Abstract
Rectus sheath hematoma (RSH) develops due to rupture of epigastric arteries or the rectus muscle. Although RSH incidence rate is low, it poses a significant diagnostic dilemma. We evaluated the risk factors for RSH, its presentation, management, and outcomes for 115 patients hospitalized with confirmed RSH by computed tomography scan between January 2005 and June 2009. More than three-fourth (77.4%) of the patients were on anticoagulation therapy, 58.3% patients had chronic kidney disease (CKD) stage ≥3, 51.3% had abdominal injections, 41.7% were on steroids/immunosuppressant therapy, 37.4% had abdominal surgery/trauma, 33.9% had cough, femoral puncture was performed in 31.3% of patients, and 29.5% were on antiplatelet therapy. Rectus sheath hematoma was not an attributable cause in any of the 17 deaths. Mortality was significantly higher in patients with CKD stage ≥3 (P = .03) or who required transfusion (P = .007). Better understanding of RSH risk factors will facilitate early diagnoses and improve management.
Introduction
Rectus sheath hematoma (RSH) is an uncommon occurrence which develops due to rupture of epigastric arteries or the rectus muscle itself within the rectus sheath. Rectus sheath hematoma often poses a significant diagnostic dilemma, 1 –5 resulting in unnecessary invasive interventions. Although it is usually characterized by a benign course, severe adverse outcomes including mortality have been reported. 6 –8
There is a limited amount of information available concerning RSH in the medical literature and most comes from case reports. 6,7,9 Cherry et al reported important RSH risk factors including female gender, elderly age, anticoagulation (AC) or antiplatelet therapy (APT), abdominal injections, comorbid conditions, and cough or other abdominal trauma. 6 At our institute, we had 2 consecutive reports of adverse drug events resulting in RSH. Both of these events occurred in middle-aged female patients with chronic kidney disease (CKD) stages 4 and 5 while receiving prophylactic AC with unfractionated heparin (UFH). These patients had none of the other previously described risk factors for RSH besides prophylactic AC. However, both patients were on long-term steroids or immunosuppressant medications. Commonality of these cases ignited interest in evaluating additional risk factors for RSH such as CKD and the use of steroids/immunosuppressant medications which might contribute to an increased risk of bleeding. Better understanding of RSH risk factors will enable physicians to diagnose RSH early in its course, avoid unnecessary invasive procedures, allow for the implementation of prevention strategies, and improve management. Our study aims to evaluate the risk factors for RSH, the outcomes of patients with RSH, and the management of this undesirable complication that could be serious.
Methods
Retrospective evaluation of RSH risk factors was conducted in hospitalized patients identified from the computed tomography (CT) scan reports for RSH recorded in the hospital Medical Archival Retrieval System (MARS) laboratory data system. The MARS query included the CT reports from January 2005 to June 2009. Three investigators, a research faculty, risk management nurse specialist, and an advanced practice nurse, in cardiovascular medicine reviewed the patient charts. All chart reviewers were given the same detailed instructions and list of risk factors as well as other variables for data collection.
The investigators ascertained that each patient has a recent RSH by including only patients with an abdominal CT scan performed no later than 1 day prior to admission to the hospital. Patients with chronic RSH or inadequate documentation were excluded from the analysis. Selected patient charts were reviewed for demographic data (age, gender, and race), body mass index (BMI), pertinent medication (AC, APT, and steroids and immunosuppressant) information, indication for AC, coagulation laboratory parameters for the day the CT scan, RSH risk factors, presenting symptoms, management, and outcome. Patient’s AC regimens were categorized as therapeutic or prophylactic based upon the dose of parenteral AC administered or, in the case of warfarin, the target international normalized ratio (INR). No patient in this series was receiving one of the several novel oral ACs available in our institution.
The RSH risk factors evaluated in our study were demographic characteristics, BMI, AC administration (therapeutic/prophylactic), corticosteroid or immunosuppressant use, abdominal surgery, abdominal injections, femoral puncture, cough, presence of CKD, and cancer. Immunosuppressants were medication classes for malignancy treatment, disease modifying for rheumatoid arthritis and other immunological diseases, and to prevent transplant rejection. Management was categorized as follows: (1) observation (if no additional treatment was required), (2) medical (if pain management, transfusion and coagulopathy reversal, emergency team response, and intensive care unit [ICU] transfer were required), and (3) surgical (if surgical exploration and/or embolization/ligation of bleeding vessel were required). Mortality in the hospital was also collected. The research faculty reviewed records of patients who died to determine whether RSH was a contributing factor. Among those patients in whom AC was reinitiated, the time from the detection of the RSH to reinitiation was recorded.
Statistical Analyses
Descriptive statistics were used to report the demographic characteristics, BMI, RSH risk factors, presenting symptoms of RSH, and its management. Differences in these variables were examined by chi-square or t test as appropriate by AC status and mortality. Factors associated with mortality in patients with RSH were examined using stepwise logistic regression analysis. Factors examined were demographics, BMI, renal failure of CKD stage ≥3, whether patient was on ACs, APT, or steroids/immunosuppressants, all other RSH risk factors, the type of RSH treatment, and whether transfusion was required.
Results
The MARS query from January 2005 to June 2009 for CT diagnosis of RSH returned 146 patients. In all, 31 patients were excluded from the analysis because chart review identified either no recent RSH or inadequate documentation and 115 patients were included in the study. Of the patients, 52 (45.2%) had RSH at the time of admission and 63 (54.8%) developed RSH during the hospital course.
The demographic data and clinical characteristic of the 115 patients are shown in Table 1. In our patient cohort, 73 (63.5%) were females, median age was 65 years (range 19 to 88 years), and 28 (24.3%) were obese with BMI >30. The median INR and activated partial thromboplastin time were 1.3 (range 0.9-9.1) and 65.3 (range 20-150) seconds for patients on warfarin and UFH, respectively. The presenting symptoms of RSH were abdominal pain or ecchymosis in 63 (54.8%), drop in hematocrit/hypotension/mental status changes in 36 (31.3%), or incidentally diagnosed on CT in 16 (13.9%) patients. Risk factors evaluated are shown in Table 2. None of the patients were pregnant or lactating at the time of RSH diagnosis.
Demographic and Clinical Characteristics.

Abbreviations: BMI, body mass index; INR, international normalized ratio; PTT, partial thromboplastin time; UFH, unfractionated heparin.
aMedian (range).
bPatients on warfarin.
cPatients on UFH.
Risk Factors of RSH.
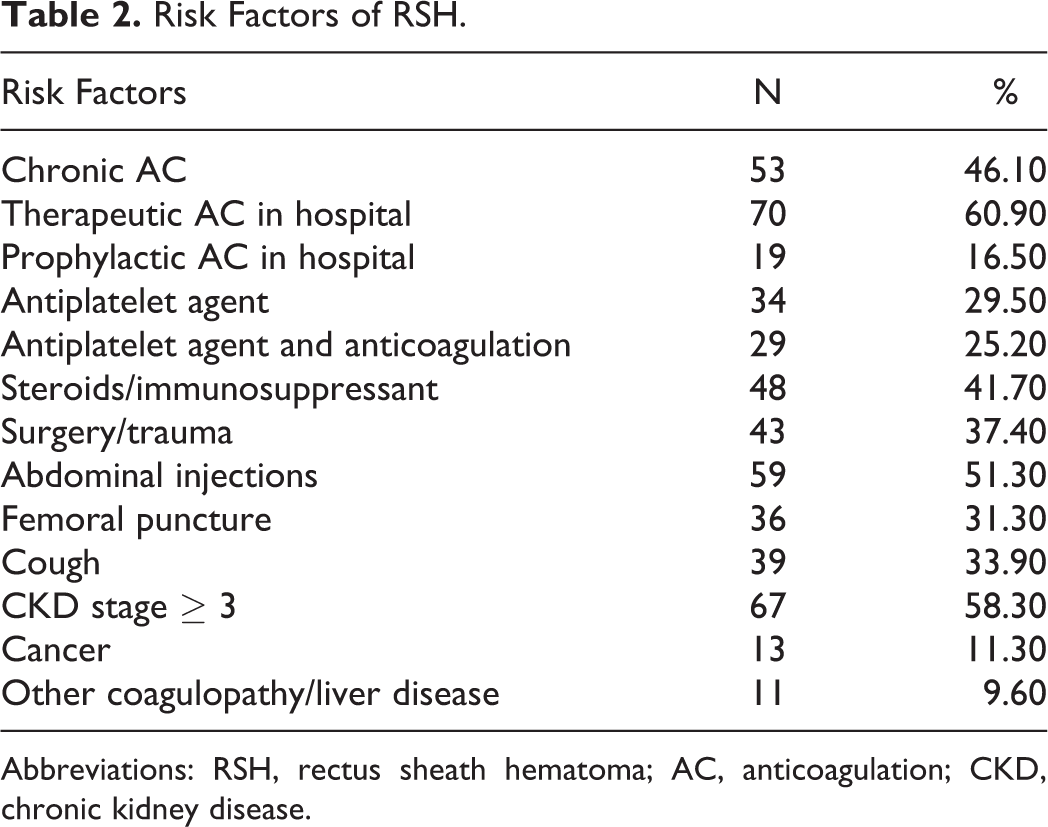
Abbreviations: RSH, rectus sheath hematoma; AC, anticoagulation; CKD, chronic kidney disease.
From the cohort, 53 (46.1%) patients were on chronic AC (defined as AC administered to the patient as an outpatient) and 89 (77.4%) patients received AC in the hospital. Anticoagulation therapy was therapeutic in 70 (60.9%) and prophylactic in 19 (16.5%) patients. In all, 42 patients were on warfarin, 37 patients were on UFH, 17 patients were on low-molecular-weight heparin (LMWH). Twenty (22.5%) patients were on oral and parenteral AC simultaneously. The indications for therapeutic AC were venous thromboembolism in 28 (40%), atrial fibrillation in 25 (35.7%), cardiac valve replacement in 8(11.4%), myocardial infarction or angina in 3 (4.3%), and thrombophilia in 3 (4.3%) patients. In our study, 34 (29.6%) patients were on APT (aspirin or clopidogrel). Twenty-nine (25.2%) patients received AC and APT simultaneously.
Anticoagulation was the most common risk factor (77.4%) followed by CKD (58.3%), abdominal injections (51.3%), and steroids/immunosuppressant medications (41.7%). Patients who were given subcutaneous abdominal injections: anticoagulant injections were SQ UFHs 6.8%, SQ LMWHs 28.8%, and 64.4% were other medications.
Risk factor differences by AC status are reported in Table 3. There were significantly more caucasian (72 of 92 [78.3%]) patients compared to African American patients (6 of 12 [50%]) who had received any form of AC (P = .03). Additionally, more patients had undergone abdominal surgeries in the no AC group and more patients received abdominal injections in the AC group.
Patient Characteristics Based on Anticoagulation Status.
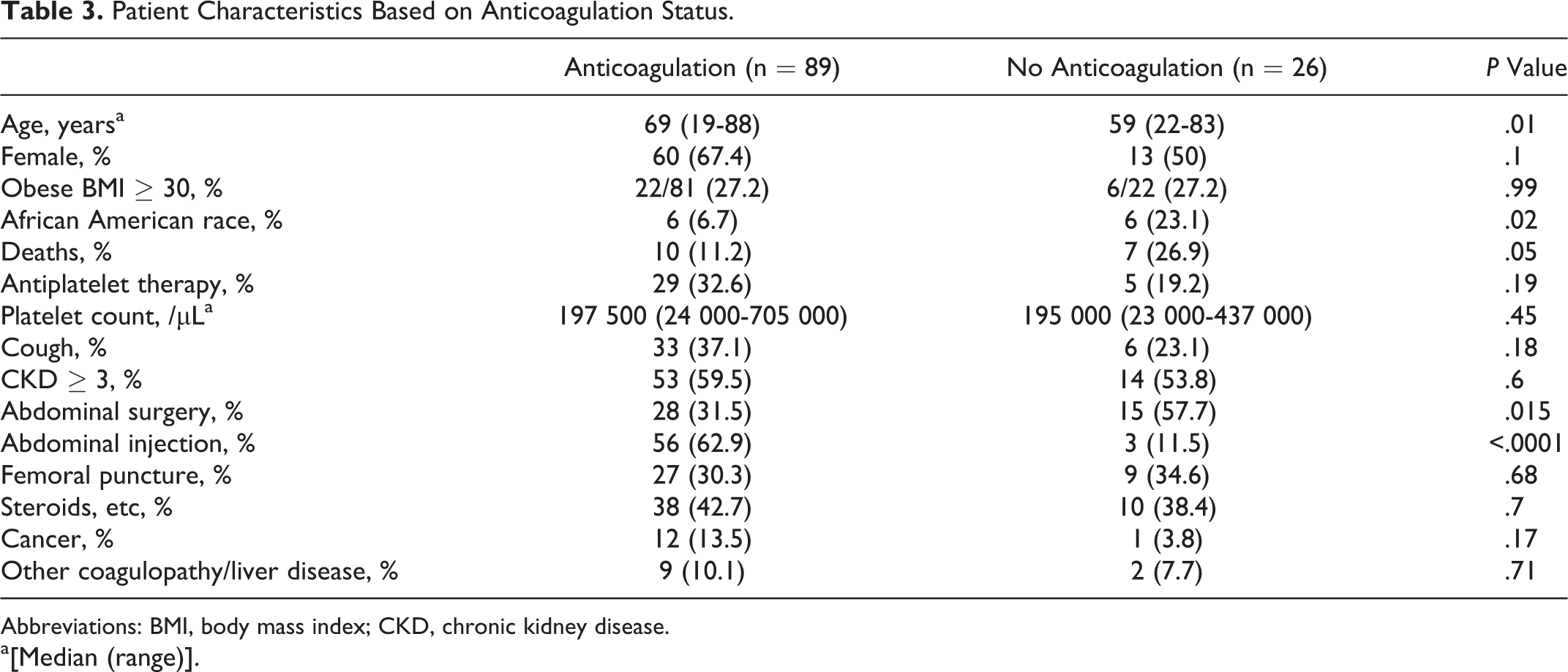
Abbreviations: BMI, body mass index; CKD, chronic kidney disease. a[Median (range)].
Rectus sheath hematoma treatment was observation in 44 (38.3%) patients, medical in 57 (49.6%) patients (pain management, transfusion and coagulopathy reversal, emergency team response, and ICU), and surgical in 14 (12.1%) patients.
There were 17 (14.8%) deaths; however, RSH was not a direct attributable cause in any patient. Patients who died were similar in demographic characteristics to those who were discharged from the hospital. Discharged patients had more frequently received chronic AC and AC therapy in the hospital. More patients with renal failure died in the hospital. In stepwise logistic regression, mortality was significantly associated with stage 3 or higher CKD (odds ratio [OR] 6.0, P = .03, 95% confidence interval [CI] 1.18-30.4) or required transfusion for RSH treatment (OR 9.3, P = .007, 95% CI 1.83-47.5); chronic AC was less frequently prescribed in patients who died (OR 0.2, P = .02, 95% CI 0.05-0.75).
In 43 (48%) patients, AC was reinitiated on average 7 days (range 0-24 days) after the event. None of these patients were readmitted with recurrence of RSH.
Discussion
Rectus sheath hematoma is often associated with damage of either the superior or inferior epigastric arteries or their branches that form numerous anastomoses between the rectus abdominis muscle and the posterior layer of the rectus sheath 10 or tear of a rectus abdominis muscle itself.
The presenting symptoms and signs in our study group were abdominal mass or pain in 54.7% and drop in hematocrit or hypotension in 31.3%. Other signs and symptoms reported are abdominal distension or ecchymosis, peritoneal irritation, nausea/vomiting, constipation, scrotal swelling, and tachycardia. 6,7 Frequently RSH is diagnosed as an incidental finding (13.9% of patients in our review) or on imaging studies. Diagnosis of RSH can be difficult because the presenting signs and symptoms mimic other more common etiologies like appendicitis, abdominal hernias and diverticular, biliary, or gynecological diseases. 1 –5
Abdominal ultrasonography, although cost effective, has a sensitivity of 70% for RSH and may not distinguish between RSH and intra-abdominal pathologies. 11 Computed tomography scan is the gold standard diagnostic modality with reported sensitivity of up to 100%. 11 Computed tomography scan findings have also been used to categorize RSH on basis of its severity and coupled with the proposed treatment. 12 Computed tomography scan diagnosis of RSH was the inclusion criteria for this review.
Risk factors for RSH are AC, subcutaneous abdominal injections, blunt trauma, muscular exertion (cough, exercise, etc), surgery, pregnancy, and medical comorbidities. 1,6,7 Rectus sheath hematomata occur mostly in the elderly patients. The median age in our study group was 65 years (range 19-88 years). The elderly patients are likely more vulnerable due to progressive weakening of abdominal wall with age and greater use of AC. Females (63.5%) were more prone to RSH in our study. The increased risk in females may be related to anatomic differences in rectus muscle between the 2 genders and weakening of the abdominal wall muscles due to excessive stretch during pregnancy. These findings are consistent with past studies reporting higher incidence of RSH in the female and elderly population. 6,13 None of our patients were pregnant at the time of diagnosis but pregnancy has been reported as a risk factor for RSH. 14
Anticoagulation therapy is the most common risk factor reported for RSH. 6,9,15 In our study, 89 (77%) patients were on some kind of AC therapy. International normalized ratio was supratherapeutic (>3) in only 5 patients. Subcutaneous UFH and LMWHs, in addition to its anticoagulant effect, increase the risk of RSH due to direct trauma to abdominal wall during administration. 8
Abdominal trauma is an important risk factor for RSH. Nonsurgical trauma especially due to coughing or strenuous exercise is attributed to RSH formation in multiple cases. 6,7,16,17 One-third of our patients had coughing episodes. Coughing paroxysm can cause severe contractions of the abdominal muscles which can injure the epigastric vessels or the rectus muscle itself. The injury is predominantly in the lower part of the rectus muscle as it lacks posterior support below the arcuate line. Fifty-one percent of our patients were given subcutaneous abdominal injections. In all, 37% underwent abdominal surgery or had catheter placement for peritoneal dialysis and femoral puncture was performed in 31.3% of the patients. All these procedures can either directly cause the RSH or weaken the abdominal wall leading to RSH from a minor trauma or coagulopathy.
Concomitant use of AC and APT in patients with atrial fibrillation increased the risk of major bleeding (adjusted hazard ratio [HR] = 1.53; 95% CI 1.20-1.96) and bleeding-related hospitalizations (HR = 1.52; 95% CI 1.17-1.97) were significantly higher in those on both oral AC and aspirin. 18 One-fourth of our patients received both AC and APT. The incidence of RSH may increase as use of AC and APT therapy increases in the elderly patients who concurrently have a compromised abdominal wall and declining renal function.
Our study showed a high number of patients with CKD who had RSH. Besides increased drug exposure due to decreased renal excretion, CKD also affects the metabolism of anticoagulants in the liver by downregulating hepatic cytochrome P450 which may lead to overanticoagulation. 19 Manufacturers (Lovenox) recommend dose adjustment in patients with creatinine clearance (CrCl) <30 mL/min due to decreased drug clearance. 20 Some authors recommend dose adjustment of LMWHs in patients with CrCl <40 mL/min due to RSH formation. 15 Other important contributing factor to bleeding risk in uremia is platelet dysfunction. Platelet aggregation is decreased in patients with uremia due to decreased thromboxane production, impaired platelet granule release, and an acquired dysfunction of the platelet membrane glycoprotein IIb/IIIa. 21 The use of APT in patients with CKD further increases the risk of bleeding episodes. The bleeding risk is high with any stage of CKD and increases with severity of CKD. 19,22,23
Chronic corticosteroids may increase the risk of bleeding through its interaction with warfarin on metabolic enzymes and coagulation factors. 24,25 Forty-two percent of our patients were on either steroids or immunosuppressant medications, and 2 of 5 patients with supratherapeutic INRs were on steroids.
The severity of the RSH dictates the required treatment. In clinically stable patients, conservative therapy with rest and analgesics may suffice. Large hematomas can cause hemodynamic instability and may require fluid resuscitation, blood transfusions, and AC reversal agents. It is advisable to discontinue any AC or APT until the active bleeding is controlled. Platelet transfusion should be considered in any bleeding patients taking antiplatelet agents after the antiplatelet agents have been withheld for the appropriate length of time.
Interventional measures may be required if conservative treatment fails or there is significant, active bleeding. Historically, surgical evacuation of the hematoma and ligation of bleeding vessels have been the mainstay treatment modality in such cases. Percutaneous embolization of bleeding vessels either by gel foam or by coil has been reported as an alternative treatment that reduces the recovery time of the patients. In this review, RSH treatment was observation in 44 (38%) patients, medical in 57 (49.57%) patients (pain management, transfusion and coagulopathy reversal, emergency team response, and ICU), and surgical in 14 (12.2%) patients.
Although RSH is rarely fatal, few cases of mortality have been reported. Rectus sheath hematoma was not a cause of death in any patient in our study; however, it may have contributed to the risk of death in patients with comorbidities such as CKD. Patients who died had less frequently received chronic or inpatient AC. Similar to previous studies, 6 no adverse events were reported after restarting AC in the above-mentioned patients. Physicians should decide whether the benefits of reinitiating AC outweigh the risk of bleeding on an individual basis.
Limitations
This study was exploratory based on retrospective review and has limitations of inadequate information in the charts. We excluded patients with incomplete documentation to minimize this limitation. There was no comparison to control matched patients without RSH which makes it difficult to study the association of risk factors. Study aim was to inform all possible risk factors for RSH. The study describes diagnoses, risk factors, management, and mortality in patients with RSH at our institute. The findings may not be generalizable.
Conclusion
Rectus sheath hematoma is an unusual complication of AC and/or APT and is related to the intensity and type of therapy. Other identified risk factors include old age, female gender, abdominal surgery/trauma/injections, cancer, and coagulopathies. Our study demonstrated the risk of developing RSH is related to age and the presence of renal impairment according to its severity. Immunosuppressant and steroids may increase the risk of bleeding particularly in patients with multiple risk factors. Patients with renal impairment may be at higher risk of developing RSH and worse outcomes, so prompt diagnoses and management are necessary for these patients. Reinitiation of AC therapy should be individualized, but from our experience, it seems safe to initiate therapy 7 days after bleeding remits.
Footnotes
Authors’ Note
The poster presentation of this study was presented at American Society of Nephrology (ASN) Kidney week, 2012, San Diego, California.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
