Abstract
This study aimed to investigate the oxidative stress, hypoxia biomarkers, and circulating microparticles (MPs) in β thalassemia major. The study included 56 children with thalassemia and 46 healthy controls. Hypoxia biomarkers, oxidative stress biomarkers, and total plasma fragmented DNA (fDNA) were detected by the standard methods. The MPs were assessed by flow cytometry. Hypoxia and oxidative stress biomarkers, fDNA, and MPs were higher and total antioxidant capacity (TAC) was lower in patients with thalassemia than the controls. In splenectomized patients and those who had complications, vascular endothelial growth factor (VEGF), malondialdehyde, fDNA, endothelial, platelet, and activated platelet MP levels were higher while, TAC was lower than the nonsplenectomized patients. In conclusion, the increased tissue hypoxia, oxidative stress in β thalassemia, and its relationship with DNA damage and MPs release could explain many complications of thalassemia and may have therapeutic implications. The VEGF could serve as an important indicator for adequacy of blood transfusion in thalassemia.
Introduction
The profound anemia of homozygous β thalassemia and the high oxygen affinity of the blood produced combine to cause severe tissue hypoxia. Tissue hypoxia increases the expression of erythropoietin (EPO). In thalassemia, due to ineffective erythropoiesis, the imbalance between erythrocyte supply and demand persists despite increased tissue hypoxia and increased EPO. As a result, EPO level remains high and the marrow typically becomes hypercellular. 1,2 Over time, the combination of tissue hypoxia, increased EPO, and ineffective erythropoiesis creates a vicious cycle that may ultimately lead to a massive expansion of erythroblasts. Eventually, secondary bony pathologies 3 and extramedullary erythropoiesis 4 can also develop.
The hypoxia-inducible factor (HIF) pathway plays a protective role in regulating genes that mitigate the effects of low oxygen tension. Under normoxic conditions, oxygen-sensitive HIF-α isoforms are rendered inactive via proline hydroxylation by HIF-specific prolyl hydroxylases. Under hypoxic conditions, HIF-α isoforms are stabilized, heterodimerize with HIF-β, and translocate to the nucleus where they bind to hypoxia-responsive element motifs. 5 –7 In cooperation with other transcriptional coactivators, HIF induces transcription of genes that ameliorate the effects of hypoxia, including EPO and its receptor, transferrin and its receptor, glucose transporters, glycolytic enzymes, and vascular endothelial growth factor (VEGF). 8 The VEGF is a chemoattractant that enables the homing of regenerative endothelial cells into local vascular lesions. 9 There is still a considerable debate over the vasculoprotective versus proinflammatory effects of VEGF. 10
Oxidative stress in thalassemia primarily caused by the red blood cell (RBC) abnormalities, the oxidation of globin subunits in thalassemia erythroid cells, leads to the formation of hemichromes. Hemichromes bind to or modify various components of the mature RBC membrane, such as protein band, ankyrin, and spectrin. 11 After the precipitation of hemichromes, heme disintegrates and toxic nontransferrin-bound iron (NTBI) species are released. Other contributing factors to iron overload are increased intestinal absorption and regular blood transfusions. Consequently, free iron species, NTBI and labile plasma iron catalyze the formation of reactive oxygen species (ROS). 12 This oxidative stress may contribute to shortened life span of erythrocytes, primary or secondary amenorrhea, hypogonadism, osteoporosis, heart failure, endocrine abnormalities like diabetes, hypothyroidism, liver failure, and ultimately early death. 13
Cell membrane microparticles (MPs) are phospholipid microvesicles shed from the plasma membrane of cells undergoing activation or apoptosis. The MPs are present in healthy individuals. However, an increase in their release is a controlled event and is considered a hallmark of cellular alteration. The MPs display cell surface proteins that indicate their cellular origin. In addition, they may also express other markers, for example, markers of cellular activation. 13 Most MPs are highly procoagulant, expressing annexin V binding sites, and are capable of interacting with other cells to participate in various physiologic and pathologic processes, especially thrombosis and inflammation. 15,16 Oxidative stress is one of the risk factors involved in increasing the MPs formation. 17 At the same time, oxidative stress is generated by MPs. 18
To date there is no prior study investigating oxidative stress, hypoxia biomarkers, fDNA and circulating MPs together in children with thalassemia in Upper Egypt. We supposed that impaired response to tissue hypoxia, subsequent oxidative stress, and MPs release would be major determinants of the clinical outcome of patients with thalassemia. This study aimed to investigate the oxidative stress, hypoxia biomarkers, and circulating MPs in β thalassemia major.
Materials and Methods
Patients
A total of 56 pediatric patients with thalassemia were voluntarily recruited to this prospective cross-sectional controlled study in the Thalassemia Centre of Assiut Children University Hospital, Assiut, Egypt, during the period from January 2011 to September 2011. Investigations were compared to the levels in 46 socioeconomically , age- and sex-matching healthy control group of volunteers. Controls and patients were on a normal diet without vitamin supplementation and were free from any infection or medication. The study was approved by the local ethics committee and was conducted in accordance with the Declaration of Helsinki and informed consent was obtained in every case from their parents. Demographic, clinical, routine, and investigative biochemical characteristics of all participants were reported.
Inclusion Criteria
Patients with β-thalassemia who visit the Pediatric Hematology unit for follow-up and for blood transfusion. They were on irregular blood transfusion and chelation therapy with subcutaneous administration of desferrioxamine with variable compliance.
Exclusion Criteria
Patients on antioxidants or with known cardiac, infectious, inflammatory, or pulmonary diseases were excluded from the study.
Biochemical Investigations
Blood samples (5 mL) were collected from fasting and resting participants through venipuncture in pyrogen-free EDTA/sodium fluoride (3 mL) or plain tubes. Serum was separated and aliquot frozen at −20°C till used. Specific enzyme-linked immunosorbent assay (ELISA) was utilized to measure the plasma levels of each of VEGF (Cat# ELH-VEGF-001, RayBiotech, Inc, Norcross, Georgia) and HIF-1α (Cat# DYC1935-5, R&D Systems, Minneapolis, Minnesota) with specific capture/biotinylated detection antibodies and streptavidin–horseradish peroxidase/tetramethylbenzidine as the detection system. Serum total antioxidants capacity (TAC) and total peroxides (TPs; organic and inorganic) were colorimetrically measured according to the methods described by Koracevic et al 19 and Harma et al. 20 Oxidative stress index (OSI) was the ratio of TP/TAC. Serum thiobarbituric acid (TBA) reactive substances and malondialdehyde (MDA) were measured in a reaction mix containing 50 μL of sample and 25 µL of 10% sodium dodecyl sulfate, mixed with 100 µL TBA (0.530 g/dL in 100 mmol/L sodium acetate buffer, pH 3.2) and 10 µL butylated hydroxytoluene (0.3 mmol/L in absolute ethanol). The mixture was incubated at 90°C for 1 hour, then cooled and centrifuged at 10 000 rpm for 10 minutes at 4°C. Optical density (OD) of the pink chromogen was measured against H2O as blank at 540 nm. 1,1,3,3-Tetraethoxypropane (100 mmol/L). 21 Plasma pyruvate was measured following the decrease in ultraviolet absorption by NADH/H+ according to instructions of the manufacturer (Cat# 180 000, Greiner Diagnostics GmbH, D-79353 Bahlingen, Germany). Plasma lactate was estimated colorimetrically according to instructions of the manufacturer (Cat# 274 001, Spectrum Biodiagnostics, Cairo, Egypt). The lactate/pyruvate (L/P) ratio was calculated. Plasma total fDNA was measured after precipitation with perchloric acid (20 µL 7 mol/L perchloric + 260 µL sample) for 10 minutes, then mixed and centrifuged at 10 000 rpm for 10 minutes at 4°C. One hundred fifty microliters of supernatant was mixed with 300 µL of diphenylamine (0.088 mol/L in 98%, v/v, glacial acetic acid: 1.5%, v/v, sulfuric acid: 0.5%, v/v, of 1.6% acetaldehyde), incubated at 50°C for 1 hour, and the OD of the purple chromogen was measured at 575 nm against H2O as blank and using DNA as a standard. 22 Determination of serum ferritin was done by ELISA kit manufactured by Diagnostic Biochemist Canada Inc (41 Byron Avenue, Dorchester, Ontario Canada) (cat. No. CAN – f – 4280 version 5.0).
Microparticles Isolation and Characterization
Blood samples were collected into a 5 mL tube containing 3.2% citrate. In order to isolate the MPs, cells were removed by centrifugation for 20 minutes at 1550g at 20°C within 15 minutes after collection. Then, 250 µL of plasma were centrifuged for 30 minutes at 18 800g at 20°C. After centrifugation, the supernatant was removed and the pellet was resuspended in phosphate-buffered saline (PBS) and centrifuged for 30 minutes at 18 800g at 20°C. The supernatant was removed again and MPs pellet was resuspended in PBS. 23
Flow cytometric analysis was used to quantify and characterize MPs. Five microliters of MPs sample were diluted in 35 µL of PBS containing 2.5 mmol/L calcium chloride. The samples were then incubated for 20 minutes at room temperature in the dark with 5 µL of fluoroisothiocyanate-conjugated annexin V (IQ products, Netherland) and 5 μL of phycoerythrin and peridinium-chlorophyll-protein-conjugated cell-specific anti-human monoclonal antibody. After incubation, PBS/calcium buffer was added and the samples were analyzed on a fluorescence activated cell sorter (FACS) Caliber flow cytometry with Cell Quest software (Becton Dickinson Biosciences, San Jose, California, USA). Fifty thousand events were analyzed, and MPs were reported as a percentage of the total events. Anti-human immunoglobulin G was used as an isotype-matched negative control for each sample. 23,24
Microparticles were identified on the basis of their forward scatter compared to that of calibrate reference beads of 1.0 µm to calibrate the size range of MPs (latex beads, amine-modified polystyrene, fluorescent red aqueous suspension, 1.0 μm mean particle size, Sigma-Aldrich Chemie Gmbh Munich, Germany) and positivity for annexin V. The microparticles express phosphatidylserine, which is detected by annexin V labeling. 23 Microparticles subpopulations were identified by their ability to bind cell-specific monoclonal antibodies. Detection of endothelial MPs was performed using anti-CD146 labeling (Beckman coulter, France). Detection of platelet MPs was performed using CD41 and/or CD61 (Becton Dickinson Biosciences). Erythrocyte, monocytes, and leukocyte MPs were detected by anti-glycophorin A (Becton Dickinson Biosciences), CD14, and anti-CD45 (Beckman coulter), respectively. Anti-CD62P (Becton Dickinson Biosciences) labeling was used to detect P-selectin positive MPs (activated platelet MPs). 25,26
For detection of MPs and MPs subpopulation, forward- and side-scatter histogram were used to define the MPs (R1) according to the size of the reference calibrate bead. Events defined as MPs (R1) were then selected for their annexin V binding, determined by positivity for annexin V (R2). Then annexin V positive MPs (R2) was further examined for expression of cell-specific antibodies such as CD 61, CD41, and glycophorin A (Figure 1). Total MPs are reported as percentage of total events. Annexin V positive was reported as the percentage of total MPs. The percentage of MPs subpopulation such as platelet, endothelial, and other are expressed as percentage of annexin V positive population.
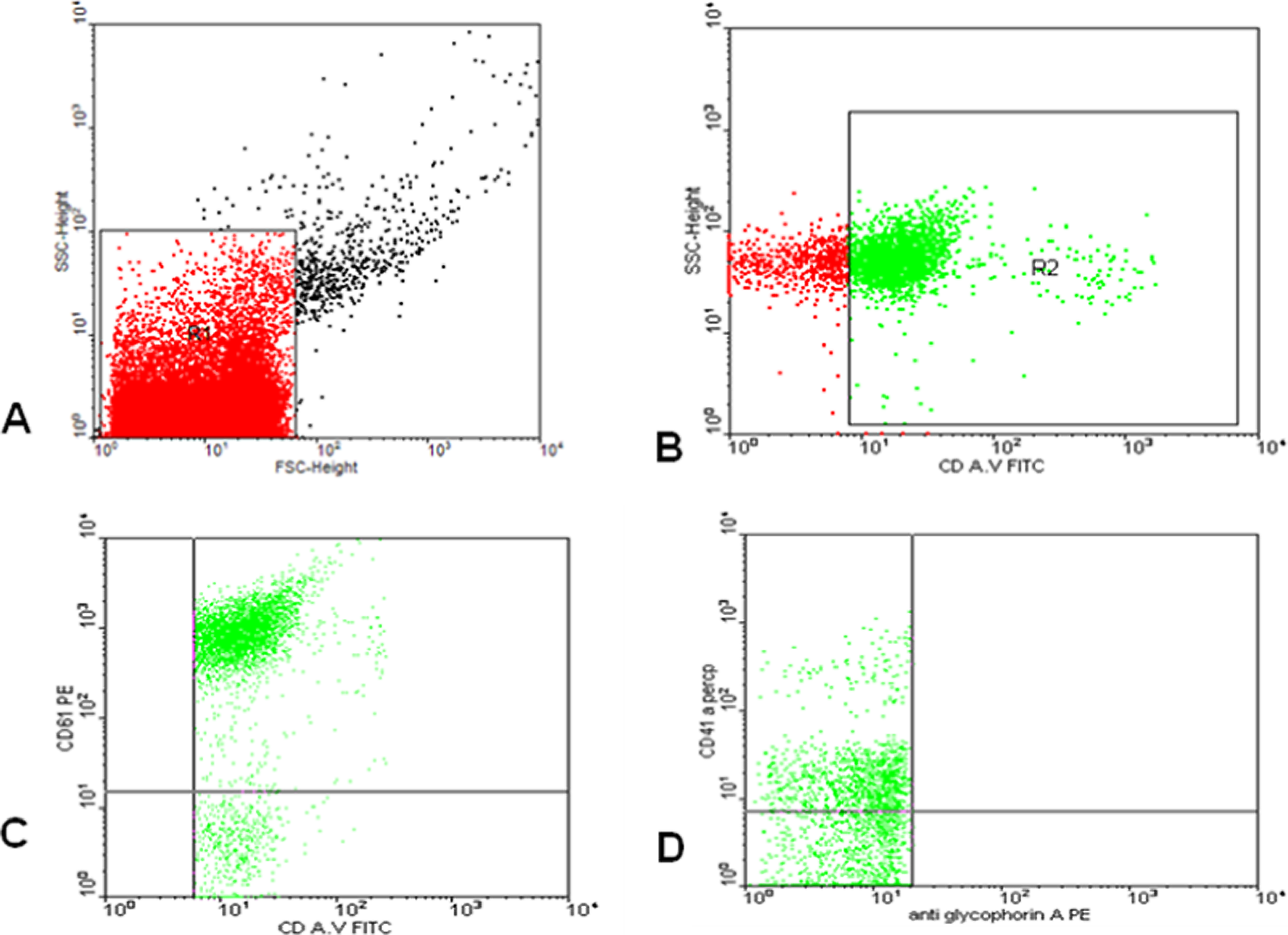
Flow cytometric analysis of microparticles: a representative set of scattergrams of a sample from a patient with thalassemia showing microparticles (MPs) and MPs subpopulations. Forward and side scatter histogram was used to define the MPs (R1) according to the size of reference calibrate bead (A). Events defined as MPs (R1) were then selected for their annexin V binding, determined by positivity for annexin V (R2) (B). Then annexin V positive MPs (R2) were further examined for expression of cell-specific antibodies such as CD 61, CD41, and anti glycophorin A (C, D) compared to the negative isotype control (not shown).
Statistical Analysis
Data analysis was done using statistical package for social sciences (SPSS), version 16. Data were expressed as the mean ± standard error of mean. Results were analyzed statistically using Mann-Whitney U analysis. Correlation among the investigated parameters in each group was tested by the nonparametric Spearman analysis. A P value ≤.05 denoted the presence of a statistically significant difference.
Results
The most relevant characteristics of patients are shown in Table 1. Differences between healthy controls and pediatric patients with thalassemia in the levels of the investigated hypoxia and oxidative stress biomarkers are shown in Table 2. There was a significantly higher fDNA, hypoxia biomarkers (lactate, L/P ratio, VEGF, HIF-1α), and oxidative stress markers (OSI, MDA) and significantly lower TAC in patients with thalassemia than the healthy controls. The differences in some clinical characteristics and the investigated hypoxia and oxidative stress biomarkers between the splenectomized patients and nonsplenectomized patients with thalassemia are shown in Table 3. There was a significantly lower TAC and significantly higher VEGF, MDA, and fDNA levels in splenectomized than in nonsplenectomized patients with thalassemia.
The Relevant Clinical, Anthropometric, and Some Laboratory Characteristics of the 56 Patients With β Thalassemia.a
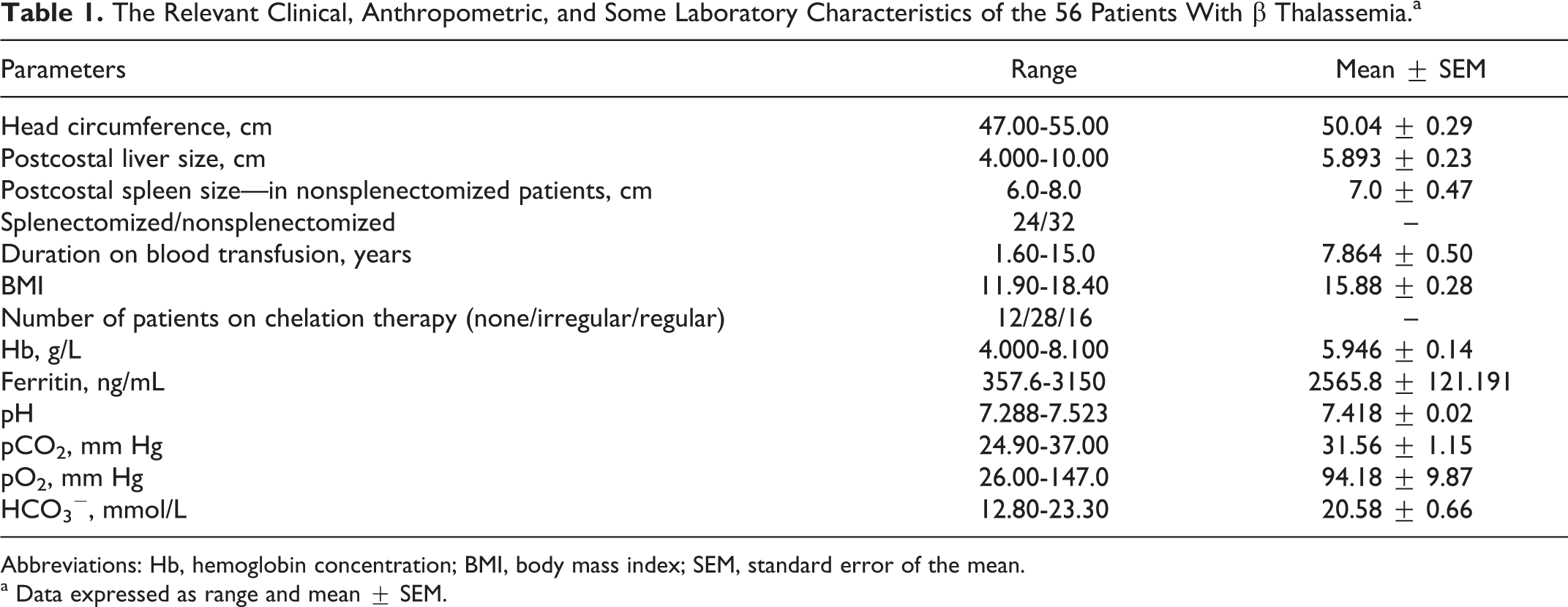
Abbreviations: Hb, hemoglobin concentration; BMI, body mass index; SEM, standard error of the mean.
a Data expressed as range and mean ± SEM.
Differences Between Healthy Controls and Pediatric Patients With Thalassemia in the Levels of the Investigated Hypoxia and Oxidative Stress Biomarkers.a
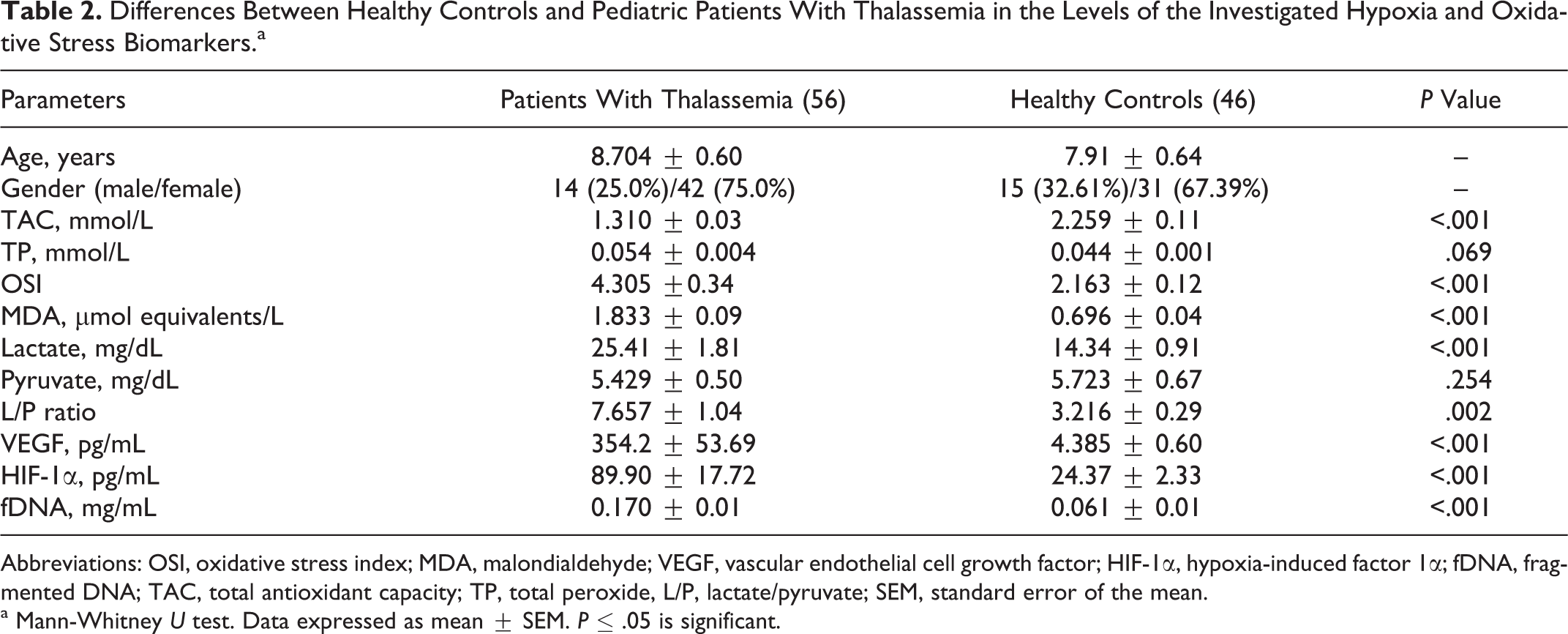
Abbreviations: OSI, oxidative stress index; MDA, malondialdehyde; VEGF, vascular endothelial cell growth factor; HIF-1α, hypoxia-induced factor 1α; fDNA, fragmented DNA; TAC, total antioxidant capacity; TP, total peroxide, L/P, lactate/pyruvate; SEM, standard error of the mean.
a Mann-Whitney U test. Data expressed as mean ± SEM. P ≤ .05 is significant.
The Differences in Some Clinical Characteristics and the Investigated Hypoxia and Oxidative Stress Biomarkers Between the Splenectomized Patients and Nonsplenectomized Patients With Thalassemia.a
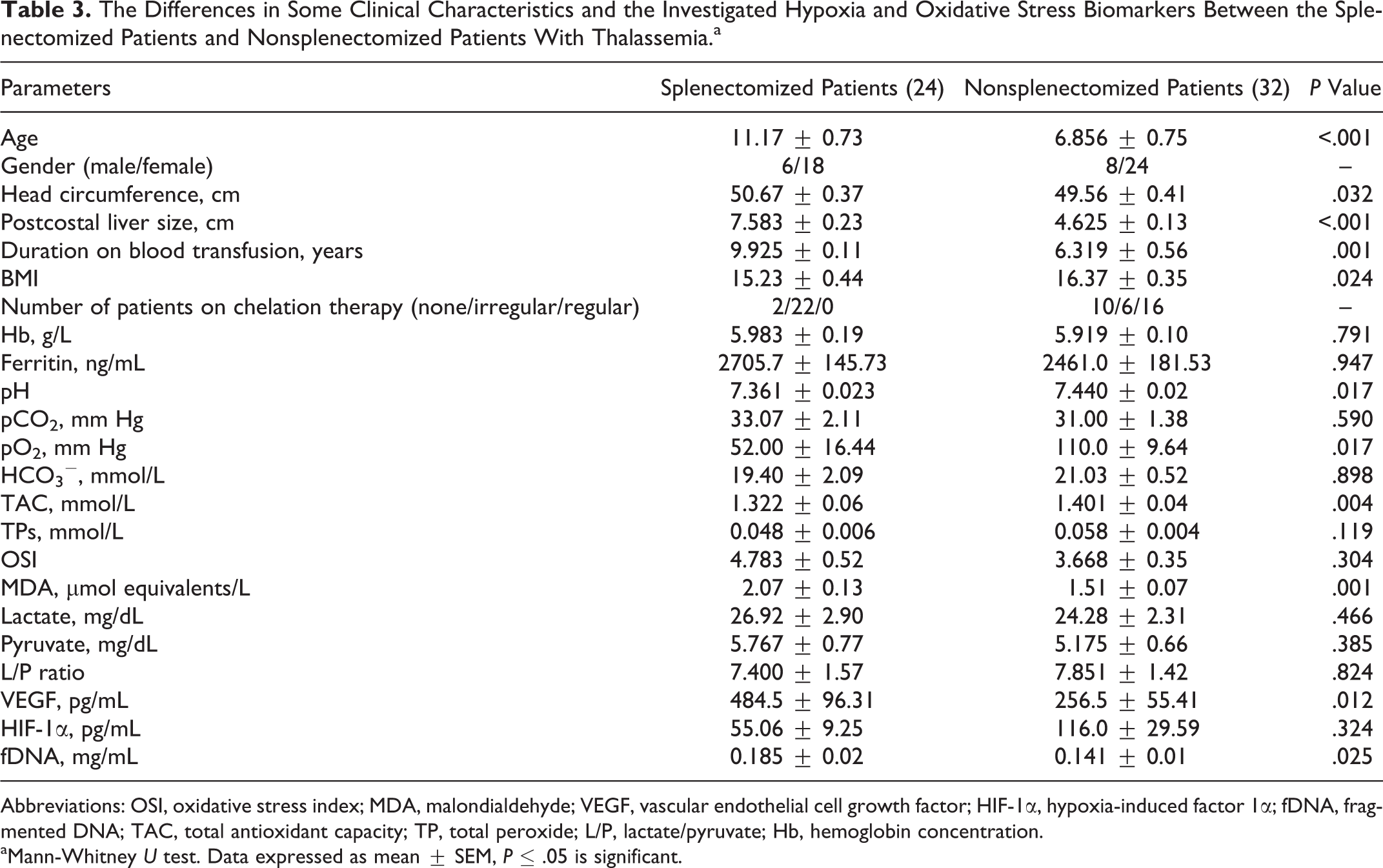
Abbreviations: OSI, oxidative stress index; MDA, malondialdehyde; VEGF, vascular endothelial cell growth factor; HIF-1α, hypoxia-induced factor 1α; fDNA, fragmented DNA; TAC, total antioxidant capacity; TP, total peroxide; L/P, lactate/pyruvate; Hb, hemoglobin concentration.
aMann-Whitney U test. Data expressed as mean ± SEM, P ≤ .05 is significant.
The percentage of total number of circulating MPs was significantly increased in patients with thalassemia than in the controls. The expression of annexin V (procoagulant MPs) in the patients’ MPs was significantly increased than the controls. The expression of the platelet markers, CD61 and CD41 on MPs (platelet MPs) and platelet activation markers, CD62P were significantly higher on MPs from the patients than on those from the controls. At the same time, the expression of the endothelial marker, CD146 (endothelial MPs) and erythrocyte marker, antiglycophorin A+ (erythrocyte MPs) were significantly higher in the patients than in the controls. Microparticles of other cellular origins such as leukocytes (CD45+) and monocytes (CD14+) were not significantly different between the patients and the controls (Table 4).
Circulating Microparticles in Patients With Thalassemia and Controls.a
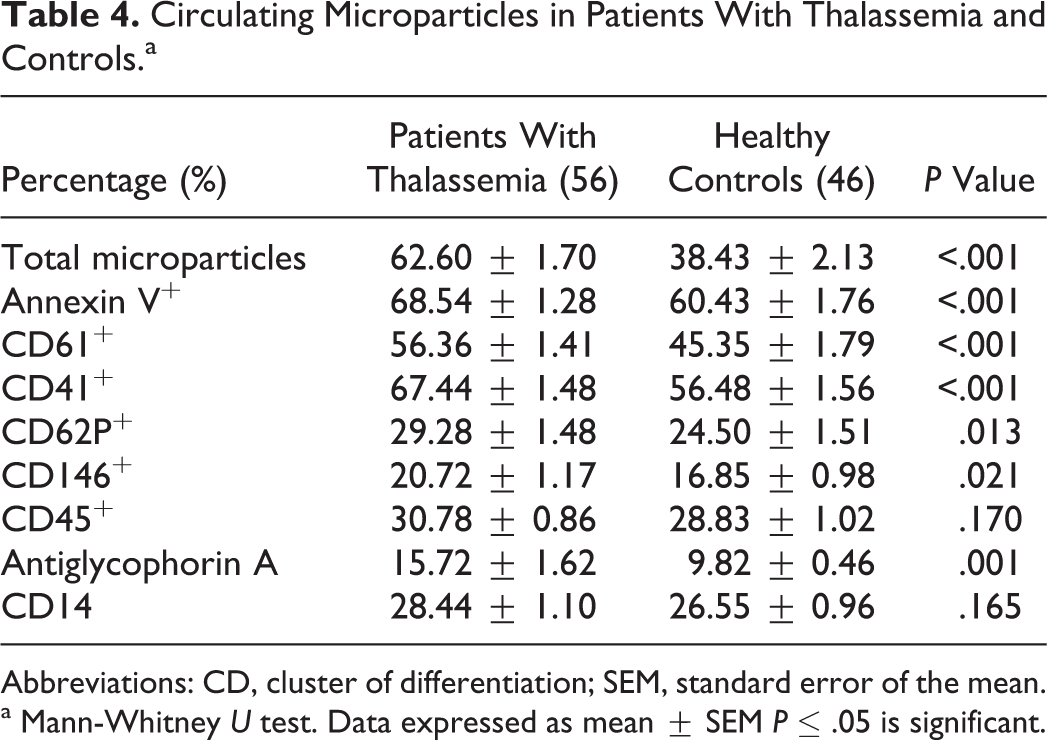
Abbreviations: CD, cluster of differentiation; SEM, standard error of the mean.
a Mann-Whitney U test. Data expressed as mean ± SEM P ≤ .05 is significant.
When comparing splenectomized with nonsplenectomized patients, the percentage of total number of circulating MPs was significantly higher in splenectomized patients than in the nonsplenectomized patients. The MPs of endothelial, platelet, and activated platelet origin were significantly higher in splenectomized patients than the nonsplenectomized patients (Table 5). Many significant correlations were present between the studied parameters (Table 6).
Circulating Microparticles in Splenectomized and Nonsplenectomizd Patients with thalassemi.a
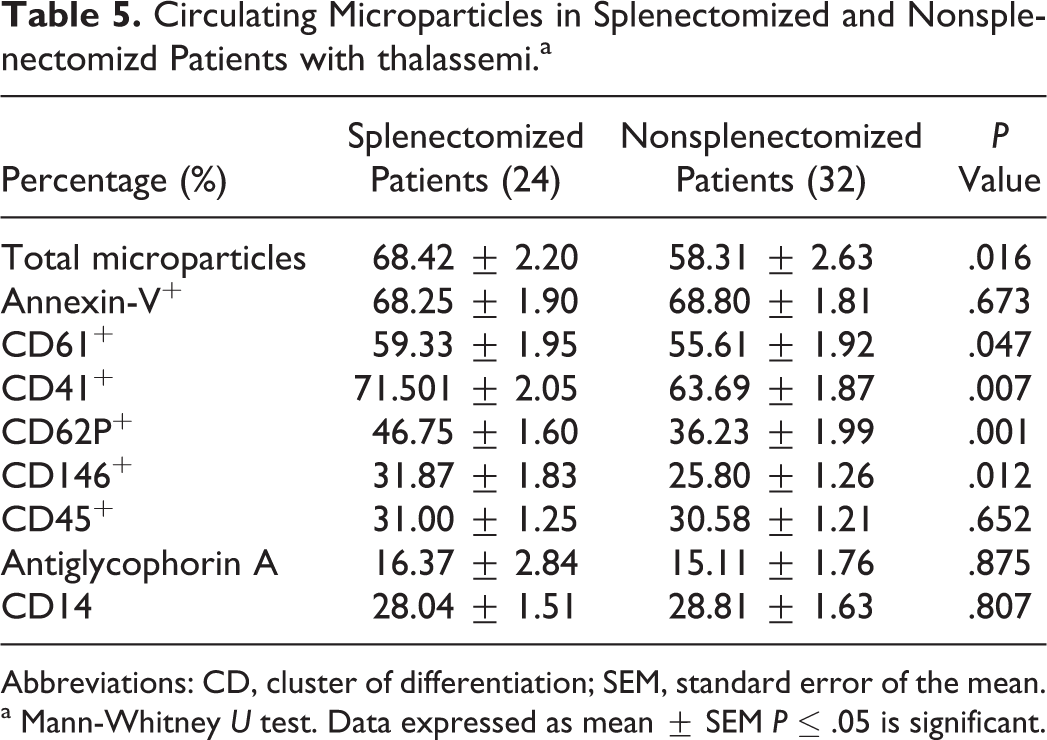
Abbreviations: CD, cluster of differentiation; SEM, standard error of the mean.
a Mann-Whitney U test. Data expressed as mean ± SEM P ≤ .05 is significant.
Correlation Results Among Some Investigated Parameters in Patients With Thalassemia.
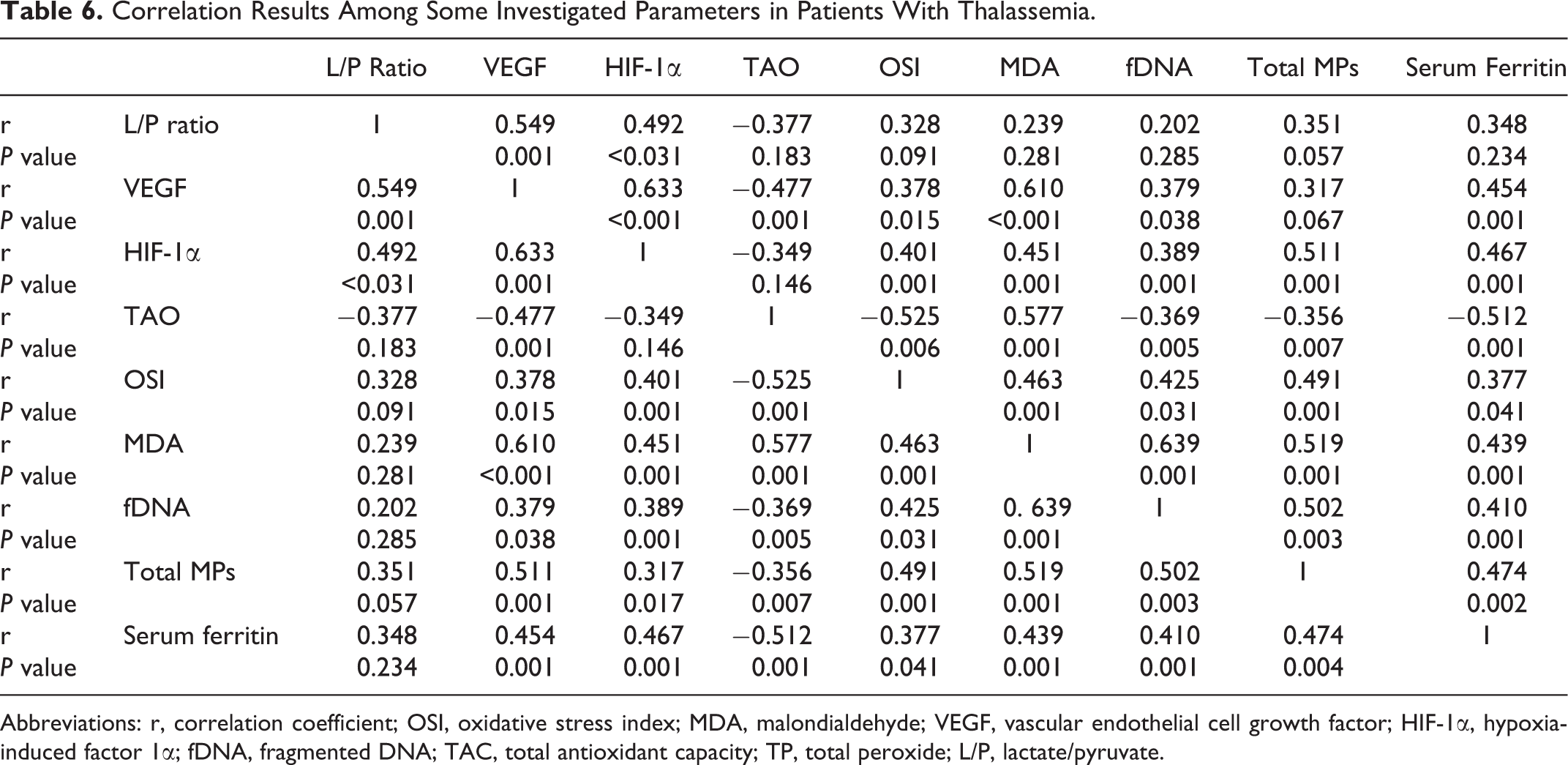
Abbreviations: r, correlation coefficient; OSI, oxidative stress index; MDA, malondialdehyde; VEGF, vascular endothelial cell growth factor; HIF-1α, hypoxia-induced factor 1α; fDNA, fragmented DNA; TAC, total antioxidant capacity; TP, total peroxide; L/P, lactate/pyruvate.
Discussion
Patients with β thalassemia major accumulate body iron over time which causes hepatic, endocrine, and cardiac complications. 27,28 In our patients, the serum level of ferritin was markedly increased. The increased serum ferritin in patients with thalassemia was due to repeated blood transfusion. 29 The other potential cause for hyperferritinemia in patients with thalassemia includes the fact that these patients had anoxia due to low hemoglobin level in their blood. Ferritin concentration has been shown to increase in response to stresses such as anoxia. 30
In this study, we investigated hypoxia, oxidative stress, DNA damage, and procoagulant biomarkers. Although all these markers have been previously investigated in patients with thalassemia, in our study all these factors are simultaneously analyzed and possible correlations among them are demonstrated. Tissue hypoxia is a common feature of anemia. Hypoxia is accompanied by a significant increase in blood lactate and systemic acidosis as a direct effect of anerobic metabolism. Besides the direct effects of anaerobic metabolism, catecholamine induced stimulation of cellular glycolysis and subsequent synthesis of lactate. In such conditions, accumulated pyruvate is metabolized into lactate. So determination of the L/P ratio provides a more accurate statement of tissue metabolism and the cytosolic redox condition. 31 In our patients, the increased level of lactate and L/P ratio could be explained by the above-mentioned mechanism.
Under hypoxic/ischemic conditions, HIF induces upregulation of transcription of various cytokines including EPO and VEGF. 32 In contrast to the rapid regulation of lactate in response to hypoxia, changes in protein transcription require more time. In patients with thalassemia, the peripheral oxygen demand is greater than the supply for a prolonged period and the resulting tissue hypo oxygenation induces HIF-1-regulated VEGF synthesis. 33
Although high lactate values suggested tissue hypoxia in patients with anemia, it failed to predict the need for RBCs transfusion. 34,35 In addition, this parameter responds quickly to hypoxia, reasons other than anemia, such as hemodynamic alterations, can lead to an elevation in lactate level. In contrast, HIF-regulated changes in VEGF concentration occur relatively slowly. Thus, VEGF is a more appropriate indicator of chronic hypoxic conditions such as anemia. 36,37 The VEGF may serve as an excellent surrogate parameter for identifying tissue hypoxia in patients with anemia and, thus help in determining the need for RBCs transfusion. The marked elevation of VEGF concentrations in our patients with thalassemia suggests insufficient peripheral tissue oxygenation and the need for RBCs transfusion. These data are in accordance with that of the previous studies which showed elevated VEGF concentrations in adult patients with renal 36 or sickle cell disease (SCD). 37
In splenectomized patients with thalassemia, VEGF level was higher than in nonsplenectomized patients which means more tissue hypoxia in splenectomized patients with thalassemia. This could be due to more severe pulmonary affection of splenectomized patients and pulmonary arterioles occlusion which cause more hypoxia. In accordance with our finding, Butthep et al 38 found higher concentration of VEGF in splenectomized patients with thalassemia. Splenectomized patients have higher plasma hemoglobin level than nonsplenectomized patients with thalassemia and higher circulating hemoglobin containing vesicles which worsen the pulmonary hypertension after splenectomy. 39 In previous work on our patients with thalassemia, echocardiographic evidence of pulmonary hypertension was detected in 68.75% of the cases. It is more common in splenectomized than nonsplenectomized patients. 40
In this study, HIF-1α was investigated as a hypoxia biomarker for the first-time in patients with thalassemia. Plasma HIF-1α positively correlated with L/P ratio and VEGF. This indicates the utility of HIF-1α as plasma biomarker of cellular hypoxia in thalassemia which reflects the disease outcomes.
Peroxidative damage of lipids is indicated by the increase in serum MDA levels which is a good indicator of oxidative damage. The extent of lipid peroxidation denotes the amount of free oxygen radicals generated, which have not been scavenged by the defense mechanism. In the present study, MDA in our patients with thalassemia showed a significant increase relative to the control group, indicating an increase in the superoxidation of lipids which results from increased oxidative stress in thalassemia. In accordance with our results, Walter et al 41 found that MDA was significantly increased in patients with thalassemia compared to controls. Cighetti et al 42 also found elevated level of MDA in thalassemia. Consequently, there is a rationale for iron chelation to eliminate the free iron species, which in this respect, act like antioxidants. 43
In the present study, the antioxidant defense was evaluated by measuring TAC in serum. The measurement of different antioxidant molecules separately is labor intensive, time-consuming, and costly. Moreover, some investigators suggest that assessment of TAC of plasma may be more useful than measuring each antioxidants individually since their synergistic interaction could be determined. 44,45 In our patients, the TAC was significantly lower with increased levels of TP and OSI. There are limited studies about the assessment of serum TAC in patients with thalassemia. Ghon et al showed the depletion of antioxidants in patients with thalassemia. 46 In a study in Italy, 47 a significant decrease in TAC in patients with thalassemia was reported compared to controls. However, Cakmak et al 48 reported no significant differences in TAC between thalassemic and control groups, in spite of increased level of oxidant status and OSI. In our splenectomized patients with thalassemia, TAC is markedly reduced and MDA is increased than in nonsplenectomized patients. This finding can be explained by increased hypoxia and oxidative stress present in splenectomized patients. In our patients with thalassemia, there were positive correlations between serum ferritin, VEGF, HIF-1α, MDA, and OSI. All these parameters correlated negatively with TAC. These correlations indicate that more tissue hypoxia in patients with thalassemia induce more iron overload that induces more oxidative stress. In 2 of our females patients, there was a delay in pubertal onset and one of them had cardiomyopathy. In addition, one of our male patient had portal vein thrombosis and 2 had paraparesis. These 5 patients had the highest level of serum ferritin, VEGF, HIF-1α, MDA, OSI, and fDNA, while TAC demonstrated the lowest level.
The elevated level of fDNA in our patients with thalassemia was in accordance with that of Kunwittaya et al 49 who found increased DNA damage in patients with thalassemia. Also, Söker et al 50 found elevated DNA damage in patients with thalassemia. Patients with thalassemia have coexisting nutritional deficits 51 and iron-induced oxidative stress, 52,53 both of which are expected to increase DNA damage. In splenectomized patients, fDNA is higher in splenectomized than in nonsplenectomized patients. The fDNA was positively correlated with serum ferritin, MDA, and OSI. Iron overload can stimulate lipid peroxidation, hence generating miscoding DNA adducts in patients with thalassemia and more DNA fragmentation. In accordance with our finding, Meerang et al 54 found positive correlation between DNA damage with MDA and serum ferritin in patients with thalassemia.
The percentage of total MPs was significantly higher in patients with thalassemia than the controls and higher in splenectomized than in nonsplenectomized patients. Patients who had cardiomyopathy, portal vein thrombosis, or paraparesis had the highest level of total MPs. Patients with β thalassemia have a hypercoagulable state and this hypercoagulabilty is more in splenectomized patients. 55,56 The MPs participate in hemostasis and have procoagulant potential in thalassemia. 57 The higher expression of annexin V (procoagulant MPs) in our patients means that MPs of patients with thalassemia contain more phosphatidylserine in their outer surfaces. 58 Such phosphatidylserine may contribute to the pathogenesis of thrombosis. 59
The elevated endothelial MPs in our patients not only constitute an emerging marker of endothelial dysfunction, but also considered to play a major biological role in inflammation, vascular injury, angiogenesis, and thrombosis. Recent studies indicate that endothelial MPs are able to decrease nitric oxide-dependent vasodilatation, increase arterial stiffness, promote inflammation, and initiate thrombosis. 60 Platelet MPs supposed to contribute to the development of thrombotic complications in the pathologic states associated with the increase in their blood concentration. 61 Activated platelets MPs provide a procoagulant aminophospholipid surface for the assembly of the specific enzymes of the coagulation cascade. Erythrocyte-derived MPs enhance coagulation activation. 62 Endothelial MPs, platelet MPs, and activated platelet MPs were significantly higher in our splenectomized patients. Habib et al 63 reported that the values of circulating MPs in patients with thalassemia intermedia than the controls with highest statistical significance in MPs of RBCs, endothelial, and leukocytic origins, while platelet MPs were significantly higher in splenectomized patients versus nonsplenectomized patients. Different types of MPs were presents in other types of hemolytic anemia; Wun et al 64 and van Beers et al 65 found high numbers of both erythrocyte-derived and platelet-derived MPs in patients with SCD. Nantakomol et al 66 found increased MPs in individuals with glucose-6-phospate dehydrogenase deficiency; MPs were largely derived from RBCs and platelets. Pattanapanyasat et al 67 showed that splenectomized patients with β-thalassemia/HbE had significantly higher levels of MPs than nonsplenectomized patients. Shet et al 68 found endothelial- and monocyte-derived MP in SCD. In our study, we were not able to detect a significant different between patients and controls in the small subset of MPs such as leukocyte or monocyte MPs, this might be due to the differences in the centrifugation forces used to isolate the MPs.
The percentage of total MPs was correlated positively with, serum ferritin, VEGF, OSI, and MDA, suggesting that both hypoxia and oxidative stress are responsible for increasing MPs in thalassemia. During periods of hypoxia, cells are metabolically compromised resulting in cellular dysfunction that can eventually lead to cell death through metabolic failure. Neutrophils bound to the endothelial surface accelerate endothelial cell dysfunction and tissue destruction through a variety of cytotoxic mechanisms. Activated neutrophils mediate a variety of harmful responses, such as production of ROS, cytotoxic enzymes, and cytokines. 69 The ROS, in large quantities, disrupt the redox balance of cells resulting in oxidative stress and damage to membrane structures possibly leading to membrane MPs release. 70
Conclusion
The increased tissue hypoxia, oxidative stress in β thalassemia, and its relationship with DNA damage and MPs release could explain many complications of thalassemia and may have therapeutic implications. The VEGF could serve as an important indicator for adequacy of blood transfusion in thalassemia.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
