Abstract
Objective:
To evaluate the performance of methylenetetrahydrofolate reductase (MTHFR) gene C677T polymorphism in predicting hyperhomocysteinemia (HHcy) in Chinese patients with hypertension.
Methods:
We measured plasma total homocysteine tHcy level and C677T genotype in 1058 Chinese patients with hypertension from 4 previous studies. We used 10, 15, and 20 μmol/L as cutoff values for the definition of mild, modest, and severe HHcy, respectively. Logistic models for HHcy were built from the study sample using the C677T genotype as well as age and gender as predictors. The receiver–operating characteristics of the models were evaluated.
Results:
Our major findings are that (1) C677T TT genotype is consistently associated with a higher tHcy across the 4 studies, with an increase in size ranging from 38% to 68% in the 4 studies and 51% overall. The C677T polymorphism independently explained about 14% of the total variance of the normalized tHcy. (2) The TT genotype is associated with a large increase in odds ratio (OR) for HHcy. Overall, the multivariate-adjusted ORs for the TT genotype are 3.9 (95% confidence interval [CI]: 2.4-6.4), 6.5 (95% CI: 4.0-10.6), and 17.9 (95% CI: 8.4-38.1) for mild, modest, and severe HHcy, respectively. (3) Overall, the predicting performance increased with HHcy severity, with sensitivity improving from 31.0% for mild HHcy to 70.3% for severe HHcy, and with specificity slightly decreasing from 85.4% to 80.3%. Inclusion of gender and age as predictors significantly improves the sensitivity, especially for predicting mild HHcy.
Conclusion:
With an excellent sensitivity and a modest specificity, C677T could be a useful screening marker for severe HHcy.
Introduction
Hyperhomocysteinemia (HHcy), characterized by an elevated plasma level of total homocysteine (tHcy), is linked to an increased risk of cardiovascular diseases, stroke, preeclampsia, and neurological disorders. 1 –3 According to a meta-analysis, the odds of stroke increase by 59% for every 5 μmol/L increase in tHcy. 4 Furthermore, there appears to be a synergic effect between HHcy and hypertension related to the increased risk of cardiovascular diseases and stroke. 5
Elevated tHcy level usually results from a deficiency in vitamin B, genetic variations, and significantly reduced renal function. Of the known genetic determinants of tHcy, C677T polymorphism of the methylenetetrahydrofolate reductase (MTHFR) gene is by far the most important and the best studied. 6 –8 A recently published meta-analysis of 2.7 million single-nucleotide polymorphisms genotyped from 10 genome-wide association studies showed that the MTHFR C677T is the most significant genetic variant associated with homocysteine (Hcy) levels among common variants at 13 loci (P-meta = 4.34 × 10E-104). 9 On average, the TT genotype of C677T is associated with a 40% increase in tHcy and a 26% increase in odds for stroke. 10,11 However, the effect size of such associations varies with population 10 and the extent to which MTHFR C677T can be used to predict HHcy, especially in Chinese patients with hypertension, remains to be examined.
In the present study, we studied the association between C677T polymorphism and tHcy in 1058 Chinese patients with hypertension and examined the performance of C677T at predicting 3 different degrees of HHcy. We also built a predictive model for HHcy using age, gender, and C677T genotype and evaluated the receiver–operating characteristic (ROC) of the model.
Materials and Methods
Study Population
In this study, we pooled 1058 patients with hypertension from 4 epidemiological studies and clinical trials on hypertension treatment previously conducted in China. Brief description of these 4 studies is given subsequently. 12 –15
The Antihypertensive and Homocysteine-lowering Trial (AHT): This was a trial of the maleate enalapril and folic acid tablet in patients with hypertension conducted in 6 major medical centers in China (clinicaltrials.gov identifier: NCT00520247). This study has been described elsewhere. 12 A total of 464 patients with complete C677T genotype and baseline phenotypic data were included in this analysis.
The Combined Amlodipine and Terazosin Trial (CATT): This was a trial to study the efficacy of the combination of amlodipine and terazosin on treatment of lower urinary tract symptoms in patients with hypertension (clinicaltrials.gov identifier: NCT00693199). The detailed description of this trial was published elsewhere. 13 A total of 355 men with Stage I or II hypertension were included in this analysis.
The Antihypertensive and Homocysteine-lowering Pharmacogenomics Study (AHPS): This was a pharmacogenetic study to investigate the effect of MTHFR C677 T polymorphism on the efficacy of a combination of folic acid and enalapril in patients with stage I or II hypertension. A total of 242 patients were selected according to the C677T genotypes such that the 3 genotype groups were of roughly equal size. 14
The Benazepril Pharmacogenomics Study (BPS): This was a pharmacogenetic study to investigate the effect of MTHFR C677T polymorphism on the efficacy of benazepril in patients with stage I or II hypertension. A total of 726 patients were enrolled, 15 of which 114 were from Anqing and were selected such that the 3 C677T genotype groups were of roughly equal size. These 114 patients were used in the current analysis.
The AHT was approved by institutional review boards at the participating medical institutions, including the First Hospital affiliated with Beijing University. The other 3 studies were approved by the institutional review board at the Institute of Biomedicine of Anhui Medical University. The purpose and procedures of the study were carefully explained to all participants, and written informed consent was obtained from all the participants.
Plasma tHcy measurement
Plasma samples were prepared from 2 mL of fasting vein blood with 2% EDTA anticoagulant by centrifugation within 1 hour of blood collection and stored at −80°C until assay. Plasma tHcy level was determined using a high-performance liquid chromatography (HPLC) method with fluorescence detection. 16
Methylenetetrahydrofolate reductase C677T genotyping
Genomic DNAs were isolated from leukocytes using a salt precipitation procedure. Methylenetetrahydrofolate reductase C677T polymorphism genotype was determined by a polymerase chain reaction (PCR)-restriction fragment length polymorphism method. Forward and reverse PCR primers were 5′-CTTTGAGGCTGACCTGAAGC-3′ and 5′-CTGGGAAGAACTCAGCGAAC-3′, respectively. DNA fragments were amplified by 35 cycles of denaturation at 94°C for 30 seconds, annealing at 65°C for 45 seconds, and extension at 72°C for 45 seconds. The PCR products were digested by Hinf I at 37°C for 1 hour and analyzed by 3% agarose gel electrophoresis. C and T alleles were determined by the presence of a signature band of 274 bp and 228 bp, respectively.
Statistical analysis
Epidata 3.1 Chinese version was used for database design, data entry, and data check. Mean ± standard deviation [SD] was calculated for continuous variables. Since tHcy distribution was significantly skewed, geometric mean and SD (ie, log transformed) were used for better normality conformation. In the AHT and CATT studies in which samples were not ascertained on genotype, genotype distribution was tested for Hardy-Weinberg equilibrium using a chi-square test. The effects of MTHFR C677T genotype on tHcy were estimated using linear regression models with log transformation of tHcy and with adjustment for age, sex, body mass index (BMI), and study (in pooled analysis). Three different tHcy cutoff values of 10, 15, and 20 μmol/L were used for defining mild, modest, and severe HHcy, respectively. The odds ratio (OR) of C677T TT genotype for HHcy was estimated for each separate study using logistic regression with adjustment for age, sex, and BMI. Logistic regression models for predicting HHcy with variables, such as age and sex, and C677 T genotype were built, and their ROCs were evaluated. Unless otherwise specified, all analyses were done using R packages, including “glm” for regression analyses and “lroc” for plotting of ROC curves.
Results
All patients from the 4 studies were patients with stage I or II hypertension. The basic characteristics of the 4 study populations were summarized in Table 1. Of the 4 populations, 3 were from rural areas in Anqing, Anhui Province of China while patients in the AHT were from 6 major cities, mostly in Northern China. The phenotypic characteristics of patients in the AHT differed markedly from the other 3 populations; for example, they were much more obese and had higher tHcy levels. Overall, 68.9%, 24.4%, and 11.2% of patients had mild, modest, and severe HHcy, respectively, with the highest rate observed in the AHT. There was also a big difference in MTHFR C677T genotype frequency. Samples were not ascertained on genotype for the AHT and CATT and C677 T genotype distributions were in Hardy-Weinberg equilibrium (P > .05). However, the TT genotype was much more common in the AHT group (25.4%) than that in the CATT (14.3%).
Characteristics of the 4 Studies Used in the Analysis.a
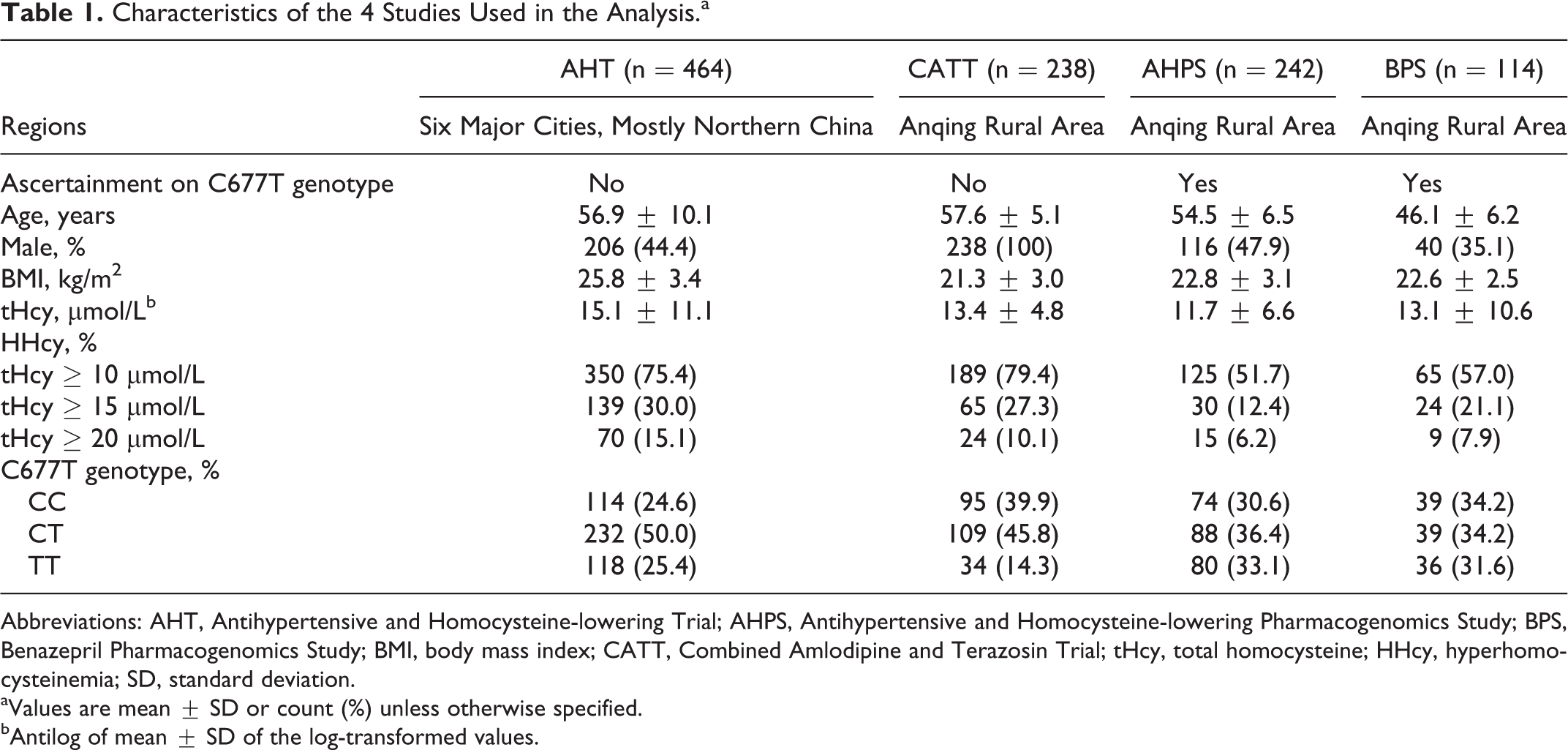
Abbreviations: AHT, Antihypertensive and Homocysteine-lowering Trial; AHPS, Antihypertensive and Homocysteine-lowering Pharmacogenomics Study; BPS, Benazepril Pharmacogenomics Study; BMI, body mass index; CATT, Combined Amlodipine and Terazosin Trial; tHcy, total homocysteine; HHcy, hyperhomocysteinemia; SD, standard deviation.
aValues are mean ± SD or count (%) unless otherwise specified.
bAntilog of mean ± SD of the log-transformed values.
A consistent relationship between C677T genotype and tHcy level was observed across all 4 studies (Table 2). The tHcy levels were essentially the same in the CC and CT groups. In comparison, the TT genotype was associated with a much higher tHcy, with the increase in size ranging from 38% to 68% in the 4 studies and 51% overall. The C677T polymorphism independently explained about 14% of the total variance of the normalized tHcy. Similarly, the TT genotype was associated with a very large increase in OR for HHcy (Table 3). Overall, after adjustment for the confounders, the ORs for the TT genotype were 3.9 (95% confidence interval [CI]: 2.4-6.4), 6.5 (95% CI: 4.0-10.6), and 17.9 (95% CI: 8.4-38.1) for mild, modest, and severe HHcy, respectively.
Regression Analyses of MTHFR C677T Genotype and tHcy.a
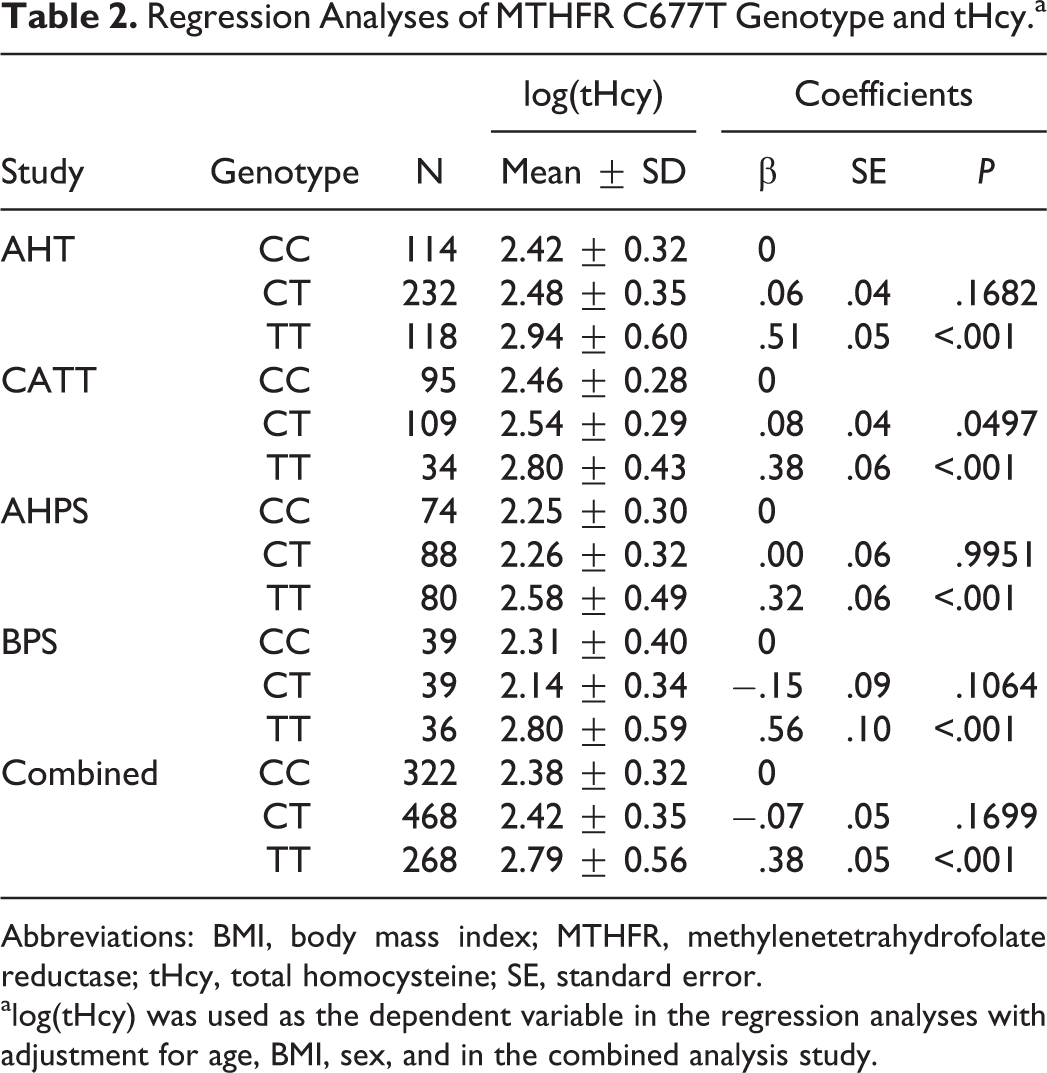
Abbreviations: BMI, body mass index; MTHFR, methylenetetrahydrofolate reductase; tHcy, total homocysteine; SE, standard error.
alog(tHcy) was used as the dependent variable in the regression analyses with adjustment for age, BMI, sex, and in the combined analysis study.
Performance of HHcy Predicting Models With MTHFR C677T Genotype Alone.a
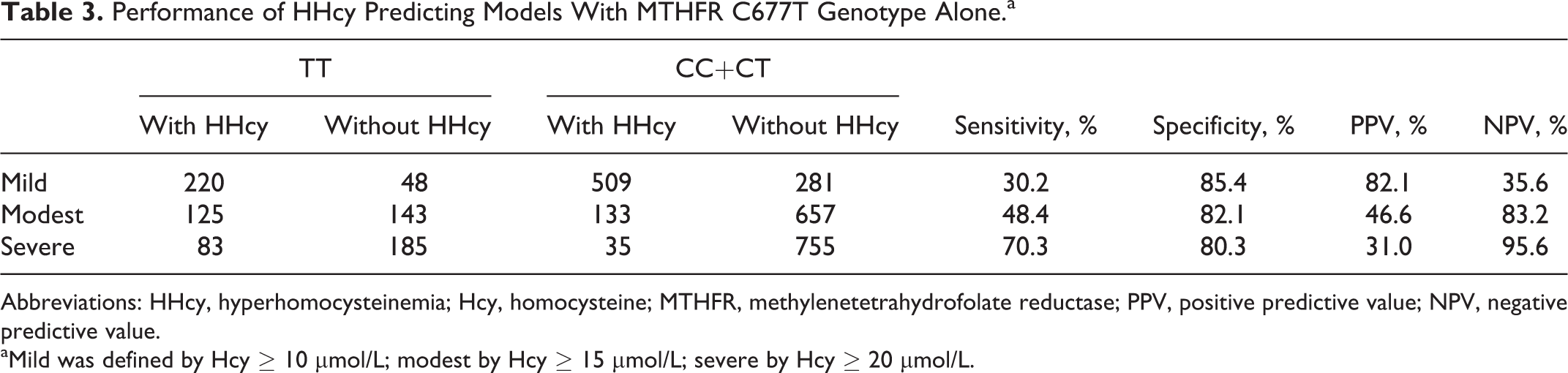
Abbreviations: HHcy, hyperhomocysteinemia; Hcy, homocysteine; MTHFR, methylenetetrahydrofolate reductase; PPV, positive predictive value; NPV, negative predictive value.
aMild was defined by Hcy
We evaluated the performance of using C677T genotype alone to predict HHcy (Table 4). Overall, the performance increased with HHcy severity, with sensitivity improving from 31.0% for mild HHcy to 70.3% for severe HHcy and with specificity slightly decreasing from 85.4% to 80.3%. We further evaluated the ROC curves of logistic regression models with age, gender, and genotype as HHcy predictors (Figure 1). The biggest performance improvement over the genotype-only model was observed for mild HHcy, while at the same specificity level (85.4%) the sensitivity increased from 31.0% to 48.7%. The performance improvement in the 3-factor model over the genotype-only model was much smaller for predicting severe HHcy.
Regression Analyses of MTHFR C677T Polymorphism and HHcy.a
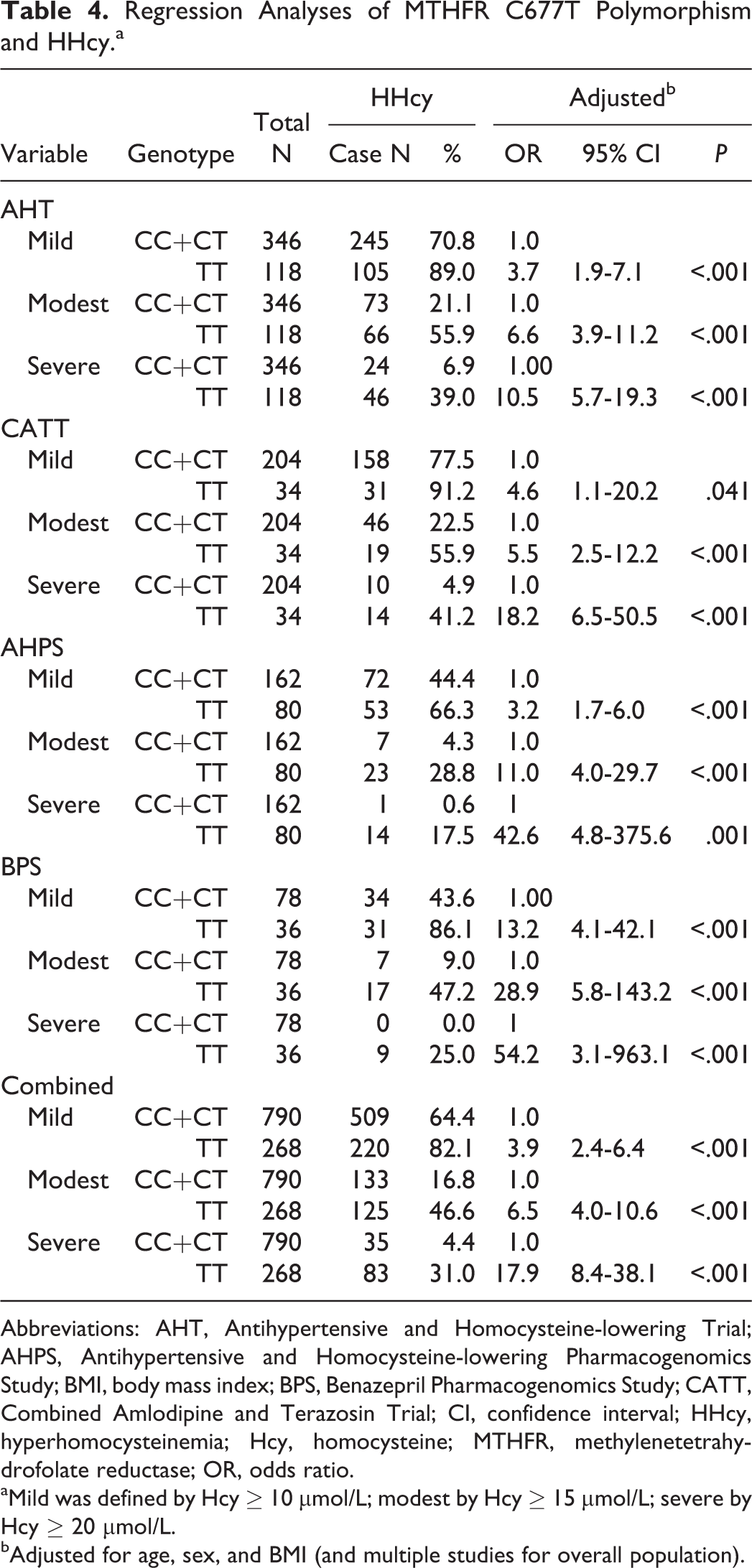
Abbreviations: AHT, Antihypertensive and Homocysteine-lowering Trial; AHPS, Antihypertensive and Homocysteine-lowering Pharmacogenomics Study; BMI, body mass index; BPS, Benazepril Pharmacogenomics Study; CATT, Combined Amlodipine and Terazosin Trial; CI, confidence interval; HHcy, hyperhomocysteinemia; Hcy, homocysteine; MTHFR, methylenetetrahydrofolate reductase; OR, odds ratio.
aMild was defined by Hcy
bAdjusted for age, sex, and BMI (and multiple studies for overall population).
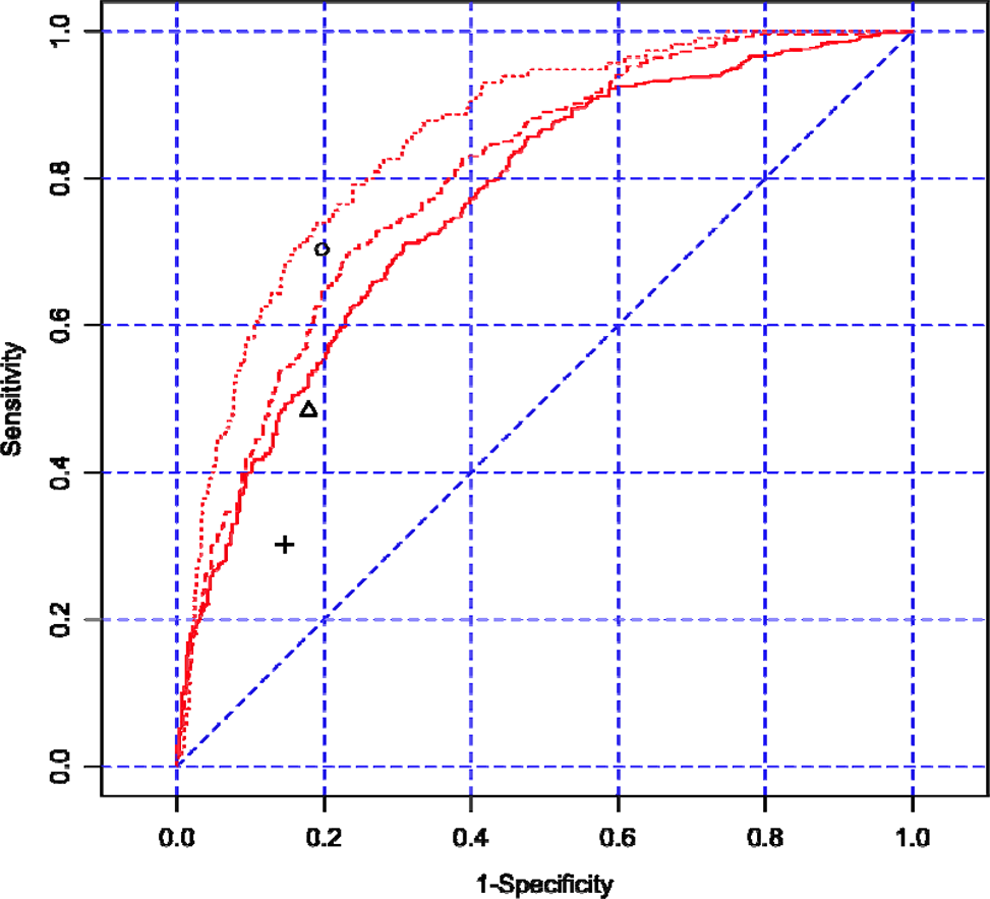
ROC curves of HHcy prediction models with age, gender, and C677T genotype. Legend: ROC curves for mild, modest, and severe HHcy are represented in solid, dashed, and dotted lines, respectively. The corresponding performance points for genotype-alone models were represented in plus sign (mild HHcy), triangle (modest HHcy), and circle (severe HHcy). ROC indicates receiver operating characteristic; HHcy, hyperhomocysteinemia.
Discussion
Consistent with previous reports, we have shown in this article that MTHFR C677T polymorphism is a significant predictor for tHcy and HHcy across all 4 study populations. C677T independently explained 14% of the total variance of the log-transformed tHcy in the study population, while age and gender accounted for another 4.5%. Compared with the CC genotype, TT confers a much higher risk of HHcy, with an adjusted OR as high as 17.9 for severe HHcy (tHcy > 20 μmol/L).
We evaluated the performance of C677T in predicting 3 different levels of HHcy in the study population. Its performance increases with the severity of HHcy, with 30.2% sensitivity and 85.4% specificity for mild HHcy, and 70.3% sensitivity and 80.3% specificity for the severe form. A 3-factor regression model with addition of gender and age as predictors significantly improves the sensitivity, especially for predicting mild HHcy. At 90% sensitivity, the model for severe HHcy has a specificity of 60.2%. With an excellent sensitivity and a modest specificity, C677T could be a useful screening tool for severe HHcy.
Homocysteine is metabolized by remethylation or transsulfuration. 17 MTHFR catalyzes the conversion of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate, a cosubstrate for Hcy remethylation to methionine. Methylenetetrahydrofolate reductase C677T polymorphism, with 2 possible nucleotides, C and T, at base 677, encodes 2 enzyme variants, 1 with alanine and the other with valine at amino acid position 222, respectively. The variant encoded by the T allele is thermolabile and has less activity compared with the one by the C allele. 8,18,19 TT genotype, especially under folate deficiency, causes reduced availability of 5-methyltetrahydrofolate and accumulation of Hcy. 8,19 A large number of epidemiological studies have shown that individuals with TT genotype have significantly higher tHcy than those with a CC or CT genotype. 20 –24 However, in a recent publication, Koç and Akar 25 found that there were 2 different populations in their Turkish data, 1 that found the MTHFR 677TT genotype to have higher tHcy levels as expected, the other showed that the patients with the TT genotype had lower tHcy levels. So the author speculated that there may exist another Hcy-determining haplotype but with no data to support this hypothesis. However, in our 4 independent studies, our data consistently showed that the TT genotype had significantly higher tHcy levels and had increased risk of HHcy. Our data supports the conclusion that the 677TT genotype is a causal marker for predisposition to severe HHcy. Furthermore, it is interesting to note that TT frequencies varied in our study populations. TT frequency was 25.4% in the AHT samples, which included patients who were enrolled from 6 major cities in China, of which 5 are in northern China, and was 14.3% in the CATT sample of patients from Anqing of southern China.
Mutations in the cystathionine synthase gene, which catalyzes the first step of the transsulfuration pathway, from Hcy to cystathionine, have been found to cause severe HHcy, 23 which can subsequently lead to atherosclerosis, thrombosis, and death. 26,27 However, these genetic deficiencies are so rare that they are unlikely to make any significant impact on the population scale. Recent genome-wide association studies on tHcy have identified some new genetic markers associated with tHcy, including CPS1, MUT, DPEP1, ZNF366, PTPRD, and NOX4. 28 –32 A meta-analysis of genome-wide association studies on tHcy concentrations in 44 147 individuals of European descent further identified that 6 novel loci in or near MMACHC, SLC17A3, GTPB10, CUBN, HNF1A, and FUT2 were associated with tHcy concentrations. 9 However, further studies are needed to verify and evaluate their effects. Until now, MTHFR C677T has remained the sole established genetic predictor for tHcy which is of population impact.
Nutritional deficiency of vitamin B and it subtypes involved in Hcy metabolism, including folic acid, B12, and B6, can also cause HHcy, and supplementation of vitamin B can effectively reduce tHcy. 2,7,24 In our study population, a limitation is that we did not measure nutritional levels of vitamin B, although C677T, age, and gender explained up to 20.5% of the total interindividual variation in tHcy. Considering the fact that folic acid deficiency is quite common in the Chinese population, 33 with up to 40% of Northerners having lower plasma folate concentrations (<6.8 nmol/L), 34 we speculate that our prediction model could be significantly improved by adding nutritional status as well as other genetic markers, though further studies are needed.
Another limitation of this study is that all 4 study samples were comprised of patients with hypertension, 3 of which were from Anqing rural areas and 1 that included patients representing 6 major cities in China. Although it would be reasonable to presume that the same prediction model would apply to nonhypertensive individuals, further validation in a more representative and independent sample is warranted.
Footnotes
Acknowledgments
We thank Dr Scott A. Venners for manuscript editing. We also gratefully acknowledge the assistance and cooperation of the faculty and staff of First Hospital affiliated with Peking University and the Anhui Medical University and thank all of the participants in our study. This study was conducted in accordance with the current regulations of the People’s Republic of China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported financially by Major State Basic Research Development Program (Grant No.2012CB517703) and supported by National Natural Science Foundation of China (Grant No. 81373484, 81141116 and 30700454).
