Abstract
In patients with liver failure complicated by acute kidney injury, renal replacement therapy (RRT) is often required to improve the internal environment. The use of anticoagulants for RRT in patients with liver failure remains controversial. We searched the PubMed, Embase, Cochrane Library, and Web of Science databases for studies. The methodological quality of the included studies was assessed using the Methodological Index for Nonrandomized Studies. A meta-analysis was performed using R software (version 3.5.1) and Review Manager (version 5.3.5). During RRT, 348 patients from 9 studies received regional citrate anticoagulation (RCA), and 127 patients from 5 studies received heparin anticoagulation (including heparin and LMWH). Among patients who received RCA, the incidence of citrate accumulation, metabolic acidosis, and metabolic alkalosis were 5.3% (95% confidence interval [CI]: 0%–25.3%), 26.4% (95% CI: 0–76.9), and 1.8% (95% CI: 0–6.8), respectively. The potassium, phosphorus, total bilirubin (TBIL), and creatinine levels were lower, whereas the serum pH, bicarbonate, base excess levels, and total calcium/ionized calcium ratio were higher after treatment than before treatment. Among patients who received heparin anticoagulation, the TBIL levels were lower, whereas the activated partial thromboplastin clotting time and D-dimer levels were higher after treatment than before treatment. The mortality rates in the RCA and heparin anticoagulation groups were 58.9% (95% CI: 39.2–77.3) and 47.4% (95% CI: 31.1–63.7), respectively. No statistical difference in mortality was observed between the 2 groups. For patients with liver failure, the administration of RCA or heparin for anticoagulation during RRT under strict monitoring may be safe and effective.
Introduction
Liver failure is a large number of hepatocyte necrosis caused by various reasons, resulting in serious dysfunction or decompensation of liver synthesis, detoxification, excretion, and biotransformation. In severe cases, liver failure may be combined with other organ dysfunction.1,2 Liver failure is associated with a high mortality rate. 3 When liver failure is aggravated or combined with acute kidney injury, medical therapy is poorly controlled, so renal replacement therapy (RRT) is often required to maintain the balance of the internal environment. 4 RRT is one of the most commonly used blood purification techniques in the rescue of critically ill patients, which mimics the principle of glomerulus work through convection and diffusion, so as to achieve the purpose of removing solutes. The water in plasma and solutes with small and medium molecular weight dissolved in it is removed by convection, and substances smaller than filter membrane holes are filtered out.5,6 At the same time, substances needed by the organism are imported into the body in the form of replacement fluid, partially replacing kidney function, creating conditions for organism recovery and prolonging survival time.
Keeping the extracorporeal circulation line of RRT without clotting is the premise to achieve the therapeutic goal, and the correct use of anticoagulants is particularly important.7,8 The balance between clotting and bleeding risk is a major concern with RRT. When a large number of hepatocytes are damaged, the procoagulant factors and anticoagulant factors produced by the liver are significantly reduced, which often leads to an increased risk of bleeding and a hypercoagulable state. 9 Due to this hypercoagulable state, the incidence of thrombosis is high even in the case of elevated international normalized ratio (INR) and thrombocytopenia. Thrombocytopenia is also common in liver failure and may be associated with an increased incidence of multiple organ failure. Because coagulopathy is the underlying pathophysiology of liver failure, anticoagulation problems are more prominent in RRT of patients with liver failure. 10
The most commonly used anticoagulation strategies in RRT include heparin, low molecular weight heparin (LMWH), and regional citrate anticoagulation (RCA). 11 Heparin and LMWH in vivo can accelerate the inactivation of coagulation factor Xa by binding with antithrombin III and prevent coagulation by inhibiting the adhesion and aggregation of platelets. 12 Heparin has wide application and rich experience in anticoagulation, and its existence period in vivo is short. There are corresponding antagonists and conventional detection experiments. 13 LMWH is more predictable than heparin's pharmacokinetics. It can be given by body weight and is more reliable without monitoring. However, both heparin and LMWH may induce adverse reactions such as bleeding and thrombocytopenia, thereby aggravating the risk of bleeding in patients with liver failure and coagulation dysfunction, 14 threatening the life safety of patients. Citrate exerts anticoagulation in the extracorporeal circulation pipeline, and the anticoagulant effect of citrate is related to its high affinity for calcium ions. 15 Calcium ions participate in the chelation of coagulation reactions, reducing the level of ionized calcium in the blood, and thereby acting as an anticoagulant. And this effect is reversible. In order to avoid negative calcium balance in patients and compensate for the calcium lost in the chelated form of calcium citrate, it is necessary to supplement the calcium ions by infusion at the end of the blood filtration circuit or directly through a separate pathway, 16 which can normalize the coagulation process. RCA is currently recognized as the preferred anticoagulant method in the RRT of critical patients with bleeding risk, 15 which can reduce the bleeding risk caused by anticoagulants such as heparin and LMWH, and obtain a long extracorporeal circuit life. 17 During the anticoagulant process of citrate in vitro, most citrate was filtered by a filter, while a small part of citrate entered the body and was degraded by liver metabolism. In severe liver failure, the metabolic function of the liver is significantly reduced. If the citrate entering the body exceeds the metabolic capacity of the liver, it can form citrate accumulation, which can cause metabolic disorders or affect liver repair. 18 Nevertheless, many studies have found that RCA does not deteriorate the liver function of patients with liver failure.19–22
The appropriate choice of anticoagulant for RRT in patients with liver failure remains controversial. Therefore, we conducted this systematic review and meta-analysis to compare the efficacy and safety of RCA and heparin anticoagulation in patients with liver failure.
Method
This systematic review and meta-analysis were performed and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement. 14 The study protocol has been registered on PROSPERO (the International Prospective Register of Systematic Reviews; registration no. CRD42022365884). Ethical approval was not required for this review of published studies.
Search Strategy
We comprehensively searched the PubMed, Embase, Cochrane Library, and Web of Science databases from inception to September 2022. The following Medical Subject Headings (MeSH) terms were used for the search: “liver failure,” “heparin,” “citrate,” and “anticoagulants”. A separate search was performed in Embase, Cochrane Library, and Web of Science using combinations of MeSH terms and PubMed entry terms. The literature search was independently performed by 2 reviewers, and any disagreements were resolved by a third reviewer. The author information section and reference list of the retrieved articles were searched for additional study information.
Study Selection
Studies that met the following criteria were included: (i) designed as a case-control study, cohort study, randomized controlled trial, or single-arm clinical trial; (ii) included adult patients (age > 18 years) with liver failure; and (iii) investigated patients with liver failure who underwent RRT with RCA or heparin (including heparin and LMWH) as the anticoagulation strategy.
Studies with the following characteristics were excluded: (i) duplicate publication; (ii) unavailable full text; (iii) no data on RCA or heparin use; (iv) sample size < 5; and (v) published as a review article, letter, case report, conference abstract, editorial, or comment.
Study Quality
Two independent reviewers assessed the methodological quality of the included studies using the Methodological Index for Nonrandomized Studies (MINORS). 23 MINORS contains 12 items, each of which can be scored from 0 to 2 (0, not reported; 1, incompletely reported; and 2, adequately reported). The specific MINORS items were as follows: (i) clearly stated study aim; (ii) inclusion of consecutive patients; (iii) prospective collection of data; (iv) endpoints appropriate to the study aim; (v) unbiased evaluation of endpoints; (vi) follow-up period appropriate to the major endpoint; (vii) rate of follow-up loss not exceeding 5%; and (viii) prospective calculation of the sample size. The remaining 4 items were designed for studies with a control group: (i) a control group receiving the gold standard intervention; (ii) contemporary groups; (iii) baseline equivalence of groups; and (iv) statistical analyses appropriate for the study design. In this meta-analysis, noncomparative studies that scored <8, 9–12, and 13 were considered to be of low, medium, and high quality, respectively. Comparative studies that scored <12, 13–18, and >19 were considered to be of low, medium, and high quality, respectively.
Data Collection
Before study screening and data collection, all criteria and required data were defined. Two reviewers independently collected the data, and a third reviewer resolved any disagreement. The article type, title, and abstract were evaluated first for study screening. Studies that were identified as eligible in the first screening round were subjected to a full-text review for final inclusion. The following data were extracted from the articles: the country where the study was performed, ethnicity of the studied patients, study type, inclusion and exclusion criteria, information relevant to the study's quality, baseline data of patients, data relevant to liver failure, and information relevant to RCA or heparin use.
Statistical Analysis
All data analyses were performed using R software (version 3.5.1; The R Foundation for Statistical Computing, Vienna, Austria) and Review Manager (version 5.3.5; The Cochrane Collaboration 2021, The Nordic Cochrane Centre, Copenhagen, Denmark). For categorical variables, incidence rates were calculated by dividing the total number of patients by the number of observed events. Continuous variables reported as median (interquartile range) were converted to mean ± standard deviation. Fixed-effect and random-effect models were used for the meta-analysis to pool the effect sizes and 95% confidence intervals (95% CIs). Heterogeneity across the included studies was assessed using the I2 statistic, as follows: I2 < 25%, low heterogeneity; 25%< I2 < 50%, medium heterogeneity; and I2 > 50%, significant heterogeneity. A random-effects model was applied if I2 > 50%; otherwise, a fixed-effects model was used. A sensitivity analysis was conducted to assess the effect of individual studies on the pooled estimates. Funnel plots analysis and Egger's and Begg's tests were performed to assess publication bias. Descriptive statistics were used to analyze the characteristics of study participants. P < .05 was considered to indicate statistical significance.
Results
Study Selection
Figure 1 shows the study inclusion flowchart. A total of 621 relevant articles were found: 118 from PubMed, 262 from Embase, 52 from the Cochrane Library, and 189 from Web of Science. The articles were screened according to the inclusion and exclusion criteria. Of the identified studies, 59 and 362 were excluded because of duplication and irrelevant article types, respectively. Additionally, 76 studies with small sample sizes, 68 studies with no related data, and 42 on intervention failure were excluded. Finally, 14 studies involving 475 patients were included in this systematic review.

The study inclusion flowchart.
Description of the Included Studies and Patients
Table 1 shows the characteristics of the 14 included studies. During RRT, 348 patients from 9 studies received RCA,24–31 and 127 patients from 5 studies received heparin anticoagulation.32–35 The studies were published between 2006 and 2022. All included studies had an observational in design, with 10 prospective and 4 retrospective studies. All studies were single-center investigations.
Characteristics of Included Studies.

Abbreviations: LF, liver failure; ALF, acute liver failure; NR, not reported; RCA, regional citrate anticoagulation; Caion, ionized calcium; LMWH, low molecular weight heparin; HE, hepatic encephalopathy; ACLF, acute-on-chronic liver failure; AKI, acute kidney injury; PTA, prothrombin activity.
Table 2 shows the baseline characteristics of the 475 patients enrolled in the included studies. The Model for End-Stage Liver Disease (MELD), Acute Physiology and Chronic Health Evaluation (APACHE-II), and Child-Pugh score was the primary tool used for liver failure severity assessment in the reviewed studies, whereas the Sequential Organ Failure Assessment (SOFA) was used to assess the severity of critically ill patients.
Baseline Data of Enrolled Patients.

Abbreviations: RCA, regional citrate anticoagulation; NR, not reported; MELD, model for end-stage liver disease; APACHE, acute physiology and chronic health evaluation; SOFA, sequential organ failure assessment.
Quality Evaluation
Table 3 shows the results of the MINORS quality assessment. Five studies were judged to have a low risk of bias according to different grading standards, and the total scores indicated that the studies were of high quality. The remaining 9 studies were determined to be of medium quality.
Risk of Bias in Each Study Assessed Using the MINORS Tool.

Note: Item 1, the aim of the study; Item 2, the inclusion of consecutive patients; Item 3, prospective collection of data; Item 4, endpoint appropriate to the study aim; Item 5, unbiased evaluation of endpoints; Item 6, follow-up period appropriate to the major endpoint; Item 7, loss to follow-up not exceeding 5%; Item 8, prospective calculation of the sample size; Item 9, a control group having the gold standard intervention; Item 10, contemporary groups; Item 11, baseline equivalence of groups; Item 12, statistical analyses adapted to the study design.
Study Results
Safety
The safety of RCA and heparin anticoagulation was evaluated according to changes in the levels of liver, kidney, and blood coagulation function indicators; the incidence of citrate accumulation; and changes in acid–base and electrolyte balance during RRT.
Liver Function
Liver function was assessed according to the levels of total bilirubin (TBIL), alanine aminotransferase (ALT), and aspartate aminotransferase (AST). Five studies reported changes in TBIL levels. We adopted a fixed-effects model based on the P and I2 values (P < .01, I2 = 0). Figure 2A shows that the pooled results indicating that the TBIL levels decrease after both RCA (mean = 1.73, 95% CI: 0.07–3.38, P < .05, I2 = 0) and heparin group (mean = 5.90, 95% CI: 3.06–8.75, P < .01, I2 = 0). A statistical difference in the changes in TBIL levels was observed between the RCA and heparin groups (χ2 = 6.18, df = 1, P = 0.01), and no significant heterogeneity existed among the studies (χ2 = 6.38, df = 4, P = .17, I2 = 37%). Additionally, there 3 studies reported changes in ALT and AST levels after RCA. Table 4 and Figure 8 in the Appendix show that no significant changes were observed in the levels of ALT (mean = −240.24, 95% CI: −673.33 to 192.84, I2 = 77%) and AST (mean = −530.95, 95% CI: −1537.59 to 475.68, I2 = 81%).
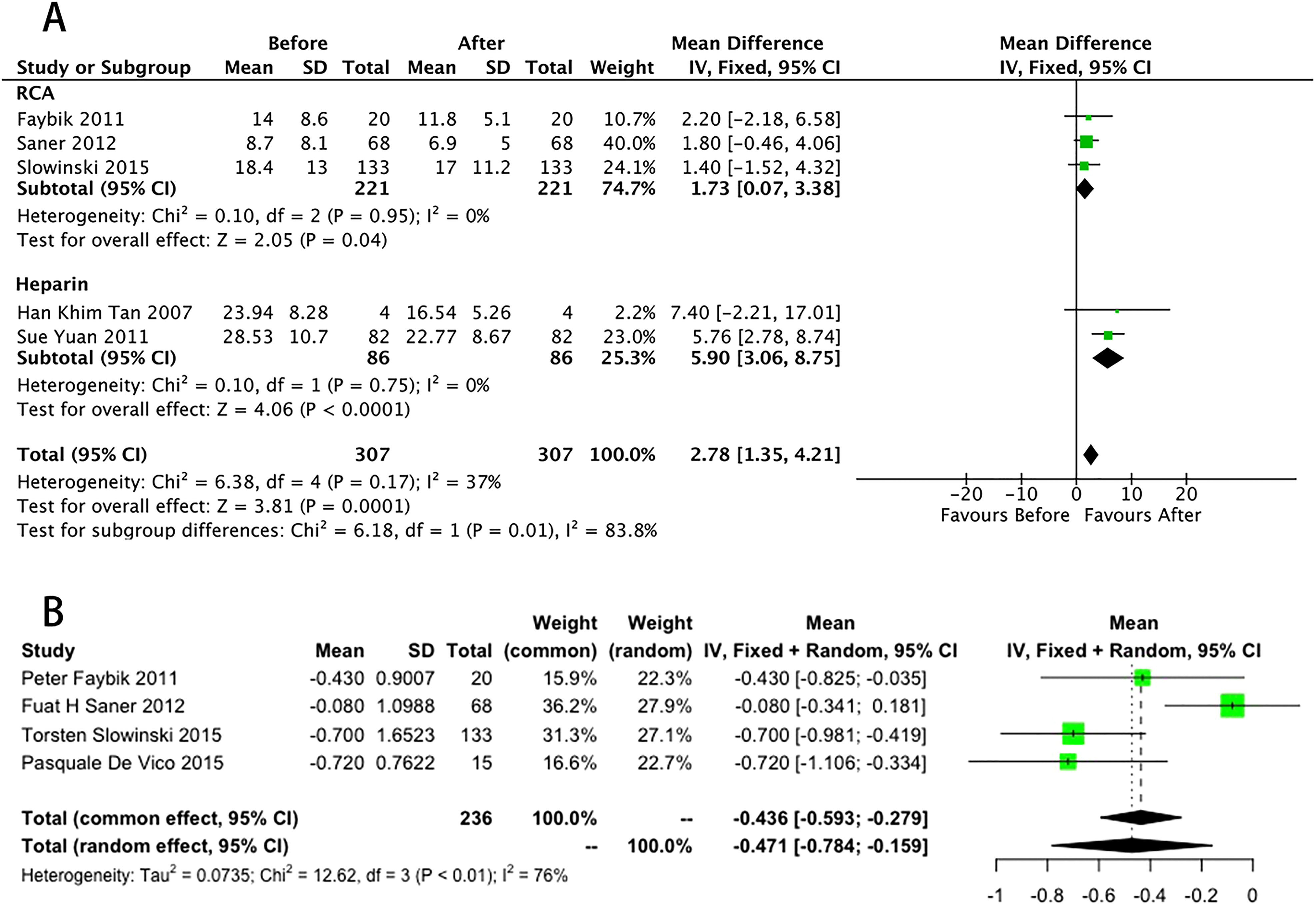
The pooled rates (95% CI) of a subgroup of TBIL (A), and the pooled results of Cr (B) between the start of using RCA and the end of observation.
Data Integration Results of Different Characteristics.

Abbreviations: RCA, regional citrate anticoagulation; Caion, ionized calcium; Caton, total calcium; ALT, alanine transaminase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; APTT, activated partial thromboplastin time; INR, international normalized ratio; BE, base excess; ICU, intensive care unit; los_ICU length of ICU stay; NR, not reported; PTA, prothrombin activity; WBC, white blood cell; PLT, platelet; TB, total bilirubin; FIB, fibrinogen; HGB, haemoglobin.
Renal Function
In our systematic review, we used a random-effects model because the complication rates were heterogeneous (P < .01, I2 = 76%). Figure 2B shows the pooled results of 4 studies, which demonstrated a decrease in creatinine (Cr) levels (mean = −0.47, 95% CI: −0.78 to 0.16) after RCA. Additionally, Table 4 and Figure 8 in the Appendix show no significant changes in blood urea nitrogen levels (mean = −35.53, 95% CI: −71.16 to 0.09, I2 = 98%) after using RCA.
Blood Coagulation Function
Changes in the levels of related blood coagulation function indicators were observed. Subgroup analysis showed a statistically significant decrease in platelet counts in the heparin group (mean = 21.22, 95% CI: 12.53–29.90), but not in the RCA group (Figure 3A). Meanwhile, subgroup analysis showed a statistically significant increase in activated partial thromboplastin time (APTT) in the heparin group (mean = −16.29, 95% CI: −21.90 to −10.69), but not in the RCA group (Figure 3B). Figure 3C shows that 1 study reported an increase in D-dimer levels (mean = 2.60, 95% CI: 1.59–3.60, P < .01) after heparin anticoagulation.

The pooled results of a subgroup of PLT (A) and APTT (B). The pooled results of D-Dimer (C) between the start of using heparin and the end of observation.
No significant changes were observed in the INR (mean = −0.24, 95% CI: −0.53 to 0.05, I2 = 89%), and prothrombin activity (PTA) (mean = 0, 95% CI: −11.18 to 11.18) after RCA. Additionally, no significant changes were observed in INR (mean = −0.75, 95% CI: −2.02 to 0.52, I2 = 97%), PTA (mean = 2.48, 95% CI: −0.16 to 5.13, I2 = 0), and prothrombin levels (mean = 1.36, 95% CI: −1.70 to 4.42, I2 = 80%) after heparin anticoagulation. The results are shown in Table 4 and Figure 9 in the Appendix.
Citrate Accumulation
Three studies reported the number of patients with citrate accumulation. The incidence of citrate accumulation was 5.3% (95% CI: 0–25.3). Figure 4A shows a statistically significant difference in the incidence of citrate accumulation and the presence of heterogeneity among the studies (χ2 = 10.90, df = 2, P < .01, I2 = 82%).

The pooled result of citrate accumulation (A), totCa/ionCa ratio (B), potassium (K+) (C), and phosphorus (P+) (D) between the start of RCA use and the end of observation.
Five studies evaluated citrate accumulation during RCA according to changes in the total calcium/ionized calcium (totCa/ionCa) ratio. Figure 4B shows the results of the pooled analysis of the involved studies. An increase in the totCa/ionCa ratio was observed after RCA (mean = 0.17, 95% CI: 0.06–0.29, P < .01, I2 = 89%).
Electrolyte status
The electrolyte equilibrium fluctuated during RRT. Five studies reported a decrease in potassium (K+) levels (mean = −0.18, 95% CI: −0.36 to −0.01, P < .01, I2 = 74%; Figure 4C), and 2 studies reported a decrease in phosphorus (P+) levels (mean = −0.35, 95% CI: −0.66 to −0.04, P < .01, I2 = 85%; Figure 4D). Table 4 and Figure 10 in the Appendix show that the pooled results demonstrate no significant differences in the levels of serum sodium (Na+) (mean = 0.07, 95% CI: −1.75 to 1.90), serum magnesium (Mg2+) (mean = −0.09, 95% CI: −0.29 to 0.10), and blood chloride (Cl−) (mean = 0.00, 95% CI: −1.37 to 1.37).
Acid–Base Status
The acid–base equilibrium fluctuated during RRT. Figure 5A shows that 5 studies reported an increase in (pH) (mean = 0.05, 95% CI: 0.03–0.08, P < .01, I2 = 82%). Figure 5B shows that 4 studies reported an increase in bicarbonate (HCO3−) levels (mean = 3.49, 95% CI: 1.59–5.38, P < .01, I2 = 85%), and Figure 5C shows that 5 studies reported an increase in base excess (BE) levels (mean = 2.83, 95% CI: 1.07–-4.59, P < .01, I2 = 91%). Figure 6A shows that 3 studies reported a decrease in the anion gap (AG) levels (mean = −1.21, 95% CI: −2.20 to −0.22, P < .01, I2 = 0). Figure 6B shows that 3 studies reported the incidence of metabolic acidosis rate of 26.4% (95% CI: 0–76.9), P < .01, I2 = 97%, and Figure 6C shows that 2 studies reported that the incidence of metabolic alkalosis rate was 1.8% (95% CI: 0–6.8), P < .01, I2 = 51%. All results are shown in Table 4.
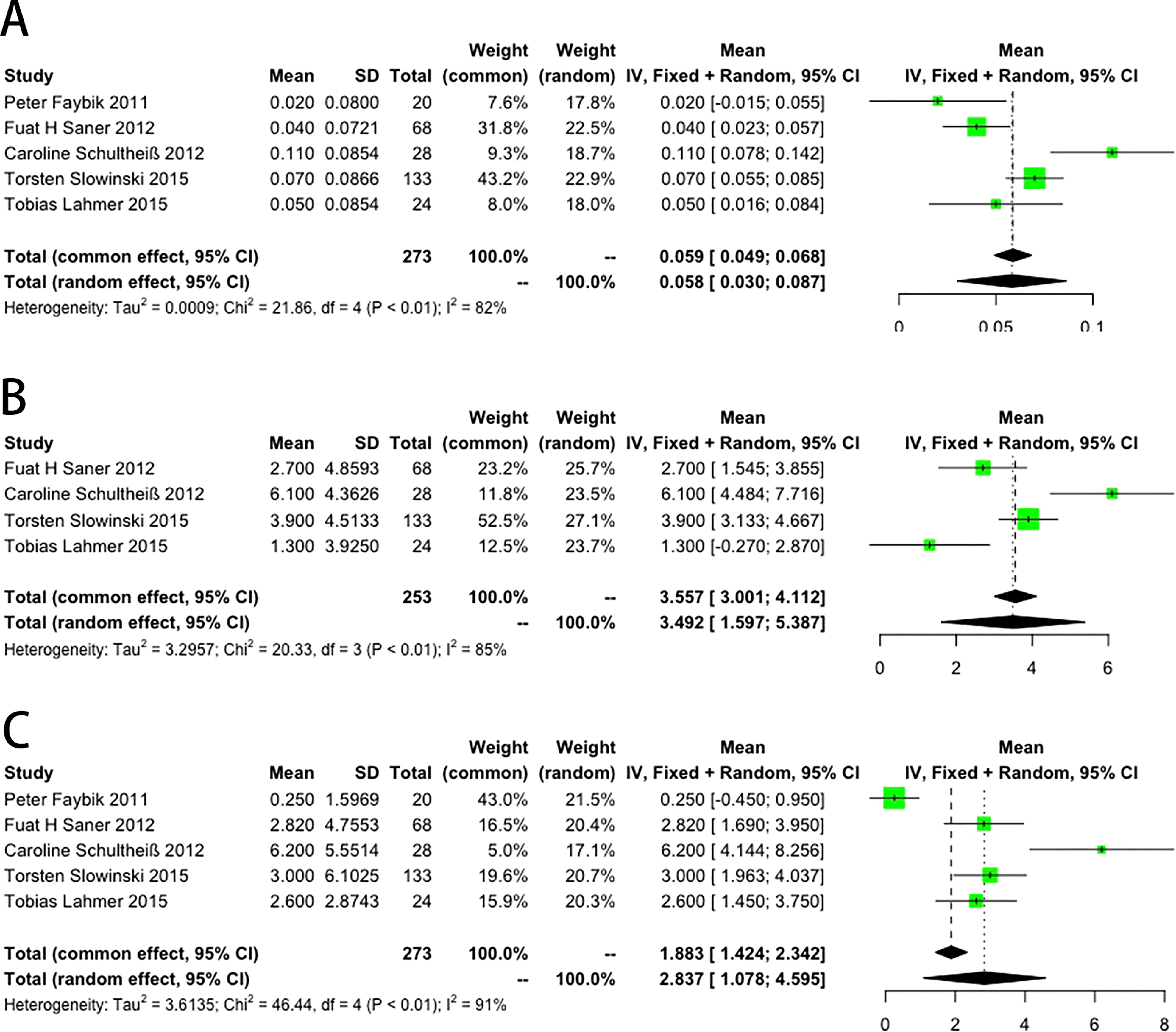
The pooled results of the potential of hydrogen (pH) (A), carbonic acid hydrogen radical (HCO3−) (B), and BE (C) between the start of using RCA and the end of observation. Abbreviations: CI, confidence interval; SD, standard deviation; BE, base excess; RCA, regional citrate anticoagulation.

The pooled results of AG (A), metabolic acidosis rate (B), and metabolic alkalosis rate (C) between the start of using RCA, and at the end of the observation.
Efficacy
The efficacy of RCA was assessed according to mortality rates. Nine studies reported mortality rates and the overall mortality rate was 55.4% (95% CI: 40.9–69.9) (Figure 7). The mortality rates in the RCA and heparin groups were 58.9% (95% CI: 39.2–77.3) and 47.4% (95% CI: 31.1–63.7), respectively. No statistical difference in the mortality rates was observed between the 2 groups (χ2 = 0.25,df = 1,P = .62), although significant heterogeneity was observed among the studies (χ2 = 33.08,df = 8, P < .01,I2 = 76%).

The pooled results of a subgroup of mortality.
Sensitivity Analysis and Publication Bias
Figure 11 in the Appendix shows the results of the sensitivity analysis of the 14 included studies for the pooled effect sizes. The pooled effect sizes did not show significant changes, indicating robust results. Funnel plot analysis and Egger's test were performed to assess publication bias. The sensitivity and publication bias of the mortality results were evaluated (Figure 12A in the Appendix). The asymmetric funnel plot indicated no significant publication bias, which was confirmed by the results of Egger's test (P = .32) (Figure 12B in the Appendix). The validity of Egger's test may be limited if <10 studies were included, making it difficult to distinguish between contingency and asymmetry. No evidence of potential publication bias was detected in any of the included studies.
Discussion
We analyzed the results of 14 studies from 4 databases to assess the efficacy and safety of citrate and heparin as anticoagulants during RRT in patients with liver failure. Among patients who received RCA, the incidence of citrate accumulation, metabolic acidosis, and metabolic alkalosis was 5.3%, 26.4%, and 1.8%, respectively. The K+, P+, TBIL, and Cr levels were lower, whereas the pH, HCO3−, and BE levels and the totCa/ionCa ratio were higher after treatment than before treatment. Among patients who received heparin anticoagulation, the TBIL was lower, whereas the APTT and D-dimer levels were higher after treatment than before treatment. The mortality rates in the RCA and heparin groups were 58.9% and 47.4%, respectively. No statistical difference in mortality was found between the 2 groups.
Liver failure is a group of clinical symptoms caused by several factors, including severe coagulation dysfunction, significant jaundice, hepatic encephalopathy, and ascites. 2 The main clinical symptoms include extreme fatigue, loss of appetite, abdominal distension, nausea, vomiting, and altered consciousness. The TBIL levels and INR are often evaluated to determine the severity of liver failure. The overall prognosis of patients with liver failure is poor owing to rapid disease progression, treatment difficulties, and high treatment costs. 3 When the disease is complicated by acute kidney injury, the management involves numerous drug therapies. This leads to the requirement for RRT, which can maintain the stability of the internal environment, remove harmful substances from the body, and partially replace damaged kidney function. 7
The goal of anticoagulation in RRT is to maintain the patency of the extracorporeal circuit. Therefore, anticoagulants must be used correctly. Blood coagulation disorder is a basic pathophysiological change in liver failure. Maintaining the balance between blood coagulation and bleeding is important for patients with liver failure who require anticoagulation. 8 Furthermore, the choice of anticoagulant in patients with liver failure may not be based on the presence of liver failure itself but on the severity of the disease. Currently, heparin and RCA are among the most commonly used anticoagulation strategies.
Heparin changes its configuration mainly by combining with antithrombin III. 37 Inhibition of various coagulation factors results in anticoagulant effects. Heparin is an inexpensive, easy-to-administer, and potent anticoagulant. However, heparin anticoagulation may lead to heparin-induced thrombocytopenia.15,17 When liver failure occurs, many hepatocytes undergo necrosis, and the production of related coagulation factors and antithrombin becomes impaired, which may increase the risk of bleeding. According to the results of our analysis, the TBIL levels were lower after heparin anticoagulation than before. The platelet counts significantly decreased and the APTT significantly increased in the heparin group. No significant changes were observed in other indicators reflecting blood coagulation function, and heparin had no significant impact on liver function. More attention should be paid to indicators of blood coagulation function and patient monitoring should be enhanced when using heparin for anticoagulation.
Sodium citrate acts as an anticoagulant by chelating calcium ions in the extracorporeal circuit.21,38 Because citric acid is a small molecule, most calcium–citrate complexes are free to leach into the waste solution, making a systemic infusion of calcium necessary to replace the calcium lost with citrate. RCA can prolong filter life and reduce bleeding and heparin-induced thrombocytopenia.39–42 Approximately 70% of sodium citrate molecules can be filtered during RCA, whereas the liver metabolizes most of the sodium citrate that enters the body. However, in the event of liver failure, a large number of hepatocytes undergo necrosis and sodium citrate metabolism disorder may lead to citrate accumulation, 16 which may aggravate the liver damage and cause an internal environment disorder. Frequent monitoring of electrolyte levels and acid–base balance is necessary because of the possibility of hypernatremia, metabolic alkalosis, and ionic hypocalcemia. Patients with severe liver failure and lactic acidosis may have difficulty metabolizing citrate and develop citrate accumulation, characterized by decreased intracellular calcium levels, increased total serum calcium levels, metabolic acidosis, and increased AG levels. In this review article, 9 studies reported that the incidence of citrate accumulation, metabolic acidosis, and metabolic alkalosis in patients with liver failure who received RCA was 5.3%, 26.4%, and 1.8%, respectively. RCA did not result in obvious electrolyte imbalances and had no significant effects on liver, kidney, or blood coagulation function. A serum totCa/ionCa ratio of ≥2.5 may be the critical threshold for potential citrate accumulation. Additionally, the presence of acidosis or elevated lactate levels needs to be part of the definition of citrate accumulation. Furthermore, citrate dose is one of the determinants of citrate toxicity. Therefore, various factors need to be considered when evaluating the presence of citrate accumulation. Close monitoring during RRT when RCA is used as the anticoagulation strategy may help avoid citrate accumulation. The TBIL levels were lower than before the RCA. Moreover, the ALT levels, AST levels, and INR showed no significant differences between before and after RCA. Taking our results together, we considered that RCA has no significant effect on liver function.
Citrate is a weak acid that acidifies the RRT solution. RCA has an acid–base effect that is not limited to citrate. Many citrate solutions have a high sodium content (1 citrate molecule requires 3 Na+). This net sodium administration tends to increase the plasma strong-ion difference, leading to plasma alkalization. 43 When citrate catabolism is normal, RCA causes plasma alkalization. This alkalization effect is maximally achieved with sodium citrate. At the same time, our systematic review demonstrated that heparin anticoagulation has no significant impact on patients with liver failure. As patients with liver failure have severe coagulation dysfunction, we considered that the increase in the APTT and D-dimer levels was related to the necrosis of many hepatocytes. 44 With respect to mortality, no statistical difference was observed between the RCA and heparin groups. Therefore, the severity of the patient's disease should be considered.
Our systematic review had some limitations. First, the patients enrolled in the included studies had different fundamental characteristics, which may have led to significant clinical heterogeneity. Second, the anticoagulation regimens (RCA or heparin) and the causes and degree of liver failure differed across the included studies. Our results might have been influenced by the inherent limitations of the nonrandomized studies included in the meta-analysis. Third, the timing of the evaluation of adverse events after RCA or heparin anticoagulation is important. Early testing may result in a higher incidence of adverse events because of the patients’ instability. Therefore, more prospective randomized controlled trials are required to validate our findings.
Conclusion
For patients with liver failure, RCA or heparin anticoagulation during RRT may be effective and safe under close supervision. More prospective studies with unified standards and randomized controls are needed to provide theoretical support for the use of anticoagulation therapy in patients with liver failure.
Footnotes
Acknowledgments
We thank the medical and nursing staff of the Department of Critical Care Medicine, Beijing Ditan Hospital, Beijing, China, for their valuable comments during this study.
Availability of Data and Materials
Not applicable.
Author Contributions
W.Q. designed and drafted the manuscript. J.L. participated in the literature search and summarized the relevant literature. A.L. provided the theme and design and edited the manuscript. All the authors have read and approved the final manuscript.
Ethical Approval
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
