Abstract
The risk of thromboemboli is increased in patients with cancer, and this is precipitated by the chemotherapeutic agents. Bevacizumab is an anti-vascular endothelial growth factor monoclonal antibody and has an importance in the treatment of metastatic colon cancer. The association between bevacizumab, which is demonstrated to increase the risk of thromboemboli, and mean platelet volume (MPV), which is a marker of thrombocyte function, has been investigated within study. A total of 74 patients with metastatic colon cancer were included in the study and the levels of platelets (PLTs), MPV, and platecrit (PCT) values were recorded in SPSS 16.0 program both at baseline and at the >third month. There were significant decreases in 3 parameters (PLT, MPV, and PCT) during the treatment period with bevacizumab (P = .009, P = .001, and P = .000, respectively). Unlike cases with thrombosis, there is a significant decrease in MPV in combination treatments with bevacizumab.
Introduction
Thrombocytes play an important role in atherosclerotic complications. 1–4 Mean platelet volume (MPV) is an indicator of thrombocyte volume. In the presence of increased MPV levels, there are wide platelets with dense granules, containing more thromboxane A2 in the blood. 5 In vitro response to adenosine 5′-diphosphate (ADP) and collagen as well as tendency to aggregation are higher. 6 Increased MPV related with thromboembolism has been reported in patients with previous myocardial infarction, 7 cerebrovascular thromboembolism, 8 diabetes mellitus, 9 and in smokers. 10 In these diseases and in smoking, which increase thromboemboli risk similar to bevacizumab, MPV levels are increased.
Vascular endothelial growth factor (VEGF) is primarily targeted in antiangiogenic treatment of solid tumors. 11 Bevacizumab, sunitib, sorafenib, and pazopanib are currently used in cancer treatment as an antiangiogenic agents. 12–14 Unlike others, bevacizumab exists in monoclonal antibody structure and causes VEGF blockage. 15 Its efficacy has been shown in colorectal cancer, 16–19 lung cancer, 20,21 breast cancer, 22 renal cancer, 23 brain tumors, 24 and ovarian cancer. 25 Thromboembolic events, delay in wound healing, gastrointestinal system perforation, hemorrhage, hypertension, and proteinuria have been observed as side effects associated with bevacizumab. 26 Thrombosis has been well investigated and arterial thrombosis has been observed to be increased in patients with metastatic colon cancer using bevacizumab. 27
In our study, we investigated the association between MPV, which is an indicator factor in thrombosis, and bevacizumab, which is known to increase the risk of arterial thromboembolism. Our goal was to determine whether or not combination therapies including bevacizumab had an effect on MPV in the treatment of metastatic colon cancer.
Methods
Patients with metastatic colon cancer receiving combination therapy with bevacizumab were included in this study. A total of 74 patients were enrolled from 2 centers (Kayseri Education and Research Hospital and Erciyes Univercity). It was observed that the patients had received combination therapy with bevacizumab for at least 3 months. Platelet, MPV, and platecrit (PCT) levels were recorded using Statistical Package for the Social Sciences 16.0 (SPSS16.0) statistical software at the time of bevacizumab initiation and at the third month and later periods of the treatment. Age, gender, and the type of combination were also included. Additionally, subgroup analysis was performed according to gender and age (younger and older than 65 years). Frequency analyses and paired sample t test were performed. P < .05 was considered statistically significant.
Results
The mean age of the 74 patients was 59 ± 20 years, with a majority of male patients (67.6%). Regarding the type of therapy, the majority of the patients received bevacizumab + folinic acid–fluorouracil–irinotecan (FOLFIRI) combination (90.5%). The patient characteristics are presented in Table 1 .
Patients and Characteristics
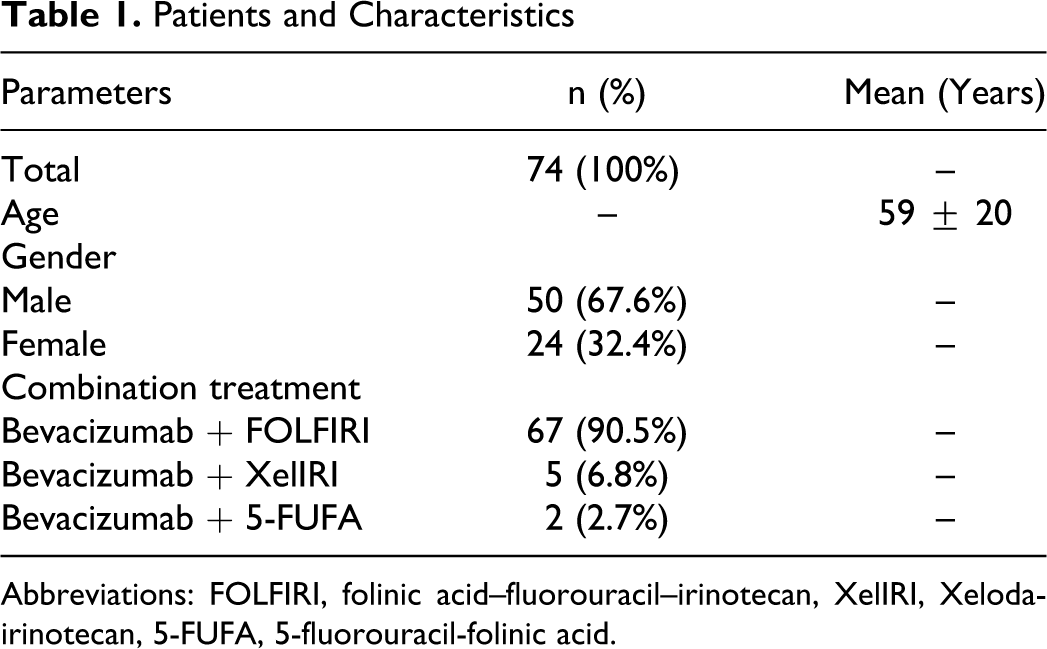
Abbreviations: FOLFIRI, folinic acid–fluorouracil–irinotecan, XelIRI, Xeloda-irinotecan, 5-FUFA, 5-fluorouracil-folinic acid.
There was a significant difference between MPV values prior to treatment, at the 3rd month of the treatment, and at following periods (P = .001). Mean platelet volume levels decreased by the treatment in combination with bevacizumab. A significant difference was also obtained regarding the other 2 parameters (platelet count and PCT). There were significant decreases in platelet (P = .009) and PCT counts (P = 0.000) following treatment. Upon examination of the subgroups, PLT, MPV, and PCT values were found to be significant (P values were .021, .004, and .001, respectively). In female patients, none of the 3 parameters were found to be significant (PLT, MPV, and PCT P values were .077, .079, and .067, respectively). While there was a significant decrease in MPV value (P = .023) in patient over 65 years of age, significant changes were not observed in PLT and PCT values. In patients younger than 65 years of age, PLT, MPV, and PCT values were statistically significant (P values were .017, .010, and .001, respectively). Values for PLT, MPV, and PCT are presented in Table 2 .
Levels of PLT, MPV, and PCT
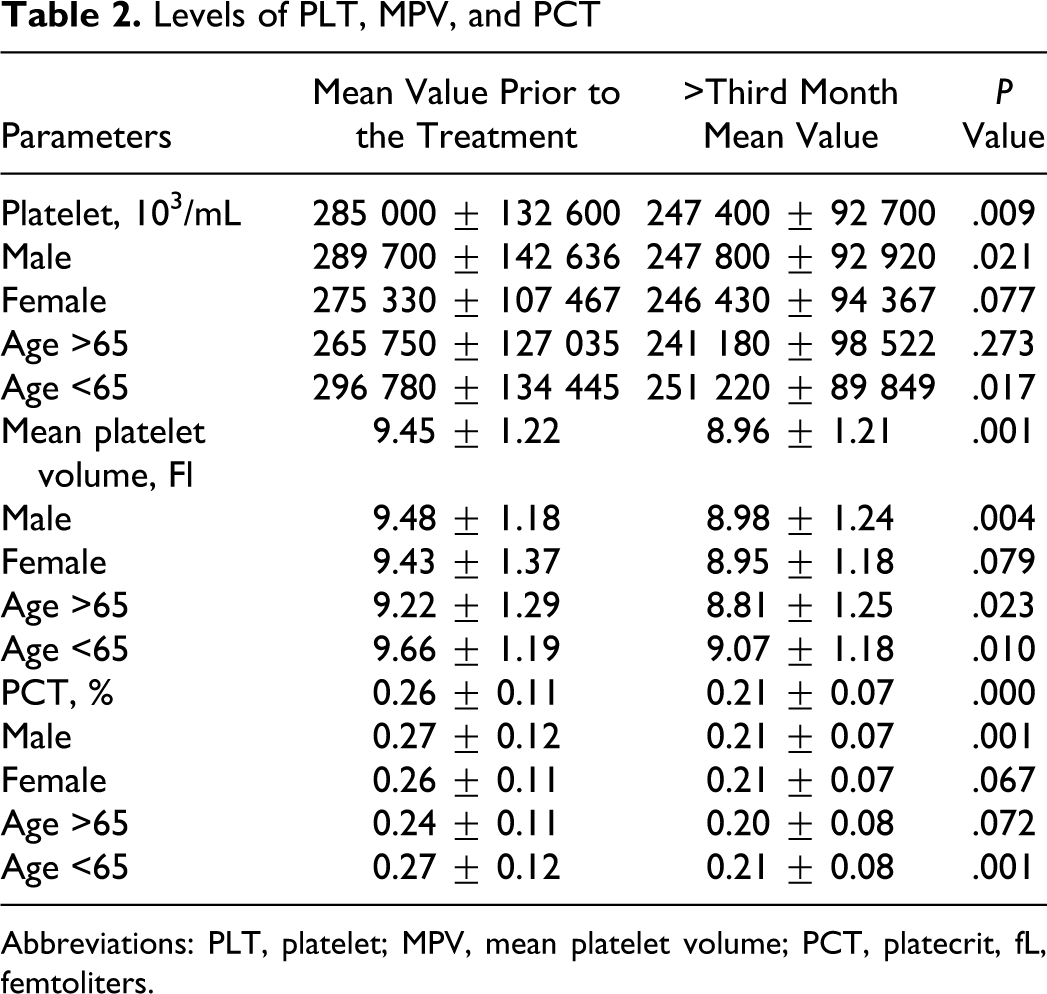
Abbreviations: PLT, platelet; MPV, mean platelet volume; PCT, platecrit, fL, femtoliters.
Discussion
Cancer is a disease with increased thrombosis risk. Infection and thromboembolic events are the 2 leading reasons related to cancer. 28,29 The importance of 3 factors in the prothrombotic period has been understood after investigating the cases with malignancy: the elevation of procoagulation factors secreted from the cancer cells, inactivation of fibrinolytic system, and reactivity of thrombocytes. 30 These 3 mechanisms initiate thrombosis formation in the presence of predisposing factors, such as stasis of the venous system, surgical intervention, chemotherapy, and radiotherapy. 31
According to the results of our study, there is a significant decrease in MPV values, which is an indicator of thrombocyte reactivity. Similarly, there was a significant decrease in thrombocyte and PCT levels. Cancer itself was a condition in which thrombosis increased. In addition, the patients in our study were receiving combination therapy with bevacizumab that was shown to increase thrombosis. In this therapy group, the number, size, and granulation of thrombocytes, which are the most important factors in thrombosis, significantly decreased after treatment. These results demonstrate that thrombocytes play a minor role in thromboembolism associated with bevacizumab. Additionally, the results were significantly affected by gender and age.
Consequently, the usage of bevacizumab in patients with metastatic colon cancer significantly decreases the MPV levels. These results may likely offer a counterview in usage of aspirin to prevent thromboembolic events in patients receiving bevacizumab due to the risk of hemorrhage.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
