Abstract
Fagonia arabica (FA) is a deobstruent and blood purifier, which possesses thrombolytic and antioxidant activities. In this study, the anticoagulant effects of FA and its derived fractions were evaluated. Plasma recalcification was performed with multisolvent extracts of FA and then with extracts prepared successively with increasing polarity of the solvents. Aqueous extract was the most potent anticoagulant extract, which was fractionated by thin-layer chromatography and column chromatography. Five fractions collected were checked for their anticoagulation effect. The most potent fraction was screened for phytoconstituents. Aqueous extract of FA is the most active anticoagulant (31 minutes). Results were statistically significant when compared to heparin (38 minutes) and saline (4.04 minutes; P > .001). The Fifth fraction (FA5), the most potent fraction (27 minutes), was found positive for flavonoids, saponin, tannin, triterpenoids, carbohydrates, reducing sugar, and monosaccharides. Aqueous FA and fraction FA5 were most active in in vitro anticoagulation, and any of the phytochemicals identified could be considered the active component.
Introduction
Anticoagulants have been widely used in both clinical and in vitro medical treatments. In clinical practice, they are the drugs of choice for the prevention and treatment of thromboembolic disorders and prophylaxis of thrombotic events both pre- and postsurgery. It is estimated that of 1000 patients, only 2 to 4 receive anticoagulant therapy. 1,2 Substances with anticoagulant activities are among the first ones to chosen as functional components that are being used to open up new areas of application for anticoagulants.
Anticoagulant monotherapy has been the basis of treatment for patients with atrial fibrillation and left ventricular thrombus as well as for prevention or treatment of deep venous thrombosis and pulmonary embolism. Considering the adverse effects of the existing anticoagulant therapy, alternative drugs from natural origin can help get the new molecular lead as a hope toward the better efficacious and safe anticoagulant.
Modern pharmaceutical industries are researching to identify new active components, and they can be either natural or synthetic used for curing several diseases. Worldwide, about 30% of the pharmaceuticals are prepared from plants. 3 Epidemiologic studies have provided evidence that foods with experimentally proved antithrombotic effect could prevent coronary events and stroke. 4 Considerable efforts have been directed toward the discovery and development of natural products from various plant and animal sources that have antiplatelet, 5,6 anticoagulant, 7,8 and antithrombotic 9 activity.
Fagonia arabica (FA) belongs to the family Zygoplyllaceae. 10 The FA is a small spiny undershrub with stiff, more or less prostrate, branches and found in northwest India and Deccan. The leaves of FA are arranged opposite to each other, with 1 to 3 foliolates, entire, linear-elliptic leaflets, and small, pink flowers. 11 Since ancient times, FA has been reported to be used as a folk medicine for the remedy of various ailments. Uses of FA as blood purifier and deobstruent were also mentioned in Ayurvedic description. 12 In recent times, FA was reported to possess antioxidant properties, 13 thrombolytic, and 14 mollucidal activities 15 and has also been used for cancer treatment and in liver empowerment. 16 We undertook the present study to evaluate the potential antithrombotic effects of the extracts of FA and its derived fractions.
Materials and Methods
Herbal Preparation
Commercially available total extract of the herb FA (Dhamasa) was purchased from Innocon Foods (Pune, India). The multiple solvent (methanol–isopropyl alcohol–acetone) extraction procedure was used to prepare the extract by the supplier, namely, 1, 2, 3, 4, 5, 10, and 20 mg/mL were suspended in distilled water in 7 different glass test tubes, and the suspension was shaken vigorously on a vortex mixer. The suspension was kept overnight and decanted to remove the soluble supernatant that was filtered through a 0.22-μm syringe filter and stored at 4°C until use.
In Vitro Anticoagulant Activity of the Herb by Plasma Recalcification Time Assay
All the participants selected for the study were healthy, did not have any previous bleeding or thrombotic history, and were not on hormone replacement or hormonal contraception (using a protocol approved by our institute’s ethics committee). Venous blood was withdrawn between 8 and 10
Fagonia Arabica
The plant Fagonia arabica was purchased and identified by Dr A. M. Mujumdar, Agharkar Research Institute Pune, Maharashtra, and the authentication number is Auth07-89.
Preparation of Extracts of the Herb
Successive Extraction
Dried and powdered whole part of the FA was extracted with different solvents of increasing polarity, namely, water, ethanol, methanol, isopropyl alcohol (IPA), butanol, ethyl acetate, acetone, chloroform, petroleum ether, and hexane. Dried and pulverized FA was obtained using soxhlet extraction, with all the 10 solvents successively at their respective boiling points and 8 cycles per solvent was done. All the extracts were filtered and stored in screw cap bottles at 4°C until used.
Test for Anticoagulation
All the 10 extracts were subjected to plasma recalcification assay to study the anticoagulant effect. The method was repeated 10 times with each extract, and the most active extract was identified and studied.
Partial Characterization of FA
The aqueous extract was prepared in bulk by taking 2 kg of dried FA. To obtain a concentrated extract, the aqueous extract was then subjected to rotary evaporator. A brown color sticky mass (paste) was obtained. Ethanol was added to the sticky mass (paste) and triturated until the solid portion separated out, and this solution was refluxed for recrystalization and filtered. Yellowish-orange filtrate portion was collected and stored at 4°C until use. Crude portion was let dried and stored in air-tight container for further studies.
Thin-Layer Chromatography
Filtrate and crude mass was subjected to thin-layer chromatography (TLC). For that a stationary phase of silica gel 60 f254 (20 × 10 cm plate) marking at 1.5 cm for start was selected. The solvent system (liquid phase) used was chloroform–ethanol in a 1:1 ratio. After the run has finished the TLC plates were subjected to developer (iodine fumes). The experiment (TLC) was repeated 10 times with each sample i.e., both filtrate and crude mass.
Column Chromatography
The TLC was followed by column chromatography to separate out and collect all the spots observed through TLC. The column was prepared with Silica Gel 60-120 mesh size, and 2 eluent systems were used, that is, ethanol–chloroform in 1:1 ratio and later ethanol–water in 3:7 ratio, to separate and collect each spots for further analyses.
Phytochemical Screening of FA Fractions
Chemical tests were carried out on the extracts using standard procedures to identify the constituents (alkaloid, saponin, triterpenoids, steroids, flavonoids, carbohydrates, reducing sugars, monosaccharide, polysaccharides, and protein) as described by Sofowora (1993) 17 , Trease and Evans (1989) 18 , and Harborne (1973) 19 .
Anticoagulation Activity of FA Fractions
All the spots collected by TLC-guided column chromatography were subjected to plasma recalcification time (RT) assay for selection of the most potent fraction having anticoagulant activity.
Statistical Analysis
Data were expressed as means ± standard deviation [SD]. One-way analysis of variance was used for repeated measurements of the same variable, and Duncan multiple range t test was used to determine which means were significantly different from that of the control. Differences were significant at P < .05
Results
Effect on Plasma RT by Multisolvent Extract of FA
The in vitro anticoagulant properties of the multisolvent extract (MSE) were assessed by RT assays using healthy human plasma (Figure 1). The normal value of RT for plasma from a healthy human was 4.2 minutes. The RT was prolonged to 6 minutes at a concentration of 1 mg/mL. Increasing the concentration of the extract led to an even higher prolongation of RT. This shows that a concentration-dependent anticoagulant activity was obtained. At 20 mg/mL concentration, the time taken for clot formation increased to approximately 32 minutes. The result was statistically significant as the P value was <.0001 when time taken for coagulation by FA was compared with that of saline (control). With heparin as positive control (0.9 µg/mL), 38.5 minutes of prolonged coagulation time was observed.
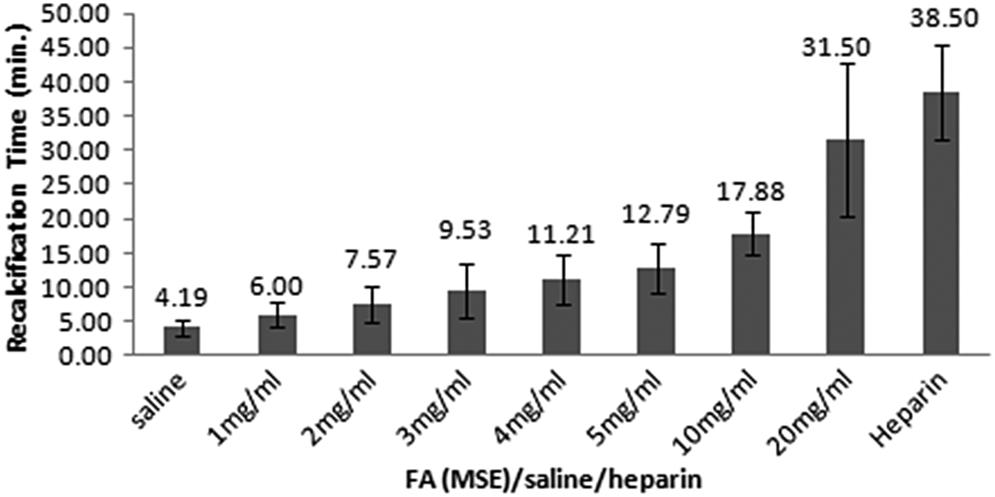
Anticoagulant effect of multisolvent extract (MSE) of Fagonia arabica (FA), positive control (heparin) and negative control (saline) by plasma recalcification assay (n = 20). Values are expressed as mean ± standard deviation. Significance was determined by 1-way analysis of variance, followed by Duncan multiple range t tests. The results were statistically significant as P < .001.
Plasma Recalcification Assay of FA Extracts Prepared With Different Solvents
Among all the extracts assayed for anticoagulation, namely, water (aqueous), ethanol, methanol, IPA, butanol, ethyl acetate, acetone, chloroform, petroleum ether, and hexane, aqueous extract was found to be most effective as it delayed the RT up to 27 minutes, which is equivalent to the MSE extract of FA, that is, 27.34 minutes (n = 10; P < .001). Anticoagulant effects of all the extracts are tabulated in Table 1. Negligible activity was observed with other extracts (Table 1).
Anticoagulant Effect of the 10 Extracts Prepared by Successive Extraction, Multisolvent Extract (MSE) of FA, Heparin, and Saline by Plasma Recalcification Assay (n = 10).
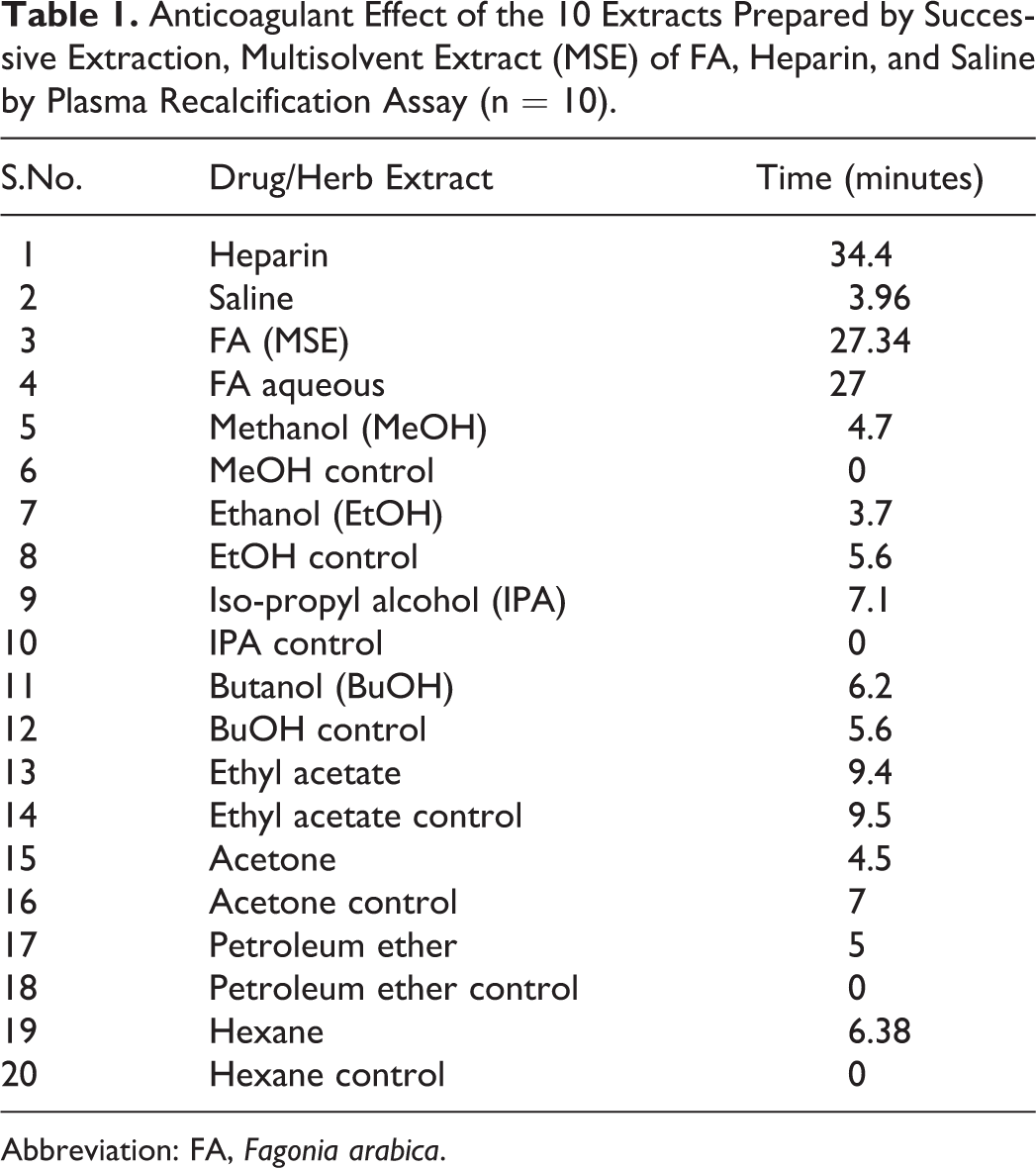
Abbreviation: FA, Fagonia arabica.
Preparation of FA Fractions
Thin-Layer Chromatography
Both filtrate portion and solid mass of FA were subjected to TLC as described earlier. According to the TLC analysis of the ethanol soluble filtrate, 5 spots were detected (n = 10), and the retardation factor values of each spot are listed in Table 2. However, the TLC of the solid portion showed only a single spot (Table 2).
Results of the Filtrate and Solid Mass of Aqueous Extract of FA Obtained by Thin-Layer Chromatography (TLC).

Abbreviation: Rf, retardation factor.
Column Chromatography
After TLC, each spot was separated and collected with the same eluent system as that of TLC. With chloroform–ethanol (1:1), 4 spots were collected and stored at 4°C for further analysis. For the collection of the fifth fraction, the eluent system was changed to ethanol–distilled water (3:7). The fractions thus collected were named as FA1, FA2, FA3, FA4, and FA5.
Effect of FA Fractions on In Vitro Anticoagulation Activity
All the 5 fractions collected by TLC-guided column chromatography of FA (aqueous) were subjected to plasma recalcification assay (Figure 2). With FA1, FA2, FA3, and FA4, 6.7, 9.92, 7.11, and 7.58 minutes of anticoagulation was noted, respectively. Maximum anticoagulation was found in FA5. The FA5 of 100 µL was equally effective as heparin (20 µg/mL), with 25.16 minutes of RT (P < .001).
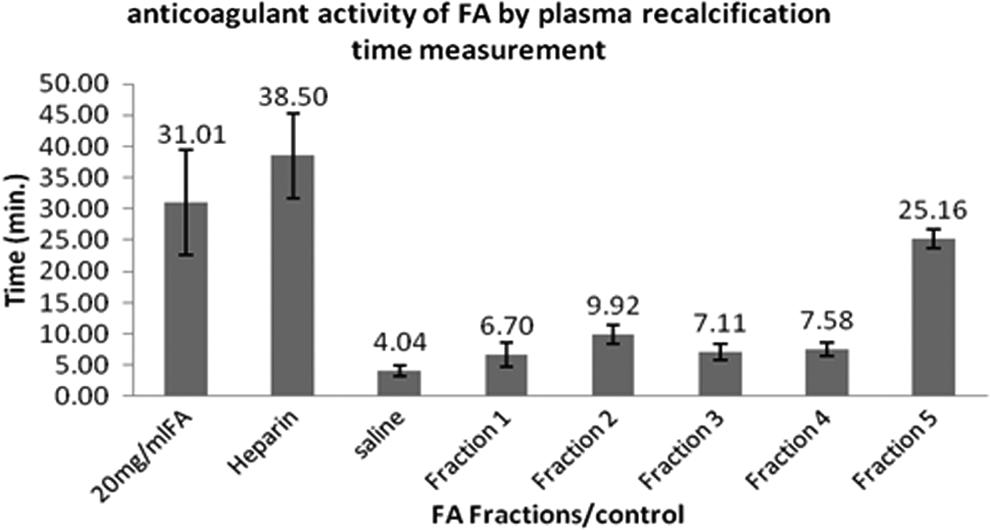
Anticoagulant effect of fractions of aqueous extract of Fagonia arabica (FA; FA1, FA2, FA3, FA4, and FA5) with heparin as positive control and saline as negative control (n = 10). Values are expressed as mean ± standard deviation. Significance was determined by 1-way analysis of variance, followed by Duncan multiple range t tests. The FA5 was found to be most active for anticoagulation and the P value was <.001.
Phytochemical Screening of FA5
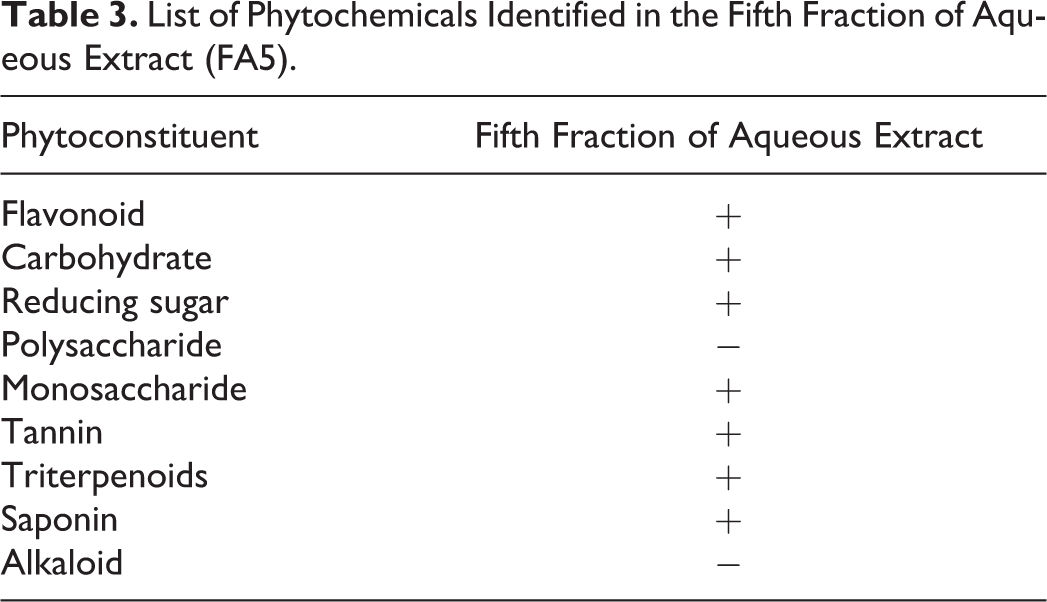
The FA5 was estimated for the presence of the following phytochemicals: flavonoids, carbohydrate, reducing sugar (monosaccharide and polysaccharide), alkaloids, protein, tannin, triterpenoid, and saponin. The most active fraction (FA5) was positive for flavonoids, carbohydrate, reducing sugar, monosaccharide, tannin, terpenoid, and saponin (Table 3). Total yield of FA5 was 20.82% extracted from 2 kg of dried FA.
List of Phytochemicals Identified in the Fifth Fraction of Aqueous Extract (FA5).
Discussion
Anticoagulants are agents that prevent the clotting of blood. A thrombus is formed only in in vivo (unlike blood clots that can form in vitro). Pieces of the thrombus may break off and form an embolism that may lodge in vessels in the lungs or brain or any other body part, causing damage to the tissues supplied. Thrombolytic drugs are able to dissolve thrombi, and the antiplatelet drugs diminish the adhesion of platelets and their contribution to thrombus formation. In some situations, for example, myocardial infarction, the 3 classes of drug—anticoagulants, antiplatelets, and thrombolytics—may be used in combination. 20
Antithrombotic, thrombolytic, and antiplatelet drugs are used to cure the patients with atherothrombosis. Through our experiments, it was found that FA possesses thrombolytic property that is proved both by an in-house developed Tube method of Central Indian Institute of Medical Sciences 21 and by Microtitre Plate Clot Lysis assay. 22 Moreover, its antioxidant property was recently reported, and its neuroprotective role was defined. 13 Anticoagulant property of FA was not studied so far; therefore, it was desirable to study the anticoagulant property of FA. Hence, anticoagulation property of FA has been studied by plasma recalcification assay along with partial characterization of FA.
The MSE of FA purchased from Innocon Foods was tested for anticoagulation by plasma recalcification assay and compared with positive (heparin) and negative controls (saline). With saline, it took 4.2 minutes to form a firm clot; however, with heparin the time taken for firm clot formation increased to 38.5 minutes. Interestingly, with increasing concentration of MSE, the time for clot formation also increased in increasing order. The highest anticoagulation activity was found with 20 mg/mL concentration of MSE that is 31.5 minutes (Figure 1). With repeated experiments, the same results were observed; thus, it was clear that MSE of FA possesses anticoagulation property in vitro. In the next phase of this study, in order to characterize the potential effect of FA, 10 raw dried FA extracts were prepared successively with increasing polarity of solvents. Aqueous extract was the most potent extract (Table 1) possessing the anticoagulation property, as its effect was almost similar to that of FA extract purchased commercially as well as with the positive control heparin. With other extracts, negligible activity was noted; thus, aqueous extract was chosen for further characterization.
Aqueous extract was reduced by rotary evaporator. It was then subjected to a series of steps including TLC and separation of collected spots by column chromatography. All the 5 spots/fractions (FA1-FA5) thus collected were subjected to in vitro anticoagulation assay to determine the most active anticoagulation fraction. Interestingly, FA5 was found to be the most potent anticoagulant fraction (27 minutes) that was very near to the effect noted with commercial FA extract (31 minutes). Thus, we can say that FA5 contains the most active component responsible for the anticoagulation effect.
To analyze the contents of FA5, it was subjected to phytochemical screening. The FA5 was found positive for saponin, flavonoids, tannin, triterpenoids, carbohydrates, monosaccharide, and reducing sugar.
Through this study, it was made clear that FA possesses anticoagulant property; and after partial characterization, it was further demonstrated that FA5 possesses the active anticoagulant property and contains several phytochemicals. Plasma recalcification assay is the most convenient method for general anticoagulation testing of newer drugs/herbs. 23,24 This test is convenient to perform and requires no analyzer or special instrument to evaluate anticoagulation. To the test plasma, CaCl2 is added and observed for coagulation by measuring the time taken for clot formation. The cost of the test is minimal and is good to study anticoagulation of newer test drugs. The anticoagulation effect can be studied in general, but there are other methods that could be used for further determination of anticoagulant mechanism, for example PT, APTT, PTT, and so on. 25,26 These assays test the intrinsic and extrinsic coagulation cascade of the newer drugs. These tests are used worldwide to study anticoagulation; however, recently 3 new tests are reported to study the intrinsic and extrinsic coagulation pathways with respect to newer drugs/herbs. These are The intrinsic coagulation activity assay (INCA), The extrinsic coagulation activity assay (EXCA), and The recalcified coagulation activity assay (RECA). 27 –29 All the 3 methods are recent global assay used to study coagulation, are more advanced techniques that require less reagents (than PT, APTT and PTT assay), and require no special analyzer to identify and calculate the coagulation time with respect to newer anticoagulant drug study in individual patients. For financially unaided laboratories, installing new analyzers or instruments and bearing the cost of reagents are difficult; hence, plasma recalcification assay is the most convenient and affordable assay to study anticoagulation initially for newer drugs. As with FA, we have found significant anticoagulation effects, and FA can be further studied using INCA, EXCA, and RECA in future for better understanding of the mechanism.
The FA as a whole or its fraction could be investigated for anticoagulant activity using in vivo experimental protocol. Further studies could also reveal whether phytoconstituents of FA5 are responsible for anticoagulation. As FA is an herb and is used for medication since ancient times, the related side effects could be negligible. Hence, FA could be used as a new effective herbal anticoagulant for the treatment of patients with thrombosis and associated diseases. Through earlier studies conducted by other researchers, it was reported that FA possesses the following components: flavonoids (6 types) isorhamnetin 3-glucoside and 3-rutinoside, herbacetin 8-rutinoside, herbacetin 8-methyl ether-3-rutinoside, 3,7-diglucoside, and 3-rutinoside-7-glucoside, triterpenoidal glycosides, sulfated triterpenes, sulfated triterpene glycosides, saturated and sulfated pentacyclic triterpene of the taraxastane series with a C-20, 28 lactone unit, monodesmosidic saponins, bidesmosidic saponins, ceryl alcohol,-sitosterol n-triacontanal chinovic acid, fagogenin, saponin, genin A genin B, stigmasterol, campesterol, and harmine. Three triterpenoid of oleanane group, triterpenoid saponin mixture containing saponin A, B, and C, 5 substituted 8-methoxyflavones, and 2 erythroxan-type diterpene, fagonone (16-O-acetylflagonone), and minerals, namely, Zn, Na, Mn, Fe, P, K, Ca, Cu, vitamin C, protein, amino acids (alanine, arginine, glycine, isoleucine, leucine, lysine, phenylalanine, proline, tyrosine, valine) were also found. 15,30 –33 Through characterization of the phytochemicals identified in FA5 with reference to the reported phytochemicals, the most active component could be identified and analyzed for anticoagulation.
Anticoagulants play a pivotal role as agents for the prevention and treatment of thromboembolic disorder. 34,35 For more than 5 decades, anticoagulant such as heparins, vitamin K-antagonists, and their derivatives have been the major players in the clinical practice. Although their efficacy remains undisputed, the fatal life-threatening side effects of these drugs have also been well documented. 36–37 The cost of developing a novel anticoagulant is also given due consideration, whereby a cheaper yet effective alternative would be of immense value. 38,39 In addition, there is a pressing need for an orally available anticoagulant agent to replace warfarin. 40 At present, the focus is on potent anticoagulant sources hailing from natural origin. There is a compelling need for the consumption of dietary anticoagulants or phytochemicals with anticoagulant properties, which can ultimately reduce or eliminate the risks of thromboembolic diseases. 41 Hence, anticoagulants of natural origin provide a good remedy/therapy for the aforementioned disorders. 42 Moreover, anticoagulants have also been identified from other natural sources such as cellulose sulfate, 43 sulfated flavonoids, 44 halothurian glycosaminoglycan, 45 a glycosaminoglycan extracted from the body wall of the sea cucumber, 8 protein from Taraxacum platycarpum, 46 and many more.
The hot water extraction of the whole plant of FA gave the highest yield and exhibited the most potent anticoagulant properties for all parameters tested in vitro. These results highlight the biochemical nature of the active compounds in FA that might contribute to anticoagulation. Since it is evident that hot water has the ability to extract the phytochemicals responsible for the activity, presumably, the active compounds extracted at this temperature are thermally labile. Many traditional medicinal plants and herbs have been reported to have various levels of anticoagulant activity. In general, plant extracts of FA have yielded promising results, hence it seems reasonable to conclude that there are probably important anticoagulant compounds present in FA, but we need to isolate and identify these active constituents. Further characterization will reveal the identity of these compounds. Hence, it could be stated that the phytochemicals thus identified in this study could be responsible for anticoagulation action of FA otherwise FA as a whole extract could also be incorporated in specific preparation for anticoagulation.
The physicians give both thrombolytic and antithrombotic drugs for patients with thrombosis. This type of combination ensures that thrombolytic drugs would dissolve the clots already formed in the veins and arteries and anticoagulant drug will prevent further clot formation. In lieu of the present study and with our earlier findings, FA possesses both thrombolytic and anticoagulant activities. There are other natural products and their components that possess both anticoagulant and thrombolytic properties such as onion, 47 Paeonia anomala extract, 48 tuftsin, 49 Paeonia lutea root bark, 50 danshen (Salvia miltiorrhiza), 51 and meadowsweet (Filipendula ulmaria (L) Maxim.). 52 –54
The FA could be a better agent for curing patients with thrombosis, as it would act as both a clot dissolver and an anticoagulant agent. Moreover, being a natural product, the related side effects would be less and therefore safer.
Conclusion
Fagonia arabica extract (aqueous and commercial) and fractions of aqueous extract (obtained by column chroma-tography based on TLC-guided results) were evaluated for anticoagulation activity based on an RT assay in an in vitro system. Prolongation of RT produced by the FA and its fractions increased with an increase in the concentration of FA. Further investigations are needed to isolate the bioactive compounds responsible for the anticoagulant activity as well as for the determination of the coagulation factors affected. This natural source is a cheaper and better alternative anticoagulant because of the abundant availability of raw material and simple extraction methods.
Footnotes
Acknowledgments
We would like to acknowledge the Indian Council of Medical Research, New Delhi, for providing Senior Research Fellowship to Sweta R. Chourasia.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sweta R. Chourasia has received Senior Research Fellowship from Indian Council of Medical Research, New delhi, India.
