Abstract
Pulmonary embolism (PE) is a common and potentially lethal form of venous thromboembolic disease in ICU patients. A limited number of risk factors have been associated with PE in ICU patients. In this study, we aimed to screen the independent risk factors of PE in ICU patients that can be used to evaluate the patient's condition and provide targeted treatment. We performed a retrospective cohort study using a freely accessible critical care database Medical Information Mart for Intensive Care (MIMIC)-III. The ICU patients were divided into two groups based on the incidence of PE. Finally, 9871 ICU patients were included, among which 204 patients (2.1%) had pulmonary embolism. During the multivariate logistic regression analysis, sepsis, hospital_LOS (the length of stay in hospital), type of admission, tumor, APTT (activated partial thromboplastin time) and platelet were independent risk factors for patients for PE in ICU, with OR values of 1.471 (95%CI 1.001-2.162), 1.001 (95%CI 1.001-1.001), 3.745 (95%CI 2.187-6.414), 1.709 (95%CI 1.247-2.341), 1.014 (95%CI 1.010-1.017) and 1.002 (95%CI 1.001-1.003) (Ps < 0.05). ROC curve analysis showed that the composite indicator had a higher predictive value for ICU patients with PE, with a ROC area under the curve (AUC) of 0.743 (95%CI 0.710 −0.776, p < 0.001). Finally, sepsis, tumor, platelet count, length of stay in the hospital, emergency admission and APTT were independent predictors of PE in ICU patients.
Introduction
Venous thromboembolism (VTE) includes deep vein thrombosis (DVT) and pulmonary embolism (PE). PE is a clinical and pathophysiological syndrome of pulmonary circulatory disturbance caused by various emboli (thrombus, bacterial emboli and so on) blocking the pulmonary artery system. Furthermore, it is the third most common cause of cardiovascular death globally, second only to stroke and myocardial infarction. 1 Although it is well-documented in the general ward, the prevalence of PE in the intensive care unit (ICU), where it is more challenging to diagnose and treat, has been largely understudied. 2
A limited number of risk factors of PE in ICU patients has been identified in previous studies.3–5 Accordingly, the database Medical Information Mart for Intensive Care III (MIMIC-III) was used in the present study to analyze the risk factors for PE in ICU patients. To the best of our knowledge, this is the first study to investigate risk factors for pulmonary embolism in ICU patients based on the MIMIC-III database. We aimed to screen the independent risk factors for PE in ICU patients that can be used to evaluate the patient's condition and provide targeted treatment.
Methods
Data Source
The study is a retrospective cohort study based on data from a freely available intensive care database, Medical Information MIMIC-III. 6 In short, the database includes clinical data for patients hospitalized in the ICU at Beth Israel Deaconess Medical Center between 2001 and 2012. We completed an online course from the National Institutes of Health -- Protecting Human Research Participants (certification number 39861991) to apply for permission to access the database.
Inclusion and Exclusion Criteria
All adult (≥18 years of age) patients were enrolled; however, only patients admitted to the ICU for the first time were selected if they were hospitalized more than once. Besides, patients with ICU stays of less than 24 h during their first hospitalization were excluded. To avoid data entry errors, we excluded patients with a hospitalization duration shorter than the ICU duration stay. Furthermore, to ensure that pulmonary embolism occurred during hospitalization, we excluded patients hospitalized due to pulmonary embolism. Moreover, since the database protects the privacy of patients over the age of 90, the database changed the age of these patients to 300 years old. Therefore, we also excluded these patients. Furthermore, our study did not include patients without laboratory parameters (such as white blood cells [WBC]).
The inclusion criteria consisted of: (1) patients admitted to the ICU within 24 h of admission; (2) patients aged 18 to 89 years old. A flowchart of study participant selection is presented in Figure 1.
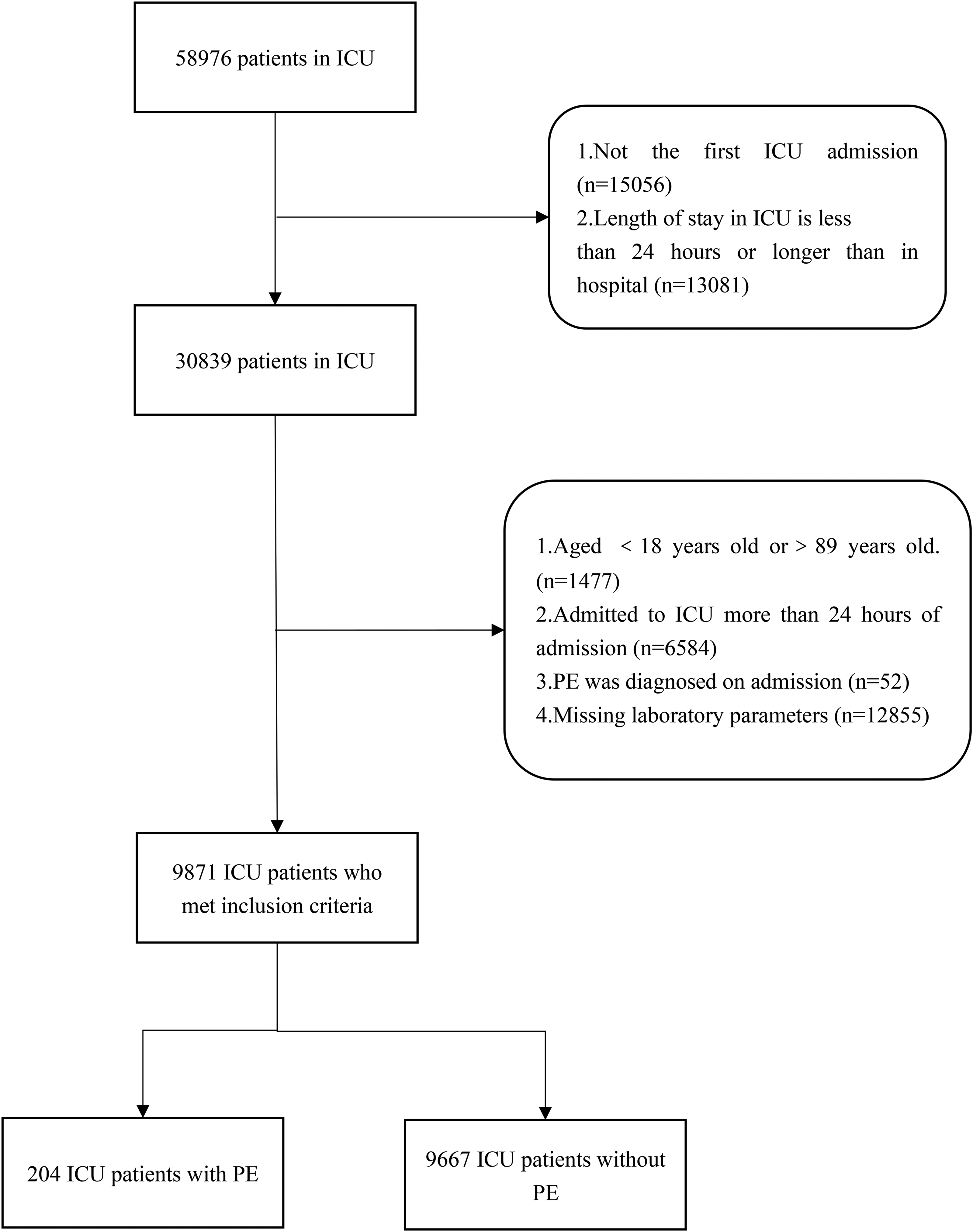
Flow chart of the study participants. Abbreviations: ICU, intensive care unit; PE, pulmonary embolism.
Data Extraction
The data extraction was performed using Structured Query Language and codes from the MIMIC Code Repository (https://github.com/MIT-LCP/mimiccode). PE was identified using the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes. In addition to clinical outcome variables, the following variables were extracted or calculated: age, sex, BMI, ethnicity, type of insurance, type of admission (emergency and non-emergency), the length of stay in ICU (ICU_LOS), the length of stay in hospital (hospital_LOS), alcohol drinking, GCS (Glasgow Coma Scale) and the SOFA (Sepsis-related Organ Failure Assessment) score 7 and laboratory parameters (select the first value during the ICU). Sepsis, fracture, hypertension, tumors, anticoagulants (prophylactic use of anticoagulants such as heparin or warfarin within 24 h after admission to ICU), and central venous catheter (CVC) were also identified using ICD-9-CM codes. All methods were conducted in accordance with relevant guidelines to protect patient privacy.
Grouping
We divided the ICU patients into two groups based on the incidence of PE. Then, we used appropriate statistical methods to analyze risk factors between the two groups.
Statistical Analysis
The data distribution was tested using the Shapiro-Wilk test. Furthermore, we used median (interquartile range [IQR]) or frequency and percentage to describe patient characteristics. A nonparametric test (Mann-Whitney U test or Kruskal-Wallis test) was used for data with abnormal distribution or heterogeneity of variances. Categorical variables were expressed as percentages and analyzed using the Pearson Chi-squared test.
Logistic regression analysis is a generalized linear regression analysis model, which is often used in the automatic diagnosis of diseases. 8 For example, to find risk factors for PE and predict the probability of PE occurrence based on risk factors. Univariate logistic regression was used to determine risk factors for PE. Furthermore, the multivariable logistic regression included the variables significantly associated with PE in the univariate analysis (P < 0.1). Receiver operating characteristic curve (ROC) curves were applied to analyze predictors of PE in ICU patients. All statistics were calculated using SPSS software (version 26.0; SPSS Inc., Chicago, IL, USA).
Results
Baseline Characteristics of the Study Population
A total of 9871 ICU patients were included, among which 204 patients (2.1%) were diagnosed with PE. The baseline patient characteristics and outcomes for the patients are summarized in Table 1. Differences in age, fracture, platelet, RBC (red blood cell), serum inorganic phosphorus, blood glucose, APTT, sepsis, tumor, type of admission, type of insurance, ICU_LOS and hospital_LOS between the two groups were statistically significant. No statistically significant difference in Body Mass Index (BMI), gender, hypertension, alcohol drink, ethnicity, SOFA, serum potassium, serum sodium, serum creatinine (Scr), Blood urea nitrogen (BUN), WBC, GCS, CVC, anticoagulants, international normalized ratio (INR) and prothrombin time (PT) difference was observed between the two groups.
Comparison of baseline characteristics between two groups.

Abbreviations: WBC, white blood cell; RBC, red blood cell; SOFA, Sequential Organ Failure Assessment; INR, international normalized ratio; PT, Prothrombin time; APTT, activated partial thromboplastin time; ICU_LOS, the length of stay in ICU; hospital_LOS, the length of stay in hospital; GCS, Glasgow Coma Scale; BMI, Body Mass Index; CVC, central venous catheter;
Logistic regression analysis for independent risk factors of PE in ICU patients.
The univariate logistics regressions analysis was applied to the baseline variables, laboratory tests and comorbidities. Serum inorganic phosphorus, BUN, Scr, blood glucose, serum sodium, serum potassium, ICU_LOS, hospital_LOS, RBC, SOFA, GCS, platelet, WBC, ethnicity, sepsis, hypertension, types of admission, types of insurance, tumor, alcohol drinking, fracture, CVC, anticoagulants, PT, INR, APTT, sepsis were analyzed during the univariate analysis (Table 2). The significant parameters (p < 0.1): sepsis, hypertension, fracture, tumor, RBC, serum inorganic phosphorus, APTT, types of admission, ICU_LOS, hospital_LOS and platelet were included in multiple logistic regression analysis. The results showed that sepsis, hospital_LOS, type of admission, tumor, APTT and platelet were independent predictors of PE in patients admitted to ICU, with OR values of 1.471 (95%CI 1.001-2.162), 1.001 (95%CI 1.001-1.001), 3.745 (95%CI 2.187-6.414), 1.709 (95%CI 1.247-2.341), 1.014 (95%CI 1.010-1.017) and 1.002 (95%CI 1.001-1.003) (Ps < 0.05) (Table 3).
Univariate logistics regressions analysis of risk factors to ICU patients with Pulmonary embolism in ICU.

Abbreviations: WBC, white blood cell; RBC, red blood cell; SOFA, Sequential Organ Failure Assessment; INR, international normalized ratio; PT, Prothrombin time; APTT, activated partial thromboplastin time; ICU_LOS, the length of stay in ICU; Hospital_LOS, the length of stay in hospital; GCS, Glasgow Coma Scale; BMI, Body Mass Index; CVC, central venous catheter
Multivariate logistics regressions analysis of risk factors to ICU patients with Pulmonary embolism.

Abbreviations: APTT, activated partial thromboplastin time; Hospital_LOS, the length of stay in hospital
Receiver operating characteristic curve (ROC) analysis was performed to assess the predictive value of the composite indicators of PE in ICU patients.
Taking whether the patient had PE as the status variable and hospital_LOS + sepsis + type of admission + platelet + tumor + APTT as test variables of the composite indicator, ROC curve analysis showed that the AUC of composite indicators for the prediction of PE was 0.743 (95%CI 0.710 −0.776, p < 0.001) (Figure 2).

Discriminatory accuracy for predicting ICU patients with PE by receiver operator characteristics (ROC) analysis calculating area under the curve (AUC).
Discussion
PE is a potentially fatal cardiovascular disorder that is common in hospitalized patients. In this regard, it has been established that PE can significantly reduce the quality of life and even increase the mortality of ICU patients. PE-related indicators are essential for early detection and timely treatment. The prevalence of PE in ICU patients was 2.1% in this study. According to our logistic regression model and ROC curve analysis, sepsis, tumor, hospital_LOS, platelet, emergency admission and APTT were independent predictors of PE in ICU patients.
Sepsis is a systemic inflammatory response syndrome caused by bacterial and other pathogenic microorganisms invading the body. 9 Kaplan et al. suggested that sepsis is a risk factor for VTE due to systemic inflammation 10 Moreover, sepsis can cause multiple organ dysfunction, mediated by activation of the coagulation system, which promotes the formation of septic pulmonary embolism (bacterial emboli block pulmonary vasculature).11,12 Furthermore, a study by Levi et al. 13 concluded that sepsis mainly affects three main anticoagulant pathways, antithrombin, the protein C system, and tissue factor pathway inhibitor (TFPI), thus promoting the formation of thrombosis and even pulmonary embolism in the body. Besides, sepsis leads to the formation of septic emboli in the circulatory system. Once the septic emboli become blocked in the pulmonary artery, it can cause a septic pulmonary embolism. Importantly, Chou et al. 14 demonstrated that septic pulmonary embolism significantly increases mortality in ICU patients. Accordingly, important measures must be taken to prevent the occurrence of pulmonary embolism in sepsis patients, including fluid resuscitation and antibiotic treatment. 15 Moreover, experimental studies have shown that heparin can inhibit the clotting activity to a certain extent in sepsis. Hence, using heparin to prevent pulmonary embolism in sepsis patients can be effective. 16 Given that sepsis is a high-risk factor for PE in ICU patients, there is an urgent need for an optimal treatment approach to restore the coagulation function of patients, thus preventing thrombosis and even pulmonary embolism.
A tumor is a mass formed by rapid cellular proliferation due to the loss of normal regulation of local tissue cells on gene-level under various tumorigenic factors. Currently, low molecular weight heparin is recommended to prevent and treat thromboembolism in tumor patients. 17 Some studies have shown that tumors are risk factors for thrombosis due to their complex physiological mechanisms. 18 Tumor cells can activate platelets directly by secreting platelet agonist 5′-diphosphate. Besides, podoplanin is a membrane protein highly expressed in cancer cells, which induces platelet activation by binding to CLEC-2 (C-type lectin-like receptor 2) and promotes thrombosis in cancer patients. 19 Besides, in a multicenter study by Trujillo-Santos et al. an elevated WBC count was found to be a risk factor for venous thrombosis in cancer patients. 20 Khorana et al. 21 showed that platelet count, tumor site, hemoglobin, erythropoietin use, and leukocyte growth factor before chemotherapy were significantly associated with VTE development in cancer patients. Last but not least, Simanek et al. showed that a high platelet count was an independent risk factor for venous thrombosis in cancer patients. 22 The above findings suggest that clinicians should pay more attention to the pulmonary condition of cancer patients in the ICU.
In our study, the PE group had a statistically significantly higher proportion of emergency admissions than the normal group (92.6% vs 72.8%, p < 0.001). Patients admitted to emergency care are more likely to have relatively poor function and require emergency surgery than patients admitted electively. Studies have shown that patients undergoing emergency surgery are more likely to develop thrombus and even pulmonary embolism than patients undergoing elective surgery.23–25 Furthermore, these patients often have an urgent or life-threatening condition that requires immediate medical attention, such as a heart attack or a ruptured artery. Importantly, these patients have a dysfunctional clotting system that can promote thrombosis and pulmonary embolism. 26 Accordingly, anticoagulants are indicated to prevent thrombosis and pulmonary embolism in this patient population. It is widely acknowledged that when warfarin is used to prevent thrombosis, monitoring of INR and PT should be conducted. Once hemorrhage occurs, clinicians can reverse the effects of warfarin with vitamin K and fresh frozen plasma. 27 There are two types of heparin for preventive treatment of thrombosis: unfractionated heparin (UFH) for injection and low molecular weight heparin (LWMH) for parenteral administration. UFH must be closely monitored for bleeding at any site and is usually monitored using APTT. LMWH has a better dose-response curve than UFH and warfarin; accordingly, it does not require close monitoring. Importantly, protamine sulfate can partially reverse the effects of heparin in the presence of acute bleeding due to heparin therapy. 28 Factor Xa inhibitors are a novel anticoagulant type that includes the direct inhibitors rivaroxaban and indirect inhibitors fondaparinux sodium. Nonetheless, there are no specific experimental parameters to monitor their anticoagulant effect. Besides, no drugs can reverse their side effects. Although little evidence is available to support this, the German Neurological Association recommends prothrombin complex concentrates to reverse the coagulopathy caused by factor Xa inhibitors.29,30 In summary, clinicians should pay more attention to patients admitted to the ICU on an emergency basis. When necessary, anticoagulants and close monitoring of coagulation can prevent the occurrence of VTE.
ICU patients often require immobilization during their stay. In our study, the length of hospital stay of patients with pulmonary embolism was much higher than patients without pulmonary embolism (338 vs 188, p < 0.001). This finding suggests that ICU patients with longer hospitalization are more likely to develop pulmonary embolism, consistent with the literature. In this regard, another study demonstrated that prolonged hospital stay is a risk factor for lower extremity deep vein thrombosis and pulmonary embolism.31,32 These findings emphasize the need to shorten the length of hospital stay to reduce the VTE incidence. Preventive education of DVT in ICU patients was recommended in a study by Boddi et al. to reduce the risk of complications and shorten the length of hospital stay. 33
A platelet count is often included in a common complete blood count; platelets are mainly involved in hemostasis and the formation of arterial thrombosis. 34 In our study, platelets were significantly higher in patients with pulmonary embolism than patients without (209 vs 191, p = 0.001). Consistently, Warny, M.et al. demonstrated that a high platelet count is closely related to the risk of thrombosis in the cerebral arteries. 35 Moreover, Telo et al. 36 proved that the higher the platelet-to-lymphocyte ratio, the worse the prognosis of pulmonary embolism. Besides, Araz et al. 37 found that an increased mean platelet volume predicted pulmonary embolism recurrence.
Activated partial thrombin time (APTT) is the most commonly used and sensitive screening test to reflect the coagulation activity of the endogenous coagulation system. It is widely used to detect endogenous coagulation factor defects and related inhibitors, monitor heparin therapy, early diagnosis of disseminated intravascular coagulation (DIC), and so on. The normal range of APTT is 22 to 35 s. 38 In our study, although the APTT of both groups was within the normal range. However, the APTT in the PE group was higher than in the normal group (32.0 vs 31.5 p = 0.014). Many studies have shown that prolonged APTT is a risk factor for thrombosis and pulmonary embolism.39–41 Accordingly, more emphasis should be placed on monitoring APTT in ICU patients to prevent pulmonary embolism.
There were also some limitations in our study. First of all, the use of ICD-9-CM codes to identify PE or other comorbidities may result in entry errors. Moreover, our study is limited by the inherent biases of retrospective study design. Furthermore, some variables related to PE, such as d-dimer and familial venous history, were not analyzed due to a missing data rate of up to 90%. These variables may have a confounding effect on this study. However, the large number of patients included in this study can reduce the bias caused by these variables. In addition, the type of PE in patients, such as fat pulmonary embolism, was not studied in this study. Therefore, it is necessary to explore the types of PE in ICU patients in the future. Moreover, only data available online were analyzed in the present study, warranting the need for external validation to increase the strength of our findings.
Conclusion
Based on the clinical data of 9871 ICU patients in the MIMIC-III database, we found that sepsis, tumor, platelet count, the length of hospital stay, emergency admission and APTT are the main predictors of PE in ICU patients.
Footnotes
Author Contributions
CBH and THX designed the study; collected, analyzed, and interpreted data; and drafted the manuscript. CXH provided important suggestions for this study. DYZ collected, analyzed, and interpreted data. ZYW collected and analyzed data. JX and CJ drafted the manuscript and interpreted data. BZW and LC designed the study, drafted the manuscript, and interpreted data. LY designed and supervised the study, obtained funding, and drafted the manuscript. All authors read and approved the final manuscript.
Ethical Approval
Ethical approval to report this case series was obtained from Laboratory for Computational Physiology at the Massachusetts Institute of Technology. Since the database does not contain protected health information, informed consent is not required.
Patient Consent for Publication
The institutional review boards of both Beth Israel Deaconess Medical Center and Massachusetts Institute of Technology Affiliates granted us access to the MIMIC-III database. Since all data has been confirmed, informed consent is not required.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 81772348).
