Abstract
Keywords
Introduction
L-Asparaginase is an important component of therapy for acute lymphoblastic leukemia (ALL). However, its use is associated with a significantly increased risk of thrombosis, with events mainly occurring during remission induction. 1 The reported incidence is between 1.7% and 36.7% depending on the individual study protocol and whether asymptomatic events are included. 2,3 The cerebral veins are a disproportionately overrepresented site of thrombosis. 4 A meta-analysis of 1752 patients from 17 prospective paediatric ALL trials demonstrated a rate of symptomatic thrombosis of 5.2%. 5 Of these 91 events, 53.8% were in the central nervous system (CNS), 42.8% in the non-CNS venous thrombosis, and 3.3% unspecified. Cerebral vein thrombosis (CVT) is therefore not a rare complication during ALL induction therapy. It is a difficult problem to manage with a risk of mortality and long-term disability. In addition, the HOVON-37 trial in adults demonstrated that patients with venous thromboembolism (VTE) in cycle 1 had a significantly poorer complete remission rate (adjusted odds ratio [OR] 0.18; 95% confidence interval [CI] 0.07-0.50), particularly patients with CVT (adjusted OR 0.17; 95% CI 0.04-0.65). 6
The pathogenesis of thrombosis is complex. A combination of the disease itself, steroids, chemotherapy, and profound asparagine depletion by
Although several clinical trials have reported the rate of thrombotic complications, there has not yet been detailed analysis of the clinical features, laboratory data, management, and outcome of individual cases, particularly cerebral thrombosis. Therefore, we performed a retrospective review of all the cases of cerebral thrombotic complications associated with
Results
Patient characteristics are shown in Table 1. Eight patients had CVT and 2 patients had CNS arterial ischemic events. Although it has not been possible to extract total numbers of patients treated at these centers over the exact 5-year period, we can estimate the incidence of CNS thrombotic complications from the average annual numbers of patients treated intensively with ALL at these centers (50 at center 1, 12 at center 2, 10 at center 3, and 5 at center 4). Therefore, over any 5-year period, an average total of 385 patients with ALL are treated at these 4 centers (135 adults and 250 children). The incidence of CNS thrombotic complications therefore is approximately 3% (6% adults and 1% children).
Patient Demographics.a
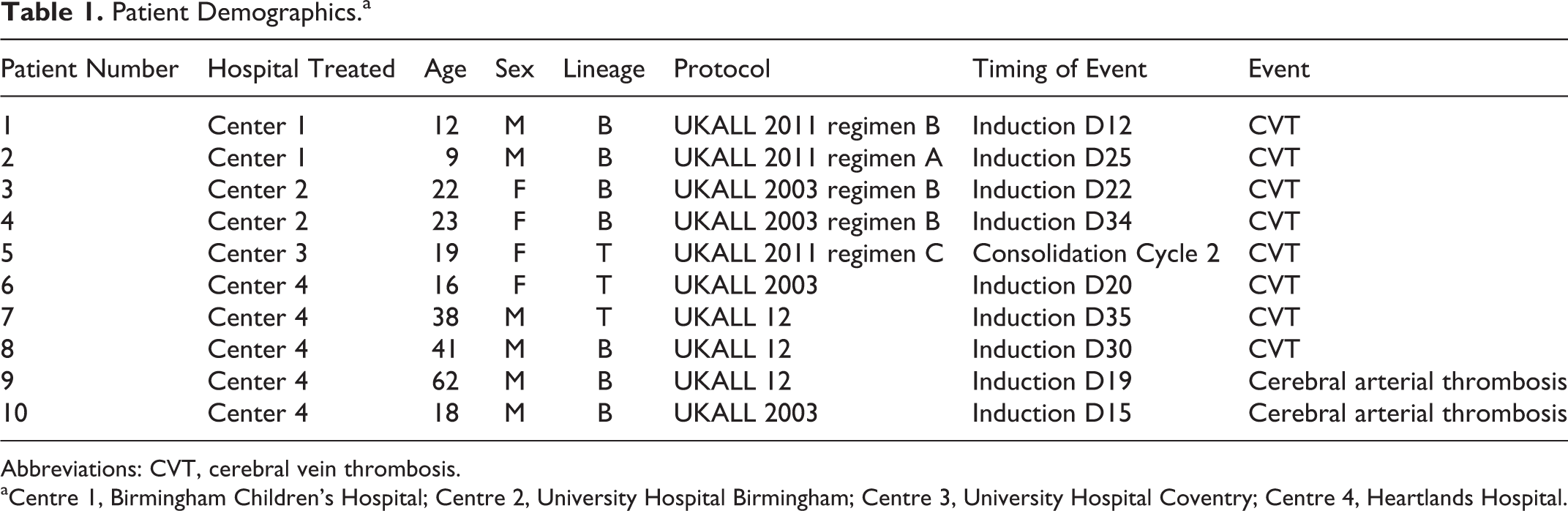
Abbreviations: CVT, cerebral vein thrombosis.
aCentre 1, Birmingham Children’s Hospital; Centre 2, University Hospital Birmingham; Centre 3, University Hospital Coventry; Centre 4, Heartlands Hospital.
Six patients were male and 4 were female. The age range was 9 to 62 years (median 20.5 years) and 7 of 10 patients were less than 25 years.
In 9 of 10 patients, thrombosis occurred during the first 35 days of the onset of therapy (induction remission). The other CVT occurred in the consolidation cycle 2 (
Clinical and Laboratory Features
Table 2 summarizes the clinical presentation and laboratory findings of patients. Patients presented with one or more of headache, seizures, reduced consciousness, confusion, and focal neurology. All patients had a platelet count above 75 × 109/L at the time of thrombosis. Of the 7 evaluable patients, 4 patients had normal antithrombin levels (0.75-1.40 IU/mL), 2 patients had borderline low levels (0.70-0.75 IU/mL), and 1 patient had a significantly reduced level (<0.70 IU/mL). All patients had normal prothrombin time (PT or international normalized ratio) levels. Three patients had shortened partial thromboplastin time (PTT) <27 seconds (2 CVT and 1 arterial ischemia). One patient with arterial ischemia had prolonged PTT >35 seconds (38 seconds). The other 6 patients had normal PTT results. Four patients had low fibrinogen levels (<1.5 g/L). The other patients had normal results (1.5-4 g/L). Central lines were present in 4 of the 10 patients.
Clinical and Laboratory Parameters.
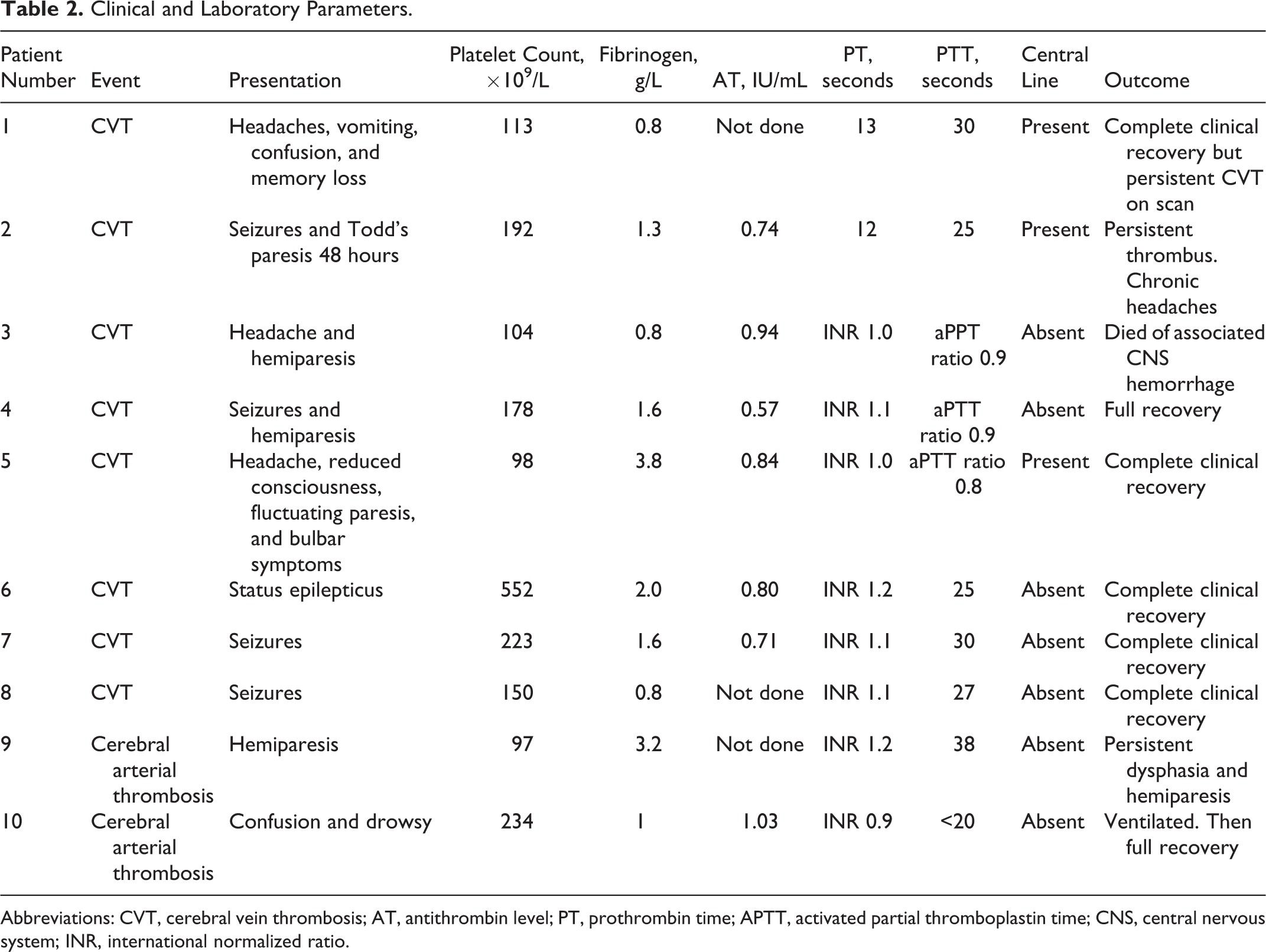
Abbreviations: CVT, cerebral vein thrombosis; AT, antithrombin level; PT, prothrombin time; APTT, activated partial thromboplastin time; CNS, central nervous system; INR, international normalized ratio.
Radiological Findings
Of the 8 patients with CVT, 5 had varying degrees of associated bleeding prior to the therapy. One of these had evidence of mass effect with increased intracranial pressure. In 4 patients who initially had a plain computed tomography (CT), 2 scans were normal, 2 revealed hemorrhages only and detection of thrombosis required further imaging (CT venogram or magnetic resonance (MR) venogram). In 1 patient with arterial thrombosis, CT head was normal and MR imaging revealed left carotid occlusion. In the other patient, CT revealed bilateral thalamic ischemia.
Therapy
All 8 patients with CVT, including those with intracerebral bleeding, were anticoagulated with heparin. Seven patients were treated with low-molecular-weight heparin (LMWH) at various doses. In the other patient with mass effect and increased intracranial pressure due to bleeding, intravenous unfractionated heparin was initially used (commenced without an initial bolus and at a rate to maintain the activated PTT ratio between 1.5 and 2.5) with fresh frozen plasma (FFP) to maintain the fibrinogen >1 g/L. She was later changed to LMWH (enoxaparin 40 mg twice daily). Only 1 patient received antithrombin concentrate (levels were maintained >0.75 IU/mL with ad hoc doses of antithrombin between 1000 u or 2000 u). No patient received neurosurgical intervention. The arterial events were managed conservatively without antiplatelet therapy or anticoagulation.
Outcome
Only 1 of 8 patients with CVT died (this is the patient who had mass effect and increased intracranial pressure). She was initially managed in the intensive care unit and made an temporary improvement and then transferred to the ward but 4 days later rapidly deteriorated with fresh occipital lobe bleeding and increased midline shift (CT report). All the other patients with CVT recovered without obvious neurological deficit, although one has had persistent headaches. One patient with arterial ischemia has been left with hemiparesis and dysphasia and the other made a full neurological recovery.
Discussion
Since the 1980s,
In the United Kingdom national pediatric trial, UKALL 2003, the overall rate of thrombosis has been reported as 59 (3.2%) of 1824 patients
8
with a generally good outcome of CVT and safe, feasible reexposure to
Over this 5-year period, based on average annual rates of ALL at these 4 centers, we estimate the incidence of CNS thrombotic complications was 3% overall (1% in children and 6% in adults). Therefore, this serious complication of treatment is not rare for patients with ALL. These 10 cases highlight some important features of
Second, the platelet count was consistently over 75 ×109/L, suggesting that those most at risk of this complication are eligible for thromboprophylaxis. Third, antithrombin levels were significantly low in only 1 patient, emphasizing the complexity of pathogenesis and contribution of other factors in the etiology. Fourth, low fibrinogen levels are not protective against thrombosis and were present in 4 of 10 patients. Fifth, plain CT is usually inadequate to pick up CVT, and further imaging (CT venogram or MR venogram) should be specifically requested. Sixth and finally, the 8 cases of CVT presented here support the use of anticoagulation in accordance with national guidelines, 9 with 7 of 8 patients making a full neurological recovery.
There are numerous reasons why cerebral thrombotic events may predominantly occur in the induction phase of chemotherapy. For example, the leukemic blasts or other features of the leukemic state (stress response, infection, etc) may contribute to the pathogenesis. Similarly, patients at greatest VTE risk may develop thrombosis when first challenged by asparaginase. Alternatively, fewer patients survive to subsequent rounds of treatment, which may bias the number of events to the induction phase.
Patients with ALL should be assessed for risk of VTE and bleeding in line with guidelines,
10
with the knowledge that asparaginase is an additional risk factor for thrombosis.
1
It is worth considering fastidious thromboprophylaxis in patients considered at low risk of bleeding (including platelet count is >50 × 109/L) for both in- and outpatients during
The majority of patients in our group had relatively preserved antithrombin levels, suggesting that thrombosis would not have been prevented by measuring and replacing antithrombin prophylactically. This approach is also expensive and of unproven efficacy in some studies, 12 while others have shown a modest reduction in VTE. 13 –15 The evidence for prophylactic FFP is also conflicting. 16,6 Therefore, larger randomized controlled trials will be required to define the role of these interventions.
In conclusion, as the efficacy of treatment for ALL improves, the prevention and management of toxicities related to therapy will become as important as the management of the leukemia itself. In particular, there is a growing international concern regarding the thrombotic side effects of
Footnotes
Authors’ Note
Dr Hipkins collected data on the patients at Heartlands Hospital. Dr Bradbury collected data on the patients at University Hospital Birmingham and University Hospital Coventry and wrote the article. Dr Eden collected data at the Birmingham Children’s Hospital. We are especially grateful to Dr A. Mumford and Prof D. I. Marks for their helpful comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
