Abstract
Introduction:
Since the introduction of computed tomography pulmonary angiography, isolated subsegmental pulmonary embolism has become a commonly recognized clinical problem, but its clinical relevance remains unclear. The objective of the present study was to evaluate the extent to which the simplified Wells score discriminates between patients with varying levels of risk of complications after presenting with subsegmental pulmonary embolism.
Materials and Methods:
Retrospective cohort study. Patients included had subsegmental pulmonary embolism (1 or multiple emboli limited to subsegmental arteries). Primary explanatory variable was the simplified Wells score, categorized as high (>4) or low (≤4). The primary outcome was time to death or new venous thromboembolism. Kaplan-Meier techniques and Cox regression analysis were used to compare the survival experience of patients with high versus low Wells score with and without adjustment for active malignancy, age, Charlson score, previous venous thromboembolism, and previous major surgery in the last 30 days.
Main Results:
Seventy-nine patients with subsegmental pulmonary embolism were included. Patients with a high Wells score had a 4-fold increased risk of the composite outcome (hazard ratio = 4.2, 95% confidence interval [CI] = 2.0-8.9, P < .001). Other covariates significantly associated with increased risk in univariate analyses included active malignancy, a low serum albumin, and an increased Charlson score. In multivariate Cox regression analyses adjusting for these other factors, a high Wells score remained significant (hazard ratio 5.5, 95% CI 2.4-12.6, P < .001).
Conclusion:
High Wells score is associated with death or new venous thromboembolism during follow-up among patients with subsegmental pulmonary embolism.
Clinical trial registration:
ClinicalTrials.gov number, NCT01372514.
Introduction
Since the introduction of computed tomography pulmonary angiography (CTPA) for the diagnosis of pulmonary embolism (PE), 1–2 isolated subsegmental PE (SSPE) has become a commonly recognized clinical problem (accounting for 5% to 15% of all PE cases). 3 –5 Despite this, the clinical relevance of SSPE remains unclear. 6 The majority of studies that have addressed this question have suggested that SSPE is associated with a low risk of complications during subsequent follow-up, similar to that of patients with suspected PE and negative diagnostic studies (2% of recurrence risk at 3 months). 7 –9 However, a recent study has shown SSPE to have a risk of complications similar to that of patients with more proximal PE. 10–11 This discrepancy may reflect 2 distinct scenarios, both of which could be at play. First, that patients with SSPE can belong to distinct risk categories based on individual baseline characteristics and clinical severity. Second, much SSPE may be incorrectly classified (ie, false positives) due to the limitations of the diagnostic procedure. 9,12,13
We are unaware of any previous studies that have attempted to stratify the risk of poor clinical outcomes in patients with SSPE. If such a risk stratification measure was available, it could have important clinical implications for treatment. For example, most patients with isolated SSPE are treated with anticoagulation therapy although they might face low risk of new thromboembolic events. Current recommendations on anticoagulation strategies or withholding treatment (eg, in patients with negative leg ultrasound [US], good respiratory reserve, low D-dimer, and absence of classic risk factors) are not evidence based. 6,5,14 The Wells score was designed to stratify patients with suspected PE according to their underlying risk of acute PE. 15 However, given this pretest stratification, CTPA false-positive results among patients diagnosed as having SSPE could be higher in patients with low pretest probability. These patients might therefore face lower risk of poor outcomes during follow-up.
We report on a cohort study of 79 patients with SSPE to evaluate the extent to which the simplified Wells score 15 discriminates between patients with varying levels of risk of death and new venous thromboembolism (VTE). Our overall goal was to identify patients who might benefit from a distinct treatment strategy (eg, initiating or withholding anticoagulation therapy).
Materials and Methods
Study Design
We conducted a cohort study among patients with SSPE who were attending a tertiary teaching hospital in Buenos Aires, Argentina. This study was conducted in accordance with the amended Declaration of Helsinki. The study protocol was approved by the hospital’s ethics review board (protocol number: 2212), and all patients included in this analysis provided informed consent.
Study Population
Using an electronic registry (ClinicalTrials.gov number, NCT01372514) of all patients who present with suspected VTE in any hospital department (including outpatient clinics, inpatient general wards, and critical care units), we identified 4800 consecutive patients aged 18 years or older who presented to the Hospital Italiano de Buenos Aires with suspected VTE between June 2006 and August 2013 (Figure 1). Of these, 2108 had newly diagnosed VTE defined as new deep vein thrombosis (DVT) confirmed by leg US or PE confirmed by either ventilation/perfusion scans or CTPA. Of these patients, 839 had PE and 79 presented with isolated SSPE confirmed by CTPA. These latter 79 patients are the focus of this analysis.
Flowchart.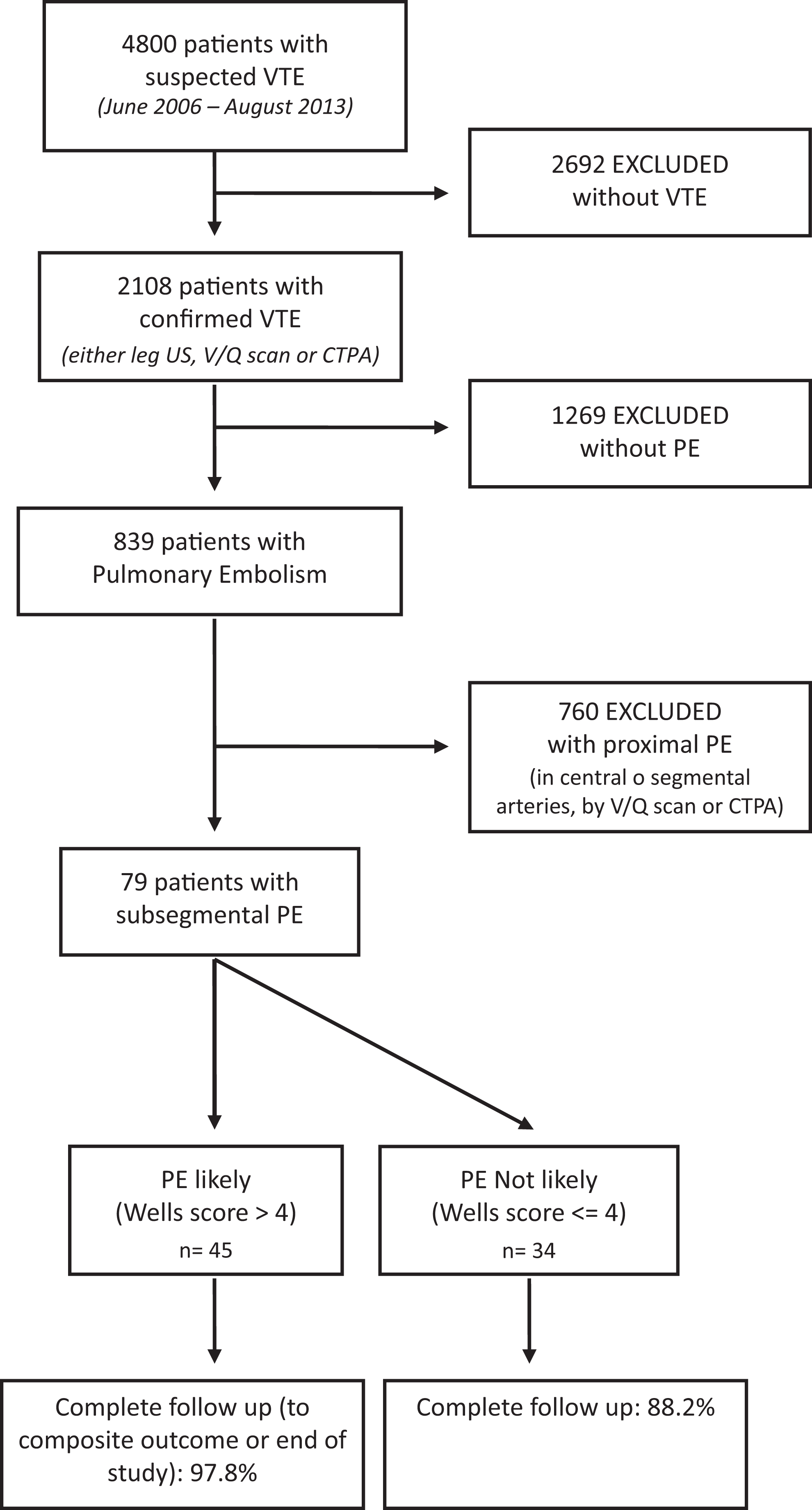
Diagnostic Criteria
Evaluations of CTPA were made by 1 of the 3 trained attending radiologists. Pulmonary arteries were evaluated and embolus localization was classified as central, segmental, or subsegmental. Patients classified as having isolated SSPE had 1 or multiple emboli limited to subsegmental arteries and were included in the present analysis.
Data Collection and Management
A full-time trained research fellow screened all patients and completed baseline evaluation at the time of initial diagnosis and updated the registry database during all follow-up visits. Complete follow-up was defined as follow-up till death, first recurrence of VTE, or study termination.
We used the hospital’s VTE registry to gather baseline information on demographics, clinical history and comorbidities, physical examination, and laboratory and radiological data. We also used the registry to capture information on vital status and new VTE events during follow-up.
Explanatory Variables
Our primary exposure of interest was the simplified Wells score for PE at entry into the cohort, dichotomized as high (>4) or low (≤4). 16,17 This score represents a simple clinical model to calculate clinical pretest of patients presenting with suspected PE. Additional covariates included in the analysis and assessed at study entry were Charlson comorbidity 18 score (a method of classifying comorbidity and estimating risk of death from comorbid disease), age, active malignancy (treatment within previous 6 months or palliative stage), immobilization for 3 or more days within the previous 4 weeks, trauma or major surgery in the past 30 days, any previous VTE, bilateral SSPE, anticoagulation treatment, and pulmonary hypertension at the time of VTE diagnosis. Baseline demographics, past clinical history, and comorbidities were obtained through both patient self-report and electronic chart review. Laboratory and radiological data were extracted from electronic medical records.
Outcome Variables
The primary outcome was time to the composite of death or occurrence of a new VTE during follow-up (defined as new symptomatic DVT or PE by leg US or CTPA, respectively). Major bleeding (defined as fatal or symptomatic bleeding in a critical organ; fall in hemoglobin level of at least 2 g/dL or more, or bleeding leading to transfusion of two or more units of red blood cells or hospitalization) served as a secondary outcome.
Statistical Analysis
Quantitative variables are presented as mean and standard deviation or, for noticeably skewed data, median and interquartile range (25th-75th percentile). Baseline differences between Wells score groups were assessed using Fisher exact test for categorical variables and Mann-Whitney test or Student t test for continuous variables as appropriate. We used Kaplan-Meier techniques to estimate unadjusted survival curves and the log-rank test to compare them. We used Cox regression analysis to compare the survival experience of the 2 Wells score groups after adjusting for covariates that were significant at the .05 level in univariate analyses. For this adjusted analysis, we retained any variable with a P value of less than .10, although we used a P value of .05 to declare statistical significance. Active malignancy was not included in the multivariate modeling since both Charlson and Wells score take this covariate into account.
Results
Baseline Characteristics of Sample.a
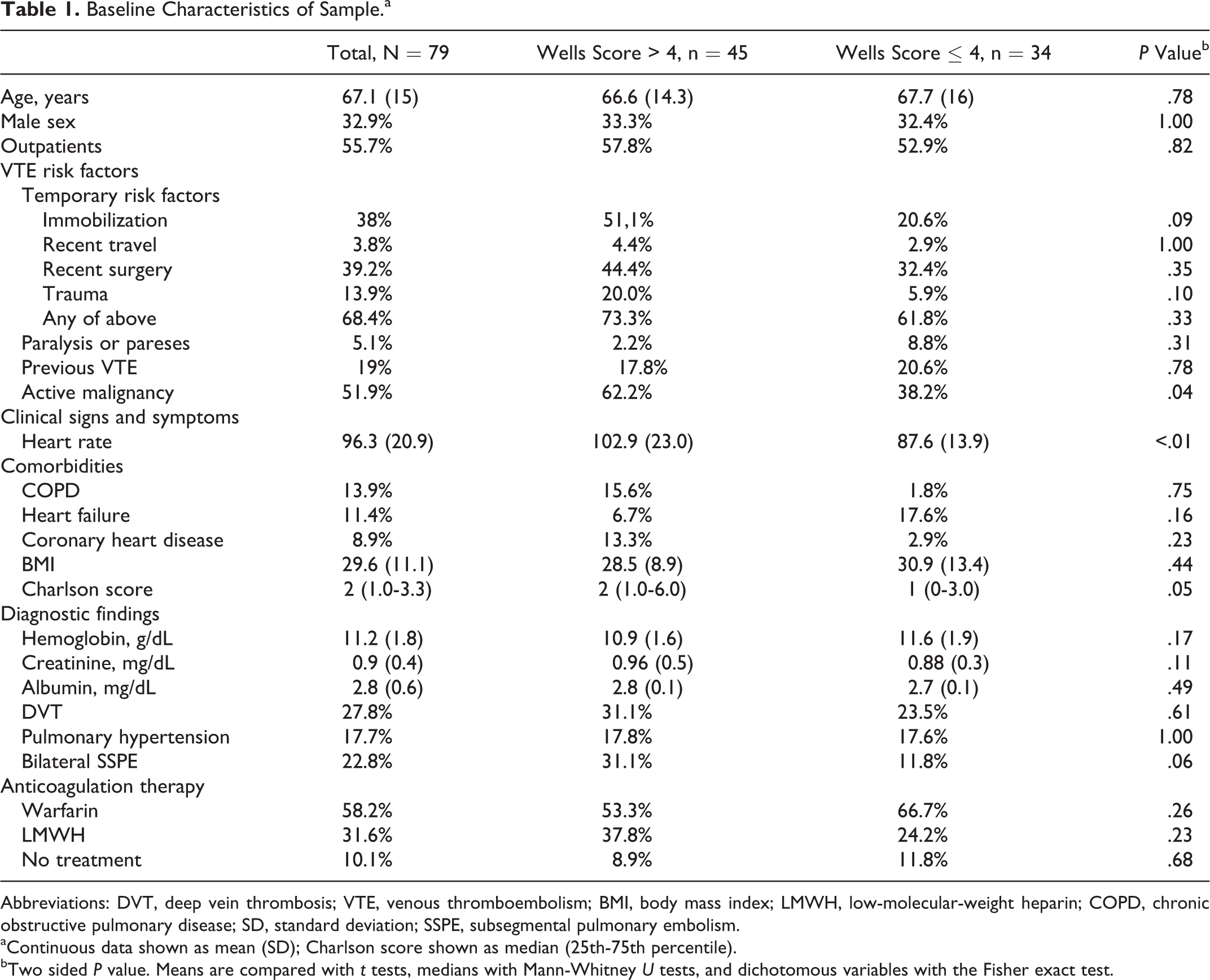
Abbreviations: DVT, deep vein thrombosis; VTE, venous thromboembolism; BMI, body mass index; LMWH, low-molecular-weight heparin; COPD, chronic obstructive pulmonary disease; SD, standard deviation; SSPE, subsegmental pulmonary embolism.
aContinuous data shown as mean (SD); Charlson score shown as median (25th-75th percentile).
bTwo sided P value. Means are compared with t tests, medians with Mann-Whitney U tests, and dichotomous variables with the Fisher exact test.
Follow-up exceeded 88% for both Wells score groups (Table 2). However, the mean duration of follow-up was lower in patients with a high Wells score, presumably reflecting the increased mortality in this group. Consistent with the elevated incidence rate in the high Wells group, recurrence-free survival was significantly higher among patients with low Wells score when compared to patients with a high score (Figure 2).
Recurrence-free Kaplan-Meier survival curves for patients with high or low simplified Wells score. Participant Follow-Up Information. Abbreviation: VTE, venous thromboembolism.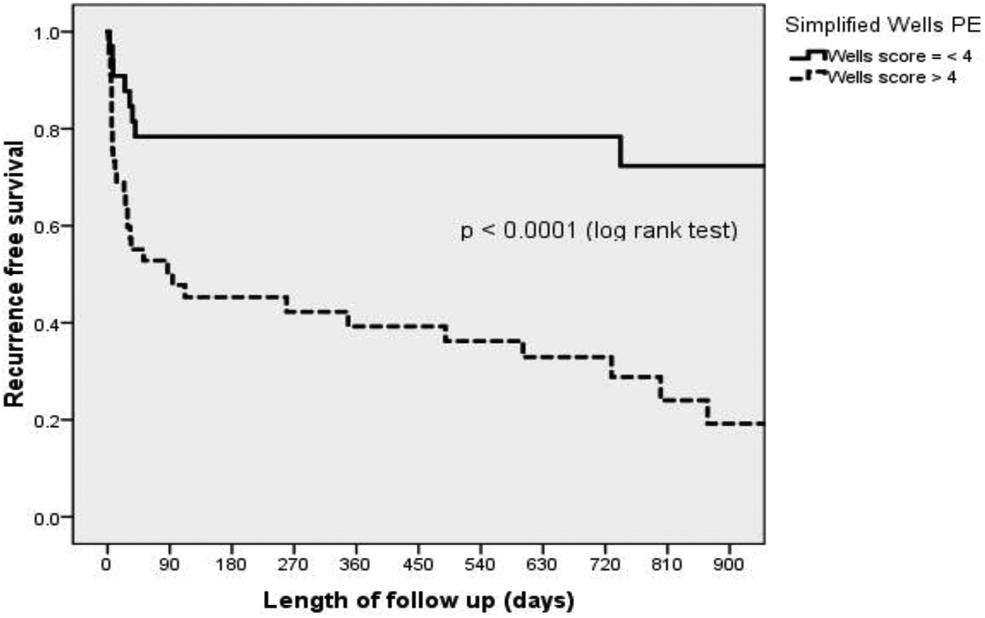
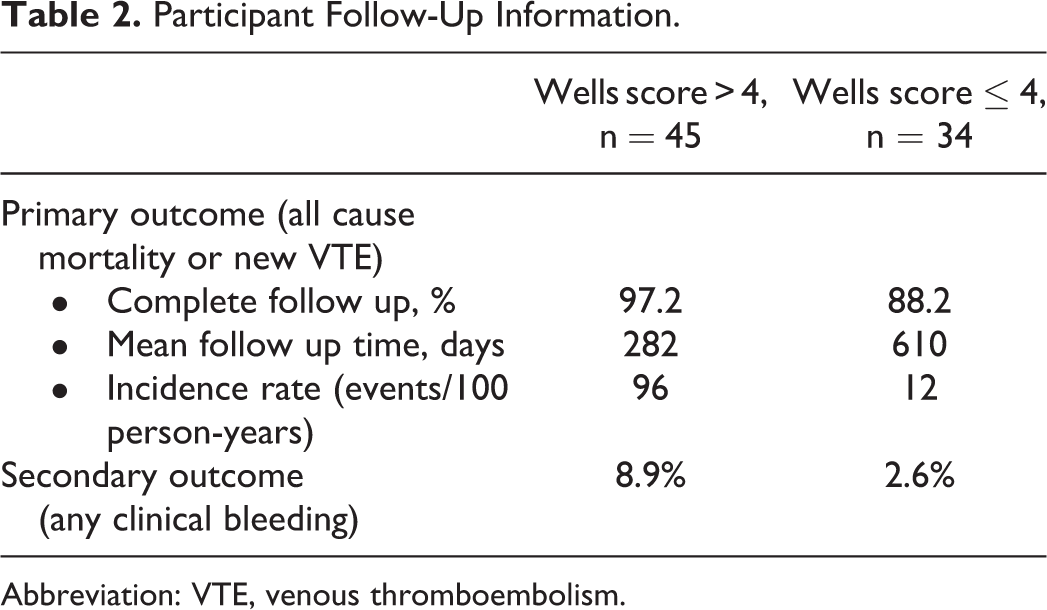
For the Wells score, the hazard ratio for a high versus low score was 4.2 (95% confidence interval [CI] 2-8.9, P < .01) in the univariate Cox regression model. This association persisted in the multivariate model, and the adjusted hazard ratio associated with the simplified Wells score remained significant and actually increased from 4.2 to 5.5, (95% CI 2.4-12.6, P < .001).
Hazard Ratios (HR) and 95% Confidence Intervals for All-Cause Mortality or New VTE During Follow-Up.
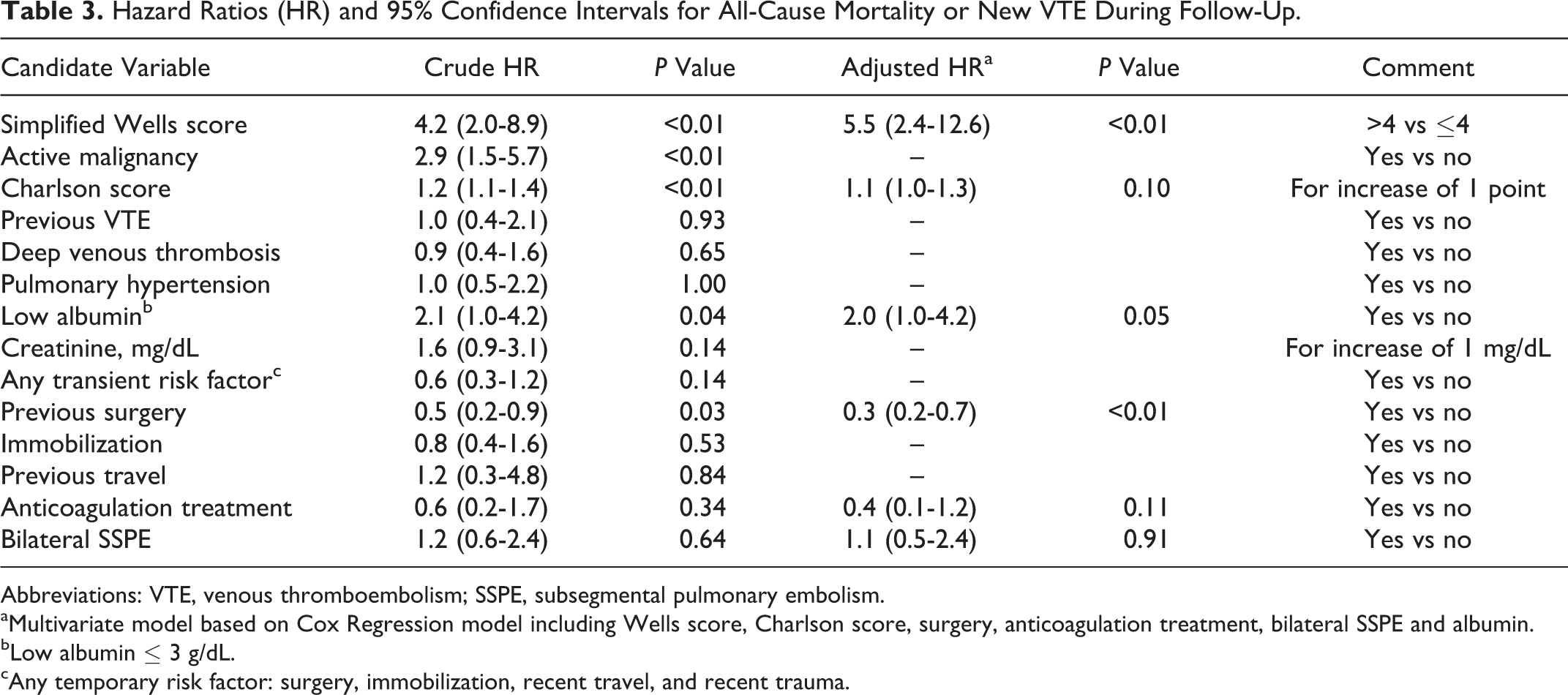
Abbreviations: VTE, venous thromboembolism; SSPE, subsegmental pulmonary embolism.
aMultivariate model based on Cox Regression model including Wells score, Charlson score, surgery, anticoagulation treatment, bilateral SSPE and albumin.
bLow albumin ≤ 3 g/dL.
cAny temporary risk factor: surgery, immobilization, recent travel, and recent trauma.
Finally, since nearly 50% of patients included had cancer, our results could be driven by overall survival. We therefore performed a stratified analysis to evaluate the relationship between the Wells score and the composite outcome of time to death and recurrent VTE in both cancer and noncancer patients (Figure e1, supplementary appendix). In noncancer patients, for the Wells score, the hazard ratio for a high versus low score was 5.1 (95% CI 1.4-19.0, P = .02) in the Cox regression model. Regarding patients with cancer, the hazard ratio for a high versus low score was 2.7 (95% CI 1.1-6.7, P = .03). To further evaluate the impact of survival in our results, we also report overall mortality and risk of recurrent VTE separately (Figure e2, supplementary appendix).
Discussion
Among patients with SSPE, we found that a high Wells score at time of VTE diagnosis is associated with a 4-fold increase in the incidence of death or new VTE during follow-up. This association persisted after adjusting for potential confounders.
As mentioned previously, the clinical relevance of SSPE is controversial. Since CTPA was first used as a diagnostic tool, PE burden, including SSPE incidence, has increased. 19,20 This higher incidence with similar remaining mortality rates has been presented as indirect evidence of possible overdiagnosis. 21 Furthermore, past retrospective studies have suggested that the risk of complications related to SSPE during follow-up was low and even proposed a conservative strategy consisting of withholding anticoagulation in this subset of patients. 5 However, den Exter et al 10 have recently shown a different risk profile of patients with SSPE, resembling patients with more proximal PE. Our study findings may shed some light on why this inconsistency is found in the literature. We show that different risk profiles may exist and the Wells score might help in finding patients at low or high risk of complications. This finding underscores the need not only to assess patients baseline characteristics and possible prothrombotic conditions at the moment of SSPE diagnosis but also the need of CTPA interpretation by experienced trained radiologists to detect false-positive results in patients with low clinical pretest. Our study thus supports the possibility that patients with SSPE and low Wells score have “dots, not clots” (eg, small peripheral emboli with low clot burden without any clinical relevance) or even just false-positive results. This warrants further evaluation and eventually withholding treatment strategies, given the possibility that the risk of new events or death during follow-up may be lower than treatment-associated risks and bleeding. On the other hand, patients with a high Wells score at baseline may benefit from anticoagulation treatment resembling patients with more proximal PE due to their higher incidence of new VTE events or death. It has been recently proposed that, in addition to a currently ongoing trial on withholding anticoagulation in SSPE, a predictive score regarding SSPE risk profile could be useful in clinical decision making. 13
Other risk factors that might be associated with poor outcomes among this set of patients are the Charlson score, low albumin, and active cancer. Moreover, patients who present with SSPE after surgery (or other transient risk factor) might face lower incidence of complications during follow-up, likely due to the lower prevalence of a permanent thrombotic risk factor such as cancer. Together, these risk factors might be useful when caring for patients and making clinical decisions regarding treatment or follow-up strategies.
Our study has several limitations. First, our population comes from a single tertiary teaching center in Buenos Aires, making our patients older and with a higher cancer prevalence than previous reports on SSPE. Moreover, the mortality rate we report is higher than commonly recognized for these patients. This potentially affects the generalizability of our results. However, the total number of events in our cohort and time to follow-up analysis make it possible to see differences in groups that might not been recognized until now. Second, CTPA analysis was done by only 1 trained radiologist at the moment of diagnosis and thus inclusion in our clinical registry. Given the documented interobserver variability in interpreting results of this test,
22
this might limit generalizability by not mimicking the variability that exists among readers. Third, information on certain covariates was not readily available in the electronic registry or had missing information and were not included in the analysis. This was particularly true for biomarkers for right ventricle disfunction: sedimentation rate, troponin,
Our study also has numerous strengths. First, although the sample size is small in absolute terms, it is nonetheless one of the largest cohort studies on patients with SSPE to date, making it possible to evaluate several risk factors regarding poor outcomes. Second, the composite primary outcome is clinically relevant and gives useful information when dealing with patients having SSPE and their prognosis. Third, the simplified Wells score is a well-known score that is easy to calculate, usually bedside. This increases the generalizability of our results. Fourth, follow-up was high, with only 4 patients censored before the 3-month threshold. Finally, our study presents a possible way of estimating different complication risks in different patients with SSPE using a simple clinical tool. The ultimate goal of estimating patient’s prognosis is to change management strategies and eventually change nonfavorable outcomes. In this case, adequate risk stratification would enable detection of those patients who would benefit from anticoagulation treatment or a distinct strategy (eg, in patients with low clinical pretest if treatment associated bleeding is greater than incidence of VTE-related complications).
Conclusion
In conclusion, our findings help to shed light on inconsistencies in the literature concerning the prognosis of patients with SSPE. Those with a high Wells score appear to have a prognosis similar to those of patients with more proximal PE and presumably would benefit from anticoagulation treatment. By contrast, patients with a low Wells score are likely to be either misdiagnosed or to have relatively mild disease with a good prognosis. In either case, the best course of treatment for such patients may be just “watchful waiting.” Thus, use of the widely available and reliably calculated Wells score appears to offer a useful tool to guide clinical management of these patients. However, these findings should probably be independently confirmed before being widely adopted into routine clinical practice.
Footnotes
Acknowledgments
The authors (FA, BLF, and MLPM) would like to acknowledge the assistance of the American Thoracic Society’s MECOR training course during the development of this study and the preparation of this manuscript.
Authors’ Note
FA contributed to concept and study design, data analysis and interpretation, and drafted the manuscript. BLF contributed to concept and study design, data interpretation, and critically revised the manuscript. MLPM and DG contributed to participant recruitment, data acquisition, and critically revised the manuscript. FV contributed to data acquisition, study supervision, and critically revised the manuscript. WV contributed to data analysis and interpretation, and critically revised the manuscript. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
