Abstract
Background:
Venous thromboembolism (VTE) is a common complication that manifests during and/or after hospitalization, as well as postsurgeries including orthopedic surgeries. Edoxaban is a new oral direct factor Xa inhibitor that has been recently approved for treating VTE in patients who have already been treated with a parenteral anticoagulant and for the prevention of stroke and non-central nervous system systemic embolism in patients with nonvalvular atrial fibrillation.
Objectives:
The purpose of this systematic review was to evaluate the safety and efficacy of edoxaban for VTE prophylaxis after lower limb orthopedic surgery.
Materials and Methods:
A comprehensive search was conducted in Google Scholar, PubMed, MEDLINE, and ScienceDirect to identify potential records, then titles, abstracts, and full texts were screened using the inclusion criteria to filter out irrelevant studies. Moreover, the data extraction and quality assessment were undertaken using standardized tools, and the results were narratively synthesized and presented in tables.
Results:
Six studies were included in this systematic review after screening 2989 records. The majority of studies demonstrated a statistically significant reduction in VTE events in the edoxaban group(s) compared to the dalteparin, placebo, or enoxaparin groups (P < .05). The differences in VTE cases in some studies reached to approximately 50% favoring edoxaban 30 mg over the comparator (P < .05). However, other studies uncovered a statistically insignificant difference between edoxaban and the comparator “enoxaparin” when used for VTE prophylaxis (P > .05). On the other hand, although edoxaban found to cause more bleeding, the differences between edoxaban and the comparator are statistically insignificant (P > .05).
Conclusion:
This study helped to amalgamate evidence with regard to the use of edoxaban for VTE prophylaxis post-lower limb orthopedic surgery. In line with the results of the reviewed studies, edoxaban seems to be highly effective in reducing VTE post-lower limb orthopedic surgery.
Introduction
Venous thromboembolism (VTE) might manifest as deep venous thrombosis (DVT) and/or pulmonary embolism (PE), where DVT is defined as the formation of blood clot in the veins of the lower extremities, while PE occurs when an embolus travels to block the arteries of the lung. 1,2 In fact, VTE is considered to be the commonest preventable complication seen during and/or after hospitalization, as VTE is documented in approximately 10 million annual cases worldwide. 2,3 This information is not new, as in the 1980s, VTE was considered to be one of the major preventable causes of death in the United States. 4 More recent data retrieved from the MarketScan Commercial and Medicare databases shed light on the annual prevalence of VTE in the United States in 2006, which was around 422 cases per 100 000. 5 The consequences of VTE are rather detrimental and create financial burden, where PE expenses for 190 patients admitted in 2008 reached up to approximately US$1.9 million. 6 Hence, VTE prevention became a quality indicator that gets assessed and investigated frequently. 1,7 Although there are plenty of VTE prophylaxis guidelines such as the 2016 American College of Chest Physicians (ACCP) guidelines 8 and the National Institute for Health and Care Excellence guidelines, 9 some studies unveiled that adherence to guidelines is yet to be achieved. 1,10 -14
The risk of developing VTE among surgical patients varies and depends on a number of factors including the type of procedure, type and duration of anesthesia, duration of immobilization, and other patient-related factors. 14 -19 More specifically, the risk of developing VTE post-orthopedic surgery can range between 40% and 60% in the absence of prophylactic measures, therefore, guidelines continue to emphasize the importance of employing prophylactic measures. 19 As per the guidelines of the ACCP, prevention of VTE in orthopedic surgery patients is achieved by the use of low-molecular-weight heparin, fondaparinux, apixaban, dabigatran, rivaroxaban, low-dose unfractionated heparin, adjusted-dose vitamin K antagonist, aspirin (all grade 1B), or an intermittent pneumatic compression device (IPCD; grade 1C). 20
Advancements in VTE prophylaxis and the introduction of new medications stem from the desire of researchers and scientists to successfully prevent and treat VTE (figure 1). 20 -22 Edoxaban (Savaysa) was the latest to be approved by the Food and Drug Administration for 2 indications:

Edoxaban is an oral anticoagulant that is administered once daily, as presented in Table 1, 23 and produces its effect by directly blocking factor Xa. 21 Additionally, it demonstrates linear pharmacokinetics (PK), rapid onset of action, and a good oral bioavailability. 21 Finally, some studies illuminate the plausibility of using edoxaban for VTE prophylaxis post-orthopedic surgeries. Therefore, the overarching aim of this systematic review is to evaluate the safety and efficacy of edoxaban for VTE prophylaxis after lower limb orthopedic surgery.
Dosage and Indication. 23

Abbreviations: CrCl, creatinine clearance; DVT, deep venous thrombosis; NVAF, nonvalvular atrial fibrillation; PE, pulmonary embolism; P-gp, P-glycoprotein.
Methods
Search Strategy
A systematic review of the literature was performed based on the Preferred Reporting Items for Systematic Reviews guidelines. 24 Four electronic databases including Google Scholar, Medline, PubMed, and ScienceDirect were searched using terms such as “Edoxaban AND VTE prevention,” “edoxaban post-orthopedic surgery,” and “edoxaban for VTE” to collate relevant studies.
Study Selection
We reviewed papers of studies that met the following inclusion criteria and were available as a full text: The study design was a randomized controlled trial. The study aim was to evaluate the safety and efficacy of edoxaban when used for VTE prophylaxis. The study population included patients who underwent hip fracture surgery, total knee replacement surgery, or total hip replacement surgery. Edoxaban and comparator(s) were prescribed for VTE prevention post-orthopedic surgery. Published in English and between the year 2010 and 2015.
We excluded papers according to the following: Study design was not a randomized controlled trial. Study was not evaluating the safety and efficacy of edoxaban when used for VTE prophylaxis. Edoxaban was not prescribed for VTE prophylaxis post-orthopedic surgery. Other agent than edoxaban was prescribed for VTE prophylaxis. Articles we were not able to source the full text.
Quality Assessment
Jadad scale was utilized to assess the qualities of the randomized controlled trials included in this systematic review. 25,26 A score of ≤2 indicates a low-quality design, while a score of ≥3 indicates a high-quality design. 25,26 These scores were calculated initially by 2 researchers, which then were validated by a third researcher to ensure reliability.
Review Procedure
Four researchers were involved in the process of data extraction, and the extracted data were reviewed afterward by a fifth researcher.
Data Extraction and Synthesis
Data were extracted from studies using a standardized document, where it was oriented to obtain information such as author, year of publication, country, aim, number of patients, dose, study design and duration, comparator(s), and main findings. Moreover, the results of the studies were synthesized narratively, with all results presented in a tabulation format.
Results
Identification of Studies
The searching process went through a series of phases as displayed in Figure 2. In the first phase, specific terms were used to locate studies, and this yielded in retrieving 2700 studies from Google Scholar, 57 studies from MEDLINE, 61 studies from PubMed, and 171 studies from ScienceDirect, as presented in Figure 2. In phase 2, the aim was to screen titles and then use the inclusion criteria to filter out irrelevant studies either via abstracts or full texts, and this has resulted in identifying 6 studies to be incorporated in this systematic review.

Preferred Reporting Items for Systematic Reviews (PRISMA) flowchart.
All the studies included were exploring the efficacy and safety of edoxaban post-orthopedic surgery. 27 -32 The efficacy parameter explored here was the total VTE prophylaxis, while bleeding/all bleeding were the safety parameters. 27 -32 To further elaborate, the efficacy end point “VTE” includes symptomatic DVT, asymptomatic DVT, and symptomatic PE, whereas the safety end point “bleeding or all bleeding” includes major bleeding, clinically relevant nonmajor bleeding, and minor bleeding. 27 -32 Some of these studies used different doses of edoxaban, whereas other studies were testing a single dose of edoxaban “30 mg.” 27 -32 Besides, the Jadad score of the vast majority of these studies fell within the range of 3 to 5, except 2 studies that got a lower score “2.” 27 -32 Key data and findings with regard to the efficacy and safety parameters are summarized in Table 2. 27 -32
Results Gleaned From RCTs.
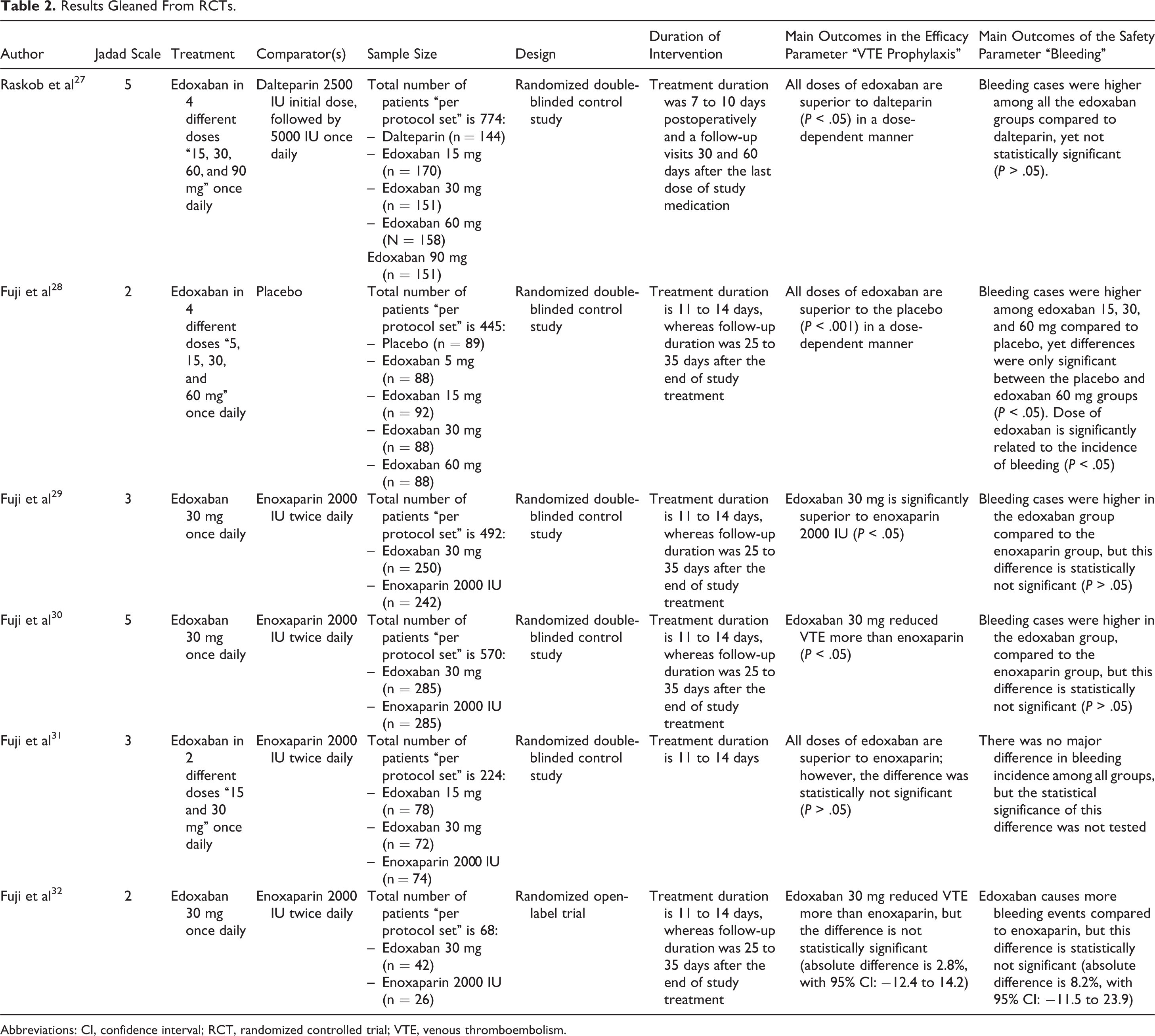
Abbreviations: CI, confidence interval; RCT, randomized controlled trial; VTE, venous thromboembolism.
Efficacy
Raskob and colleagues studied 774 participants who were randomly assigned to receive either dalteparin 5000 IU once daily or edoxaban 15 mg, 30 mg, 60 mg, or 90 mg once daily. 27 Efficacy and safety end points were monitored for 7 to 10 days after the surgical intervention. 27 When comparing edoxaban “in different doses” to dalteparin, it has been found that edoxaban is significantly superior to dalteparin, where the incidence of VTE were 43.8%, 28.2%, 21.2%, 15.2%, and 10.6% in the edoxaban 15 mg, 30 mg, 60 mg, 90 mg, and dalteparin 5000 IU groups, respectively, as seen in Table 2 (P < .05). 27 Furthermore, a dose-dependent VTE prophylactic effect was observed in the edoxaban groups, hence increasing the dose provides a better protection against VTE. 27 Another study conducted by Fuji and colleagues incorporated 445 participants who were randomly assigned to receive either a placebo or exodaban 5 mg, 15 mg, 30 mg, or 60 mg. 28 This study uncovered the ability of edoxaban “in various doses” to provide a significantly better prevention against VTE compared to placebo (P < .001), as seen in Table 2. 28 Where, the incidences of VTE were 48.3%, 29.5%, 26.1%, 12.5%, and 9.1% in the edoxaban 5 mg, 15 mg, 30 mg, 60 mg once daily and placebo groups, respectively. 28 Moreover, the VTE prophylactic effect was found to be dose dependent. 28
On the other hand, when enoxaparin was compared to edoxaban, the VTE incidence was significantly lower among patients who received edoxaban 30 mg once daily compared to patients who received 2000 IU of enoxaparin twice daily, “equivalent to 20 mg twice daily,” with a difference of 4.5% (P < .05). 29 These findings go in consistent with another study that was done by Fuji et al, which revealed the existence of statistically significant difference between edoxaban and enoxaparin in terms of VTE prevention, as the number of VTE cases seen in the edoxaban 30 mg group was 7.4% compared to 13.9% in the enoxaparin group (P < .05). 30 On the other hand, Fuji and colleagues conducted another study on 224 patients who were randomly assigned to receive either edoxaban 15 or 30 mg once daily or enoxaparin 2000 IU twice daily. 31 This study brought to light the ability of all agents used to reduce VTE, yet the differences among groups were statistically insignificant (P > .05). 31 Besides, a randomized open-label study, which included 68 patients and was conducted to compare edoxaban 30 mg once daily to enoxaparin 2000 IU twice daily, showed that enoxaparin is more effective than edoxaban in preventing VTE (absolute difference is 2.8%, with 95% confidence interval [CI]: −12.4 to 14.2). 32 However, the differences were found to be statistically insignificant. 32
Safety
When the safety profile of dalteparin was compared to edoxaban “in different doses,” bleeding was observed more among the edoxaban groups compared to the dalteparin, as 0.6%, 2.1%, 1.8%, 4.9%, and 4.0% cases of bleeding were seen in the dalteparin and edoxaban 15 mg, 30 mg, 60 mg, and 90 mg groups, respectively. 27 However, the differences were not statistically significant (P > .05). 27 Furthermore, the bleeding incidence was not related to the doses of edoxaban, which confirms the absence of a dose-dependent relationship with regard to bleeding incidence. 27 In addition, when the safety profile of edoxaban “in different doses” was compared to placebo, it has been found that bleeding incidence occurred more in the edoxaban 15 mg, 30 mg, and 60 mg groups compared to the placebo, where 6.9%, 10.4%, 10.7%, and 17.0% cases were documented in the placebo, edoxaban 15 mg, 30 mg, and 60 mg, respectively. 28 However, placebo was found to be associated with more bleeding events (6.9%) compared to edoxaban 5 mg (5.8%). 28 Nevertheless, all these findings were statistically insignificant, except the differences in bleeding incidence between the placebo and the exodaban 60 mg groups (P < .05). 28 Additionally, the incidence of bleeding increased by increasing the dose of edoxaban, therefore indicating a dose-dependent relationship (P < .05). 28 When edoxaban was compared to enoxaparin with regard to the bleeding risks, edoxaban was found to cause more bleeding, yet these differences were statistically not significant (P > .05), as seen in Table 2. 29 -32
Discussion
It is essential to have a rudimentary understanding about the risks of VTE when undergoing major orthopedic surgeries. 33,34 For that reason, the ACCP advised for providing a sufficient prophylaxis against VTE. 34 Although guidelines are available, adherence to these guidelines is still considered to be an insurmountable challenge. 1,35 Despite the fact that the majority of studies included in this systematic review brought to light the effectiveness of edoxaban in preventing VTE post-lower limb orthopedic surgery in a statistically significant manner, there are certain aspects that need careful attention and which will be discussed in the next paragraph.
Efficacy
Two studies in this systematic review were conducted on patients who underwent knee surgeries, where findings postulated that VTE is better prevented with the use of edoxaban compared to placebo or enoxaparin. 28,30 Indeed, the difference in findings indicates a significant noninferiority and superiority of edoxaban (P < .05). 28,30 Besides, the anti-Xa effect provided by edoxaban was found to be dose dependent, therefore giving more flexibility when changing doses. 28 These findings go in parallel with a study that was conducted by Hokusai VTE Investigators, where edoxaban was found to be noninferior to warfarin in the treatment of symptomatic VTE. 36 However, in these 2 studies that were done on patients who underwent knee surgeries, a nonpharmacological prophylactic measure “IPCD” was utilized during the study, thus intensifying the VTE prophylactic effect. 28,30 Unfortunately, these salient nonpharmacological interventions were not incorporated in the analysis, thus impinge upon the genuineness of these findings. 28,30 In fact, evidence illustrating the effectiveness of pneumatic compression is clear, as studies found that when adding it to a pharmacological prophylactic agent, it can reduce PE by 1% to 3% and DVT by 1% to 4%. 34,35
The remaining 4 studies included in this systematic review investigated patients who underwent hip surgeries. 27,29,31,32 Two studies illustrated that the reduction of VTE risk in the edoxaban group(s) was significantly higher than the comparator(s) “dalteparin or enoxaparin.” 27,29 Additionally, patients in these 2 studies were not placed on IPCD, therefore reducing the impact of confounders on the findings. 27,29 Besides, in the study by Raskob et al (2010), the VTE prophylactic effect was found to be closely related to the dose of edoxaban, hence showing a dose dependent response.” 27 These findings are consistent with a number of studies that investigated the PK and pharmacodynamics profile of edoxaban to unleash the existence of a dose-dependent VTE prophylactic effect. 37,38 On the contrary, in the other 2 studies, the difference in VTE incidence between edoxaban and enoxaparin was found to be statistically insignificant. 31,32
Safety
When exploring the safety profile “bleeding” of agents utilized in these studies, data confirmed that these medications are considered to be well tolerated, as the differences in bleeding incidence were statistically insignificant. 27 -32 In the same vein, a study done by Hokusai-VTE Investigators, to compare the safety profile of edoxaban and warfarin, found that edoxaban was associated with less bleeding risks compared to warfarin when used in the treatment of symptomatic VTE. 36 Yet, since these studies were conducted postoperatively, expectations that these patients have been placed on pain management “nonsteroidal anti-inflammatory drugs (NSAIDs),” and these medications theoretically are expected to potentiate bleeding by blocking the production of thromboxane A2. 39 Unfortunately, this information was not addressed in some studies included in this systematic review, 28,30 and even if this information was shared, it was not part of the analysis. 29,32 Therefore, consensus regarding the bleeding incidence with edoxaban is fogged by the use of NSAIDs. 29,32
Pharmacokinetics
These studies were conducted in a population that is known to have a smaller body frame compared to Western populations, afflicting dose requirements. 27 -32 Therefore, the plausibility of funneling the results to a population that has a larger frame needs to be done with caution. 27 -32 In addition, since the creatinine clearance (CrCl) is crucial for dosing, all patients included in the randomized controlled trials reviewed here had a CrCl > 30 mL/minutes, and therefore, a dose of edoxaban ≥30 mg is considered to be suitable. 40
Limitations
Having a rudimentary understanding about the limitation of this design is imperative. Publication bias will always be a concern in systematic review as it might lead to false-positive overall conlusions. In fact, one of the major limitations of this systematic review is having a limited access to certain studies that were not purchased by both academic institutions. Furthermore, some studies might have still been in the publication process and not yet available on the web to encompass their findings in this review. With regard to the studies included in this systematic review, a number of limitations were encountered such as the small sample size 32 ; hence, the statistical comparison between groups might not be sufficiently powered. 32 Furthermore, the vast majority of these studies were sponsored by the manufacturer producing Savaysa, and this might afflict the authenticity of results.
Conclusion
In conclusion, the findings of this systematic review decipher the efficacy of edoxaban in preventing VTE post major orthopedic surgeries, specifically post-lower limb orthopedic surgeries. Besides, it is considered to be an attractive alternative due to the convenience of administering “oral” compared to parenteral anticoagulants. However, findings gleaned from this systematic review have to be used with caution due to the small frame of the investigated population and the use of NSAIDs and IPCD during the period of intervention. Also, findings amalgamated brought to light that although high doses of edoxaban are more effective in reducing VTE, the risks of bleedings get intensified. Therefore, when prescribing the dose, all parameters have to be taken into consideration such as the PK and the weight to ensure an effective and safe use of medication.
Footnotes
Author Contributions
All authors contributed to the study design. All authors were involved in the literature review and data extraction process. Besides, all were involved in the writing and revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
