Abstract
We measured levels of a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS13) in 50 children and adolescents with β-thalassemia major (25 without hepatitis C virus [HCV] infection and 25 with HCV infection) compared to 25 healthy controls and assessed their relation to iron overload, HCV infection, and liver cirrhosis. Hematological and coagulation profiles, serum ferritin, and von Willebrand factor antigen were assessed. Levels of ADAMTS13 were significantly lower in β-thalassemia major with and without HCV infection compared to healthy controls, with a more significant reduction in levels among patients with HCV (P < .001). HCV-positive patients with thalassemia having liver cirrhosis had the lowest ADAMTS13 levels than those without cirrhosis (P = .012) or HCV-negative patients with thalassemia (P < .001). Levels of ADAMTS13 were positively correlated with platelet count while inversely correlated with partial thromboplastin time, serum ferritin, and VWF: Ag (P < .05).
Conclusion:
Patients with β-thalassemia major infected with HCV have low ADAMTS13 levels, and a marked reduction was observed among patients with liver cirrhosis and, therefore, may be liable to thromboembolic manifestations.
Introduction
Patients with β-thalassemia major are at high risk of acquiring a number of viral infections during multiple blood transfusions. Of these infections, hepatitis C is the most important. 1 Hepatitis C virus (HCV) is considered the principal etiologic agent of posttransfusion hepatitis and is the main cause of chronic liver disease in multitransfused patients. 2 The highest HCV prevalence in the world occurs in Egypt, where the prevalence of infection increases steadily with age, and high rates of infection are observed among people in all age-groups. 3
Every liver disease is accompanied by multiple changes in the hemostatic system, and this alteration may lead to either bleeding or thrombosis. 4 Although bleeding is the most common clinical manifestation, hypercoagulability may have an important role in many aspects of chronic liver disease such as portal vein thrombosis, occlusion of small intrahepatic vein branches, and deep vein thrombosis. The exact mechanism of hypercoagulability is not fully understood. 5
Circulatory disturbances have been observed in patients with thalassemia, manifested by transient ischemic attacks and stroke, arterial and venous thrombosis, and microcirculatory obstruction, which lead to the appearance of chronic leg ulcers as well as pulmonary thromboembolism. 6 Although microcirculatory obstruction can be explained by red blood cell membrane rigidity and reduced surface charge, the mechanism responsible for the increased incidence of thrombotic events in thalassemia remains unclear. 7
von Willebrand factor (VWF), a plasma glycoprotein that is a mediator of platelet adhesion, becomes available when the endothelium is damaged. The VWF is released as ultralarge (UL) multimers, which form spontaneous high-strength bonds with the platelet glycoprotein 1b-IX-V complex, resulting in platelet aggregation and thrombus formation. 8 A disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS13) is a metalloproteinase produced in hepatic stellate cells. Deficiency of ADAMTS13 results in accumulation of unusually UL VWF multimers (UL-VWFM) in the plasma, inducing platelet thrombi under high shear stress, followed by sinusoidal microcirculatory disturbances, and subsequent progression of liver injuries, eventually leading to multiorgan failure. 9 Portal or hepatic vein thrombosis is often observed in advanced cirrhosis, and microthrombi formation was found in 1 or multiple organs in half of autopsied patients with cirrhosis. This hypercoagulable state in liver disease may be involved in hepatic parenchymal extinction, the acceleration of liver fibrosis, and disease progression. 10
Although a decreased ADAMTS13 level has been reported in sickle cell disease, 11 it has not been explored in thalassemia, and its relation to HCV infection and liver cirrhosis remains to be elucidated. Therefore, this study aimed to determine levels of ADAMTS13 in young patients with β-thalassemia major and assess its relation to iron overload, HCV infection, and liver cirrhosis.
Patients and Methods
This cross-sectional study included 50 patients with β-thalassemia major (24 males and 26 females with a male to female ratio 1:1.1) recruited from the regular attendants of the Pediatric Hematology Unit, Children’s Hospital, Ain Shams University. Patients were diagnosed with β-thalassemia major based on complete blood picture, reticulocyte count, and markers of hemolysis as well as hemoglobin analysis using high-performance liquid chromatography. 12 Their age ranged between 1 and 14 years with a mean of 9.2 (±3.0) years. Twenty-five age, sex, and blood group–matched healthy participants (unrelated to the patients) were enrolled as a control group, with 13 males and 12 females (male to female ratio, 1:1.1), with a mean age of 7.1 (±3.4) years (range, 2-14 years). An informed consent was obtained from the guardian of each patient or control before participation. The procedures applied in this study were approved by the Ethical Committee of Human Experimentation of Ain Shams University and are in accordance with the Helsinki Declaration of 1975. Patients with coexisting malignant, acute infectious conditions, renal impairments, or hepatitis B virus infection were not included.
A standardized questionnaire was completed for all study patients, providing detailed medical history and thorough clinical examination with emphasis on disease duration, anthropometric measures, blood pressure, history of splenectomy, evidence of liver disease and HCV infection, transfusion history, and chelation therapy.
Sample Collection
Peripheral blood samples were collected on EDTA (1.2 mg/mL) for complete blood count (CBC) and on 0.2 mL 3.8% trisodium citrate in a ratio of 9 volumes of blood to 1 volume of citrate for ADAMTS13 assay and VWF antigen (VWF: Ag). For chemical and ADAMTS13 analysis, clotted and citrated samples were obtained, and both serum and plasma were separated by centrifugation for 15 minutes at 1000g and stored at –20°C till subsequent use.
Laboratory Investigations
Laboratory investigations included CBC using Sysmex XT-1800i (Sysmex, Kobe, Japan), examination of Leishman-stained smears for differential white blood cell count, peripheral blood staining by brilliant cresyl blue and examination of a stained smear for reticulocyte count, coagulation profile (prothrombin time [PT] and activated partial thromboplastin time [APTT]) using STA-Compact Coagulation Analyzer (Diagnostica Stago, Parsippany, New Jersey), liver and kidney function tests as well as serum ferritin on Cobas e411 (Roche Diagnostics, Mannheim, Germany). Calculation of the mean serum ferritin levels was done during the year prior to the study to identify the ferritin trend. Levels of VWF: Ag were measured by quantitative enzyme linked immunosorbent assay in plasma. REAADS® von Willebrand Factor Antigen Test Kit (Corgenix, Westminster, CO, USA).
Determination of ADAMTS13
Plasma ADAMTS13 levels were determined using a quantitative sandwich enzyme immunoassay technique (TECHNOZYM, Technoclone, Austria) with intra- and interassay coefficients of variation of 2.6% and 5.1%, respectively. A monoclonal antibody specific for ADAMTS13 had been precoated onto a microplate. Standards and samples were pipetted into the wells and any ADAMTS13 present was bound by the immobilized antibody. After washing away any unbound substances, an enzyme-linked polyclonal antibody specific for ADAMTS13 was added to the wells. Following a wash to remove any unbound antibody-enzyme reagent, a substrate solution was added to the wells and color developed in proportion to the amount of ADAMTS13 bound in the initial step. The color development was stopped and the intensity of the color was measured.
Radiological Examination
Abdominal ultrasonography was performed for all studied patients using Hitachi EUB-5500 ultrasound system (Hitachi Medical, Inc, Tokyo, Japan). All ultrasound scans were performed by an experienced operator who was unaware of children’s clinical details. Measurements were performed after overnight fasting and the patient in supine position with emphasis on the liver span (measuring the span of the right lobe in the mid-clavicular line on oblique view) and splenic bipolar diameter measured in a coronal plane, from the upper to the lower pole of the spleen. 13
Group Classification
Patients with β-thalassemia major were divided into 2 groups; group 1 included 25 patients without chronic HCV infectionand group 2 included 25 children with chronic HCV infection (anti-HCV antibody and/or HCV-RNA was positive at least 6 months prior to the study).
Statistical Analysis
Analysis of data was done using SPSS version 15 (SPSS Inc, Chicago, Illinois). Quantitative variables were described in the form of range, mean, and standard deviation or median and interquartile range (IQR, 25th-75th percentiles). Qualitative variables were described as number and percentage. In order to compare quantitative parametric variables between the 2 groups, Student t test was applied, while comparison of nonparametric variables was carried out using Mann-Whitney U test. Categorical variables were compared using chi-square test or Fischer exact test when frequencies were below 5. Spearman rank correlation coefficient was employed to measure the strength and direction of the linear relationship between 2 variables. A P value of <.05 was considered significant in all analyses.
Results
Clinical and Laboratory Data of Patients With β-Thalassemia Major With and Without HCV Infection.a

Abbreviations: IQR, interquartile range; WBC, white blood cell; PT, prothrombin time; APTT, activated partial thromboplastin time; ALT, alanine aminotransferase; VWF: Ag, von Willebrand factor antigen; SD, standard deviation.
aData were expressed as mean ± SD and comparisons were made using Student t test or median (IQR) where Mann-Whitney U test was used for comparisons unless specified as n (%) using chi-square test for comparisons.
Levels of ADAMTS13 were significantly lower in patients with β-thalassemia major with and without HCV infection (median [IQR], 290 [190-370] ng/mL and 490 [340-730] ng/mL, respectively) compared to healthy controls (median [IQR], 610 [490-1070] ng/mL), with a more pronounced decrease in levels among patients with HCV (P < .001; Table 1). Moreover, HCV-positive patients with thalassemia having liver cirrhosis had lower ADAMTS13 levels than their noncirrhotic counterparts (median [IQR], 160 [130-180] ng/mL vs 320 [250-410] ng/mL, respectively; P = .012). Notably, the lowest ADAMTS13 concentrations were observed among HCV-positive patients with thalassemia having liver cirrhosis compared to other studied patients’ groups (P < .001). Although ADAMTS13 level was lower in splenectomized patients with thalassemia compared to nonsplenectomized ones, however, the difference did not reach a significant level (median [IQR], 350 [240-760] ng/mL and 490 [340-640] ng/mL; P = .304).
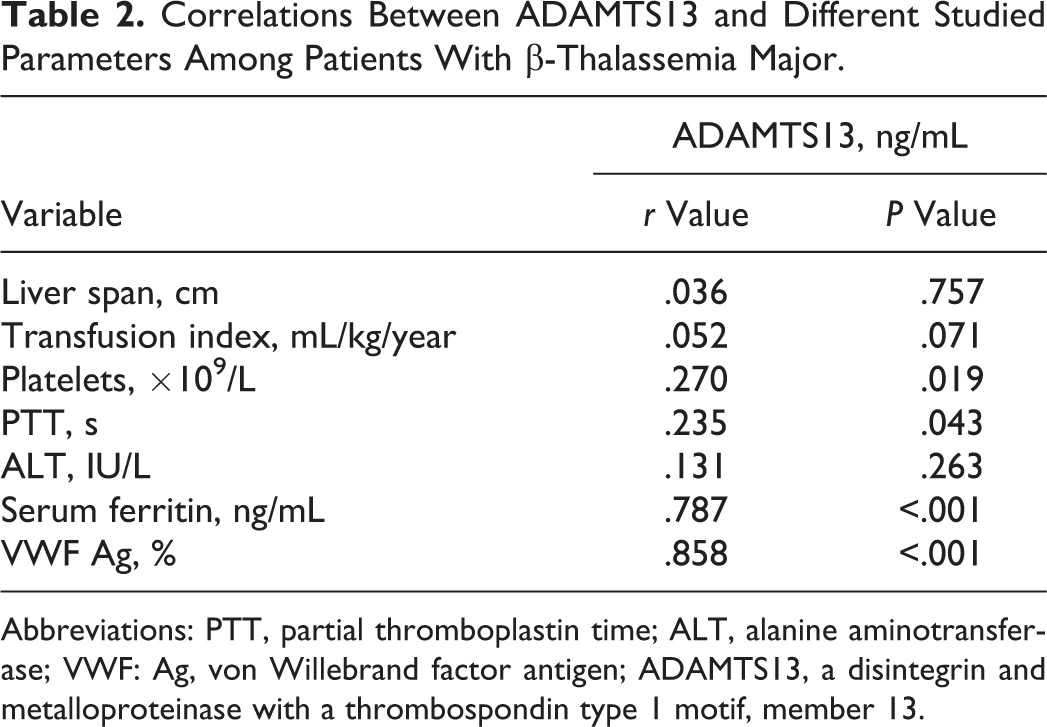
Correlation studies revealed significant positive correlation between ADAMTS13 and platelet count, while it was inversely correlated with APTT (Table 2). Moreover, ADAMTS13 showed significant negative correlation with serum ferritin and VWF: Ag (P < .05; Table 2 and Figure 1).
Negative correlation between ADAMTS13 and some laboratory variables among patients with thalassemia. Open circles indicates patients without hepatitis C virus (HCV); solid circles, patients with HCV. Correlations Between ADAMTS13 and Different Studied Parameters Among Patients With β-Thalassemia Major. Abbreviations: PTT, partial thromboplastin time; ALT, alanine aminotransferase; VWF: Ag, von Willebrand factor antigen; ADAMTS13, a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13.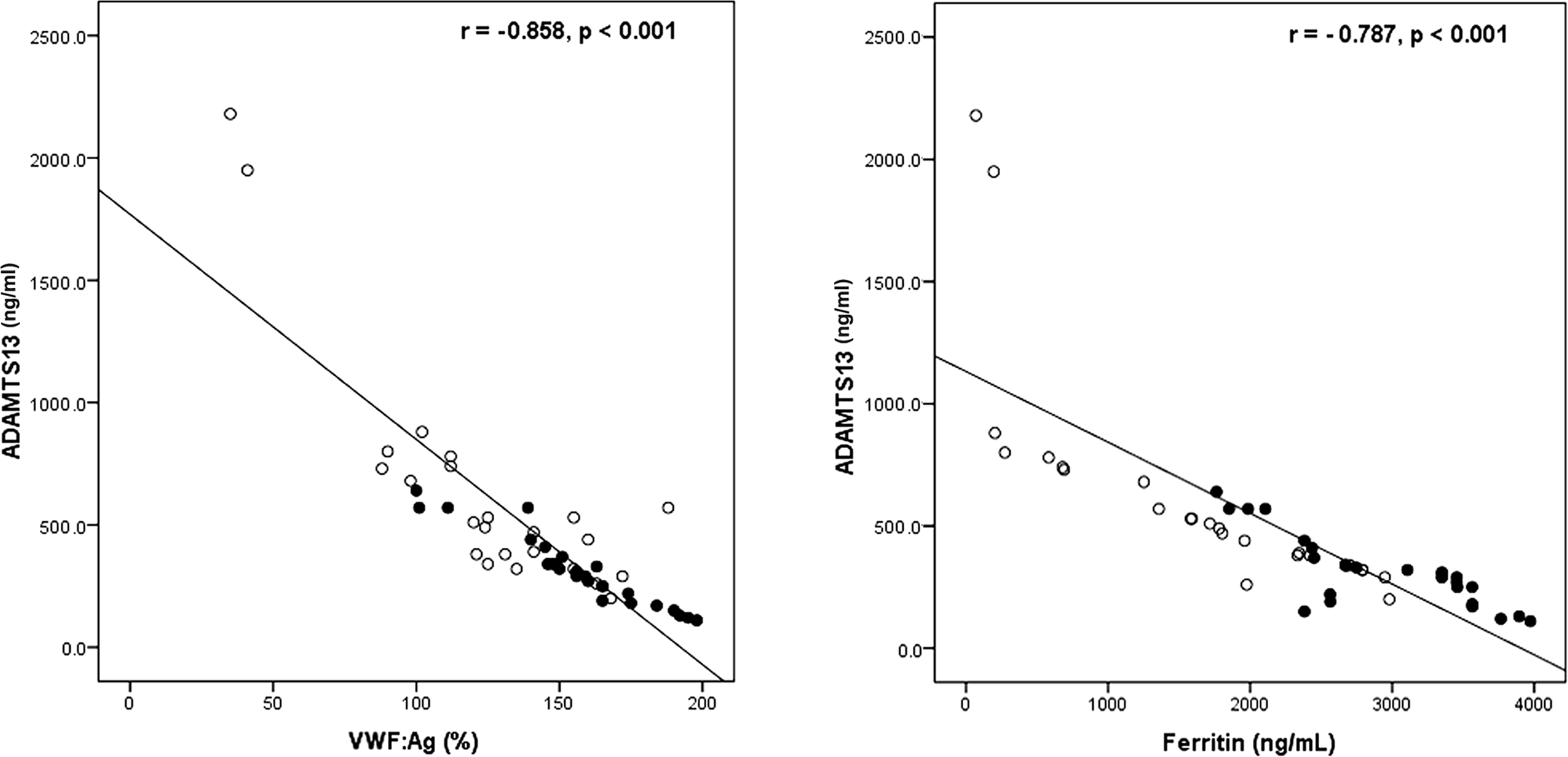
Discussion
Alterations in coagulation profile have been reported in patients with β-thalassemia. 14 –17 Reduced activity of ADAMTS13 was postulated as a pathogenic mechanism underlying many hematological disorders. It was reported that decreased plasma ADAMTS13 activity in patients with liver cirrhosis results in the accumulation of UL-VWFM and the formation of platelet thrombi. 18 Therefore, we measured the levels of ADAMTS13 among patients with β-thalassemia major and explored relation to HCV infection and liver cirrhosis.
In the current study, a significantly higher transfusion index was found in HCV-infected patients with thalassemia compared to patients without HCV. Similarly, Ataei et al 1 found that HCV-positive patients had a significantly higher number of transfused units and increased number of transfusions per month. These findings were also in agreement with other studies. 19 –21 This implies the importance of providing safe blood to decrease the incidence of HCV infection in the population with thalassemia and substantiates the important role of blood donors’ screening in the prevention of viral transmission. 22
Increased duration of disease requiring transfusion therapy and number of blood transfusions (more than 8 per year) are predictors of iron overload and have been associated with greater risk of endocrine organ failure. 23 In this study, serum ferritin was higher in HCV-infected patients with thalassemia. Similarly, Ragab et al 24 reported a positive correlation between serum ferritin (representing iron load from multiple transfusions) and HCV infection. HCV infection is often associated with an elevation in iron parameters. Free liver iron causes liver damage and liver fibrosis, preferentially through induction of reactive oxygen species. 25 In contrast, Kountouras et al 26 reported that ferritin was not significantly different among HCV-positive and HCV-negative patients, and advanced fibrosis was present with even minimal hemosiderosis, independent of ferritin values or HCV history.
Importantly, VWF: Ag levels were elevated among the studied patients with thalassemia infected with HCV compared to the noninfected group. In agreement, Bernardo et al 27 found that plasma levels of VWF: Ag substantially increases as liver diseases progress, presumably due to sinusoidal and/or extrahepatic endothelial damage induced by endotoxin and cytokines. Hugenholtz et al 28 reported that high VWF: Ag levels are associated with progressive liver failure, which might be attributed to platelet-induced microthrombus formation in the diseased liver, resulting from a substantially unbalanced VWF–ADAMTS13 ratio.
In our study, levels of ADAMTS13 were decreased in patients with thalassemia with and without HCV compared to controls. Several studies reported decreased ADAMTS13 in acquired thrombotic thrombocytopenic purpura, 29,30 idiopathic thrombotic thrombocytopenic purpura, 31,32 and sickle cell anemia. 33
Furthermore, we found that patients with thalassemia having HCV had significantly lower ADAMTS13 levels compared to HCV-negative group with thalassemia, with the lowest levels encountered among HCV-positive patients with thalassemia having liver cirrhosis. This further supports the results of Uemura et al 18 who found that the severity of ADAMTS13 deficiency was related to the severity of liver disease.
The mechanism responsible for the decrease in ADAMTS13 in liver disease may include enhanced consumption due to the degradation of large quantities of VWF: Ag, 34 inflammatory cytokines, 35 and/or ADAMTS13 plasma inhibitor. 36 However, Matsumoto et al 37 demonstrated that decreased plasma ADAMTS13 level in patients with cirrhotic biliary atresia can be fully restored after liver transplantation, indicating that the liver is the main organ producing ADAMTS13.
In the present study, a significant positive correlation was found between ADAMTS13 and platelet count, while ADAMTS13 was inversely correlated with serum ferritin, partial thromboplastin time, and VWF: Ag. This implies a link between iron overload, endothelial dysfunction, and coagulation abnormalities. Vascular endothelial cells play a pivotal role in hemostasis and thrombosis. The VWF is a marker of endothelial cell activation and plays an essential role in hemostasis. In the normal state, VWF immunostaining is usually positive in large vessels but negative in the sinusoidal endothelial cells. 38 On the occurrence of liver injury accompanied by a necroinflammatory process, the sinusoidal endothelial cells become positive for VWF, presumably in association with the capillarization of hepatic sinusoids. Subsequently, platelets adhere to subendothelial tissue mediated by UL-VWFM. The ADAMTS13 then cleaves UL-VWFM into smaller VWF multimers. This interaction between ADAMTS13 and UL-VWFM is, indeed, the initial step in hemostasis. 39
In conclusion, we suggest that ADAMTS13 may have a role in sinusoidal microcirculatory disturbance and progression of liver disease as evidenced by decreased levels among patients with β-thalassemia major having HCV infection and a marked reduction among those with liver cirrhosis. Levels of ADAMTS13 are closely related to iron overload, endothelial dysfunction, and coagulation abnormalities. Its measurement could provide utility for early detection of progressive liver disease among patients with thalassemia.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
