Abstract
Immune thrombocytopenia (ITP) is a rare autoimmune disorder with an incidence of 3 to 5 per 100 000 individuals. In children, the disease is self-limited and is most commonly virus related (acute ITP) whereas in adults, the disease is typically chronic. The age distribution of adult ITP displays 2 peaks; the first in younger adults aged 18 to 40 with a female predominance and the second in people aged older than 60 with men and women affected equally. Our approach to ITP has evolved over the past several years: there has been a change in nomenclature and ITP now denotes “immune thrombocytopenia” (the “I” no longer denoting “idiopathic”) and “purpura” no longer features in the name of the disease; new insights into the pathogenesis of ITP have revealed the importance of impaired megakaryocytopoiesis in the condition; underlying mechanisms of secondary ITP have been elucidated and finally novel thrombopoietic agents have been shown to be effective in the treatment of ITP in randomized clinical trials. In this article, we review important recent advances in the pathogenesis and treatment of ITP.
Pathogenesis
Platelet Kinetics and Mechanisms of Thrombocytopenia
Although megakaryocyte number in the bone marrow of patients with immune thrombocytopenia (ITP) is often increased, it has been observed that in fact megakarocytes may be present in normal or even in reduced numbers in ITP. This variability implies that at least in some patients, decreased platelet production may be a key element in the pathogenesis of ITP. In order to better understand the predominant mechanism of thrombocytopenia in ITP, platelet kinetic studies using indium-111-labeled autologous platelets have been used. These studies provide an estimate of platelet life span, as well as of platelet turnover, which is a measure of platelet production. Interestingly, in addition to decreased platelet life span in most patients with ITP, these studies demonstrate inappropriately normal or even low platelet turnover, indicating that platelet production is actually decreased in ITP. 1 –3 Thus, it has been shown that the pathogenesis of ITP involves both increased peripheral platelet destruction and decreased bone marrow platelet production.
Humoral Response and Platelet Antibodies
The first evidence for platelet antibodies in ITP came from Harrington seminal experiments in which he infused 500 mL of whole blood from a patient with ITP into a healthy participant and after 2 hours demonstrated a sharp decrease in platelet count from 800 × 109/L to 25 × 109/L. After 24 hours, the platelet count was 0 with recovery occurring after a number of days. 4 Later, it was shown by Shulman et al that the “plasma factor” causing thrombocytopenia could be adsorbed by platelets and that this factor was present in the immunoglobulin G (IgG) fraction of plasma. 5 This led to the hypothesis that the antiplatelet factor was an antibody. It should be noted, however, that in Harrington experiments, not all patients had a dramatic decrease in platelet count. Some developed only mild thrombocytopenia, again raising the possibility of additional mechanisms for thrombocytopenia in ITP.
Several studies performed in 1970s demonstrated the presence of IgG on platelets in patients with ITP; however, these assays had low specificity and could not discriminate between patients with ITP and those with other causes of thrombocytopenia. With improvements in serologic techniques, these antibodies were later shown to be predominantly directed against glycoprotein (GP) IIb/IIIa and less frequently against other platelet GPs such as GP Ib/IX. 6,7
The spleen is the main site of antibody production 8,9 and also the main site responsible for the elimination of antibody-coated platelets. 1,10 Thus, the spleen was recognized as an important element in the pathophysiology of ITP.
Impaired megakaryocyte production and survival have been recently highlighted as being an important component of ITP pathogenesis. Morphologic changes in megakaryocytes were noted using light microscopy as early as in the 1940s. Abnormal megakaryocytopoiesis with increased numbers of young megakaryocytes lacking cytoplasmic granularity and evidence of platelet formation and degenerative changes in the nucleus and cytoplasm were observed in the bone marrow of patients with ITP. 11,12 Notably, the same changes were reproducible in healthy patients infused with serum of patients with ITP. 13 Studies using electron microscopy show additional megakaryocyte abnormalities that are indicative of apoptosis including distended demarcation membranes, vacuolized cytoplasm, swollen mitochondria, a disrupted peripheral zone, and positive immunohistochemical staining for caspase 3. 14 Phagocytosis of megakaryocytes was also seen in some cases. 15 Megakaryocyte survival in ITP has also been shown to be reduced with antibodies directed against megakaryocytes resulting in suppression of megakaryocyte maturation and reduced platelet production. 16 –18 Studies using rats showed impairment of megakaryocyte maturation when infused with antiplatelet serum and antibodies directed against GP IIb/IIIa and GP Ib/IX bind to megakaryocytes in addition to platelets as these molecules are expressed on developing megakaryocytes. 19 Experiments using thrombopoietin-induced CD34+ stem cells showed decreased production of megakaryocytes when antibodies directed against GP IIb/IIIa and GP Ib/IX were present while antibody adsorption restored megakaryocyte production, demonstrating the specificity of the antibody effect. 20 Antibodies directed against the thrombopoietin receptor, c-Mpl, have been detected in patients with ITP, and in these cases, the antibodies suppressed megakaryopoiesis. 21
T Cell-Mediated Immune Response
The absence of demonstrable autoantibodies in about 40% of patients with ITP suggests the existence of an alternative pathway for platelet destruction. The cytokine profile of patients with ITP is typically type 1 helper, consistent with a proinflammatory response. 22 The CD4+ T cells with reactivity against platelets have been found in the peripheral blood of patients with ITP, with GP IIb/IIIa being the major target of the T cell receptor. 23 In addition, the presence of CD8+ cytotoxic T cells with platelet autoreactivity has been shown in patients with ITP. 24 Since T cells directed against GP IIb/IIIa have also been found in the blood of healthy individuals, it is presumed that impaired peripheral tolerance may underlie the immune dysregulation in ITP. 25
Regulatory T cells (Tregs) are an increasingly appreciated subset of T lymphocytes, which function as suppressors of immune response and help maintain peripheral immune tolerance. Defects in Tregs are thought to play a central role in the development of autoimmune diseases. 26 In patients with ITP, Tregs have been found to be both functionally impaired and reduced in number. 27 In an animal model, 36% of Treg-deficient mice developed thrombocytopenia that lasted 5 weeks. The thrombocytopenic mice also developed antiplatelet antibodies, and transfer of Tregs to the deficient mice prevented thrombocytopenia. 28
Impaired apoptosis of T cells is also described in ITP. The CD3+ lymphocytes in patients with ITP display aberrant apoptosis-associated gene expression and are less susceptible to dexamethasone-induced apoptosis, permitting the survival of aberrant autoreactive T cell clones in the circulation. 29
The existence of both humoral and cellular immune response abnormalities in ITP was supported by a murine model of ITP. Splenocytes from CD61 knockout mice immunized against CD61+ platelets were transferred into severely immunocompromised SCID mice with resultant development of severe thrombocytopenia. Depletion of donor mice lymphocytes subsets before transfer showed that CD4+ T cells were crucial for the development of thrombocytopenia and that both antibody-producing B cells and cytotoxic T cells were involved in the pathogenesis of thrombocytopenia. 30
Other Cellular Mechanisms
Regulatory B cells (Bregs), like Tregs, suppress autoimmune responses and maintain peripheral immune tolerance, and Bregs have been shown to have impaired function in autoimmune disease. 31 In a recent study examining the role of Bregs in patients with chronic ITP, it was shown that individuals with lower platelet counts had lower levels of Bregs in their peripheral blood. 32 In the same study, there was also evidence for impaired function of Bregs in patients with ITP.
Antigen-presenting cells have a role in the pathogenesis of ITP. Antigen-presenting cells function by presenting antigens to autoreactive T cells. Myeloid dendritic cells overexpress costimulatory molecules in patients with ITP, enhancing the immune response of autoreactive B and T cells against platelets. 33
Finally, splenic macrophages may also have a role in the pathogenesis of ITP. These cells obtained from patients with ITP with active disease have shown to induce proliferation of GP IIb/IIIa—reactive T cell lines without the presence of an exogenous antigen. 34 This may result in increased clearance of antibody-coated platelets in these patients. 35
Secondary ITP
Secondary ITP is a heterogeneous group of disorders, where the specific factor causing ITP is known. Unraveling the mechanism of immune dysregulation in these conditions may further the understanding of ITP pathogenesis in general.
Childhood ITP is typically preceded by a febrile illness and is presumed to be triggered in many cases by an acute viral infection. Possible mechanisms for thrombocytopenia in these cases include the aberrant expression of viral antigens by platelets, binding of immune complexes to the platelet surface, and the formation of antibodies with cross-reactivity between viral and platelet antigens. 36
Immune thrombocytopenia is associated with several chronic infections including Helicobacter pylori (HP), human immunodeficiency virus (HIV), and hepatitis C virus (HCV). The association of ITP with HP has been shown in several studies, and HP eradication may be associated with ITP remission although clinical trials of HP treatment in ITP have yielded contradictory results. 37 The supposed mechanism is molecular mimicry, resulting in cross-reactive antibodies between bacterial and platelet antigens. 38 Genetically determined host factors such as expression of the Lewis blood group antigen and human leukocyte antigen type may predispose to HP-associated ITP. 39
The pathogenesis of thrombocytopenia in both HCV and HIV infection is probably multifactorial. As in the case of HP, both HCV and HIV can provoke the formation of antiviral antibodies that cross-react with platelet antigens. 40 In addition, HCV binds to platelets causing the formation of an antibody–antigen–platelet complex leading to platelet destruction. 41 Finally, both HCV and HIV may directly impair megakaryocyte maturation and proliferation. 42
Treatment
Treatment of patients with ITP is determined by 2 variables: bleeding and platelet count. Severe cutaneous bleeding, prolonged epistaxis, gingival bleeding, overt hematuria or menorrhagia, and intracranial hemorrhage or other internal bleeding may develop at platelet counts less than 10 × 109/L. Patients typically present with petechiae or purpura when the platelet count is less than 20 × 109/L. At platelet counts between 30 and 50 × 109/L, patients may note easy bruising, and at platelet counts above 50 × 109/L, patients are usually asymptomatic. 43 Therapy is indicated in all patients who present with significant bleeding and is often given in patients with a platelet count less than 30 × 109/L although there is no data to indicate that treatment in the latter circumstance alters the course of ITP. 44 Those with a platelet count of more than 50 × 109/L can almost always be observed. In general, immediate therapy is not required for patients with platelet count between 20 and 50 × 109/L in the absence of bleeding or predisposing comorbid conditions such as uncontrolled hypertension, active peptic ulcer disease, anticoagulation, recent surgery, or head trauma that may predispose to bleeding. 45 Relevant factors that contribute to management decisions include the extent of bleeding, comorbidities predisposing to bleeding, complications of specific therapies, degree of physical activity and lifestyle, tolerance of side effects, potential interventions that may cause bleeding, accessibility of care, patient expectations, patient worry or anxiety about disease burden, and patient need for non-ITP medications that may create a bleeding risk. 46 Regarding the latter, Cines et al recommend maintaining platelet counts above 40 × 109 to 50 × 109/L for patients requiring aspirin, nonsteroidal anti-inflammatory drugs, warfarin, or other antithrombotic agents. 47
First-Line Treatment
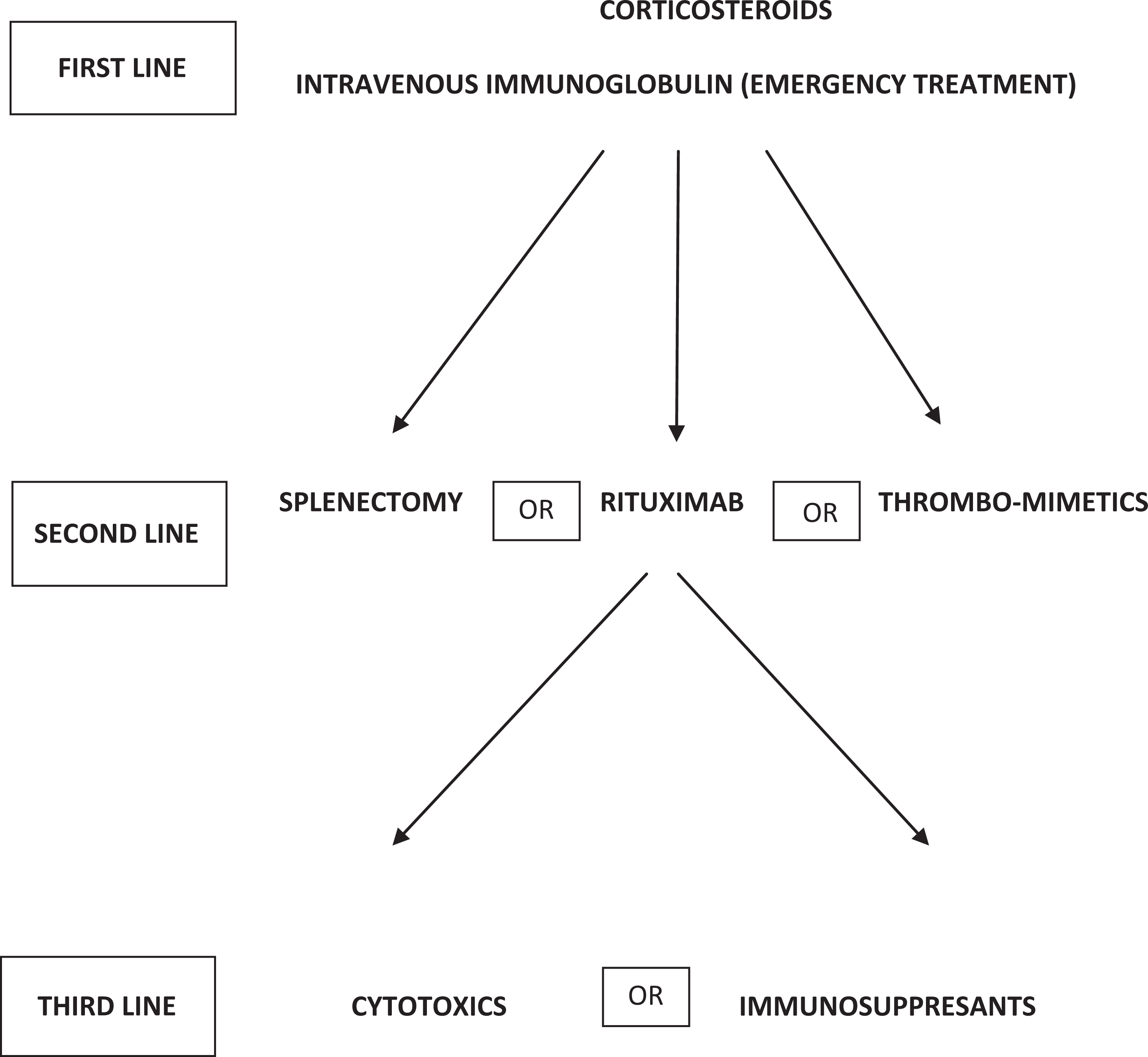
Corticosteroids are the standard initial treatment for ITP. Unfortunately, their adverse effects rapidly become apparent and may create significant complications. With time, the detrimental effects of corticosteroids often outweigh their benefits. 48 Early studies suggested that the optimal treatment is prednisone 1 mg/kg, to which 50% to 75% of patients respond with an increase in platelet counts usually within 10 to 14 days. 49 The American Society of Hematology ITP guideline published in 2011 recommends corticosteroids (prednisone 1 mg/kg orally for 21 days followed by a period of dose tapering) with longer courses being associated with a longer time to the loss of response (Figure 1). 44
High-dose dexamethasone has been evaluated as first-line therapy. Cheng et al compared high-dose dexamethasone with standard dose prednisolone. A total of 151 patients with newly diagnosed ITP and platelet counts <30 × 109/L were randomized to either dexamethasone 40 mg/d for 4 days or oral prednisolone 1 mg/kg/d. Despite a high response rate among dexamethasone-treated patients, this study showed no major difference in the sustained response rate defined as maintenance of a platelet count >30 × 109/L for 6 months by intent-to-treat analysis: 25% with dexamethasone versus 36% with prednisolone (P = .16). 50 The conclusion from this study is that both high-dose dexamethasone and prednisolone are equally effective as first-line therapy and have comparable toxicities, but no long-term benefit or cure of ITP results from a single-pulse dose of dexamethasone. An ongoing randomized trial of repeated doses of high-dose dexamethasone versus prednisolone conducted by the Italian GIMEMA study group will further contribute to define the relative efficacy of the 2 regimens in the frontline setting. 51
Novel approaches aiming to improve response to first-line therapy have used a combination of immunosuppressive drugs. A recent study prospectively evaluated the efficacy, safety, and response duration of low-dose rituximab plus high-dose dexamethasone as frontline therapy in newly diagnosed patients with ITP. One cycle of dexamethasone, 40 mg/d given intravenously for 4 consecutive days plus weekly intravenous (IV) rituximab 100 mg for 4 doses, was delivered. Twenty-one consecutive adults were enrolled. The overall response at day 28 was 90.5%. Complete sustained response at 6 months and relapse rate were 76.2% and 15.8%, respectively, compared with 30% and 62.5% for a historical group who had received standard treatment with prednisone (P = .005 and P = .004). There was a 9.5% incidence of adverse effects. This study demonstrated efficacy and safety of the drug combination but needs to be repeated in a larger population and in a randomized fashion in order to better assess its potential utility. 52
Intravenous immunoglobulin (IVIG) and IV anti-D are frequently used in the treatment of ITP. Because of their rapid onset of action, they can be used prior to invasive procedures such as surgery or delivery and are not typically considered as a “line” of therapy. In certain diseases, such as HIV, anti-D may be more efficacious and in common variable immunodeficiency, IVIG is likely to be more useful. 53 Intravenous immunoglobulin is generally well tolerated. Several regimens for IVIG administration have been utilized with comparable clinical outcome but many centers find it convenient to give 1 g/kg/d for 1 or 2 days. 54 Adverse effects occur in approximately 5% of patients, including headaches, chills, myalgia, fatigue, arthralgia, and back pain. Headaches may be severe and rarely may be associated with aseptic meningitis occurring within 72 hours of the administration of IVIG. Symptoms usually resolve spontaneously. More serious adverse events, such as intravascular hemolysis, renal failure, stroke, and myocardial infarction, have been reported. 55 These are rare and usually occur in patients with preexisting risks factors, such as the elderly patients and patients with diabetes or impaired renal function. In these patients, administration over a longer period of time (3-5 days) may be advisable. Transmission of viruses remains a theoretical risk with the use of IVIG, which is a plasma-derived product. However, since initial description of transmission of hepatitis C in the 1990s, no transmission of hepatitis, HIV, or variant Creutzfeldt-Jakob disease has been reported. 56
Severe intravascular hemolysis including 5 fatal cases has been reported following anti-D administration. The precise cause of this rare but devastating complication is unknown but is presumed to be related to complement activation. 57 Elderly patients (older than 65 years), patients with active infection, autoimmune hemolytic anemia (Evans syndrome), autoimmune disorders, or lymphoproliferative diseases appear to be at greater risk of more brisk extravascular hemolysis. 58 Anti-D is contraindicated in patients with a positive direct antiglobulin test because of risk of developing intravascular hemolysis and in patients with a hemoglobin level below 10 g/dL because of risk of developing symptomatic anemia.
Second-Line Treatment
Despite the introduction of novel agents and new information regarding response to treatment, there is no evidence to suggest a preferred sequence of treatments for patients who have recurrent or refractory ITP associated with bleeding after an initial treatment course with corticosteroids. 45,49 The central issue when approaching a decision regarding second-line therapy for an individual patient is whether to favor what has classically been considered “definitive” therapy, namely, splenectomy or whether to pursue long-term medical therapy with one of the novel thrombomimetic agents. For some patients, particularly those with an underlying autoimmune diathesis, further immunosuppressive treatment with rituximab may be preferred. In all patients who fail to respond to first-line therapy and agents such as IVIG or anti-D, it is prudent to review the diagnosis and to exclude other causes for thrombocytopenia such as primary bone marrow abnormalities, for example myelodysplastic syndromes or myelofibrosis.
Splenectomy
The initial complete response rate (platelet count above 150 × 109/L) to splenectomy is 65% to 70%, with 60% to 70% of responders maintaining a normal platelet count for several years; thus, approximately 50% of patients with chronic ITP may effectively be cured with splenectomy. A long-term retrospective analysis by Vianelli et al 59 confirmed these findings: of 233 patients with ITP who underwent splenectomy between 1959 and 2001 in 6 European hematologic institutions and who have now a minimum follow-up of 10 years from surgery, 180 (77%) achieved a complete response and 26 (11%) a partial response. Of the 206, 68 (33%) responding patients relapsed, mostly (75%) within 4 years from first response. In 92 (39.5%) patients, further treatment was required after splenectomy that was effective in 76 (83%) patients. In 138 (59%) patients, response was maintained free of any treatment at last contact. Patients who have had a splenectomy have a small risk of overwhelming infections, which is greatest in children younger than 5 years who are rarely candidates for splenectomy. Although there are no data on the efficacy of vaccination, immunizations against encapsulated bacteria (Streptococcus pneumoniae, Hemophilus influenzae B, and Neisseria meningitides) are generally recommended and should be administered at least 2 weeks before splenectomy. 60 The usefulness of postoperative antibiotic prophylaxis remains a contentious issue. 61 Laparoscopic splenectomy has become accepted as an alternative to the open procedure. In a systematic review published in 2004, 62 mortality was 1.0% (48 of 4955 patients) with laparotomy and 0.2% (3 of 1301 patients) with laparoscopy. Complication rates were 12.9% (318 of 2465) with laparotomy and 9.6% (88 of 921 patients) with laparoscopic splenectomy. Compared with open splenectomy, laparoscopic splenectomy results in a better cosmetic result and is associated with a shorter hospital stay.
Another recent study examined a cohort of 9976 patients with ITP, 1762 of whom underwent splenectomy. The cumulative incidence of abdominal venous thromboembolism was 1.6% compared to 1% in patients who did not undergo splenectomy, and deep venous thrombosis and pulmonary embolus after splenectomy occurred in 4.3% compared to 1.7% in patients who did not undergo splenectomy. While abdominal thromboses tended to occur soon after splenectomy (<90 days), deep vein thrombosis and pulmonary embolus occurred both early (hazard ratio, HR 5.2 [confidence interval, CI, 3.2-8.5]) and late (HR 2.7 [CI, 1.9-3.8]) after splenectomy. The cumulative incidence of sepsis was 11.1% among patients with ITP who underwent splenectomy and 10.1% among the patients who did not. Splenectomy was associated with a higher adjusted risk of sepsis both early (HR 3.3 [CI, 2.4-4.6]) and late (HR 1.6 or 3.1, depending on comorbidities). 63
Thrombomimetic Agents
The development of thrombopoietin mimetic agents, which increase the platelet count in ITP by increasing platelet production, began a new era in the management of the disease. Two agents, romiplostim and eltrombopag, have been approved by the Food and Drug Administration and the European Medicines Agency for use in treating ITP on the basis of randomized controlled trials. Both drugs exert their effect by activating the thrombopoietin receptor present on megakaryocytes, thus providing a good example of translational medicine: the mechanism of action of the drugs reflecting the recent advances in pathophysiology of ITP, namely, the understanding the decreased megakaryocytopoiesis, is an important factor in the disease.
In a randomized, open-label, 52-week study, 234 adult patients with ITP who had not undergone splenectomy were assigned to receive standard-of-care treatment (77 patients) or weekly subcutaneous injections of romiplostim (157 patients). The rate of a platelet response in the romiplostim group was 2.3 times that in the standard-of-care group (95% CI, 2.0-2.6; P < .001). A lower incidence of treatment failure and splenectomy, less bleeding and fewer blood transfusions, and improved quality of life was achieved in romiplostim-treated patients compared to patients treated by standard of care. 1
Two randomized, phase III, double-blind, and placebo-controlled studies of eltrombopag have been published. In the first, the response rate (platelet count > 50 × 109/L) was 59% in eltrombopag patients treated for 6 weeks and 16% in placebo patients. Platelet counts generally returned to baseline values within 2 weeks after the end of treatment. The frequency of bleeding events at any time during the study was lower in patients receiving eltrombopag than did those receiving placebo. 64
In the second trial, irrespective of splenectomy status, more eltrombopag-treated patients responded (platelet count > 50 × 109/L) during the 6-month study period compared with placebo-treated patients and experienced fewer clinically significant bleeding events (33% vs 53%), less need for rescue medication (18% vs 40%), and a reduction in concomitant treatments (59% vs 32%). 65
Long-term follow-up studies of patients treated with both romiplostim 66 and eltrombopag 67 have been published. These studies confirm the sustained response to the thrombomimetic agents and perhaps more importantly provide information regarding side effects associated with long-term use of the drugs. Increased marrow reticulin fibrosis (less than grade 3) was observed in 10 of the 271 patients in the romiplostim trials and in 7 of the 299 eltrombopag-treated patients in long-term follow-up. The clinical significance of this histologic finding is unclear, but the fibrosis appears to be nonprogressive and reversible. Hepatotoxicity is important to monitor, since approximately 3% of eltrombopag-treated patients will have an increase in serum transaminases to at least 3 times the upper limit of normal compared to 0% to 2% for controls. In the majority of patients, this is nonprogressive or resolves. 67 Cataracts that were reported to occur in animal studies of eltrombopag have not been reported in humans. There has been no report of the development of leukemia in patients with ITP receiving thrombomimetic drugs. 66 –68
Rituximab
Initial data regarding the efficacy of rituximab treatment for adults with chronic ITP were published more than 10 years ago, 69 and a meta-analysis of randomized clinical trials and observational studies on the usefulness of rituximab as means to avoid splenectomy in adult chronic ITP was published recently. 70 In this analysis of 368 nonsplenectomized patients, the overall response rate of rituximab was 57% (95% CI, 48-65), complete remission being attained in 41% of patients (95% CI, 0.33-0.51). Recently, the outcome at 5 years following response to rituximab therapy in children and adults with ITP has been published. 71 Patients received IV rituximab at the dose of 375 mg/m2 weekly for 4 weeks. The overall response rate was 52%. In 7 cases, responses were sustained (6 months or longer). In 2 patients with relapsed disease, retreatment with rituximab induced a new response. In patients with a complete or partial response, a significant rise in platelet concentrations was observed early during the course of treatment, usually 1 week after the first rituximab infusion. No clinical or laboratory parameter was found to predict treatment outcome, although there was a suggestion that women and younger patients have a higher response rate. Progressive multifocal leukoencephalopathy has recently emerged as a rare but devastating complication of rituximab treatment for rheumatologic diseases, including ITP. 72
Conclusion
Unlike the first-line treatment recommendations, which are clear, the optimal second-line treatment for ITP remains to be defined. An international consensus report published in 2010 considered second-line treatments to include azathioprine, cyclosporine A, cyclophosphamide, danazol, dapsone, mycophenolate mofetil, rituximab, splenectomy, and thrombopoietin receptor agonists eltrombopag and romiplostim. 47 The options were listed alphabetically to reflect the authors' opinion that no second-line treatment has been proven to be superior to other treatment options. The ASH 2011 guidelines recommend splenectomy for patients who have failed corticosteroid therapy (grade 1B) and thrombopoietin receptor agonists for patients at risk of bleeding who relapse after splenectomy or who have a contraindication to splenectomy and who have failed at least one other therapy (grade 1B). According to these guidelines, eltrombopag and romiplostim may be considered for patients at risk of bleeding who have failed 1 line of therapy such as corticosteroids or IVIG and who have not had splenectomy (grade 2C). Rituximab may be considered for patients at risk of bleeding who have failed 1 line of therapy such as corticosteroids, IVIG, or splenectomy (grade 2C). 1
Variability in practice regarding the choice of second-line therapy for relapsed or refractory ITP has been well illustrated in a recent survey of Oklahoma hematologists who responded to a survey regarding this issue. When presented with an adult with a platelet count of 9 × 109/L who had failed to respond to initial treatment with corticosteroids and IVIG, 32 (39%) selected splenectomy (recommended by the ASH guideline), 30 (37%) selected rituximab, and 13 (16%) selected thrombopoietin receptor agonists (both recommended by the International Consensus report). Hematologists–oncologists who had more years in practice were more likely to select splenectomy (P < .047). 73
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
