Abstract
Purpose
The purpose of this study was to investigate the clinical characteristics and risk factors for patients with lung cancer complicated by pulmonary embolism and to provide a reference for the early clinical identification of these patients.
Methods
Eighty patients with lung cancer complicated with pulmonary embolism who were treated at Bethune Hospital of Shanxi from October 2018 to October 2021 were compared with 80 patients with lung cancer without pulmonary embolism. The clinical data of the two groups of patients were collected and analysed.
Results
Compared with that in patients in the LC group, the proportion of patients with pulmonary interstitial fibrosis in the LP group was significantly greater (p < 0.05). The incidence of dyspnoea in the LP group was significantly greater than that in the LC group (p < 0.05). Compared with that in the LC group, the proportion of pulmonary artery compression in the LP group was significantly greater, and the difference was statistically significant (p < 0.05). In terms of pathological type, the proportion of adenocarcinoma patients in the LP group was significantly greater than that in the LC group (p < 0.05). In terms of tumor stage, the proportion of patients with stage III/IV disease in the LP group was significantly greater than that in the LC group, while the proportion of patients with stage I/II disease was significantly lower than that in the LC group, and the difference was statistically significant (p < 0.05). The neutrophil [NEUT (%)], prothrombin time (PT), white blood cell (WBC), carcinoma embryonic antigen (CEA) and D-dimer (DD) levels were significantly greater in the LP group than in the LC group (p < 0.05). In terms of treatment, the proportion of patients receiving systemic chemotherapy in the LP group was significantly greater than that in the LC group (p < 0.05). Logistic regression analysis revealed that adenocarcinoma, systemic chemotherapy and tumor stage III-IV were independent risk factors for lung cancer complicated with pulmonary embolism.
Conclusion
(1) Tumor stage (III/IV), systemic chemotherapy, and adenocarcinoma were independent risk factors for pulmonary thromboembolism in patients with lung cancer. (2) In addition, patients with LP were more likely to have pulmonary interstitial fibrosis, dyspnoea, compression of the pulmonary artery by the tumor location, biological targeted therapy, and abnormal increases in D-dimer, WBC, NEUT (%), CEA and PT levels as laboratory indicators. (3) Pulmonary thromboembolism should be considered in lung cancer patients with a combination of the factors described above.
Introduction
Pulmonary embolism (PE) is a life-threatening disease, is the third most common cause of cardiovascular death after stroke and myocardial infarction and has a high incidence worldwide. There are many risk factors leading to PE, among which malignant tumors are the identified risk factors for PE, and the highest incidence of malignant tumors is in lung cancer. Studies have reported that the incidence of PE in patients with lung cancer is approximately 10%–20%.1,2 Lung cancer is a common malignancy with high morbidity and mortality worldwide. PE is a serious cardiovascular disease in which blood circulation to the lungs is blocked by a blood clot in the pulmonary artery or its branches. When PE occurs in patients with lung cancer, the mortality rate increases significantly. According to research, the mortality rate of patients with lung cancer and PE is approximately 50%, while the mortality rate of patients with lung cancer alone is approximately 30%. This is because PE can block blood circulation in the lungs, making it easier for lung cancer cells to spread to other parts of the body, thus accelerating the worsening of the disease. 3 Therefore, if a patient has both lung cancer and PE, their survival may be shorter and they need to be treated and their treatment managed more radically.
With respect to the risk factors for LP, existing studies have shown that the risk factors for LP can be divided into three categories: tumor-related factors, patient-related factors and treatment-related factors, among which tumor stage IV (non-small cell lung cancer), extensive stage (small cell lung cancer) and tumor pathological type of adenocarcinoma are considered independent risk factors.4,5 However, owing to limited clinical data, few large clinical studies on the risk factors for lung cancer with PE exist, and there is no unified conclusion at present. In addition, different study designs and different groups of participants may lead to inconsistent conclusions. 6 In view of this, this study retrospectively selected lung cancer patients with PE in our hospital as research subjects, analysed the clinical characteristics of these patients to evaluate the risk factors for LP, and provided a theoretical basis for the early identification of LP.
Materials and Methods
Research Subjects
Ninety-eight patients with LP who were treated at Bethune Hospital of Shanxi from October 2018 to October 2021 were selected retrospectively. Among them, 4 patients were unwilling to participate in the study, 8 patients had incomplete clinical data, and 6 patients were lost to follow-up. Therefore, 80 patients were included in the study. In addition, the control group included 80 patients with lung cancer without pulmonary embolism who were matched 1:1 according to the principles of the same department, same period, same sex and age ± 3 years.
The inclusion criteria were as follows: (1) A pathological diagnosis of lung cancer and stage meeting the staging criteria as defined by the eighth Edition of the American Joint Board on Cancer Staging Manual. (2) The diagnostic criteria for PE were the 2018 edition of the Chinese Guidelines for Diagnosis, Treatment and Prevention of Pulmonary Thromboembolism. The exclusion criteria for patients were as follows: (1) Had metastatic lung cancer. (2) Suspected of having a tumor thrombus by imaging. (3) Cachexia caused by other diseases. (4) Patients with a previous history of thromboembolism. (5) Abnormal coagulation status in the patient. This study was approved by the Ethics Committee of Shanxi Bethune Hospital, and all patients or their families provided written informed consent.
Methods
Cases were retrospectively collected via the hospital information system (HIS) in our hospital, and relevant medical history and auxiliary examination results, including sex, age, BMI, living habits, underlying diseases, clinical symptoms, specific anatomical location of lung cancer, adjacent relationship with pulmonary vessels, pathological types and stages, laboratory results and treatment methods, were recorded. The staging of lung cancer was performed in accordance with the revision of the TNM staging system in the eighth edition formulated by the International Association for the Study of Lung Cancer. The severity of pulmonary embolism was assessed via the simplified pulmonary embolism severity index (sPESI) score.
Statistical Analysis
SPSS 25.0 software was used for data analysis. A normality test was performed for continuous variables. Continuous variables with a normal distribution or approximate normal distribution are presented as the means ± standard deviations, and two independent samples t tests were used for intergroup comparisons. Continuous variables that were not normally distributed were described by the interquartile range, and the U test was used for intergroup comparisons. Categorical variables are described as frequencies or percentages, and the χ2 test was used for comparisons between groups. Logistic regression was used for multivariate analysis of variables with statistical significance in univariate analysis and indicators with general clinical significance, and independent risk factors were obtained. For logistic regression analysis, we choose “forward: LR” for logistic stepwise regression modeling. The test level was a = 0.05, and p < 0.05 was considered statistically significant.
Results
Comparison of the General Data Between the two Groups
There was no significant difference in sex, age and body mass index (BMI) between the two groups (p > 0.05). In terms of living habits, there was no significant difference in smoking and drinking history between the two groups (p > 0.05). In terms of medical history, compared with that in the LC group, the proportion of patients with pulmonary interstitial fibrosis in the LP group was significantly greater (p < 0.05). There was no significant difference in other underlying diseases between the two groups (p > 0.05) (Table 1).
Comparison of General Data Between the two Groups.

Comparison of Clinical Symptoms Between the two Groups
The incidence of dyspnoea in the LP group was significantly greater than that in the LC group, and the difference was statistically significant (p < 0.05). There was no significant difference between the two groups in terms of other clinical symptoms, including cough and expectoration, stethalgia, haemoptysis and triad syndrome (a combination of haemoptysis, stethalgia and dyspnoea) (p > 0.05) (Table 2).
Comparison of Clinical Symptoms Between the two Groups.

Comparison of the Anatomical Position, Pathological Type and Clinical Stage of Lung Cancer Between the two Groups
The specific anatomical locations of lung cancer included the left upper lobe, left lower lobe, right upper lobe, right middle lobe and right lower lobe. In addition, there was an obvious mass shadow, and the difference was not statistically significant between the two groups (p > 0.05). Compared with that in the LC group, the incidence of pulmonary artery compression in the LP group was significantly greater (p < 0.05). In terms of pathological type, the proportion of adenocarcinoma patients in the LP group was significantly greater than that in the LC group, and the difference was statistically significant (p < 0.05).There was no significant difference in other pathological types between the two groups (p > 0.05). In addition, in terms of tumor stage, the proportion of patients with stage III/IV disease in the LP group was significantly greater than that in the LC group, while the proportion of patients with stage I/II disease was significantly lower than that in the LC group, and the difference was statistically significant (p < 0.05). In terms of tumor size and lymph node metastasis, the difference between the two groups of patients was not statistically significant (P > 0.05) (Table 3).
Comparison of Anatomical Position, Pathological Pattern and Clinical Stage of Lung Cancer Between the two Groups.

Comparison of Laboratory Indices Between the two Groups
In terms of laboratory indicators, the levels of NEUT (%), PT, WBC, CEA and D-dimer in the LP group were significantly greater than those in the LC group (p < 0.05). There was no significant difference in the other indices between the two groups (p > 0.05) (See Table 4).
Comparison of Laboratory Examination Indexes Between the two Groups.
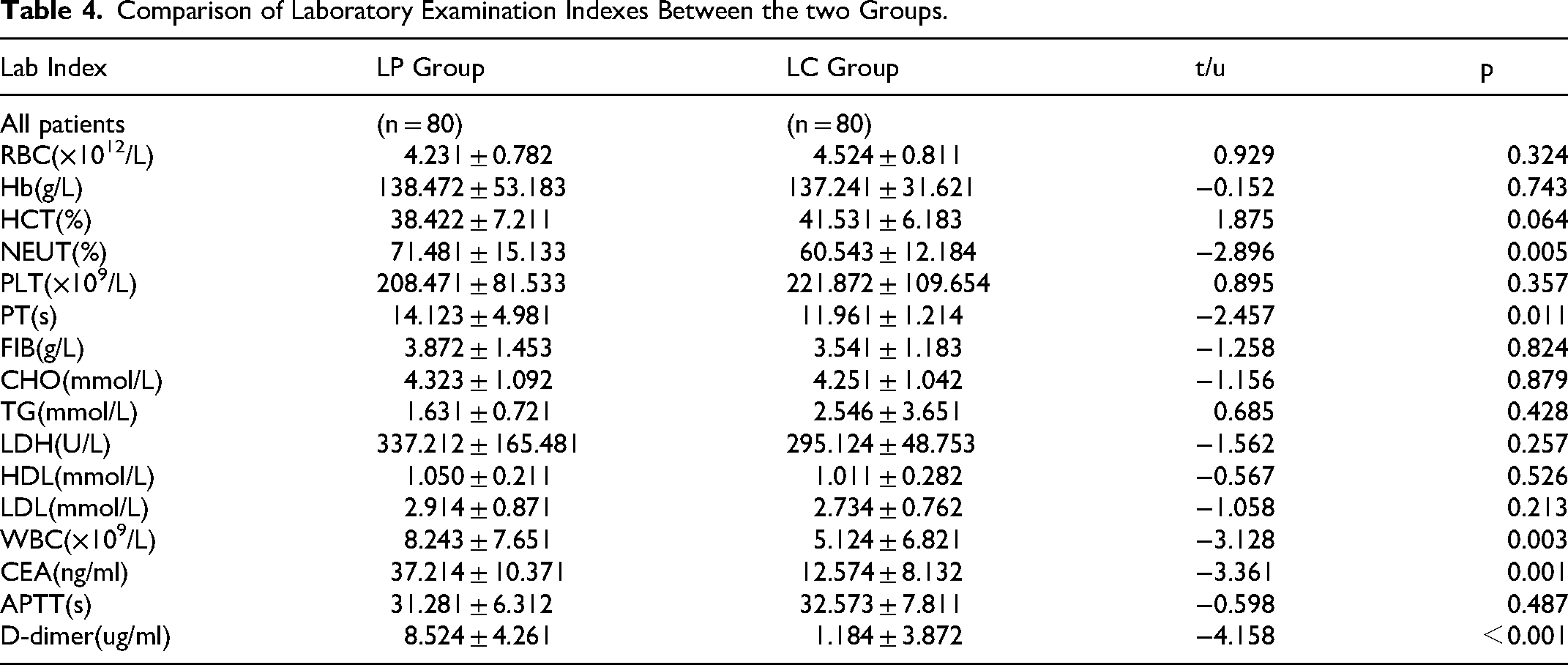
Comparison of Treatment Methods Between the two Groups
In terms of treatment methods, the proportion of patients who received systemic chemotherapy in the LP group was significantly greater than that in the LC group (p < 0.05). There was no significant difference between the two groups in terms of radiotherapy, surgery and biological targeted therapy (p > 0.05) (Table 5).
Comparison of Treatment Methods Between the two Groups.

Logistic Regression Analysis of the Clinical Data of Patients with LP
Logistic regression was used to analyse the variables with statistical significance in the univariate analysis and the indicators with general significance in clinical practice and to obtain independent risk factors. The results suggested that adenocarcinoma, systemic chemotherapy, and stage III-IV disease were independent risk factors for LP (Table 6).
Logistic Regression Analysis of Clinical Data of Patients with Lung Cancer Complicated with Pulmonary Embolism.

Relationship Between the CEA Level and sPESI Score in Patients with LP
There was no correlation between CEA levels and sPESI scores in patients with LP (r = 0.068, p = 0.545) (Figure 1).

Relationship between CEA level and sPESI score in patients with lung cancer complicated with pulmonary embolism.
Discussion
The incidence of PE in patients with lung cancer is relatively high, its pathogenesis is complex, and symptoms overlap with cancer symptoms. The tumor microenvironment, individual differences in patients and some treatment methods might be risk factors. Because of the poor prognosis, early clinical identification of such cases is essential. In addition, the occurrence of PE might increase the difficulty of treating malignant tumors, delay the treatment of primary tumors, and shorten the survival time of patients. 7 Therefore, early detection and diagnosis of LP are highly important for clinical treatment. However, owing to the limited clinical data available for patients with LP and few studies on these patients, there is a lack of unified and standardized understanding of diagnosis and treatment. The purpose of this study was to compare the clinical characteristics of these patients to identify relevant risk factors, providing a reference for the early clinical identification of PE in patients with lung cancer.
Pulmonary fibrosis is a respiratory disorder characterized by damage to alveolar epithelial cells and endothelial cells, inflammatory reactions, abnormal coagulation function and fibrosis material deposition. Abnormal coagulation plays key role in the occurrence and development of pulmonary fibrosis.8–10 Previous studies have revealed that chronic obstructive pulmonary disease (COPD) is a risk factor for patients with LP, whereas diabetes mellitus, hypertension and tuberculosis are not risk factors for patients with LP.11,12 In our study, lung cancer patients with pulmonary interstitial fibrosis were more likely to have PE. However, no significant differences were observed in the presence of hypertension, diabetes mellitus, COPD, and pulmonary tuberculosis. Although some of the results were consistent with those of previous studies, the COPD results were not consistent with those of previous studies, which might be due to the small sample size of patients in this study.
Studies have shown that lung cancer patients with adenocarcinoma are more likely to develop PE. In this series, all patients were in an advanced stage when they were diagnosed with PE. This study revealed that independent risk factors for thrombosis included adenocarcinoma, stage III-IV lung cancer, and low PaO2 levels.13,14 Adenocarcinoma cells might secrete mucin to activate platelets, leading to activation of the coagulation system. The activation of coagulant-promoting factors by mucin production might also lead to thromboembolic events. 15 In addition, mucin might be involved in the process of activating thrombogenic mediators, leading to fibrin degeneration, epithelial cell shedding, and subsequent microthrombus formation in microvessels. 16 In this study, the incidence of pulmonary embolism in adenocarcinoma patients was greater than that in other lung cancer patients and was still significantly correlated with thromboembolism according to multivariate analysis. Adenocarcinoma was an independent risk factor for patients with LP. Our results were consistent with those of previous reports. Stage III/IV lung cancer was an independent risk factor for patients with LP, and advanced lung cancer patients were more likely to have PE, which is consistent with the findings of previous studies.
In this study, specific anatomical locations of the tumor, including the left upper lobe of the lung, left lower lobe of the lung, right upper lobe of the lung, right middle lobe of the lung, right lower lobe of the lung, and no obvious mass shadow, were associated with PE in patients with lung cancer. However, tumor compression to the pulmonary artery was associated with PE in lung cancer patients, which is consistent with the findings of previous studies, suggesting that the specific location of solid tumors in lung cancer patients might have little relationship with PE. In contrast, the location of the tumor and pulmonary vessels might affect the occurrence and development of PE. Tumor compression of pulmonary blood vessels might lead to haemodynamic changes, such as local blood stasis, thus accelerating the process of thrombosis. 17
Platinum-containing chemotherapeutics might significantly increase the risk of thromboembolic events in patients with small cell lung cancer. 18 In previous studies, chemotherapy increased the risk of VTE in cancer patients. Specifically, the risk of patients with LP after chemotherapy increased with threefold. 19 Chemotherapy drugs might increase coagulation activity in lung cancer patients by affecting the tissue factor activities of endothelial cells, A549 cells and monocytes. 20 Cisplatin-based chemotherapeutic drugs might generate disulfide bonds via the protein disulfide isomerase-dependent pathway, increasing the coagulation-promoting activity of endothelial cells. 21 Our multivariate analysis revealed that systemic chemotherapy was associated with thrombus, which is consistent with the findings of previous studies.
Lung cancer with PE was associated with D-dimer, WBC, NEUT (%), CEA and PT levels, and other laboratory indices were not significantly different between the thrombus group and the nonthrombus group. Leucocytosis is significantly correlated with an increased risk of deep vein thrombosis in lung cancer patients. 22 Leucocytosis, specifically neutrophilism, is associated with venous thromboembolism in cancer patients undergoing systemic chemotherapy and is associated with inflammation, cancer invasion, and disease progression, increasing the risk of thrombosis. Neutrophils enhance thrombosis by producing neutrophil–extracellular traps, in animal models, neutrophil-released reticular cells contribute to venous thrombosis. In addition, reticular cells increase TF activity by binding elastase and cathepsin G to reduce the activity of tissue factor pathway inhibitors (TFPIs). 23 The current data were consistent with those of previous studies.
Patients with lung cancer have hypercoagulability, and an imbalance between coagulant-promoting factors and anticoagulant factors might lead to PE in lung cancer patients.23,24 Numerous risk factors for hypercoagulation in lung cancer patients, including a shortened clot formation time, maximum coagulation hardness, elevated fibrinogen and platelet levels, and thrombin activation by fibrinolytic inhibitors, increased activity levels and inhibited fibrinolytic system activity. 24 Activated partial thromboplastin time (APTT) and fibrinogen (FIB) might be increased in lung cancer. 25 In the present study, the PT and D-dimer levels were significantly greater in the LP group, whereas the FIB and APTT levels were not different between the two groups. One explanation is that most patients with LP have advanced-stage disease, and most patients receive systemic chemotherapy, which affects the clotting function in cancer patients.24,25
In patients with lung cancer, tumor markers, such as cytokeratin 19 fragment (CYFRA 211), CEA, neuron-specific enolase (NSE) and squamous cell carcinoma antigen (SCC), are often used as prognostic indicators and therapeutic monitoring indicators. Serum CEA is an effective marker for adenocarcinoma in lung cancer patients. 26 Studies have shown that the incidence of PE in lung cancer patients with elevated CEA is greater than that in patients without distant metastasis or leucocytosis.26,27 For a single marker, CEA had the strongest predictive value for LP. Scholars have further shown that an increase in the CEA level is linearly related to the risk of PE in lung cancer patients. 27 Compared with other tumor markers, CEA is a sensitive marker of lung adenocarcinoma, a histology significantly associated with PE, and CEA levels often reflect the tumor load. 27 Owing to a lack of data, other tumor markers, such as NSE, CYFRA211, and SCC, were not included in this study. Other tumor markers, such as NSE, CYFRA211, and SCC, might play a certain role in LP.
The clinical manifestations of patients with LP are difficult to distinguish from those of patients with lung cancer alone.28,29 In this study, the rate of dyspnoea in the LP group was greater than that in the LC group, suggesting that lung cancer patients should be highly vigilant with respect to new-onset symptoms, which is consistent with obstructive pneumonia and pleural effusion. 29
In this study, fewer patients in the LP group presented with triad syndrome, suggesting that most patients with LP clinically presented with typical symptoms. The majority of DVT and PE events in patients with lung cancer were asymptomatic or nonspecific symptoms before they started anticancer therapy. Patients might have been hypercoagulable before starting lung cancer treatment, and a diagnosis of PE might occur before cancer diagnosis. 30 Large tumors and metastatic lymph nodes in lung cancer patients might compress blood vessels in the lung, leading to blood stasis. All these factors might increase venous thrombus embolism (VTE) risk in newly diagnosed lung cancer patients, however, there is no clear definition of VTE in lung cancer patients prior to the initiation of antitumor therapy.29,30
In addition, this study had several limitations. First, as a retrospective analysis, the study was inevitably subject to selection bias. Second, this study could not establish a precise causal relationship between LP and differences in associated clinical symptoms. In addition, the number of patients with LP included in this study was small, and a larger sample size is needed for further research verification. Early identification of such patients and standardized anticoagulation therapy are highly important for the prognosis of patients.
Conclusion
Tumor stage (III/IV), systemic chemotherapy, and adenocarcinoma are independent risk factors for LP. In addition, patients with LP are more likely to have pulmonary interstitial fibrosis, dyspnoea, compression of the pulmonary artery by the tumor location, biological targeted therapy, and abnormal elevation of D-dimer, WBC, NEUT (%), CEA and PT levels according to laboratory indicators. It was suggested to be alert to pulmonary thromboembolism for lung cancer patients with combination of factors above.
Footnotes
Abbreviations
Author Contributions
Conceptualization: Wei Duan. Pengfei Wang and Lei Liang.Data curation: Wei Duan. Kejing Ma and Lei Liang.Formal analysis: Wei Duan. Investigation: Pengfei Wang. Methodology: Kejing Ma and Lei Liang.Project administration: Pengfei Wang and Lei Liang. Software: Kejing Ma. Supervision: Wei Duan.Validation: Wei Duan. Kejing Ma and Lei Liang. Visualization: Wei Duan. Pengfei Wang and Lei Liang.Writing—original draft: Pengfei Wang. Writing—review and editing: Wei Duan. Kejing Ma and Lei Liang.Approval of final manuscript: Wei Duan. Pengfei Wang, Kejing Ma and Lei Liang.
Data Availability Statement
The data supporting this study's findings are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
This study was performed in line with the principles of the Declaration of Helsinki. This study was approved by the Ethics Committee of Shanxi Bethune Hospital. Written consent was obtained from the patients before enrolling in this study. All methods were carried out following the necessary laws and regulations.
