Abstract
Intracerebral hemorrhage (ICH) is associated with a higher mortality rate among stroke subtypes. The amount of hematoma at baseline and subsequent expansion are considered strong independent markers for determining poor clinical outcome. Even though reduction in blood pressure to prevent and control the amount of bleeding in ICH has received considerable amount of attention, the impact of coagulopathy and platelet dysfunction, on the bleeding diathesis has not been extensively investigated. With the increasing use of antiplatelets and/or anticoagulants, given the aging population, a deeper understanding of the interactions between ICH and hemostatic mechanisms is essential to help minimize the risk of a catastrophic coagulopathy-related ICH. In this review article, etiology and risk factors associated with coagulopathy-related ICH are discussed. An overview of coagulation abnormalities, hemostatic agents, and blood biomarkers pertaining to ICH is included.
Keywords
Introduction
Intracerebral hemorrhage (ICH) accounts for 15% to 20% and 20% to 30% of strokes in Western and Asian populations, respectively, and it is associated with a high mortality rate of up to 50% at 30 days. 1 –3 The most common spontaneous ICH results from the rupture of small (lacunar) vessels generally located deep within the brain in the basal ganglia, thalamus, pons, or cerebellum, related to long-standing hypertension (HTN). Intracerebral hemorrhage may extend into the ventricles in about 40% of cases. 4 Alternatively, lobar ICH is usually related to cerebral amyloid angiopathy (CAA). 1 –3 Other etiologies for ICH include arteriovenous malformation, tumor, ischemic stroke with hemorrhagic conversion, and coagulopathy. 1 –3 The incidence of oral anticoagulation therapy (OAT)-associated ICH (OAT-ICH), which accounts for 10% to 20% of all ICH, has increased significantly in the last few decades, probably due to increased warfarin use for the treatment of atrial fibrillation. Approximately 8000 to 10 000 cases of warfarin-associated ICH are estimated to occur annually in the United States. 5 Compared with other patients with ICH, those who are on anticoagulant therapy have a greater risk of hematoma expansion (HE), subsequent clinical deterioration and death, necessitating vigorous reversal of their coagulopathy. 6 Hematoma expansion in OAT-ICH may be more common and occur over a prolonged time frame, perhaps 24 to 48 hours longer than that in non-OAT-ICH, because of persistent coagulopathy. This prolonged natural course of HE in OAT-ICH provides a longer time window for treatment with hemostatic therapy. 7 In this review, we will focus on ICH associated with coagulation system disturbances as well as platelet dysfunction.
Pathophysiology of ICH
Acute ICH presents with both nonfocal and focal neurological signs and symptoms. Nonfocal symptoms include a decreased level of consciousness, headache, nausea, and vomiting. Most of the symptoms are related to either elevated intracranial pressure or brain herniation. Focal symptoms are variable, depending on the location of ICH. Progressive deterioration often mirrors expansion of the hematoma and brain herniation. Hematoma expansion occurs in about one-third of patients, usually within the first 12 to 24 hours of symptom onset. 7 Figure 1 shows computed tomography (CT) scans of ICH at initial presentation and its evolution with HE at 8 hours. The role of aggressive blood pressure lowering is debated; however, recommendations from the American Heart Association suggest that lowering blood pressure to 140 mm Hg is relatively safe. Randomized controlled trials for aggressive reduction of blood pressure for ICH, including ATACH II, are ongoing to assess the efficacy of blood pressure reduction in improving the outcome among patients with ICH. 8
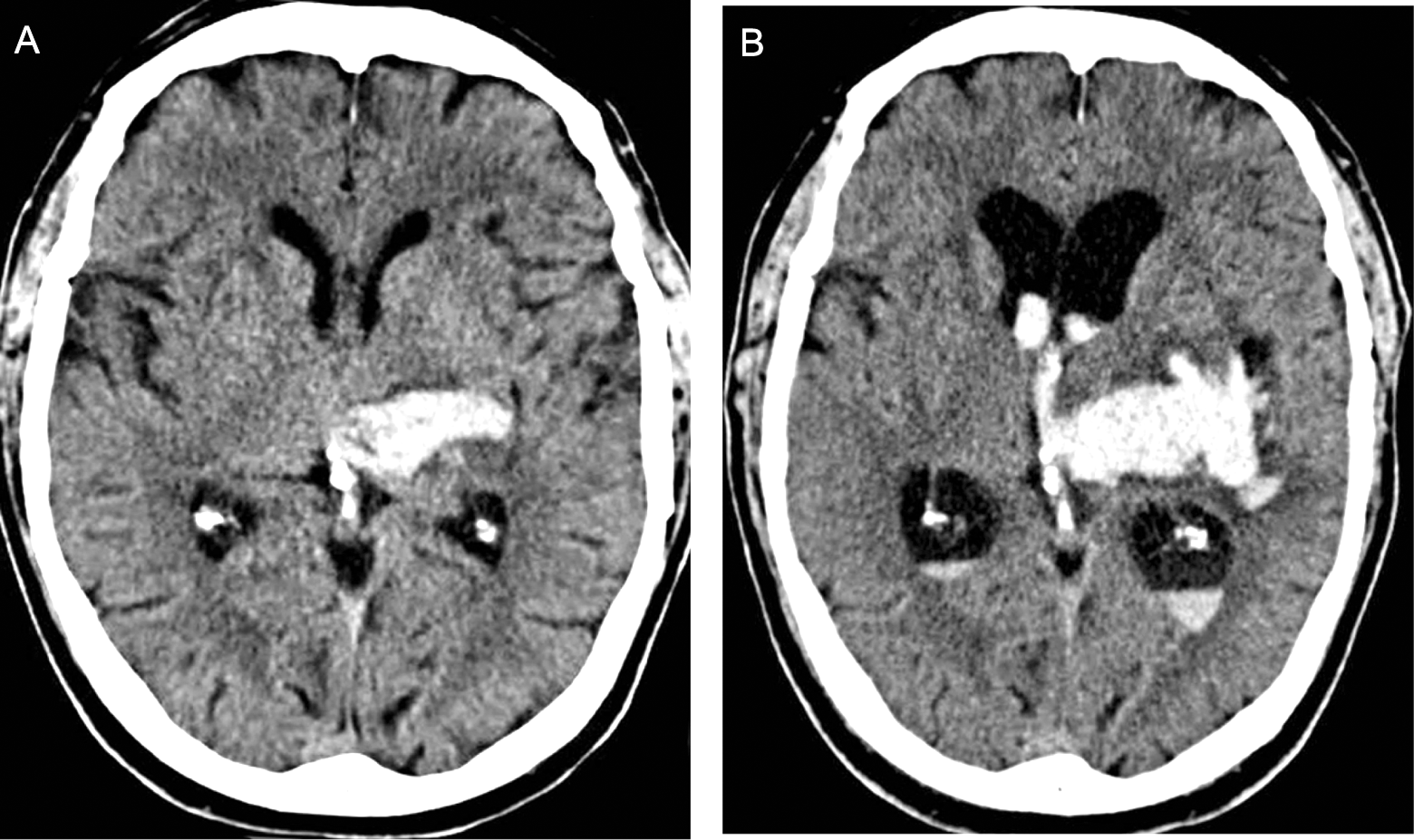
Progression of left thalamic intracerebral hemorrhage (ICH) in a 56-year-old patient with history of heavy alcohol consumption and regular aspirin use shown on computed tomography (CT) scan. A, At initial presentation. B, Hematoma expansion with intraventricular hemorrhage and hydrocephalus at 8 hours of follow-up.
Risk Factors for ICH
The 2 most common causes of ICH are HTN and CAA. Lobar ICH predominantly results from CAA, which may be recurrent and multifocal. 9 –11 Other risk factors for ICH include advanced age, diabetes mellitus, low serum cholesterol, smoking, heavy alcohol intake, leukoaraiosis on imaging, microbleeds, prior ICH, renal failure, and use of anticoagulant or antiplatelet agents. 12,13 Cerebral microbleeds are common findings in patients with ICH, occurring in up to 68% of patients. 14 In a large meta-analysis, cerebral microbleeds were found in only 5% (95% confidence interval [CI]: 3.9-6.2) of healthy adults but 60% (95% CI: 57.2-63.6) of people with ICH. Furthermore, incidental microbleeds were also associated with HTN both in healthy adults (odds ratio [OR]: 3.9, 95% CI: 2.4-6.4) and in adults with cerebrovascular diseases(OR: 2.3, 95% CI: 1.7-3.0). 15 The higher number of microbleeds may increase the risk of ICH, especially in patients taking antiplatelet agents. A study that compared microbleeds seen on MRI in patients with ICH taking antiplatelet agents found more frequent microbleeds in these patients than in matched antiplatelet users without ICH (81% vs 19%, respectively, P = .004) and nonantiplatelet users with ICH (81% vs 45%, respectively, P = .03). 16
Oral Anticoagulant Therapy–Associated ICH
Intracerebral hemorrhage is the most feared complication of OAT. It is associated with a high early mortality rate of approximately 50%. 5 The duration of bleeding is usually longer (12-24 hours) in patients on OAT, 5 with warfarin the most frequent agent. The rate of OAT-ICH ranges from 0.1% to 0.9% per year and depends on a number of factors such as the target range of international normalized ratio (INR), advanced age, underlying cerebrovascular disease, HTN, concurrent antiplatelet therapy or nonsteroidal anti-inflammatory drugs, metabolic genotypes for OAT metabolism, and comorbid illnesses. 17
A retrospective study of 303 patients with ICH found that warfarin-associated cases had a greater baseline median ICH volume (30.6 vs 14.4 mL, P = .03), an increased rate of HE defined as >33% volume increase from baseline (56% vs 26%, P = .006), and a higher mortality rate (62% vs 17%, P < .001) compared to nonwarfarin patients with ICH. 18 A study by Zubkov et al 19 looked at 88 consecutive patients with warfarin-associated ICH who presented with INR ≥1.5 and found that the mortality rate was 39.7% at 7 days and 43.2% at 30 days; in addition, there was a significant association between the initial ICH volume and 7-day mortality. The level of INR is an important factor in determining the likelihood of HE in warfarin-associated ICH. A retrospective review of 47 Japanese patients with warfarin-associated ICH found that an INR <2.0 at admission or for 24 hours after immediate treatment with prothrombin complex concentrate (PCC) was associated with unchanged hematoma size (OR: 0.069, 95% CI: 0.006-0.789, P = .031). 20 Another retrospective study that looked at 258 patients with ICH (51 of whom were on warfarin) found that there was no difference in hematoma size for patients with INR <1.2 compared to INR of 1.2 to 2.0 (P = .25) or INR of 2.1 to 3.0 (P = .36), but patients with INR >3.0 had greater hematoma volume (P = .02). 21
The emergence of new oral direct thrombin and factor Xa inhibitors (eg, dabigatran, rivaroxaban, and apixaban) will likely change the incidence of OAT. In patients with atrial fibrillation, dabigatran, rivaroxaban, and apixaban were noninferior or even superior to warfarin for prevention of stroke and systemic embolization, while the rate of ICH was lower. 22 –25 When there is bleeding into the brain, the large amount of tissue factor in the brain leads to significant thrombin generation, promoting clot formation. Warfarin causes greater impairment of thrombin generation during the propagation phase of coagulation compared to the direct Xa and thrombin inhibitors. Therefore, the use of new oral anticoagulants may lead to a lower risk of clinical ICH. 26
Antiplatelet Therapy and Platelet Dysfunction in ICH
Antiplatelet therapy is often prescribed to patients with cerebrovascular and cardiovascular disease. However, platelet dysfunction as a result of antiplatelet therapy is associated with early ICH volume growth and worse functional outcome. A prospective study measured platelet activation admission using AccumetricsVerifyNow-ASA assay (Accumetrics, California) in 68 patients with ICH. 27 This study found that reduced platelet activity (defined as ≤550 aspirin reaction units [ARUs]) was associated with increased ICH volume growth (1 [−0.05 to 7.3] vs −0.1 [−0.6 to 0.5] mL; P = .05) and worse functional outcome, defined as a modified Rankin Score (mRS) >3, at 3 months (P = .02). In addition, 16 (24%) patients received a platelet transfusion at a mean of 21.2 ± 11.4 hours after symptom onset, leading to an increase in mean platelet activity (448 ARUs, range [414-479] to 586 ARUs, range [530-639], P = .001) but without impact on outcomes. 27 In another study of 282 patients with ICH, there were no significant differences in hemorrhage volume at baseline or 72 hours, mortality at 90 days, and mRS <3 at 90 days between those who were taking (N = 70) or not taking (N = 212) antiplatelet therapy at the time of ICH. 28
Intraventricular hemorrhage (IVH) is a predictor of higher mortality in patients with ICH, and it may occur isolated or as a result of expansion of a parenchymal focus. Reduced platelet activity has been associated with increased rate of IVH, independent of ICH location. 27 Naidech et al 27 prospectively studied a series of 73 patients with ICH, where 37 had IVH. They found that reduced platelet activity was significantly associated with IVH severity (P = .01). Presence of IVH was also associated with worse National Institutes of Health Stroke Scale score (P = .002) and mRS score (P = .001) at 14 days, as well as with mRS scores at 28 days (P = .02) and 3 months (P = .008). 27
A low platelet count may also presumed to be a risk factor for ICH. There are few studies specifically regarding ICH and thrombocytopenia. One retrospective study 26 of 114 consecutive patients with ICH found that 29 of these patients had thrombocytopenia (platelet count <150 000/mm3). Fourteen of these patients had platelet counts below 10 000/mm3. Of these patients with thrombocytopenia, 6 had idiopathic thrombocytopenic purpura (ITP) and the remainder had aplastic anemia or hematologic malignancy. A review of case reports 29 of ICH in children with ITP indicated that in 71% of cases the platelet count was <10 000/mm3. Thrombocytopenia may be unlikely to result in spontaneous ICH, unless the platelet count is extremely low, or there are other contributing factors.
Thrombolysis-Associated ICH
Hemorrhagic conversion, particularly ICH, is a well-known risk of thrombolytic treatment of ischemic stroke. In the National Institute of Neurological Diseases and Stroke Tissue Plasminogen Activator (tPA) trial, the incidence of symptomatic ICH in patients given intravenous (IV) tPA between 0 and 3 hours of ischemic stroke symptom onset was 6.4% compared to 0.6% in patients given placebo, P < .001. 30 In the European Cooperative Acute Stroke Study III trial, the incidence of symptomatic ICH was higher for the tPA group (2.4%) vs placebo group (0.2%), p = .008, in patients with ischemic stroke treated from 3 to 4.5 hours of symptom onset. However, mortality did not differ significantly (7.7% and 8.4%, respectively, P = .68). 31
Complication risk of ICH for intra-arterial (IA) tPA treatment is also of concern. In the Prolyse in Acute Cerebral Thromboembolism II trial, symptomatic ICH occurred in 10% of treated patients and 2% of control patients (P = .06) with symptomatic ICH occurring in patients with severe baseline deficits. 32 A study that looked at 207 patients treated with IV/IA thrombolysis or IA alone found that there was no significant difference in symptomatic ICH (20% in IV/IA group vs 15% in IA only group). 33
Coagulation Function and HTN
Hypertension is associated with abnormalities in the vessel wall, abnormalities in blood constituents, and abnormal blood flow; therefore, the common complications of HTN are paradoxically thrombotic rather than hemorrhagic. 34 –37 Hypertension mediates a prothrombotic state that leads to changes in the platelets, vascular endothelium, and the coagulation and fibrinolytic pathways. 35,37 First, HTN causes morphological, biochemical, and functional changes in platelets. Platelets from hypertensive patients show increased aggregation and activation. High levels of platelet microparticles have procoagulant effects and may promote platelet adhesion to the vessel wall. They promote thrombin generation which in turn leads to more platelet aggregation. Platelet microparticles also contain platelet glycoprotein Ib, IIb, and IIIa, as well as P-selectin, and thrombospondin which promotes adhesion of platelets to the endothelial matrix. In addition, HTN also exposes circulating platelets to increased shear force which leads to platelet activation. 37 The renin–angiotensin–aldosterone system leads to increased platelet aggregation due to the increased levels of catecholamines often seen in patients with hypertension. 35,37 Angiotensin-converting enzyme (ACE) catalyzes the conversion of angiotensin I into angiotensin II, which is a potent vasoconstrictor and it breaks down bradykinins vasodilators. The overall effect of ACE inhibitors therefore is to cause vasoconstriction and maintain vascular tone. The activity of ACE in hypertensive blood vessels increases as the blood pressure is elevated. The oxidative stress induced by angiotensin II stimulates growth and proliferation of vascular smooth muscle cells and migration of inflammatory markers. 34 The fibrinolysis cascade is also dysfunctional in patients with high blood pressure. The Framingham Offspring Study 38 demonstrated a correlation between increased blood pressure and plasminogen activator inhibitor (PAI)-1 and tPA antigen levels, suggesting that there is impaired fibrinolysis in patients with hypertension without known cardiovascular disease. High diastolic pressure has high correlation with increased blood viscosity and hematocrit level. 36 Other comorbid conditions, such as dyslipidemia, atrial fibrillation, diabetes, and congestive heart failure, are common in patients with hypertension, where all of these conditions are known to activate the platelets. 37 Many studies have shown that the benefit of antihypertensive agents is probably beyond the blood pressure–lowering effect as they promote reversal of abnormal hemostasis, endothelial dysfunction, and platelet activation. 34,35,37 For example, ACE inhibitors and angiotensin receptor blockers have beneficial effects in reversing endothelial dysfunction and improving vascular tone by direct inhibition of the effects of ACE. 34 However, in many hypertensive cases with other cardiovascular risk factors, patients are prescribed antiplatelet agents to counteract abnormal coagulation activation and impaired fibrinolysis that lead to increased thrombogenesis.
Hemostatic Agents for ICH
Impaired coagulation can potentially facilitate HE. The hemostatic impairment may be due to the intake of anticoagulants, antiplatelet agents, or inherited or acquired abnormalities in the coagulation system. The true prevalence of HE after the onset of ICH is unknown for many reasons including variable time to presentation at hospital, inconsistent neuroimaging, and heterogeneous ICH etiologies. 39,40 However, clinical studies suggest that about one-third of the patients with ICH present further enlargement of hematoma within the first 24 hours of initial ictus. 41 –44 Rapid reversal of coagulopathy is desirable to prevent ICH expansion; nonetheless, evidence-based trials supporting the efficacy of reversal strategies are still lacking.
Recombinant activated factor VIIa (rFVIIa) has been tried in the treatment of patients with spontaneous ICH not receiving oral anticoagulants. A multicenter, randomized, double-blind, placebo-controlled, dose-escalation phase II trial of rFVIIa with 40 patients treated with placebo or 5, 20, 40, or 80 μg/kg of rFVIIa (N = 8 per group) found 33 patients experienced 186 adverse effects, including deep vein thrombosis, cerebral infarction, pulmonary embolism, ischemic electrocardiographic changes or cardiac enzyme elevation, with similar frequency in the 5 groups. 45 A second phase II study with dose escalation from 10 to 160 μg/kg versus placebo in a total of 48 patients had similar findings. 46 A third study randomized 399 patients to receive either 40, 80, or 160 μg/kg rFVIIa or placebo within 3 hours after symptom onset and observed treatment with rFVIIa decreased hematoma volume expansion, reduced mortality, and improved functional outcomes, but noted an increased frequency of thromboembolic events which occurred in 7% of rFVIIa-treated patients compared to 2% of placebo-treated patients. 47 Diringer et al 48 pooled the data from these 3 randomized placebo-controlled studies of patients with ICH diagnosed within 3 hours, who received a single dose of rFVIIa (5-160 µg/kg; n = 371) or placebo (n = 115) and concluded that rFVIIa decreased HE, but there was an increased risk of arterial thromboembolic events associated with higher doses of rFVIIa in patients with ICH as compared with placebo. Subsequently, the Factor Seven for Acute Hemorrhagic Stroke trial randomized 841 patients presenting <3 hours after ICH to 20 or 80 µg/kg of rFVIIa or placebo found treatment with 80 µg/kg rFVIIA resulted in significant reduction in HE with similar rates of venous thromboembolism. However, there were higher rates of arterial thromboembolism in the rFVIIa-treated patients with 47 (26%) in the 20 µg/kg group and 82 (46%) in the 80 µg/kg group compared to 49 (27%) in the placebo group (P = .04). 49 The study also suggested that higher doses of rFVIIa in a high-risk population are associated with a small increased risk of cardiac events. This study did not find improved survival or functional outcome in the rFVIIa treatment groups. 50 In addition, a review of 17 randomized controlled trials in which rFVIIa was used to reduce bleeding in patients with a variety of clinical conditions including ICH concluded that this therapy reduced ICH expansion as well as mortality, but it continues to present a risk of thromboembolic complications. 51
The optimal strategy for OAT reversal in ICH is not known; however, the benefits of reversal must be weighed against the potential risk of thrombotic events. Current OAT reversal treatment options include vitamin K, fresh frozen plasma (FFP), rFVIIa, and PCCs. 52 Vitamin K is essential but does not have rapid effect, often taking 24 hours for INR reversal. 5 Traditionally, FFP has been the mainstay of therapy, but it has many drawbacks including large volume, slow thawing time, and transfusion-related complications such as infections and lung injury. A retrospective study that compared 15 patients who received vitamin K and FFP with 12 patients who also received rFVIIa in addition to vitamin K and FFP found that the median times from presentation to an INR <1.3 were higher for the FFP group compared to the rFVIIa group (32.2 and 8.8 hours, P = .016), in addition, the INR normalized slowly (at 110 and 130 hours, for the FFP group compared to the rFVIIa group, respectively), concluding that rFVIIa may be an effective adjunct to FFP in warfarin-associated ICH, facilitating faster correction of INR and decreasing FFP requirements. 53 The evidence to use rFVIIa for warfarin reversal is limited. 54 While rFVIIa alone can rapidly correct the INR, this treatment may not be sufficient to reverse the clinical effects of warfarin. In a human skin punch biopsy model, no difference was seen between placebo and 5 to 80 µg/kg rFVIIa treatment on bleeding duration and blood loss, despite improved laboratory results including thromboelastography, thrombin generation, and INR with rFVIIa. 55
The PCCs have recently gained attention in patients with warfarin-associated ICH due to its rapid reversal of the INR. These are plasma-derived products containing 3 (ie, II, IX, and X) or 4 factors (ie, II, VII, IX, and X). The PCC products may also contain protein C, protein S, and heparin. 56 All PCCs are administered IV and several preparations are available that contain 1 to 4 of all the factors with viral inactivation steps (filtration, pasteurization, or solvent or detergent treatment) to minimize the risk of pathogen transmission; Recommended doses and rates vary between products, but they are generally calculated from body weight and are based on the units of factor IX. 57 The PCCs have been favored over the traditional vitamin K and FFP in urgent reversal of coagulation due to their short preadministration time as well as administration time and the volume of product given to patients; however, they have been known to carry a risk of thromboembolic complications and disseminated intravascular coagulation. 58
The optimal dosing and infusion rates of PCC to rapidly but safely reverse the INR are not clearly defined. 52 A study 59 investigating optimal dose of a 4-factor PCC in 42 patients for acute reversal of oral anticoagulation in patients with major hemorrhagic complications or who required invasive procedures measured INR before and 10 to 60 minutes after the administration of PCC with or without vitamin K. The study found that the 500 IU of PCC is likely to be the optimal dose of this PCC for emergent reversal of INR in patients requiring rapid correction of INR below 5.0 but to be an inadequate dose in patients with an INR of 5.0 or more. A prospective cohort study that looked at the use of a 3-factor PCC infused in patients whose INR was higher than or equal to 2.0 (median INR 3.5, range 2-9) at doses of 35 to 50 IU/kg, stratified according to the initial INR, found that 30 minutes after the administration of the PCC, the median INR was 1.3 (range 0.9-3), and the INR then declined to less than or equal to 1.5 in 75% of patients with the median INR remaining lower than or equal to 1.5 (median, 1.16; range, 0.9-2.2) at 96% of all postinfusion time points up to 96 hours. 60 However, another study found that infusion of a 3-factor PCC alone was suboptimal in lowering the INR in patients with INR >5.0 and bleeding or high risk of bleeding. 61 Addition of plasma as a source of factor VII improved the rate of INR correction. A retrospective analysis of 19 patients with warfarin-associated ICH treated with FFP and vitamin K (N = 9) compared with those who were treated with FFP and vitamin K plus factor IX complex concentrate, a 3-factor PCC (N = 10), concluded that factor IX complex concentrate in combination with FFP and vitamin K may result in a shorter time required for correction of warfarin-associated coagulopathy when compared to FFP and vitamin K alone. 62 A retrospective study of 55 patients who were grouped into: (1) patients treated with PCCs alone or in combination with FFP or vitamin K, (2) patients treated with FFP alone or in combination with vitamin K, and (3) patients treated with vitamin K as a monotherpay found that PCC was associated with reduced incidence and extent of HE compared with FFP and vitamin K due to its effect on a more rapid INR reversal. 52
The recent introduction of the novel oral anticoagulants, including the direct thrombin inhibitor dabigatran and the factor Xa inhibitors rivaroxaban and apixaban, has created a new challenge for controlling HE. Very limited data are available on the reversal of the drugs in humans, thus the optimal management in the ICH setting is unknown. Animal studies with dabigatran have been contradictory. A study by Lauer et al 65 did not find increased HE in a murine model of ICH when dabigatran was compared to warfarin. However, in a second study by Zhou et al, 66 HE was seen in a murine model of ICH in dabigatran-treated mice; furthermore, PCC and FFP, but not rFVIIa, limited HE. A randomized, placebo-controlled crossover study comparing dabigatran and rivaroxaban in healthy volunteers found a 4-factor PCC could reverse the anticoagulant effect of rivaroxaban but not dabigatran on coagulation tests. 67
There is much less evidence regarding whether platelet transfusion improves outcome in patients with platelet dysfunction-associated ICH, mainly due to the lack of prospective randomized clinical trials. In a retrospective study of 65 patients with ICH receiving aspirin and/or clopidogrel, there was no significant difference in HE in the first 24 hours, discharge mRS, or rate of systemic complication in subjects who were transfused (n = 35) versus nontransfused (n = 31). 68 However, the timing of platelet transfusions was unclear. A more recent study looked at 45 patients with ICH and platelet dysfunction as measured by VerifyNow-ASA assay. 69 They found that patients who had platelet transfusion within 12 hours of symptom onset compared to those transfused after 12 hours had a smaller hemorrhage size on average on follow-up (8.4 vs 13.8 mL, P = .04) and increased odds of independence (P = .01), as defined by mRS <4 at 3 months. A summary of prospective clinical trials on anticoagulant/procoagulant therapy in ICH are shown in Table 1. As shown in Table 1, several studies on anticoagulant therapy in ICH included lack of information on the time of symptom onset to treatment and lack of randomization, insufficient sample size, heterogeneous cohorts, and inadequate statistical analysis (such as lack of multivariate analysis) to accurately assess the impact of therapy on the outcome.
Published Prospective Clinical Studies on Anticoagulant/Procoagulant Therapy in Spontaneous Intracerebral Hemorrhage.
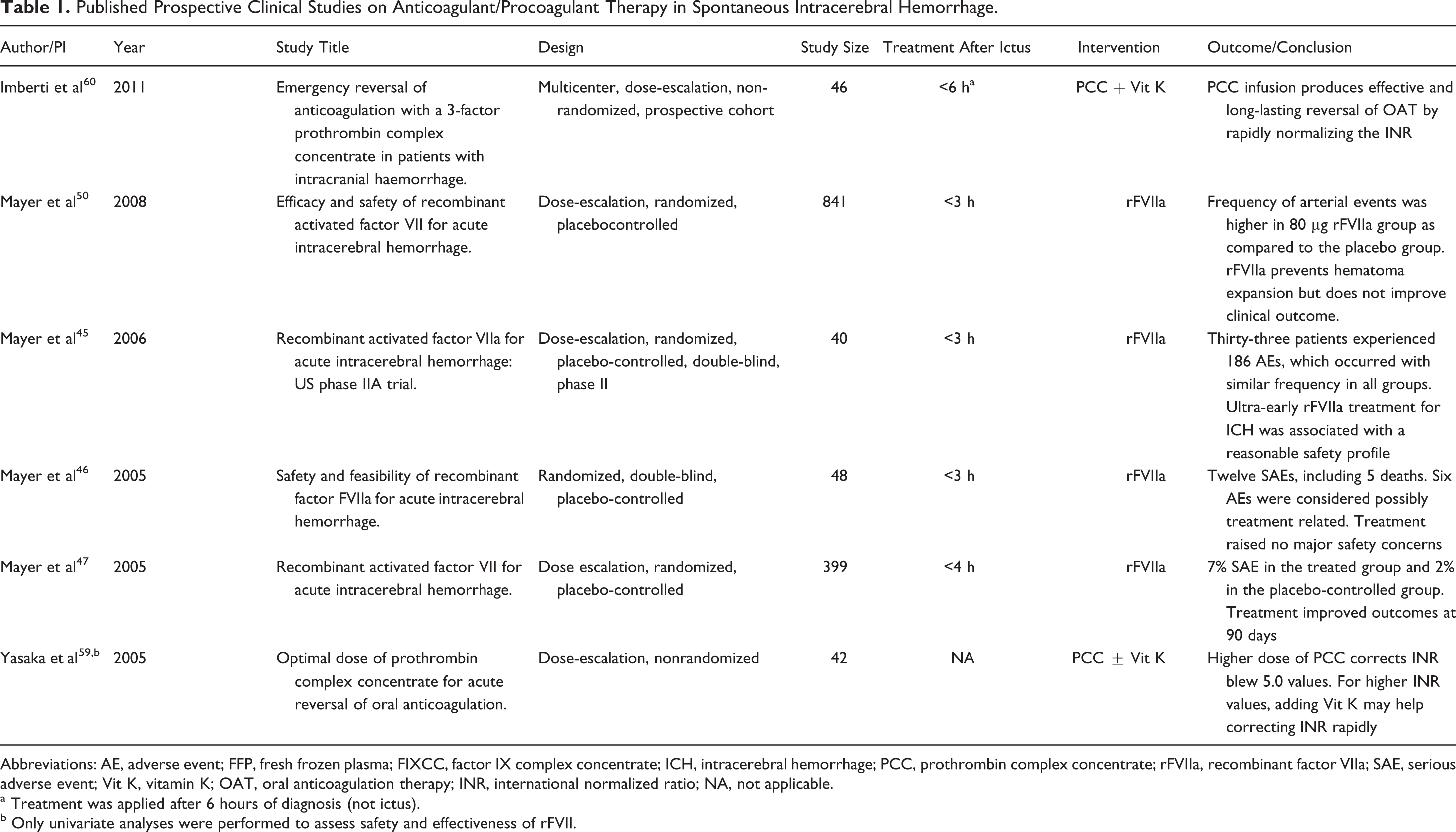
Abbreviations: AE, adverse event; FFP, fresh frozen plasma; FIXCC, factor IX complex concentrate; ICH, intracerebral hemorrhage; PCC, prothrombin complex concentrate; rFVIIa, recombinant factor VIIa; SAE, serious adverse event; Vit K, vitamin K; OAT, oral anticoagulation therapy; INR, international normalized ratio; NA, not applicable.
a Treatment was applied after 6 hours of diagnosis (not ictus).
b Only univariate analyses were performed to assess safety and effectiveness of rFVII.
Blood Biomarkers to Predict Risk of ICH, HE, and Outcome
Assessment of plasma levels of the coagulation and inflammatory biomarkers on admission could be useful to identify those who are at increased risk of HE, select treatments, quantitatively follow the efficacy of treatments, and to possibly predict a poor outcome.
70
Multiple studies have tried to identify biomarkers, but most have small numbers of patients and vary in the timing of biomarker analysis in relation to time of presentation with ICH (see Table 2). One prospective study measured the following blood biomarkers on admission and at 24 to 72 hours: fibrinogen, factor XIII, thrombin activated fibrinolysis inhibitor, PAI-1, plasminogen, α2-antiplasmin, tPA,
Published Studies on Blood Biomarkers and Their Relationship With HE in Spontaneous Intracerebral Hemorrhage.

Abbreviations: ACE, angiotensin-converting enzyme; aPTT, activated partial thromboplastin time; ARUs, aspirin reaction units; CAA, cerebral amyloid angiopathy; FFP, fresh frozen plasma; GFAP, glial fibrillary acidic protein; LDL, low-density lipoprotein; HDL, high-density lipoprotein; HE, hematoma expansion; IA, intra-arterial; INR, international normalized ratio; IV, intravenous; IVH, intraventricular hemorrhage; mRS, modified Rankin Score; PAI-1, plasminogen activator inhibitor 1; PAP, plasmin–antiplasmin; PCC, prothrombin complex concentrate; FVIIa, recombinant factor VIIa; TAT III, thrombin–antithrombin III; tPA, tissue plasminogen activator; CT, computed tomography.
a Only univariate analyses were performed to assess association between the blood biomarkers and HE.
b It was only significant for the 24 patients with CT scans performed within 3 hours of symptom onset.
Serum
Biomarkers have been studied to differentiate ICH from ischemic stroke and as predictors of risk of developing ICH. Serum glial fibrillary acidic protein (GFAP) released from astrocytes has been found to be substantially increased in patients with ICH (median 11 ng/L, range 0-3096 ng/L) compared to patients with ischemic stroke (median 0 ng/L, range 0-14 ng/L, P = .001), suggesting that serum GFAP can reliably detect ICH in the acute phase of stroke. 77 A prospective case–control study of 74 000 people in Sweden drew baseline blood studies and recorded medical histories and followed them from 1985 through 2000. First-ever spontaneous ICH occurred in 39 cases. On univariate analysis, systolic blood pressure, diastolic blood pressure, and tPA/PAI-1 complex were associated with ICH. Multivariate analysis showed associations of HTN and low von Willebrand factor with ICH. There was no association with ICH for tPA, PAI-1, or soluble thrombomodulin. 78
Conclusions
Intracerebral hemorrhage is a devastating condition that often results in high morbidity and mortality. Coagulopathy-associated ICH carries an even worse outcome due to larger hemorrhage size, expansion of hematoma, and ultimately higher mortality. Therefore, identifying patients with ICH having coagulopathy and its rapid reversal should be one of the main targets for this condition. The most promising agents for rapid reversal include rFVIIa and PCCs; however, randomized controlled trials have produced conflicting results. Therefore, further studies to determine the clinical utility of these agents in patients with ICH having coagulopathy are indicated. Finally, there have been very few studies with inadequate sample size, looking at the relation between coagulation markers and initial hematoma volume and its subsequent expansion. Accurate assessment of the relationship between these factors requires neuroimaging studies and blood tests at the early onset (<3 hours). The use of blood biomarkers to identify patients with ICH at higher risk of HE requires further investigation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Nicole D. Zantek has an equity interest in Endo Health Solutions, which manufactures some of the products discussed in this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: a generous support from Stanley S. Hubbard’s family.
