Abstract
Hemodynamic fluctuations and thromboembolic complications are significant areas of concern during the postoperative management of patients with univentricular hearts. The objective of this study is to review the incidence and risk factors associated with thrombosis and thromboembolic complications following total cavopulmonary anastomosis, the third stage of the palliative surgical procedure. A literature search of published evidence was conducted on OvidSP MEDLINE(R) and Embase followed by paired title, abstract, and full-text screening based on specific inclusion criteria. High risks of thromboembolic outcomes were identified across studies, with variable incidences between 3% and 20%, high mortality rates up to 38%, and an inverse relationship with prophylaxis treatment administration. Several risk factors for thrombotic complications, including chronic systemic venous hypertension, protein-losing enteropathy, passive blood flow, atrial arrhythmias, conduit stenosis, prosthetic material use, coagulation factor abnormalities, and several patient characteristics were identified. Based on these findings, a prophylactic anticoagulation algorithm has been proposed.
Introduction
A century ago, only approximately 25% of the individuals born with congenital heart disease (CHD) survived past their first year of life. 1 However, with advancements in surgical interventions, cardiopulmonary bypass, and intensive care, current approximations indicate that 85% of patients with CHD can survive until adulthood. 2 Univentricular congenital heart anomalies such as hypoplastic left heart syndrome and tricuspid atresia result in conditions where there might be 2 anatomical ventricles but only 1 functional ventricle. Such univentricular physiology is corrected by an invasive 3-staged palliative procedure in which the systemic and pulmonary circulations are separated in order to reestablish stable cardiac hemodynamics.
The present Fontan procedure, the final step in the palliation, typically entails the use of a lateral tunnel Fontan (LTF) or a prosthetic extracardiac conduit Fontan (ECF) in order to establish total cavopulmonary anastomosis and facilitate the redirection of blood from the inferior vena cava (IVC) to the pulmonary arteries (PAs) in order to separate the 2 circuits (Figure 1A and B). 3 The right atrium (RA)-PA connection has also been a popular variant of the procedure but has been largely replaced by the aforementioned methods in modern practice.
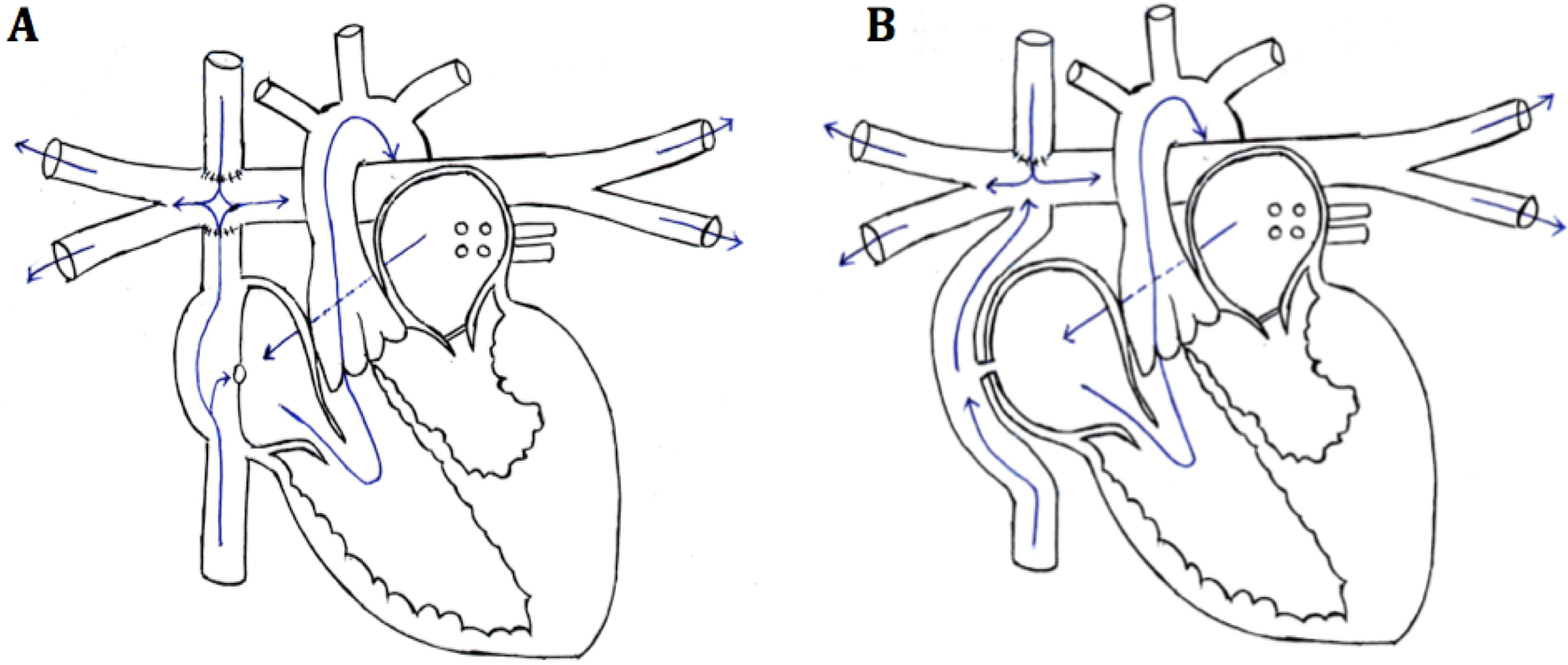
A and B, The variations in the final stage of the palliation—lateral tunnel and extracardiac conduit, both of which are fenestrated.
Although the Fontan procedure has undergone numerous improvements and modifications over the years, significant risks of postoperative complications and mortality have been reported in previous literature. These complications often include hemodynamic abnormalities, conduit stenosis, low cardiac output, arrhythmias, liver dysfunction, protein-losing enteropathy, and thrombotic complications. 4 –6 Despite the reporting of these outcomes across numerous studies, there is considerable incongruity in reported data, anticoagulation treatment and detection of thrombotic and thromboembolic end points, and significant variance in institutional practices.
This literature review attempts to summarize a large body of disparate literature regarding coagulopathy and thrombosis in patients with single-ventricle heart disease who have undergone Fontan palliation. The purpose of this review is to (1) discuss and compare thrombotic and thromboembolic incidences across the 3 variants of the procedure; (2) assess risk factors associated with thrombotic and thromboembolic end points; and (3) propose an algorithm for future administration of prophylactic anticoagulation therapy.
Methods
A systematic search was conducted to identify primary and secondary literature published in the OvidSP MEDLINE(R) and Embase databases between 2005 and 2012, pertaining to thrombotic and thromboembolic complications in patients following the Fontan procedure. A comprehensive and consistent search strategy was implemented in both databases, using the search terms “Fontan” AND “thrombosis” AND “thromboembolism” AND “anticoagulation” to identify relevant studies. Randomized controlled trials, observational, and reviews relevant to the final Fontan stage in particular were considered to be eligible for inclusion. Case reports and case series were considered ineligible study designs due to their generally limited sample sizes. In addition, letters, conferences, editorials, opinion papers, protocols, and commentaries were excluded a priori due to the lack of representative or sufficient primary data. In vitro, animal, and non-in vivo human studies were also excluded a priori.
A pair of screeners (M.F. and A.A.) then conducted blinded title and abstract screening to exclude duplicate articles and literature that did not meet the aforementioned inclusion criteria. Articles included in this preliminary stage of screening underwent blinded full-text screening by a pair of screeners to ensure they met the a priori eligibility criteria. In addition, a hand search of review references from potentially relevant studies and an independent search on PubMed were conducted to complement our literature search. No specific limitations were placed on follow-up period or study sample cohorts considered.
Results
The review conducted in OvidSP MEDLINE(R) and Embase yielded a total of 165 potentially relevant papers based on the specified search strategy. In all, 108 articles were excluded based on the a priori eligibility criteria, and 57 articles underwent paired full-text screening to yield a total of 38 articles. Additional sources were identified following a hand search of references and independent PubMed search and used to supplement the findings of the search (Figure 2).
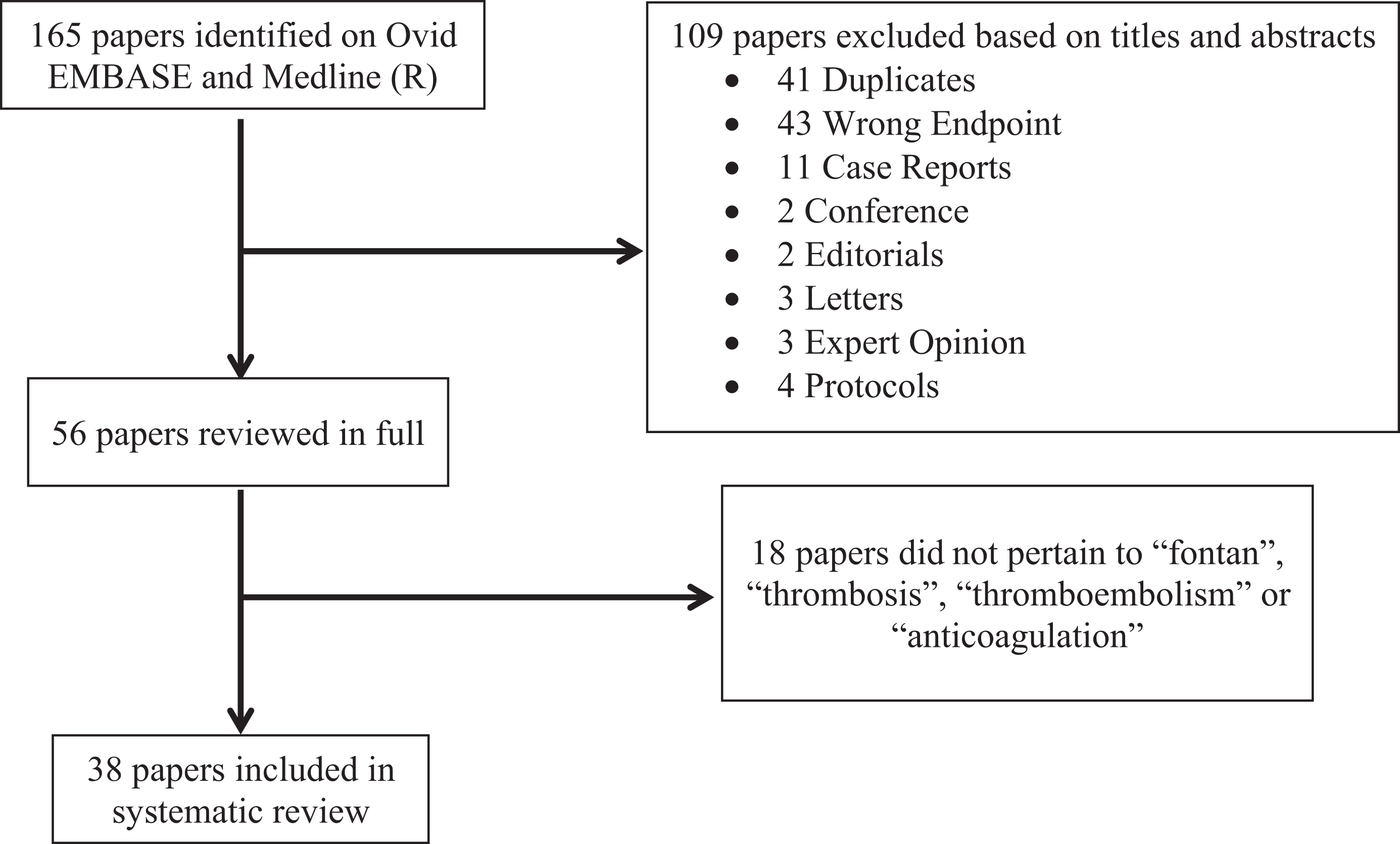
Summary of literature search.
Incidence of Thrombosis and Thromboembolism in the Fontan Circulation
Incidences of thrombosis in the Fontan circulation have been reported as high as 33% in past literature. 7 In a prospective, multicentered, randomized controlled trial assessing primary thromboprophylaxis following the completion of the Fontan procedure, Monagle et al indicated a cumulative thrombosis rate of 23% within a 2-year period and reported a symptomatic thrombosis event rate of 8% based on clinical checkups at 5 time points in conjunction with routine checkups. 8 The systemic venous atrium (48%) and pulmonary venous chamber (44%) have been noted as common sites of intracardiac thrombi formation over 22 months of follow-up postsurgery. 9 A secondary analysis by McCrindle et al on an international multicenter randomized controlled trial in 111 patients identified a time-related freedom from 69% thrombosis rate 2.5 years following randomization and a 28% incidence of clinically detectable thrombosis. 10
Studies indicate thrombus formation is most significant immediately following the procedure 10 and has been noted to peak within the first postoperative year in particular. 11 Several authors have also observed a plateau in thrombus formation after 3.5 years, followed by a second peak after 10 years. 12,13 The authors speculate that differences in reported thrombosis are largely influenced by different degrees of diagnosis of thrombus formation over time; a large decline following the first few postoperative years can potentially account for some of the variance in long-term follow-up data. In addition to a high clinical event rate, silent thrombosis events have also been discussed in several studies. The majority of past literature, however, has not considered asymptomatic cases. 14 –16 Consequently, total cardiac thrombus incidence remains undetermined. 11
Incidence of thromboembolism in the Fontan circulation has been reported to range from 3% to 20%. 14,15,17 Systemic venous and arterial thromboembolic complications have been associated with mortality rates of 25% in pediatric patients and 38% in adult patients in the literature, 18,19 indicating an increased prevalence observed with longer follow-up. Although the majority of studies identify that the incidence of thrombosis is highest within the first year following operation, strokes have been reported up to 9 years postoperation, with an incidence ranging from 3% to 19%. 18 Cheung et al identified a thromboembolic complication prevalence of 4.5% in their cohort between 0.14 and 7.7 years postoperatively, or 0.74% per patient-year. 20 A thromboembolic event incidence of 8.8% was also reported by Coon and colleagues in their study involving 592 patients. 9 Kaulitz et al found differing rates of systemic venous (5.6%) and arterial emboli (1.4%) resulting in strokes within 91.1 ± 43.9 months of follow-up following total cavopulmonary anastomosis. The study also identified a thrombotic event incidence of 7% among 142 patients with prophylaxis administration. 11 However, the study did not distinguish between thrombus formation and thromboembolic events; this inconsistency makes it difficult to compare findings across such studies. Seipelt and colleagues reported a thromboembolic event rate of 3.3 per 100 patient-years in their study and an incidence of 15.3% in its analysis of 85 patients over a mean follow-up period of 5.7 ± 3.5 years. The study reported a high incidence of thromboembolic events, with peaks after 1 and 10 years postoperatively. 21 Therefore, it is clear that there is significant variance in thromboembolic complication incidences from study to study.
Table 1 summarizes the primary study incidences of thrombosis, thromboembolism, early and late mortality, and additional data on thromboprophylactic intervention and follow-up period from a short listed set of literature based on the search conducted and the references manually identified from reviews included in the study.
Summary of the Review’s Findings.
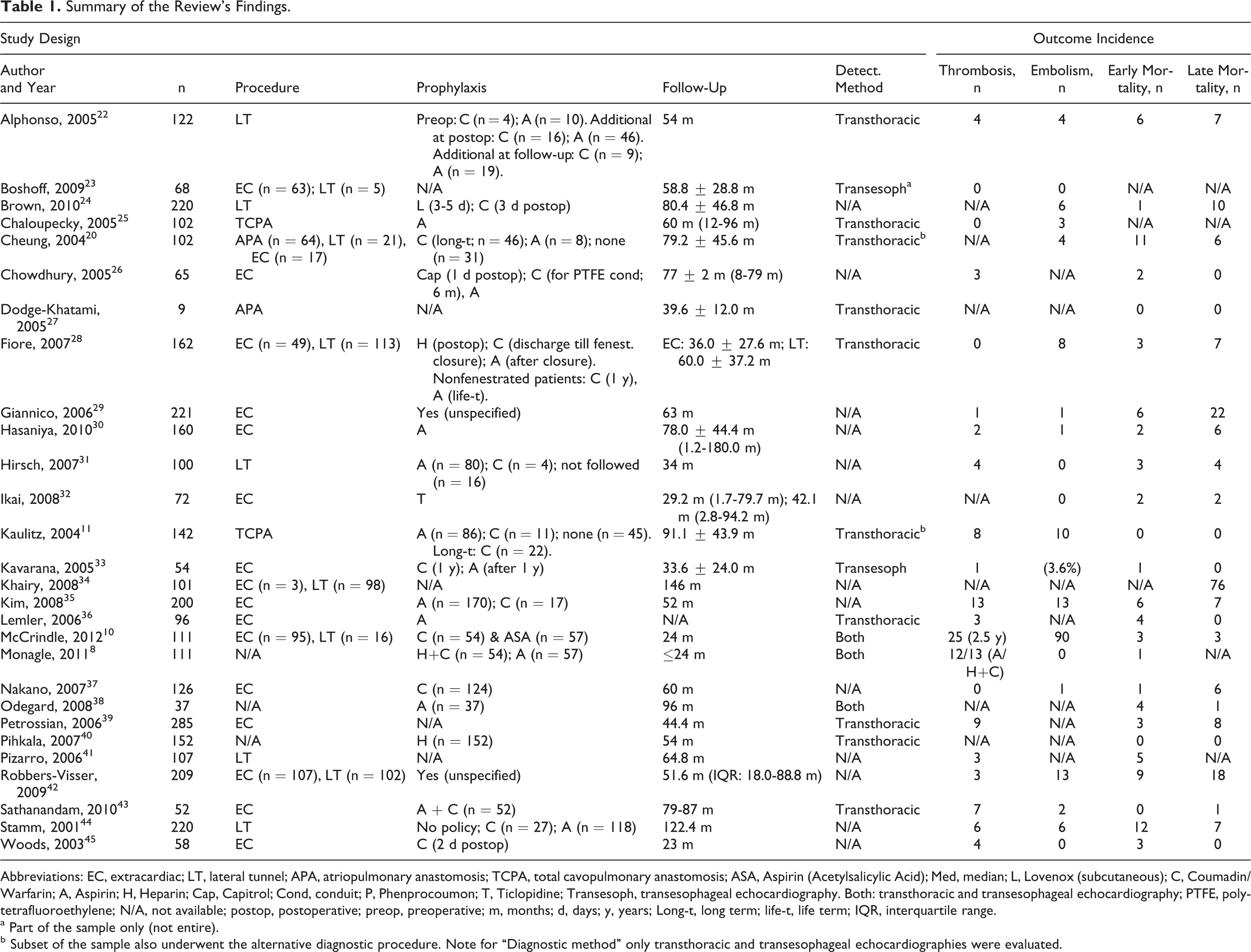
Abbreviations: EC, extracardiac; LT, lateral tunnel; APA, atriopulmonary anastomosis; TCPA, total cavopulmonary anastomosis; ASA, Aspirin (Acetylsalicylic Acid); Med, median; L, Lovenox (subcutaneous); C, Coumadin/Warfarin; A, Aspirin; H, Heparin; Cap, Capitrol; Cond, conduit; P, Phenprocoumon; T, Ticlopidine; Transesoph, transesophageal echocardiography. Both: transthoracic and transesophageal echocardiography; PTFE, polytetrafluoroethylene; N/A, not available; postop, postoperative; preop, preoperative; m, months; d, days; y, years; Long-t, long term; life-t, life term; IQR, interquartile range.
a Part of the sample only (not entire).
b Subset of the sample also underwent the alternative diagnostic procedure. Note for “Diagnostic method” only transthoracic and transesophageal echocardiographies were evaluated.
Surgical Procedures and Related Risk Factors
The 3 primary Fontan variants are associated with different adverse events and postoperative complication risks. Reported thrombotic and thromboembolic risks associated with the RA-PA connection, LTF, and ECF are discussed subsequently.
The RA-PA Fontan procedure
The RA-PA Fontan surgery directly connects the RA to the PA lying adjacent to it. Khairy et al study involving 135 patients with RA-PA procedure noted that while cardiac transplantation-free survival was similar between RA-PA Fontan patients and other variants, the procedure was an independent predictor of perioperative mortality, with a crude rate of 27.4%. 34 Pulmonary embolism, thrombus presence within the Fontan circuit, and lack of warfarin and aspirin prophylaxis were noted as predictors of all-cause mortality or transplantation in perioperative survivors, and the latter 2 were highly associated with thromboembolic death. 34 Importantly, thromboembolism has been reported among the 3 most common causes of death in RA-PA patients. 34 As this procedure involves turbulent blood flow from a large RA that delivers blood directly to the PA, the authors speculate other Fontan variants involve improved cardiac efficiency, a primary reason for the reduction in the use of this procedure and the shift in palliative practice over time.
The LTF procedure
The LTF operation was introduced by de Leval and colleagues to address the increased risk of thrombus formation and other complications as a result of the turbulent blood flow in RA-PA connection. The operation traditionally follows the bidirectional Glenn shunt or hemi-Fontan procedures and involves the connection of the IVC to the PA, with the placement of an intra-atrial baffle that may or may not be fenestrated. 46 This procedure has been noted to be more effective than the aforementioned RA-PA direct connection. Brown et al reported a thromboembolic event rate of 3%, with occurrences in 6 patients in their study of 219 patients who underwent fenestrated or nonfenestrated LTF and were followed up for an average of 6.7 ± 3.9 years. The study also reports a combined early and late mortality of 5% postoperatively, and a significant increase in thromboembolic complication frequency among patients undergoing fenestrated LTF procedures (22%, or 4 of 18 patients) as opposed to nonfenestrated LTF procedures (1%, or 2 of 201 patients). 24 Stamm et al assessed 220 patients undergoing the LTF procedure and described excellent long-term survival and freedom from failure, reporting 0.67 cases per 100 patient-years, in which thrombotic and thromboembolic complications were not noted as the primary cause relative to complications such as ventricular dysfunction. 47 The authors speculate this relatively low rate of thromboembolic complications and mortality to be associated with improvements in the hemodynamics of the circuit in comparison to the RA-PA connection, resulting in its increased use in modern practice.
The ECF procedure
The ECF procedure was developed in response to a concern over systemic venous hypertension and arrhythmia-related complications associated with the LTF procedure. The prosthetic extracardiac conduit was introduced in 1990 in an attempt to decrease the incidence of late atrial arrhythmias postoperatively. The ECF procedure involves anastomosing the IVC to the PA via a polytetrafluoroethylene tube or an alternate prosthetic material located outside the heart. 48 Despite their worldwide use, prosthetic conduits have been associated with an increased risk of stenosis and incidence of obstruction, primarily as a result of thrombosis among other complications such as fibrosis and calcification. 15,49 These risks have not been reported universally across recent literature, with several studies indicating a minimal risk of thrombotic and other obstruction-related complications with the use of the ECF. A study by Kim et al with 200 patients undergoing the ECF reported a 10-year survival rate of 92.4% ± 2.1%, thromboembolic events in 6.5% of the patient population, of which 84.6% were identified in the first year following the procedure. Furthermore, the study reported a 92.9% ± 1.9% Kaplan-Meier estimate of freedom from thromboembolism and low rates of cavopulmonary obstruction, thromboembolism, and late death. 35 Based on the finding that the risk of thromboembolism incidence was comparable to past studies despite the use of minimal anticoagulation with aspirin and the use of an extracardiac conduit, Kim et al also inferred that thromboembolism risk might be more attributable to suboptimal hemodynamics or underlying coagulopathy over conduit use. 35 These inferences of low complication and thromboembolic rates are supported by Nakano et al study involving 96.4 ± 23.0 months of follow-up for 126 ECF patients, where actuarial survival was noted to be 95.2% at 5 years and 93.6% at 10 years of follow-up. Late complications were seen in 7 patients, which included bleeding complications (n = 1), protein-losing enteropathy (n = 2), and thromboembolism (n = 1). 37 This indicates that suboptimal hemodynamics and coagulation abnormalities might be comparable to thromboembolic risk with the prosthetic conduit and might be a risk factor for the latter as well. No conduit stenosis or thrombosis were reported in this study similar to other literature considered, and freedom from Fontan-related events were reported as 88.8% at 5 years and 84.3% at 10 years postoperatively. 37
Pedicled pericardial tissue has been proposed as a potential conduit material in the past based on its nonimmunogenic and nonthrombogenic properties as well as its ability to adjust in shape based on relative flow volume. 50 Despite the risks of adverse events such as conduit stenosis, which have been discussed and reported in line with several studies, 26,51 , the use of this autologous material has been supported in the literature as well, with reports of minimal occurrence of thrombotic, thromboembolic, and stenotic complications. 30 Rosenthal et al demonstrated a difference between synthetic conduits and homografts or pericardial conduits, reporting no thrombi in the latter compared to 35% of patients developing thrombi in the former. 14 The findings of several of these studies are indicative of potential benefits to the use of autologous material in the ECF procedure as well as in other Fontan variants and might warrant greater attention in future surgical practice.
Comparison of Complications and Outcomes Between Fontan Variants
The LTF and ECF procedures, associated complications, and thrombotic risks have been evaluated and compared in several studies. Fiore et al study, with 49 patients undergoing the ECF and 113 patients undergoing the LTF procedure, reported an overall operative mortality of 1.8%, with 1 event in ECF patients versus 2 in LTF patients (P = ns). No significant difference in recognized thrombosis in the Fontan pathway (n = 0) and actuarial survival at 5 years (ECF 90% and LTF 95%) was observed, but a greater number of neurologic events with a thromboembolic origin were reported in the ECF patient group (6 events of a total of 8). 28 A similar study, including 209 patients after staged total cavopulmonary anastomosis (102 LTF and 107 ECF) with a median follow-up period of 4.3 years, reported a low incidence of thromboembolic events with no difference between the ECF and LTF groups, irrespective of the prophylaxis administered. 42 While thromboembolic complications were relatively low in both patient cohorts and freedom from late reoperations and Fontan failure at 6 years of follow-up, the study identified a notably lower incidence of arrhythmia in ECF patients. 42 Both studies discuss the presence of different risk factors associated with both procedures. 28,42 Coon et al study of LTF (n = 480), RA-PA Fontan (n = 73), and ECF (n = 32) patients reported 36 detected cases of thrombosis in the former cohort, 12 in the RA-PA connection and 1 in the latter. A slightly higher freedom from thrombosis at 1, 3, and 8 years postoperatively was observed in LTF over RA-PA patients; however, this difference was statistically insignificant. 9 Taken together, these studies demonstrate that no variant has been identified as standard in terms of their postoperative complication risks, and significant variance exists in past findings; however, the literature reviewed tends to favor the LTF procedure, particularly in terms of thrombotic 9 and thromboembolic outcomes. 28
Additional Risk Factors Associated With Postoperative Complications
The prevalence of thrombus formation in the Fontan circulation varies with the anticoagulation treatment provided, conduit length and size match with IVC, 32,52,53 conduit material and related growth potential, Fontan pathway obstruction and thromboembolic risks, 21,43,54 –57 the patient population, 20 and the presence of related risk factors. These risk factors include passive blood flow, atrial arrhythmias, the presence of a thrombogenic “blind” PA stump or cul-de-sacs, 11 prolonged postoperative immobilization, altered resting venous tone, coagulation factor abnormalities, 38 pulmonary atresia with intact ventricular septum, PA distortion, higher preoperative unconjugated bilirubin, and lower postoperative fraction of inspired oxygen. 10 Previous studies have identified unclear relationships between thrombotic risk and elapsed postoperative time and age, 43 low birth weight, 32 ethnicity, 20 and related environmental influences such as the anticoagulant and activated protein C resistance effects of certain dietary consumption patterns. 20
Furthermore, hepatic abnormalities and protein-losing enteropathy have been associated with coagulation imbalances. Chronic systemic venous hypertension in Fontan patients 25 has been proposed as a plausible hypothesis for abnormal liver protein synthesis, hypercoagulability, and elevated thrombosis and thromboembolic risk, with substantial differences in reported concentrations of factors II, V, VII, VIII, and X; plasminogen; antithrombin; and proteins C and S. 25,58 Moreover, protein-losing enteropathy is a condition resulting in the loss of serum proteins into the gastrointestinal tract, and subsequent impairment of the clotting cascade 59 has been documented in up to 13.4% of Fontan patients postoperatively. 60 –62 The condition is associated with a significantly high mortality rate, which has been estimated to range between 56% and 67%. 60,61
Discussion
This review of existing literature on thrombotic and thromboembolic complications associated with the Fontan procedure identified (1) a variance in complication incidence across different institutions and observational study results, typically ranging up to 33% for thrombotic end points and 3% to 20% for embolic end points; (2) a relative decline in RA-PA Fontan variant usage attributed to undesirable hemodynamic outcomes beyond thrombotic end points; (3) the lack of a “gold standard” variant of the Fontan surgery, with significant variation in the thrombotic complications associated with each per previous literature; (4) the potential for improved patient outcomes and reductions in thrombotic complications with the use of fenestration in the LTF or the use of autologous conduit material; (5) and predisposition to postoperative thrombotic complications with hemodynamic abnormalities, coagulation imbalances, and general patient characteristics.
This review points to a high inherent risk of thrombosis and thromboembolism in almost all variants of Fontan patients, particularly in their first year or 2 years following repair. 8,11 The decline in RA-PA connection use over time despite reasonable short-term thrombotic and thromboembolic findings is indicative of the interplay between complications involved with the Fontan procedure. Heart failure, arrhythmia, inefficient hemodynamics, turbulent blood flow, and other factors associated with sudden death are instances of complications that have taken precedence in surgical decision making. 34,63 The comparison between ECF and LTF did not identify a significant difference in thrombotic outcomes across the studies conducted, though insignificant benefits were generally identified with the latter procedure. The authors speculate the variance in results is likely to be attributed to the degree of surgical expertise, institutional practices, and patient samples being treated, among other factors. Furthermore, the role of fenestrations in reducing immediate postoperative complications has been identified in several studies. These fenestrations are typically closed at approximately 1 year postoperatively, which results in progressive cyanosis, polycythemia, and paradoxical embolism risk 64 –66 ; these aspects have not been explored in detail in recent literature. Lastly, there is significant misalignment between study findings and surgical practices in terms of autologous conduit material use, despite numerous benefits reported compared to synthetic conduit material use. 14,30,50 Autologous material seems to be rarely used in any variant of the procedure.
This review summarizes a large and disparate body of recent literature on a major complication associated with this pediatric palliative procedure. The inclusion of both primary studies and secondary literature sources strengthens the scope and comprehensiveness of the review. This review was able to identify several studies comparing multiple variants of the procedure, allowing for a direct comparison between the most common forms of the procedure in modern practice. Moreover, the inclusion of diverse patient cohorts comprising different age groups, weights, and ethnicities from institutions across numerous countries in this review makes the findings pragmatic. Unlike the majority of currently available recent literature on the topic, this review also incorporates a thromboprophylactic algorithm based on the findings of the anticoagulation practices and associated outcomes in the primary literature considered, with consultation of evidence-based clinical practice guidelines. 67
Several studies broadly reported both thromboembolism and thrombosis events as “thrombotic events” or “thrombosis” in general, while numerous others differentiated between the 2 complications; this inconsistency in primary literature reporting limits the clarity of the review’s findings. The lack of comparative primary studies assessing these 2 procedures also limits the applicability of the review’s findings in a comparative analysis of 2 or more variants of the procedure and can be largely attributed to the use of only 1 variant at a particular point in time in the surgical practices of most institutions. Furthermore, while thromboembolic complications are generally clinically significant, it is important to note that thrombotic complications are often asymptomatic, or “silent,” in nature. Transesophageal echocardiograms (TEEs) are often used to detect the presence of thrombi as compared to transthoracic echocardiograms due to its higher sensitivity in detecting clinically silent thrombi. 10 While many studies in this review have used TEE as a routine test, several have also used transthoracic echocardiography or have irregularly used TEE. Only 21% of the literature summarized in Table 1 explicitly reported the use of TEE, highlighting the underuse of this highly sensitive surveillance method for the detection of postoperative complications. This difference in detection techniques across studies makes it difficult to generalize an overall incidence of postoperative thrombus formation or thromboembolism for the Fontan procedure. As a result, total cardiac thrombi incidence, both symptomatic and clinically silent events, remains undetermined. 11
Conclusion
Based on the findings of this review, the risk of thrombosis and thromboembolic events in Fontan circulation will continue to remain high until further innovations in technique or management arrive. Until then, judicious but more aggressive thromboprophylactic therapy is justified, particularly because once thromboembolic complications have set in, mortality is as high as 38% even with aggressive intervention. 19 Moreover, the choice of anticoagulation therapy varies across institutions as the latest clinical practice guidelines for patients with Fontan circulation remain weak. Hence, all Fontan patients should have aggressive anticoagulation treatment in their first year postsurgery along with ongoing maintenance thereafter. We hereby propose an algorithm for administering consistent thromboprophylactic anticoagulation to Fontan patients until stronger evidence becomes available for clinical practice (refer to Figure 3). This conservative algorithm recommends standardized prophylaxis for the first postoperative year, followed by individualized therapy based on international normalized ratio and thrombosis-related risk factors.
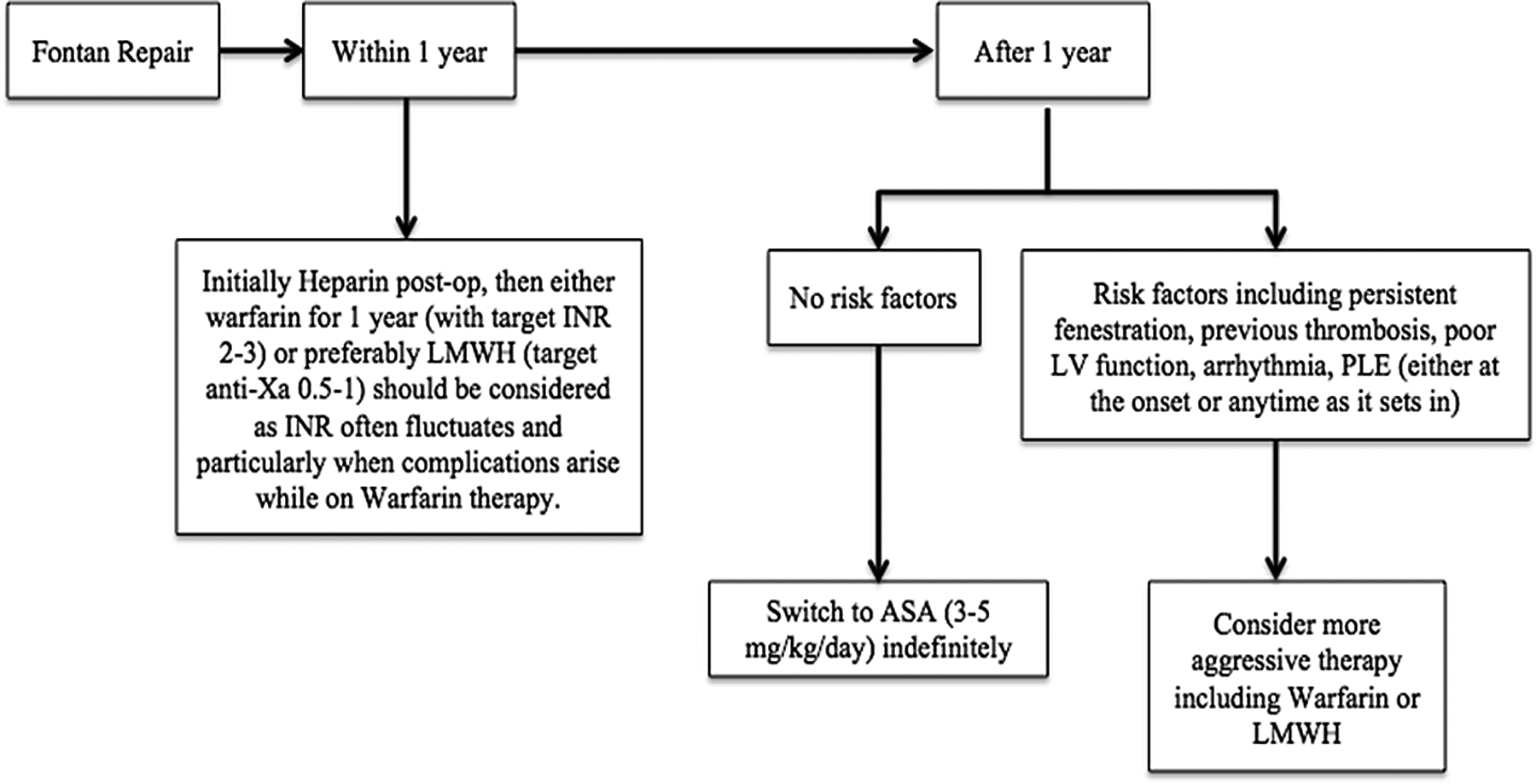
Proposed management algorithm.
Future directions include developments in pharmaceuticals for the management of abnormal hemodynamic complications, innovations in surgical techniques, and the possibility of personalized medical therapy assisted by single-nucleotide polymorphisms and other genetic screening tests to identify high-risk populations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
