Abstract
Iron-induced cardiovascular disease is the leading cause of death in iron-overloaded patients. Deferasirox is a novel tridentate oral chelator that exhibits a half-life suitable for once-daily dosing; however, little is known regarding the effectiveness of this agent in preventing iron-induced cardiovascular disease. Adult male Mongolian gerbils were randomly divided into 3 groups: control, iron overload, and iron overload followed by deferasirox treatment. Iron-overloaded animals received iron dextran 100 mg/kg intraperitoneally (ip)/5 days for 10 weeks, while deferasirox was given 100 mg/kg per d orally (po) for 9 months post iron loading. Cardiac and aortic iron levels were determined by inductively coupled plasma atomic emission spectrometry. Gerbil electro- and echocardiograms were obtained in anesthetized animals at regular intervals. Compared to control animals, iron concentration was 3.3- and 2.4-fold higher in iron-overloaded heart and aorta, respectively (P < .05). Deferasirox treatment reduced cardiac and aortic iron levels by 32% and 35%, respectively (P < .05). These results were consistent with the decrease in cellular iron deposition observed with Prussian Blue iron staining. Iron-overloaded gerbils were found to exhibit frequent arrhythmias including premature ventricular contractions, supraventricular tachycardia, and recurrent ventricular tachycardia. In addition, echocardiographic assessment demonstrated iron overload-associated increase in left ventricular dimensions including left ventricular posterior wall dimension (LVPWd: 49%), left ventricular internal dimension (LVIDd: 26%), and left ventricular septum thickness (LVSd: 42%). These parameters were significantly reduced with deferasirox treatment (LVPWd: 23%, LVIDd: 24%, and LVSd: 27%). Iron overload was also associated with reduced ejection fraction (EF: by 30%) and fractional shortening (FS: by 23%) in comparison with controls (P < .05). With deferasirox treatment, these values were higher (EF: by 30%, FS: by 28%) compared to iron-overloaded group. These findings suggest that deferasirox may be useful for attenuating iron-induced changes in cardiac structure and function.
Introduction
Despite dramatic improvement in care, iron-induced cardiac disease remains the most frequent cause of death in thalassemia major and a major life-limiting complication of other transfusion-dependent patients, hereditary hemochromatosis, and other forms of iron overload. 1 The most common form of cardiac hemosiderotic injury is dilated cardiomyopathy, which is characterized by systolic or diastolic dysfunction. 2 Signs of myocardial damage due to iron overload include arrhythmia, angina, and heart failure. 2 Iron overload can also produce conduction defects secondary to iron deposition in the bundle of His and the Purkinje system. 3 Sudden death due to arrhythmia can therefore occur among patients with advanced iron overload. 4 Although prophylactic therapy with angiotensin-converting enzyme inhibitors and β-blockers can help reduce cardiac morbidity and mortality, the mainstay of therapy for cardiac iron overload remains iron-chelating agents. 5 The goal of chelation therapy is primarily the prevention of iron overload in order to preserve organ function and improve patient survivability. To date, only deferoxamine is globally available for the first-line treatment of transfusion-related iron overload. Although effective in increasing survival rate and improving quality of life, 6 deferoxamine chelation therapy is associated with a cumbersome treatment regimen and toxic side effects, resulting in low patient adherence. In addition, treatment with deferoxamine has limited clinical outcomes and some patients still die of iron-induced cardiac disease despite apparently adequate liver iron chelation.7–9
Unlike deferoxamine, deferasirox is an orally effective iron-chelating agent with favorable patient satisfaction.10,11 The half-life of deferasirox is between 8 and 16 hours, allowing once-daily dosing.10,11 Two molecules of deferasirox are capable of binding to 1 atom of iron, which are subsequently eliminated by fecal excretion (Figure 1). 11 Deferasirox was recently approved by the Food and Drug Administration (FDA) and is the first oral medication approved in the United States for this purpose.12,13 As deferasirox is well tolerated, it is expected to greatly enhance the acceptance of iron-chelation therapy and offer a new alternative to burdensome continuous infusion therapy. The objective of this study is to investigate whether deferasirox is capable of removing excess iron from the heart and other tissues, and whether it is capable of preventing or reducing the severity of iron-induced cardiac complications in the iron-overloaded gerbil, a well-accepted animal model of iron overload that is thought to closely resemble the effects of iron overload seen in humans. 14 Our data suggest that once-daily oral deferasirox treatment appears to be effective in reducing iron-induced cardiovascular changes.

Ball and stick model of 2 molecules of deferasirox binding an iron atom (grey center). Adapted from http://upload.wikimedia.org/wikipedia/commons/a/a8/Deferasirox%E2%80%93iron%28III%29_complex.png. Accessed March 21, 2011.
Materials and Methods
Animal Selection and Care
All experiments were conducted using male Mongolian gerbils. Three-month-old Mongolian gerbils (Meriones unguiculatus) were obtained from Charles River Laboratories and housed 4 per cage in an American Association of Laboratory Animal Care (AALAC)-approved vivarium. Housing conditions consisted of a 12h:12h dark-light cycle, with temperature maintained at 22°C ± 2°C. Animals were provided food and water ad libitum. Gerbils were allowed to acclimate to the housing facilities for at least 2 weeks before the experimentation began. During this time, the animals were carefully observed. None of the animals exhibited signs of failure to thrive, such as precipitous weight loss, disinterest in the environment, or unexpected gait alterations. Animal care and procedures were conducted in accordance with the Institutional Animal Care and Use Committee of Marshall University using the criteria outlined by the AALAC as proclaimed in the Animal Welfare Act (PL89-544, PL91-979, and PL94-279).
Iron Loading and Chelation
Gerbils were divided into 3 groups (10 animals/group): control (Ctrl), iron overload (IO), and iron overload followed by deferasirox treatment (IO + DFR). Iron-treated animals received 15 intraperitoneal (ip) injections of iron dextran (Sigma Chemical, St. Louis, MO) at a dose of 100 mg/kg. After the completion of iron loading, deferasirox was administered orally for 9 months. To avoid the stress of repeated gavage feeding, deferasirox was homogenously mixed in plain peanut butter (vehicle) for oral feeding via a 1-mL syringe. Deferasirox was given at a single daily dosage of 100 mg/kg as outlined previously. 15 This dosage is based on a previously determined dose–response curve of deferasirox in iron-overloaded gerbils. 16 Oral chelator administration gave the animals approximately 0.15 mL of peanut butter per day. Age-matched Ctrl and IO groups were maintained for comparison and were given peanut butter (vehicle) only (Table 1).
Electrocardiographic Procedures
Gerbils were anesthetized with ip injections of ketamine (100 mg/mL)/xylazine (20 mg/mL) and electrocardiograph (EKG) recordings were performed using standard limb lead system (Biopac Systems, Inc., Goleta, CA). To emulate lead II, we utilized subcutaneously placed electrodes located in line with the long access of the heart. One electrode (anode) was placed in the upper right quadrant of the chest, while the other lead (cathode) was inserted in the lower left abdominal wall, just cranial to the groin area. Lead I configuration was performed by placing the cathode and the anode in the upper right and left quadrants of the chest, respectively. Lead III configuration was performed by placing the cathode in the upper left quadrant of the chest, while the anode was placed in the lower left abdominal wall. Data were collected and reviewed in a blinded fashion from each of the animals in the experimental groups (n = 10/group).
Transthoracic Echocardiography
Animals were anesthetized with ip injections of ketamine (100 mg/mL)/xylazine (20 mg/mL) and subjected to echocardiographic recordings. Gerbils were shaved in the chest area for adequate sonic transference and ultrasonic transmission gel was applied to the chest area. Two-dimensional echocardiographic measurements, 2-dimensional guided M-mode, Doppler M-mode, and other recordings from parasternal long- (PLAX) and short-axis (PSAX) views were obtained using a Phillips 5500 ECHO system with a 12-MHz transducer. The echocardiographic views were then used to position the M-mode echocardiographic line. In the long-axis procedures, the probe was oriented toward the base of the heart projecting toward the apex (x-axis) with depth along the y-axis, thus allowing pulse wave Doppler evaluation of valvular blood flow velocities. In the short-axis procedures, the probe was oriented toward the left ventricle and across the heart for evaluation of wall structure, which was utilized in the calculation of ejection fraction (EF) and fractional shortening (FS) during systole. M-mode displays were analyzed by a digital echocardiographic analysis system. Measurements from 6 gerbils were obtained for each assessment of cardiac structure and function.
Tissue Collection
Prior to sacrifice, gerbils were anesthetized with a ketamine (100 mg/mL)/xylazine (20 mg/mL) mixture and supplemented as necessary to achieve loss of reflexive response. After midline laparotomy, heart, aorta, and liver were removed and placed in Krebs Ringer bicarbonate buffer (KRB) containing: 118 mmol/L NaCl, 4.7 mmmol/L KCl, 2.5 mmmol/L CaCl2, 1.2 mmmol/L KH2PO4, 1.2 mmmol/L MgSO4, 24.2 mmmol/L NaHCO3, and 10 mmmol/L α-
Tissue Iron Assessment
Portions of the heart, aorta, and liver were weighed and analyzed for quantitative iron determination by inductively coupled plasma atomic emission spectrometry (ICP-AES). Briefly, digestions were performed using commercially available trace metal grade HNO3 (ThermoFisher Scientific, Waltham, MA). Dilutions were made with 2% (vol) HNO3 (trace metal grade) prepared with deionized water. All digestion tubes, volumetric pipettes, and volumetric glassware were soaked for 24 hours in 2% (vol) HNO3 (trace metal grade), rinsed 4 times with deionized water, and air-dried prior to use. Approximately 300 mg (wet weight) of sample was weighed into a glass digestion tube and 6.0 mL of concentrated trace metal grade HNO3 added. A 1000 mg/L Yttrium solution in 2% HNO3 (PerkinElmer, Waltham, MA), used as an internal standard (final concentration 1.67 mg/L), was added to each tube. Samples were heated at 37°C for 1 hour and then at 110°C for 2 hours. After digestion, 5.0 mL of the clear solution was transferred to a 50-mL volumetric flask and brought to volume with 2% (vol) HNO3 (trace metal grade). Solutions were filtered through a 0.22 μm syringe-driven filter unit (Millipore, Billerica, MA) and analyzed by ICP-AES. A 1.67 mg/L iron solution was used as a control for the digestion procedure. Analyses were performed on a Perkin Elmer ICP-AES (Optima 2100 DV) using a 0 to 1 mg/L, 0 to 10 mg/L, or 0 to 100 mg/L standard curve, depending upon iron concentration. Calibration and control solutions for the 0-1 mg/L and 0-10 mg/L standard curves were prepared from a multielement standard (ICP Multi-element Solution 2, Spex Chemicals, 10 mg/L). Calibration and control solutions for the 0-100 mg/L standard curve were prepared from a 1000 mg/L iron standard solution (Perkin Elmer). All calibrators and controls contained an internal Yttrium standard. The measurements for iron (238.204 nm) were conducted in the axial mode. Sample concentrations were determined from standard curves generated by linear regression analysis. The minimal detectable concentration for iron was determined using the mean plus the 3-fold standard deviation obtained from 10 blank digestions. Average iron concentration for each tissue type was determined from samples obtained from 6 gerbils in each group.
Histological Analysis
Whole hearts (bread loaf sectioned; n = 6 from each group) were fixed in 10% buffered formalin solution and processed into paraffin blocks using routine procedures. Samples were then sectioned (8 μm) with a cryostat and sections were mounted on polylysine (Sigma)-coated slides. Sections were stained with Prussian Blue iron stain. Slides were evaluated by light microscopy and images were taken at ×200, or ×400 in a blinded fashion.
Statistical Analysis
Results are presented as mean ± standard error of the mean. Data were analyzed using SigmaStat 3.0 computer software. One-way or two-way analysis of variance (ANOVA) was used for overall comparisons. The Student-Newman-Keuls post hoc test was used to determine statistical significance. The level of significance accepted a priori was P ≤ .05. Regression analysis of the dependent variables was performed across experimental groups. Significance of correlation was analyzed by 1-way ANOVA. Values of P ≤ .05 were considered statistically significant.
Results
Characterization of Animals
Total body and heart weights were obtained and compared. All animals tolerated the iron loading and chelation without any apparent ill effects. The body weights of iron-overloaded gerbils were similar to controls, indicating that the injection of iron dextran did not result in debilitation or weight loss. Compared to control animals, average heart and liver weights in the iron-overloaded group were 34% and 41% higher, respectively (P < .05). Conversely, in the deferasirox-treated animals, average heart and liver weights were 24.9% and 16% lower, respectively, than that observed in the iron-overloaded group (P < .05; Table 1 ).
Average Heart and Liver Weights of Gerbils in the Experimental Groups (N = 10/group)

Abbreviations: Ctrl, control; IO, iron overload; DFR, deferasirox; wt, weight; wk, week; mo, month.
aIndicates significant difference from age-matched control
bIndicates significant difference from iron overload groups
Cardiac, Aortic, and Hepatic Iron Levels
Compared to hearts obtained from control animals, cardiac iron concentration was 3.3-fold higher in iron-overloaded animals (P < .05). Deferasirox treatment for 9 months resulted in a 32% decrease in cardiac iron level (P < .05; Figure 2A). Similarly, aortic iron concentration was 2.4-fold higher in the iron-overloaded group compared to controls (P < .05). These levels were 35% lower in the deferasirox-treated animals than that of iron-overloaded group (P < .05; Figure 2B). In the iron-overloaded animals, hepatic iron concentration was 50-fold higher than that of controls (P < .05). Hepatic iron concentration was 47% lower after 9 months of deferasirox treatment compared to that found in the iron-overloaded group (P < .05; Figure 2C). Prussian Blue iron staining demonstrated increase in iron deposition and tissue damage in the left ventricle of the iron-overloaded group which appeared to be diminished following deferasirox treatment (Figure 3 ).

Average iron levels in mg/g tissue weight in cardiac (A), aortic (B), and hepatic (C) tissue of gerbils in the experimental groups followed for 9 months. Ctrl indicates control; IO, iron overload; IO + DFR, iron overload followed by deferasirox treatment; *,significant difference from control; †, indicates significant difference from iron-overloaded group.
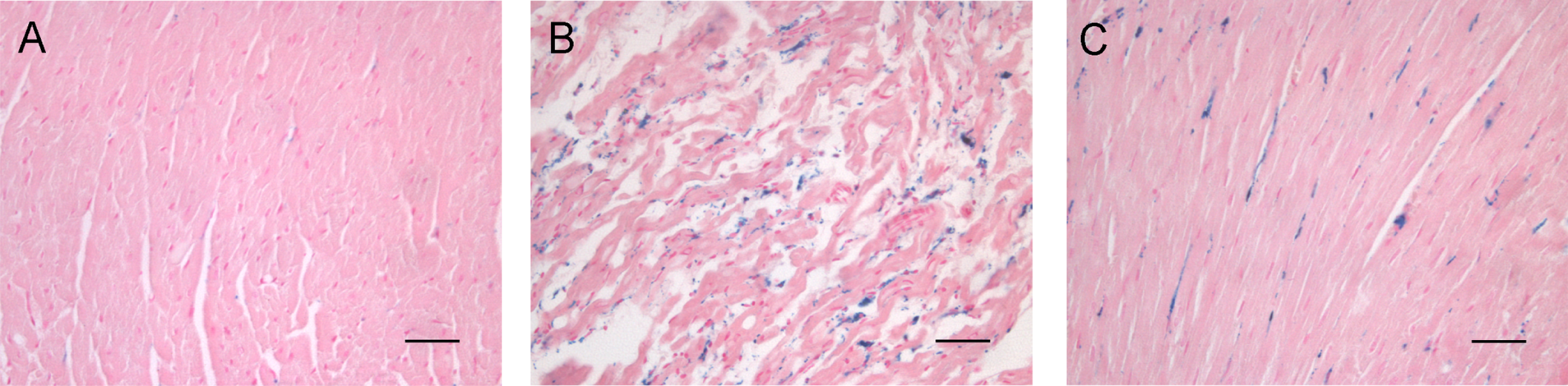
Histological analysis of cardiac tissue of Ctrl (A), IO (B), and IO + DFR (C) gerbils followed for 9 months. Ferric iron was detected by Prussian Blue staining. No apparent iron deposition was observed in control gerbils, while iron overload was associated with alterations in cardiac morphology and iron accumulation in the interstitium and cardiac myocytes. Deferasirox treatment appears to preserve tissue morphology and diminish iron deposition. Bar represents 100 µm; images were taken at ×400 original magnification. Ctrl indicates control; IO, iron overload; IO + DFR, iron overload followed by deferasirox treatment.
Electrocardiographic Evaluation
Electrocardiographic measurements suggested an increased incidence of arrhythmias in the iron-overloaded group. Representative EKGs recorded in vivo (Figure 4 , upper panel) demonstrate ventricular tachycardia in an iron-overloaded gerbil (middle) compared with normal sinus rhythm in a control (left) and a deferasirox-treated gerbil (right). Over the course of all experiments, EKGs from iron-overloaded gerbils demonstrated frequent episodes of premature ventricular contractions (PVC: 6/10), supraventricular tachycardia (SVT: 3/10), recurrent sustained and nonsustained ventricular tachycardia (VT: 4/10), and bundle branch block (BBB: 6/10; Figure 4, lower panel) compared to findings in control gerbils. The frequency of these EKG changes was relatively lower in deferasirox-treated gerbils and no evidence of ventricular tachycardia was observed (Figure 4, lower panel).

Effect of iron overload and deferasirox treatment on cardiac rhythm in gerbils from experimental groups. (Upper panel) Representative EKGs (standard leads I, II, III) from Ctrl, IO, and IO + DFR groups followed for 9 months. EKG trace from iron overloaded animal (middle) exhibits ventricular tachycardia, EKG trace from control (left) and deferasirox-treated animal (right) exhibit normal rhythm. (Lower panel) Frequency of EKG abnormalities observed in Ctrl, IO, and IO + DFR followed for 9 months. PVC indicates premature ventricular contraction; SVT, supraventricular tachycardia; VT, ventricular tachycardia; BBB, bundle branch block; EKG, electrocardiogram.
Echocardiographic Evaluation (Cardiac Structure)
Left ventricular posterior wall dimension during diastole (LVPWd) increased by 39% and 49% in the iron-overloaded group after 6 and 9 months of follow-up, respectively (P < .05; Figure 5 ). Left ventricular posterior wall dimension during diastole was 21% and 23% lower after 6 and 9 months, respectively, of deferasirox treatment (P < .05; Figure 5). Similarly, left ventricular internal dimension during diastole (LVIDd) increased by 19% and 26% in the iron-overloaded animals after 6 and 9 months of follow-up (P < .05; Figure 5). In the deferasirox treatment group, LVIDd was 17% and 24% lower than that of iron-overloaded group after 6 and 9 months respectively (P < .05). Compared to control animals, left ventricular septum thickness during diastole (LVSd) was 32% and 42% higher in the iron-overloaded group after 6 and 9 months of follow-up (P < .05), while these values were decreased by 23% and 27% following 6 and 9 months of deferasirox treatment, respectively (P < .05; Figure 5).

Effect of iron overload and deferasirox treatment on left ventricular dimensions. Ctrl indicates control; IO, iron overload; IO + DFR, iron overload followed by deferasirox treatment. BL indicates baseline (before iron loading); mo, months of follow-up; LVPWd, left ventricular posterior wall dimension during diastole; LVIDd, left ventricular internal dimension during diastole; LVSd, left ventricular septal dimension during diastole; *, significant difference from control; †, significant difference from iron-overloaded group.
Echocardiographic Evaluation (Cardiac Function)
Compared to control animals, EF was 22%, and 30% lower in the iron-overloaded group after 6 and 9 months of follow-up, respectively (P < .05; Figure 6 ). Supporting these data, FS was 13% and 23% lower than that observed in control animals after 6 and 9 months of follow-up respectively (P < .05; Figure 6). With deferasirox treatment, EF was 18% and 30% higher, while FS was 10% and 28% higher than that observed in the iron-overloaded group after 6 and 9 months of follow-up respectively (P < .05; Figure 6). In addition, gerbils in the iron-overloaded group exhibited a higher incidence of mitral valve regurgitation (MR: 6/10), tricuspid valve regurgitation (TR: 6/10), and aortic valve regurgitation (AR: 3/10). Deferasirox-treated animals exhibited lower incidence of cardiac valve regurgitations (MR: 3/10, TR: 2/10, and AR: 2/10).

Effect of iron overload and deferasirox treatment on left ventricular ejection fraction (left) and fractional shortening (right) in gerbils from experimental groups. Ctrl indicates control; IO, iron overload; IO + DFR, iron overload followed by deferasirox treatment; BL, baseline (before iron loading); mo, months of follow-up; *, significant difference from control; †, significant difference from iron-overloaded group.
Discussion
Cardiac disease is the number one health concern in patients with transfusional iron overload. To our knowledge, this is the first study to examine the long-term effect of deferasirox treatment in preventing iron-induced cardiac complications using the gerbil model of iron overload. Our data suggest that iron overload caused a dramatic increase in cardiac iron deposition and that this elevation in tissue iron was associated with increased incidence of arrhythmias, ventricular remodeling, and reduced EF. Deferasirox treatment significantly reduced cardiac iron and preserved cardiac structure and function suggesting, perhaps for the first time, that this agent may play a protective role against several types of iron-induced cardiac damage.
Deferasirox Removes Cardiac, Aortic, and Hepatic Iron
Iron distribution in the hearts of iron-overloaded gerbils is thought to be similar to that reported in iron-overloaded human hearts. 14 Our findings demonstrated that cardiac and aortic iron concentrations 9 months post iron loading were 3.3- and 2.4-fold higher than that found in the control animals. Hepatic iron levels were strikingly higher than that of heart and aorta and were 50-fold higher than that of corresponding control animals. Chronic deferasirox administration significantly reduced iron concentrations in each of these tissues (Figures 2 and 3), a finding which is consistent with previous studies suggesting that deferasirox is effective in cardiac and hepatic iron removal.15,17 Whether the disparity in iron levels that we see among tissues is related to relative differences in the rate or the mechanism of iron uptake or clearance is not clear. It has been previously suggested that the liver has especially high efficiency of iron clearance that is related to membrane potential. 18 In addition, it is thought that the rate of iron clearance from the myocardium is 6 times slower than that of liver. 19 Nonetheless, as similar findings have been observed with deferoxamine treatment, 20 it is likely that these differences are not chelator-dependent. Specific mechanisms remain to be established.
Deferasirox Treatment Preserves Cardiac Structure and Prevents Ventricular Remodeling
Iron-induced cardiac hypertrophy and ventricular remodeling is a serious long-term complication associated with iron overload. 21 Previous findings have suggested that chelation therapy with deferoxamine helps prevent iron-induced cardiac hypertrophy and failure in the clinic 22 and in animal studies. 1 Whether deferasirox produces a similar effect is not clear. Our findings suggest that iron overload was associated with increased cardiac mass, and a progressive increase in left ventricular wall dimensions, which reached significance at 6 and 9 months following iron loading. With chronic deferasirox treatment, both cardiac mass and left ventricular dimensions tended to be preserved with values similar to that found in the control animals (Table 1 and Figure 5). This finding is in agreement with recent case reports that have demonstrated that deferasirox treatment restored cardiac left ventricular diameter, LVSd, and LVPWd 23 and reversed dilated cardiomyopathy5,23 in patients with iron overload. The mechanism/mechanisms underlying this finding is not known. We have previously demonstrated that deferasirox treatment reduced iron-induced oxidative stress and MAPK phosphorylation, 24 both of which are thought to be implicated in cardiac hypertrophy. Whether these decreases in oxidative stress and MAPK phosphorylation we observe with deferasirox treatment are responsible for the diminished iron-associated cardiac remodeling cannot be established from our current data. Further experimentation examining the effect of deferasirox on ROS, MAPK, and other molecules involved in cardiac hypertrophy in isolated cardiac myocytes will no doubt help us understand the potential link between deferasirox treatment and changes in cardiac structure and function.
Deferasirox Treatment Preserves Cardiac Function and Reduces Incidence of Arrhythmia
Without adequate control of iron overload, ongoing cardiac iron accumulation results in iron deposition in cardiac myocytes and the conduction system, which is thought to predispose the heart to recurrent arrhythmias and heart failure.4,25 The mechanism of iron-induced cardiac arrhythmias is not entirely elucidated, but it has been suggested that labile “free” iron can directly interact and interfere with a variety of ion channels within cardiomyocytes including
Iron overload was also associated with progressive decline in left ventricular EF and FS accompanied with an increase in the frequency of cardiac valve regurgitation. Deferasirox treatment tended to preserve cardiac function and with treatment, only a slight decline in EF was observed (Figure 6). Similar findings have been demonstrated by Kiguchi and collegues 23 where deferasirox treatment restored EF and cardiac functions in a patient with cardiac iron overload. Whether the abnormalities in cardiac structure and function we observe are directly iron-induced is unknown. Nonetheless, the changes that we see in heart mass, left ventricular dimension, EF, and FS are highly correlated with cardiac iron concentration (Figures 7 ). In conclusion, these data suggest that deferasirox may be efficacious in the treatment and/or prevention of iron-overload-associated cardiac complications.

Correlation between cardiac iron concentration and heart/body weight or echocardiographic parameters. LVPWd indicates left ventricular posterior wall dimension during diastole; LVIDd, left ventricular internal wall dimension during diastole; LVSd, left ventricular septal dimension during diastole; EF, ejection fraction; FS, fractional shortening; R, correlation coefficient. Values of P ≤ .05 were considered statistically significant.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Funding and material from Novartis Pharmaceuticals (East Hanover, New Jersey) were used to support the costs associated with portions of this study.
