S activity (Protein S Ac, reference range 59%-118%), and antithrombin activity (Berichrom ATIII reference range 75%-125%) were determined in thawed plasma;
Statistical Analysis
For statistical analysis MedCalc version 12.2.1.0 statistical software was used. Significance for all hemostatic variables were analyzed by COCs group with Friedman analysis of variance (ANOVA) test, overall intergroup analysis was performed with Kruskal-Wallis test, and comparison of 2 COC groups with Mann-Whitney rank sum test.Determined significance level was P < .05. Relative change percentage was calculated according to formula (cycle value − baseline value)/baseline value × 100.
Results
Initial interview and sampling was carried out for 227 women, but 32 of them discontinued the study. Reasons are 21 women prematurely discontinued COCs without any additional explanation, 4 women did not take any dose of oral contraceptives because they changed their minds, 6 women did not follow the protocol correctly (eg, did not present for testing on time), and 1 woman became pregnant before starting COCs. A total of 195 participants finished the whole study.
Study participants were comparable for age and BMI, which did not change significantly by the end of study.
Results are presented as values for hemostatic tests grouped according to COC formulations: 35 EE2/NGM, 35 EE2/CPA, 35 EE2/NET, 30 EE2/LNG, 30 EE2/DRSP, 20 EE2/GSD, and 20 EE2/DRSP. Intragroup and intergroup analysis was performed. Because only a small number of participants have taken 35 EE2/CPA, their results are shown in tables but not commented.
General Tests
The PT values showed a decrease, and they were significantly changed in all groups after third and sixth cycle. Relative change was the highest at 6% in 30 EE2/LNG group after third cycle, but only in the 35 EE2/NET group the values continued to decrease after 6 cycles. Values for aPTT test were also decreased and significantly changed after 3 cycles in all groups, but only 20 EE2/GSD, 35 EE2/NET, and 30 EE2/LNG groups kept the significance of change after sixth cycle. Other groups showed tendency toward baseline values (Table 1).
Assay Values for General Tests (PT, aPTT, and Fibrinogen) in 3 Measurement Points—Baseline, After 3, and 6 Cycles.
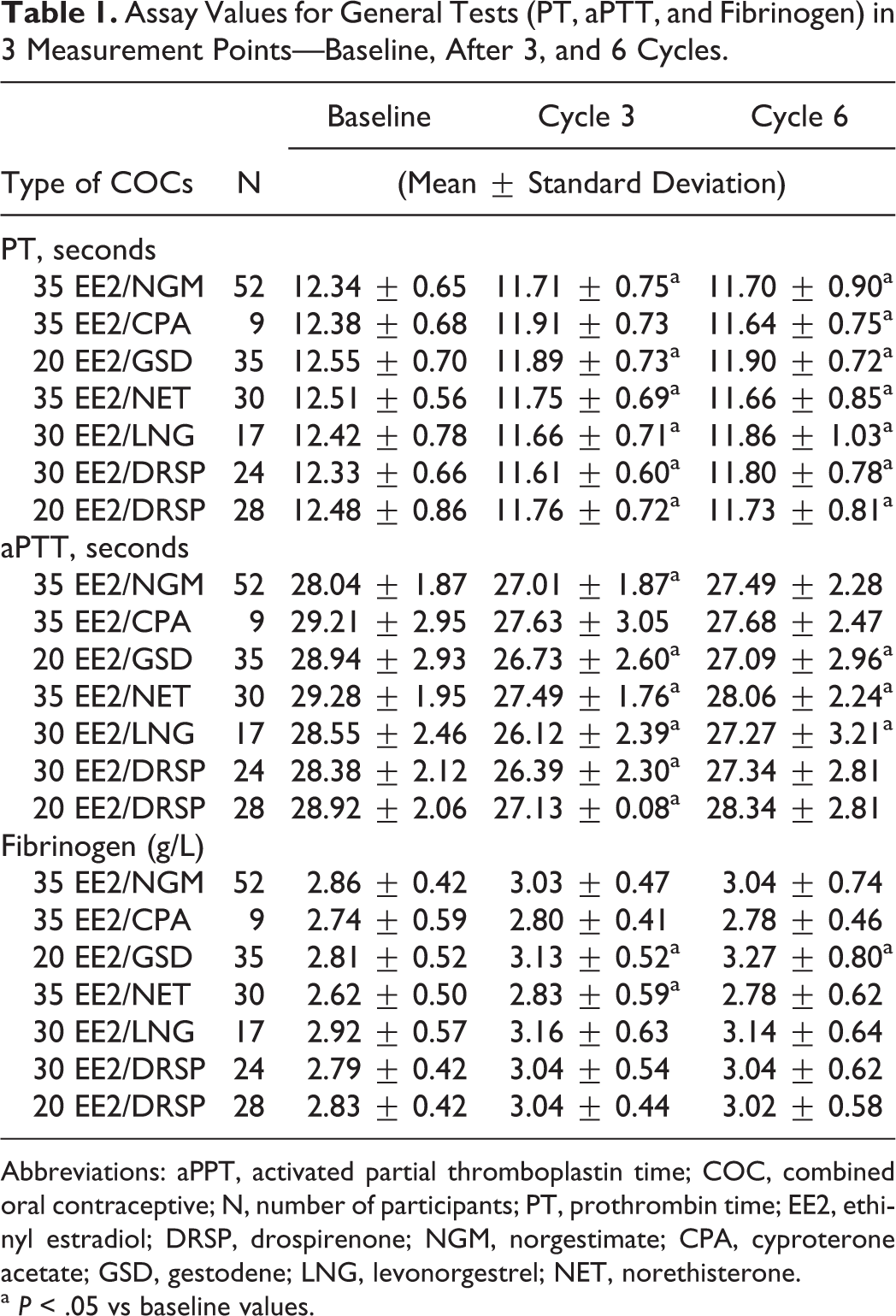
Abbreviations: aPPT, activated partial thromboplastin time; COC, combined oral contraceptive; N, number of participants; PT, prothrombin time; EE2, ethinyl estradiol; DRSP, drospirenone; NGM, norgestimate; CPA, cyproterone acetate; GSD, gestodene; LNG, levonorgestrel; NET, norethisterone.
a P < .05 vs baseline values.
Results for fibrinogen vary between groups. Significant increase after 3 cycles was shown in groups 35 EE2/NET and 20 EE2/GSD, of which only one had significance even after 6 cycles.
Coagulation Pathway Tests
Antithrombin results showed slight but notable decrease in only 2 groups—in 20 EE2/GSD group at both the time points and in 35 EE2/NGM group after the sixth cycle. All other groups of COCs did not present significant change during time of the study (Table 2).
Assay Values for Coagulation Pathway Tests (Antithrombin, Protein C, Protein S, aPCR, and FVIII) in 3 Measurement Points—Baseline, After 3, and 6 cycles.
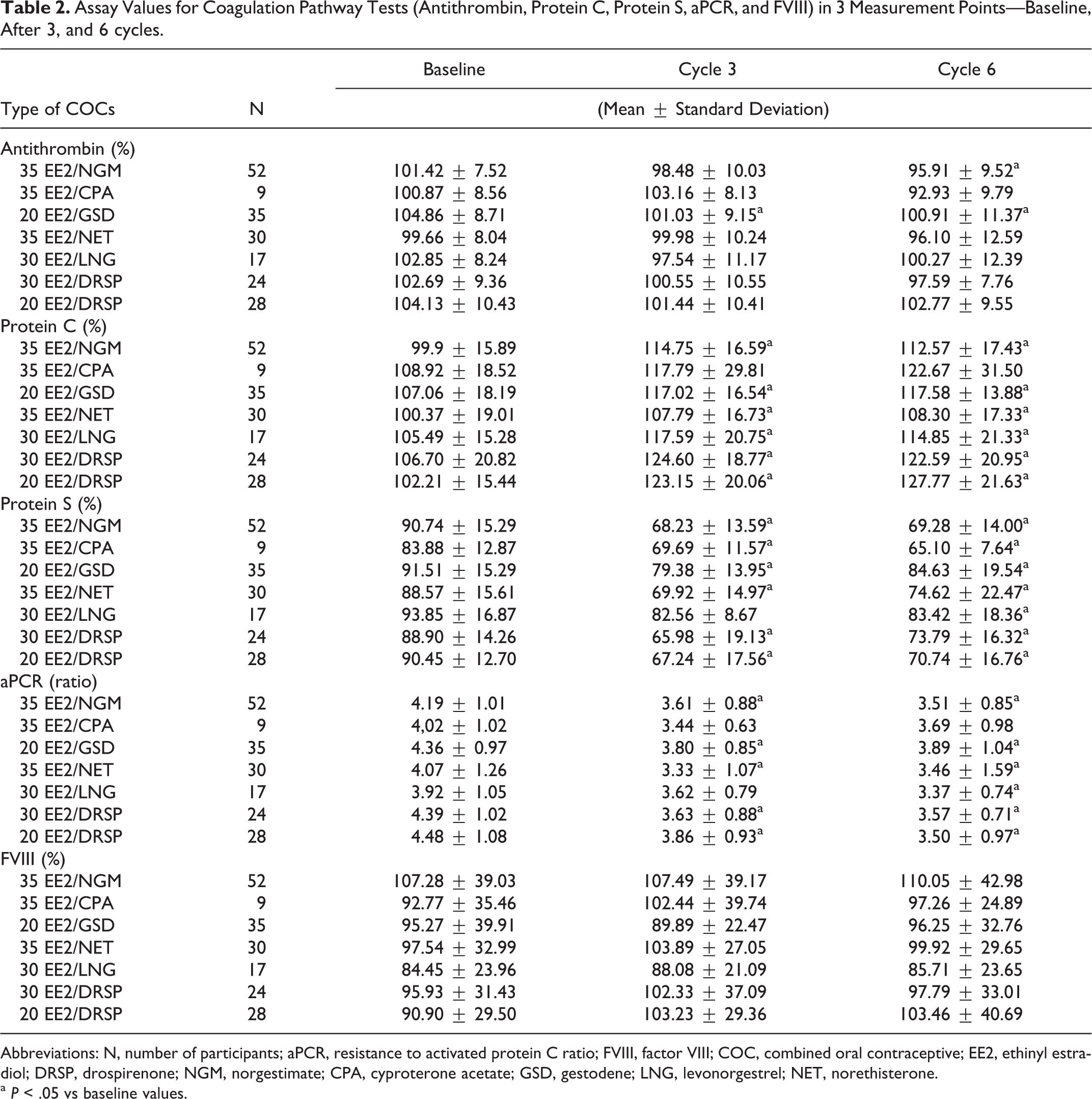
Abbreviations: N, number of participants; aPCR, resistance to activated protein C ratio; FVIII, factor VIII; COC, combined oral contraceptive; EE2, ethinyl estradiol; DRSP, drospirenone; NGM, norgestimate; CPA, cyproterone acetate; GSD, gestodene; LNG, levonorgestrel; NET, norethisterone.
a P < .05 vs baseline values.
Values for protein C activity revealed that all groups of COCs are changed. Elevated values were the most prominently changed for COCs containing DRSP 17% to 20%. Similar are the data for protein S, but here decreased values by 25% were present in COCs with DRSP and NGM. Another difference was shown by 30 EE2/LNG group, which gained significant result after sixth cycle comparing to the baseline time point.
Activated PCR ratio was measured by aPTT-based test. In study it showed significant difference in decreased values for all groups after 3 cycles, except 30 EE2/LNG, which reached significance after sixth cycle, and it had the lowest change among groups by 14%. All COC formulations further lowered values, measured at time point after sixth cycle, except 35 EE2/NET and 20 EE2/GSD groups. For the FVIII activity, none of the groups showed notable increase in values in either time point.
Fibrinolytic Pathway Tests
Increase values were recorded in all COC groups for plasminogen activity, and they stayed significantly changed after 6 cycles. All changes were greater than 25%, but the most prominent was 37% change in 20 EE2/DSP group, which further increased after sixth cycle to 44% (Table 3).
Assay Values for Fibrinolytic Pathway Tests (Plasminogen,
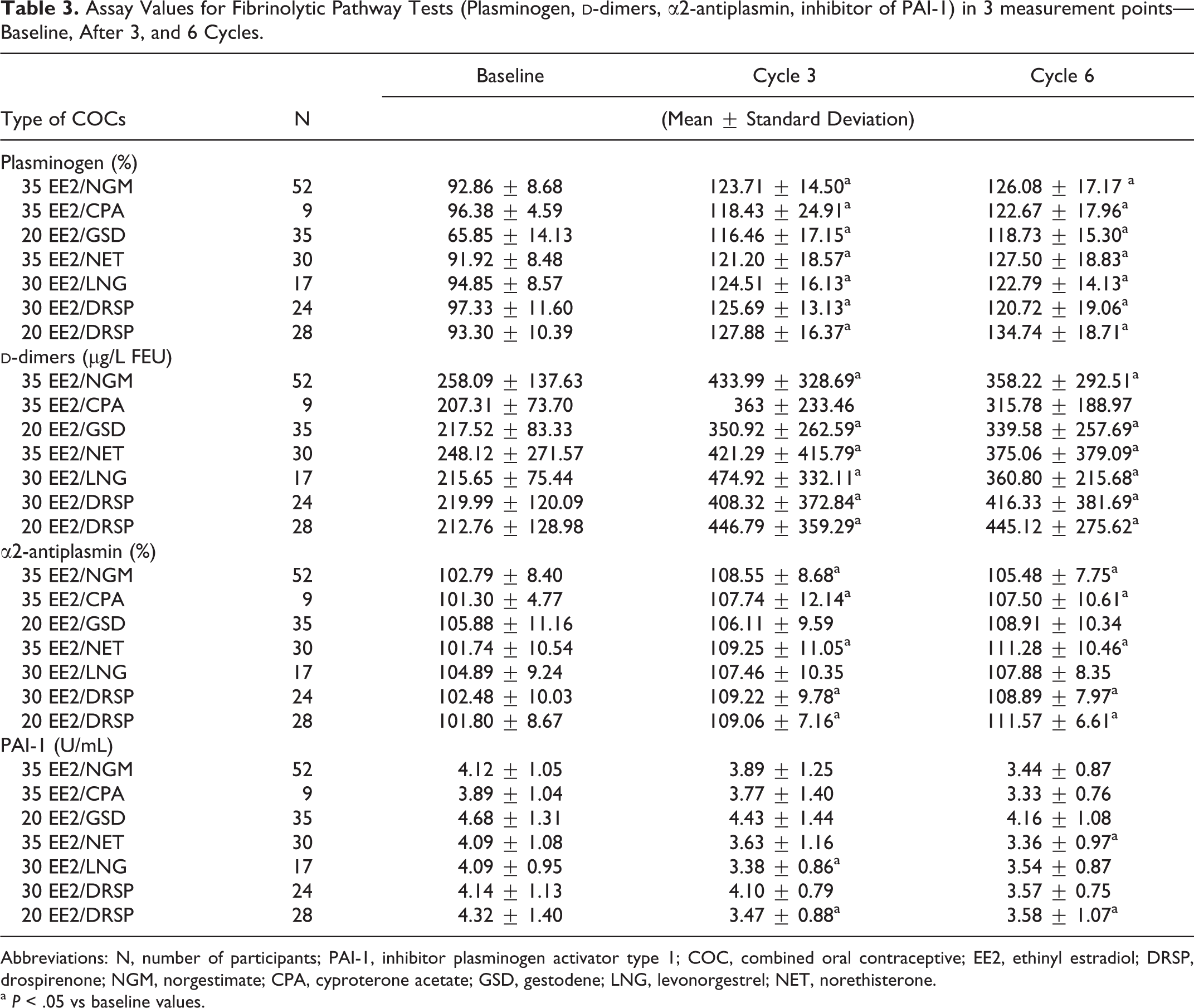
Abbreviations: N, number of participants; PAI-1, inhibitor plasminogen activator type 1; COC, combined oral contraceptive; EE2, ethinyl estradiol; DRSP, drospirenone; NGM, norgestimate; CPA, cyproterone acetate; GSD, gestodene; LNG, levonorgestrel; NET, norethisterone.
a P < .05 vs baseline values.
Most of the groups had significant elevation in α2-antiplasmin activity test values, except in 20 EE2/GSD and 30 EE2/LNG groups. The highest change was after 6 cycles by 9% in 35 EE2/NET and 20 EE2/DRSP groups.
The PAI-1 values were significantly decreased just in 20 EE2/DRSP group in both measured time points. All other groups also showed tendencies to lower the values comparing to baseline but significance was reached for 30 EE2/LNG only after 3 cycles and for 35 EE2/NET group after 6 cycles.
Intergroup Results Comparison
Overall intergroup analysis was performed after 6 cycles of COCs, and they confirmed that, although most of hemostatic tests are influenced by COCs, several of them changed significantly among themselves (Table 4). Significance was present in values for:
Intergroup Analysis of Difference Between Combined Oral Contraceptives (COCs) in Hemostatic Variables Values Measured After Sixth Cycle.
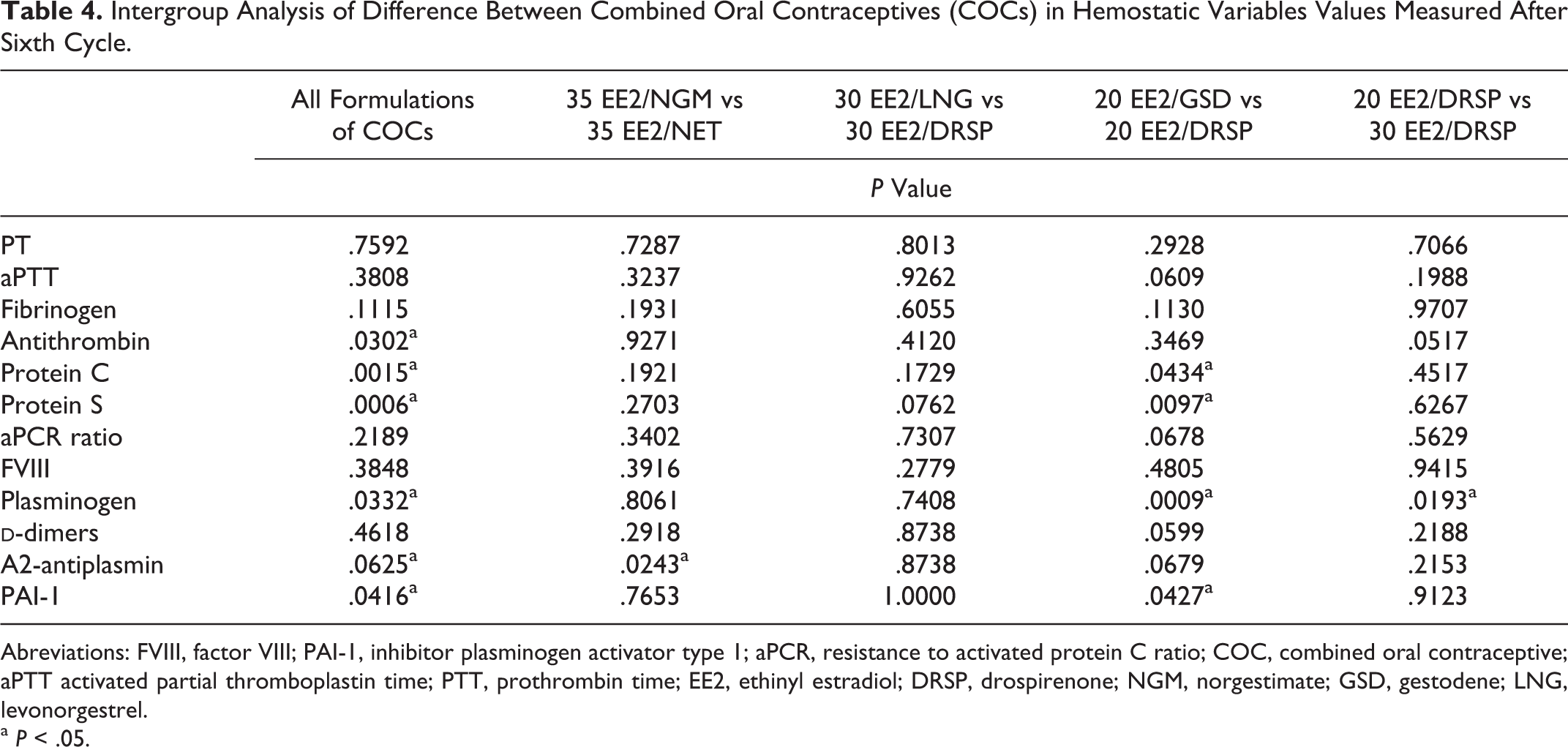
Abreviations: FVIII, factor VIII; PAI-1, inhibitor plasminogen activator type 1; aPCR, resistance to activated protein C ratio; COC, combined oral contraceptive; aPTT activated partial thromboplastin time; PTT, prothrombin time; EE2, ethinyl estradiol; DRSP, drospirenone; NGM, norgestimate; GSD, gestodene; LNG, levonorgestrel.
a P < .05.
antithrombin activity for 20 EE2/DRSP versus 35 EE2/NGM, 35 EE2/NET,
protein C activity for 35 EE2/NGM versus 30 EE2/DRSP, 20 EE2/DRSP, for 35 EE2/NET versus 20 EE2/GSD, 20 EE2/DRSP, 30 EE2/DRSP, and for 30 EE2/LNG versus 20 EE2/DRSP,
protein S activity for 20 EE2/GSD versus 35 EE2/NGM, 35 EE2/NET, 30 EE2/DRSP, 20 EE2/DRSP, and for 30 EE2/LNG versus 35 EE2/NGM, 20 EE2/DRSP, and
PAI-1 level for 20 EE2/GSD versus 35 EE2/NET, 35 EE2/NGM, 20EE2/DRSP.
To establish the difference between COCs, we have made comparison between COCs with the same amount of EE2 or progestins, after 6 cycles. In 35 EE2/NGM versus 35 EE2/NET group significant difference was only in α2-antiplasmin test—P value was .0243.
There was no significant difference between any tests of hemostasis comparing 30 EE2/LNG and 30 EE2/DRSP.
The 20 EE2/GSD versus 20EE2/DRSP showed significant difference in 4 measured tests: protein C, protein S, plasminogen, and PAI-1 with P values .0434, .0097, .0009, and 0.0427, respectively.
Comparison of 20EE2/DRSP versus 30EE2/DRSP gave us insight into hemostatic changes influenced by EE2 amount difference—and the plasminogen activity values were notably different (P value = .0193).
Discussion
Our study confirmed changes in hemostatic variables with low-estrogen COCs. Most of results per group were changed after 3 cycles in PT, aPTT, protein C, protein S, aPCR, plasminogen,
Nineteen participants (9.7%) had protein S values lower than 50%, and most of them were in 35 EE2/NGM and 20 EE2/DRSP groups (7 participants in each group). Significant intergroup difference for 30 EE2/LNG group versus 35 EE2/NGM and 20 EE2/DRSP group supports earlier studies 14,16 that DRSP-containing COCs have similar effect on protein S as the third-generation oral contraceptives like 35 EE2/NGM compared to the second-generation 30 EE2/LNG oral contraceptive.
Analysis between groups of oral contraceptives pointed out difference in protein S, protein C, antithrombin, and PAI-1 values. Decreased protein S levels have an influence on increasing resistance to activated protein C, and it is considered as one of contributing mechanisms for increased risk of VTE. 17 Although we used an aPTT-based aPCR test, which is considered as less sensitive, 18,19 we established good correlation in both measured time points (P < .05) between protein S and aPCR values but not intergroup significant difference.
Protein C activity as part of the same anticoagulant system was significantly changed, and it also presented intergroup difference for example between 35 EE2/NET and DRSP containing COCs, that could be explained with biological activities of progestin and their interaction with estrogen component. 20 Norethisterone has estrogenic and androgenic activity, and it showed the least increase in protein C activity compared to DRSP-containing contraceptives with antiestrogenic and antiandrogenic properties. 6
Decreased antithrombin activity in third generation of COCs shown in previous studies 14,21,22 coincides with our results which showed significant decrease in 20 EE2/GSD in both time points compared to baseline and 35 EE2/NGM after sixth cycle but the values remained within the reference range. However, Winkler et al, 23 established significant decrease in antithrombin activity in 30 EE2/LNG group, which we could not confirmed.
The PAI-1 levels were significantly changed in 20 EE2/DRSP group after 3 and 6 cycles, and 30 EE2/LNG after third and 35 EE2/NET after sixth cycle. Studies so far gave opposite results concerning PAI-1 activity. 23 –26 Estrogen influence on PAI-1 synthesis and its clearance in liver, 27 but polymorphism of PAI-1 locus and environmental impact may have greater influence. 28
Possible distinction between progestins was established by pairwise comparison of COCs containing same amount of EE2. The 30 EE2 group showed no difference between LNG and DRSP; 35 EE2 group showed significant difference between NGM and NET only concerning α2-antiplasmin but the values remained in reference values, and group with least EE2 showed significant difference between GSD and DRSP in protein C, protein S, PAI-1, and plasminogen, though difference found in plasminogen values can be, partly, explained with difference in baseline values. Biological properties of GSD and DRSP are distinct in androgen and glucocorticoid activity as well as high SHBG affinity of GSD, 6 resulting in less prominent changes compared to DRSP. This is particularly found in association of SHBG values and factors of protein C pathway. Elevation in SHBG is associated with increased resistance to activated protein C. Progestin that has more binding affinity for SHBG (such as GSD) reduces the effect of estrogen component in the COC formulation. Drospirenone does not bind to SHBG and therefore does not affect the estrogen effect on the hemostatic system. However, the effect of progestin on the value of SHBG in plasma is only one of the established modes of their action, while the effects of progestin on other steroid receptors and their possible impact on the hemostatic system are not fully understood. 17,29,30
Drospirenone is a novel progestin derived from spirolactone available in market in 2 formulations of 20 EE2 or 30 EE2, with antimineralocorticoid and antiandrogenic properties. 6 Antimineralocorticoid property prevents transactivation of mineralocorticoid receptors, thus reducing aldosterone effect—resulting in water and sodium excretion and consequently slight decrease in body weight and blood pressure. 31 Antiandrogenic property potentiates the use of DRSP for treatment in women with acne. However, DRSP has no binding affinity for SHBG, and therefore it does not decrease estrogen impact on SHBG levels and hemostasis. 12,32 Therefore, the overall thrombotic risk for DRSP formulations depends mostly on the amount of estrogen. Comparison of 2 formulations in our study showed notably increased plasminogen activity for 20 EE2/DRSP, suggesting its higher profibrinolytic response. None of other hemostatic variables were significantly changed. According to a study by Kluft et al, 24 only significant difference between 2 oral contraceptives persisted in protein S activity.
Factor VIII activity is only a hemostatic variable that was not changed by any type of oral contraceptives. That implies a different route for increasing resistance to activated protein C, in aPTT-based aPCR test.
Conclusion
Combined oral contraceptives represent sum of estrogen and specific progestin properties resulting in different disturbances of hemostatic variables affecting both coagulation and fibrinolytic pathways. Significance of these changes, within reference range, to increased risk of VTE is still unclear. In this study, we did not establish any major difference among COC formulations. The major difference in laboratory testing was shown in
Our findings also suggest slightly favorable hemostatic profile of 20 EE2/GSD over 20 EE2/DRSP. More studies comparing DRSP formulations are needed.
Footnotes
Authors’ Note
The study was supported by Siemens Healthcare Diagnostics GmbH, Austria.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
