Abstract
Objective
Deep venous thrombosis (DVT) is a common complication in patients with traumatic injury. Tissue factor pathway inhibitor (TFPI) is a natural anticoagulant protein in the extrinsic coagulation pathway. However, the relationship between DVT after trauma and the anticoagulant activity of TFPI remains unclear. In this prospective study, we investigated the role of TFPI in trauma patients with DVT to evaluate whether the anticoagulant activity of TFPI measured by a new functional assay can be used to help predict the risk of DVT.
Introduction
Deep venous thrombosis (DVT) and pulmonary embolism are the main causes of hospital-related morbidity and mortality and are also recognized as important complications in trauma patients. 1 Clinical data have shown that the incidence of DVT in patients with a traumatic fracture is almost 30%.2,3 The morbidity and mortality related to DVT caused by trauma is very high, although its mechanism remains unclear. Therefore, it is important to gain a better understanding of the pathophysiology of DVT caused by trauma in order to diagnose and prevent this serious condition.
Tissue factor pathway inhibitor (TFPI), also known as exogenous coagulation pathway inhibitor, is a very important natural anticoagulant in vivo. TFPI is produced by microvascular endothelial cells and pooled in the endothelium (80%), plasma (10%-20%), and platelets (<2.5%).4,5 The concentration of TFPI in plasma is very low, with the major component (80%) bound to lipoproteins (ie, high-density lipoprotein [HDL],6,7 while the full length, minor component [20%], called free TFPI, is unbound and therefore has the main anticoagulant effect. The anticoagulant effect of TFPI can be attributed to two mechanisms: inhibition of tissue factor-activated coagulation factor VII [TF-FVIIa] and activated coagulation factor X [FXa] via the tissue factor-activated coagulation factor VII-tissue factor pathway inhibitor-activated coagulation factor X [TF-FVIIa-TFPI-FXa] complex, 8 and inhibition of prothrombinase as reported in a recent study. 9
There is convincing evidence from in vitro and in vivo experiments that TFPI plays an important role in regulating the initiation of blood clotting.10,11 However, to date no study has shown a clear relationship between thrombosis and TFPI. While studies have reported that the antigen level of TFPI is related to venous thrombosis and the low antigen levels are a risk factor for venous thrombosis, 12 patients with trauma contrastingly have high levels of the antigen.13,14 The reason for this increase in TFPI levels in trauma patients with DVT is not clear. At present, the majority of studies on TFPI focus on antigen levels,15,16 a parameter, which for a number of reasons, does not provide a measure of TFPI anticoagulant activity in vivo. The anticoagulant activity of TFPI may more accurately reflect the effect of TFPI on blood coagulation, and therefore it is very important to evaluate this anticoagulant activity in patients.
The objective of our study was to develop a functional assay for measuring the anticoagulant activity of TFPI in plasma from normal and trauma patients with or without DVT, and evaluate biomarkers of DVT risk and the relationship between TFPI and DVT.
Patients and Methods
Study Design and Patients
A total of 475 traumatic patients were recruited for this prospective nested case-control study at the Traumatic Orthopedics Emergency Department of Beijing Jishuitan Hospital from November 2019 to December 2020. The inclusion criteria were: adults with traumatic lower extremity fractures of femoral shaft, distal femoral, patella, tibial plateau, tibiofibular shaft, or ankle. The exclusion criteria were: patients with diseases of the immune system, cancer, inflammation, a history of thrombosis and anticoagulant therapy, and a history of COVID19 infection. All the patients had angiographic results to estimate whether they had suffered from DVT before surgery. The trauma patients with DVT were defined as the DVT group (n = 48), while those without DVT were matched according to age and sex and called the non-DVT group (n = 48). The study also included a control group of 120 healthy individuals free from any acute or chronic disease. The study protocol is shown in Figure 1. Ethical approval for the study was given by the medical ethics committee of Beijing Jishuitan Hospital.

Study protocol.
Blood samples were collected from the trauma patients the first time they were admitted to the emergency department after being injured. The samples were collected into tubes containing either ethylene diamine tetra-acetic acid (EDTA) or 3.2% trisodium citrate. Both tubes were centrifuged for 15 min at 2500 × g to produce platelet-poor plasma (PPP) that was used to measure routine clinical laboratory parameters. The citrated plasma sample was then centrifuged twice for 15 min at 2500 × g to produce platelet-free plasma (PFP) that was stored at −80°C until analysis. The frozen-PFP samples were thawed at 37°C for 5 min before use. All the samples from each patient were tested in the same series of analyses.
Detection of Routine Clinical Laboratory Parameters
An EDTA-anticoagulated whole blood sample was used to measure red blood cell (RBC), white blood cell (WBC), and platelet (PLT) counts in a Sysmex XT4000i analyzer (Sysmex, Kobe, Japan). A coagulation profile that included activated partial thromboplastin time (APTT), prothrombin time (PT), and international normalized ratio (INR) was measured in the PPP sample by an optical method using a Sysmex CS2100i analyzer (Sysmex). A Sysmex CS-5100 analyzer (Sysmex) was used to measure fibrinogen (FIB) and D-dimer (DD) levels in the PPP sample using the Clauss and immunoturbidimetry methods, respectively.
Free-TFPI Antigen Level in Plasma
The concentration of plasma-free TFPI antigen was determined by an enzyme linked immunosorbent assay (ELISA) method (Quantikine human TFPI ELISA, R&D Systems, Minneapolis, USA) using an Infinite M200_pro reader (Tecan, Männedorf Switzerland).
Assay of TFPI Anticoagulant Activity
We developed an assay to evaluate TFPI anticoagulant activity based on the diluted prothrombin time (dPT) assay. 17 Three reagents were used for this assay. Recombinant human tissue factor lipoprotein (rhTF) protein (PT1002a, rTF1130, assaypro, USA) at a concentration of 3000 PM was dissolved in distilled water and stored at −80°C. Anti-TFPI immunoglobulin G (anti-TFPI IgG) (AF2974, R&D Systems, Minneapolis, USA) was a purified polyclonal IgG raised in a goat and was stored at −20°C at a concentration of 0.2 mg/ml reconstituted in distilled water. Calcium chloride dihydrate crystals (HuShi, China) at a Ca2+ concentration of 0.5 mol/L dissolved in distilled water was stored at 4°C.
For the assay, each plasma sample was divided into two wells, one test (with TFPI antibody), and one test (without TFPI antibody) in 96-well transparent plates (E20073F9, Greiner Bio-One, Frickenhausen, Germany). In the one test well (with TFPI antibody), 48 ul PFP, 12 μL of anti-TFPI IgG (20 μg/ml), 20 μL aliquot of rhTF (25 PM), and 20 ul Ca2+ (0.025 M) were added to a final volume of 100 ul. In the test well (without TFPI antibody), an equal volume of distilled water replaced with anti-TFPI IgG (20 μg/ml).
The assay of TFPI anticoagulant activity was performed by initially the addition of 48 μL of PFP and then incubated for 15 min at room temperature with 12 μL distilled water or 12 μL of anti-TFPI IgG (20 μg/ml). A 20 μL aliquot of rhTF (25 PM) was then added and incubated for 10min at 37°C. Finally, 20 μL Ca2+(0.025M) was added and after the mixture was shaken for 10 s the time and plasma absorbance to clotting were measured every 10 s for 2 h at 405nm and 37°C using an Infinite M200_pro reader (Tecan, Switzerland).
The reader automatically graphed two curves of time-related changes in plasma absorbance that were used to examine differences in the clotting process between plasma with or without TFPI antibodies. Another two curves were constructed using the absorbance value of the detection well subtracted from that of the blank well to examine time-related differences in absorbance difference. (Figure 2.A) We then calculated the first derivative of the two curves. (Figure 2.B&C)

The curve of TFPI anticoagulant activity.
Three parameters were defined to evaluate the anticoagulant activity of TFPI as follows.
1.TFPI initial anticoagulant time ratio (%) = (time AB−time DE)/ time AB × 100. (time AB represents the time about the coagulation without TFPI antibody from initial velocity to maximum velocity; time DE represents the time about the coagulation with TFPI antibody from initial velocity to maximum velocity)
2.TFPI whole anticoagulant time ratio (%) = (time AC-time DF)/ time AC × 100. (time AC represents the time about the coagulation without TFPI antibody from initial velocity to final velocity; time DF represents the time about the coagulation with TFPI antibody from initial velocity to final velocity)
3.TFPI anticoagulant rate (%) = (area DF-area AC)/ area AC × 100. (area DF represents the area under curve about the coagulation without TFPI antibody from initial velocity to final velocity; area AC represents the area under curve about the coagulation with TFPI antibody from initial velocity to final velocity)
To verify the coefficient of variation (CV), NPP was used in the assay of TFPI anticoagulant activity, three times a day for three consecutive days, with each experiment conducted independently.
SPSS 23.0 and GraphPad Prism 7.0 were used for the statistical analyses. The data with a normal distribution were expressed as means ± standard and non-normal data were presented as median (interquartile range). The values of RBC, WBC, PLT, PT, APTT, INR, FIB, DD, free-TFPI antigen, and TFPI anticoagulant activity were compared between healthy people and trauma patients, and between three groups including healthy people and trauma patients with or without DVT before surgery. We used univariate analysis to identify variables which showed statistically significant differences between two groups. The Kruskal–Wallis test was used to assess whether the data had normal distribution. Differences between data with an abnormal distribution were analyzed by the Mann–Whitney U test, and data with a normal distribution by the Students T test. Differences in variables between three groups were examined using analysis of variance. Multivariate logistic regression analysis incorporating a backward stepwise model was used to generate adjusted odds ratios (OR) and 95% confidence intervals (CI). To estimate the predictive value of the various indices receiver operating characteristic (ROC) curves were constructed for area under the curve (AUC) analysis with 95% CI. The correlation between variables was examined by calculating Pearson coefficients (r). For all box plots shown, the horizontal bars represent medians and the vertical bars represent the 2.fifth and 97.fifth percentiles. P values <0.05 were regarded as statistically significant.
Results
Study Population
A total of 475 trauma patients who satisfied the inclusion and exclusion criteria were enrolled in the study between November 2019 and December 2020. 48 patients were diagnosed with DVT and were age- and sex-matched to 48 patients without DVT. In addition, 120 healthy controls (males 56 and females 64) aged 26 to 93 years were enrolled in the study. The demographics and clinical characteristics of the healthy controls and patients are summarized in Table 1. During the study, the incidence of DVT before surgery in the trauma patients was 10.1% (48/475).
Characteristics of Studied Groups
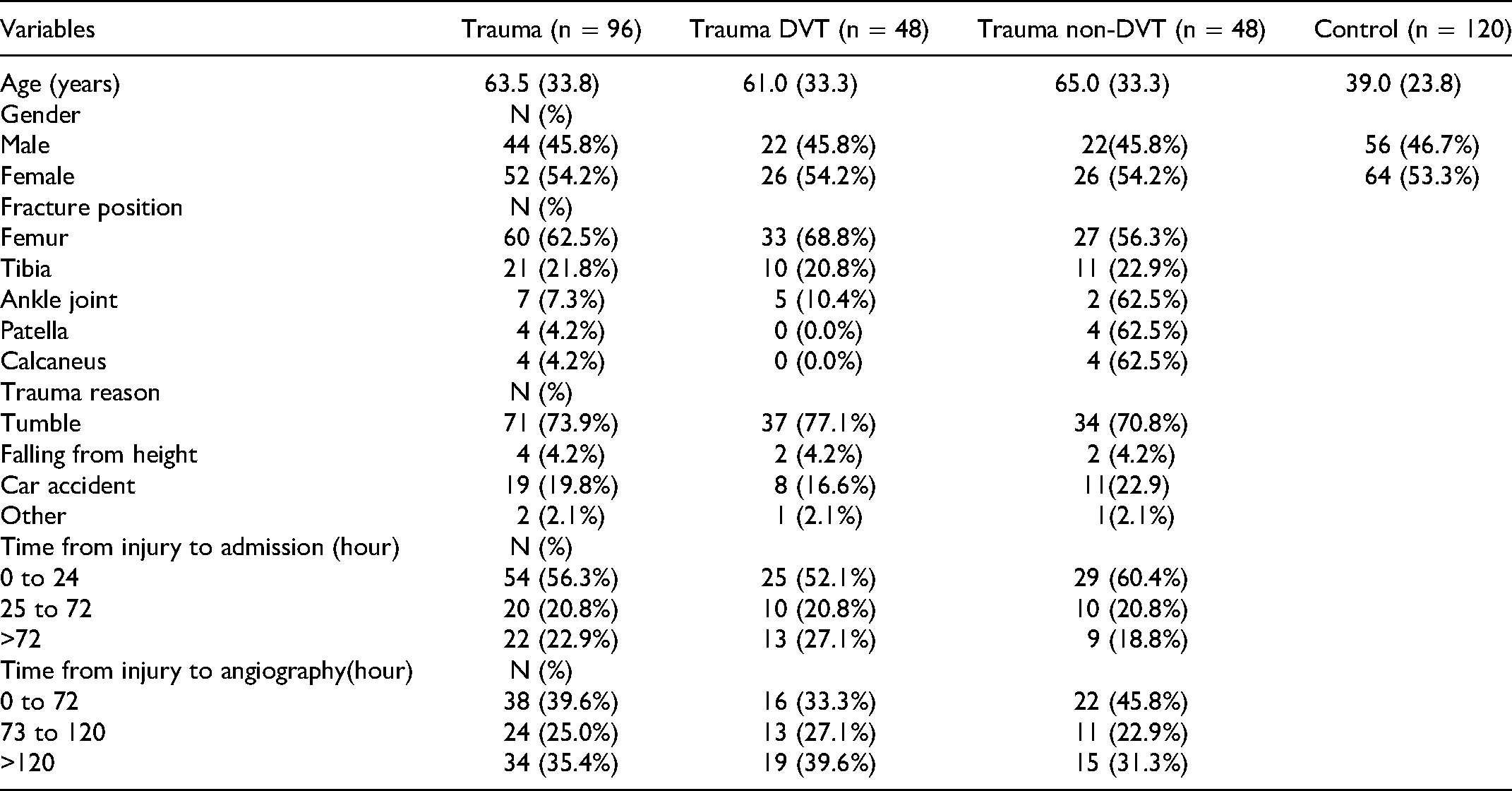
Characteristics of Studied Groups

The descriptive statistics of the three parameters of TFPI anticoagulant activity was established in 120 healthy individuals. TFPI initial anticoagulant time ratio, mean 33.81 ± 10.48%; TFPI whole anticoagulant time ratio, mean 10.45 ± 5.10%; and TFPI anticoagulant rate, mean 28.18 ± 9.22%. The three parameters’ values were also not significantly different at different ages and sexes (Table 2).
Patrameters of TFPI anticoagulant activity in Healthy Population

Patrameters of TFPI anticoagulant activity in Healthy Population

*Normally distributed data are presented as mean ± standard, and non-normally distributed data are showed as median (interquartile range).
The between-day coefficient of variation (CV) for TFPI initial anticoagulant time ratio, TFPI whole anticoagulant time ratio, and TFPI anticoagulant rate were 2.8%, 18.5%, and 9.1%, respectively, while the corresponding within-day CVs were 6.4%,19.9%, and 3.4%.
All the parameters measured in the healthy controls and trauma patients are shown in Table 3.
Comparison of Parameters in Healthy Controls and Trauma Patients

Comparison of Parameters in Healthy Controls and Trauma Patients

*Normally distributed data are presented as mean ± standard, and non-normally distributed data are showed as median (interquartile range).
For TFPI anticoagulant activity, there were significant increases in the levels of TFPI initial anticoagulant time ratio and TFPI whole anticoagulant time ratio in the trauma group (37.51 ± 12.19%; 12.77 ± 6.77%) compared to the healthy group (33.81 ± 10.48%, P = .049; 10.45 ± 5.10%, P = .045), respectively. The TFPI anticoagulant rate was not significantly different between the two groups (P = .825).
All the parameters measured in the healthy controls and trauma patients with DVT or non-DVT are shown in Table 4.
Comparison of Parameters in Healthy Controls, and Trauma Patients with or without DVT

Comparison of Parameters in Healthy Controls, and Trauma Patients with or without DVT

* Normally distributed data are presented as mean ± standard, and non-normally distributed data are showed as median (interquartile range).
a P < 0.050, and the difference is statistically significant when compared with healthy controls.
b P < 0.050, and the difference is statistically significant when compared with trauma patients with non-DVT.
For the parameters of TFPI anticoagulant activity, the levels of TFPI initial anticoagulant time ratio, TFPI whole anticoagulant time ratio, and TFPI anticoagulant rate were higher in the trauma group with DVT (39.91 ± 13.44%; 13.65 ± 7.96%; and 32.61 ± 15.43%) compared with the healthy group (33.81 ± 10.48%, P = .013; 10.45 ± 5.10%, P = .016; and 28.18 ± 9.22%, P = .029, respectively). In addition, there was no significant difference in these three parameters between the healthy group and the trauma group without DVT.
Comparison of parameters between DVT and non-DVT patients
The univariate analyses shown in Figure 3 indicated that TFPI initial anticoagulant time ratio, TFPI whole anticoagulant time ratio, TFPI anticoagulant rate, free-TFPI antigen, PT, and D-dimer were significantly associated with the occurrence of DVT in trauma patients.

Comparison of parameters between the DVT group (n = 48) and the non-DVT group (n = 48), including the (A) TFPI initial anticoagulant time ratio (P = .022), (B) TFPI whole anticoagulant time ratio (P = .048), (C) TFPI anticoagulant rate (P = .034), (D) free-TFPI antigen (P = .035), (E) Prothrombin time (P = .030), and (F) D-dimer (P = .039). * P < .05, and the difference is statistically significant.
For the parameters of TFPI anticoagulant activity, all three parameters were increased markedly in the DVT group compared with the non-DVT group. The level of TFPI initial anticoagulant time ratio was higher in the DVT group (39.91 ± 13.44%) than in the non-DVT group (32.19 ± 10.28%, P = .022). The level of TFPI whole anticoagulant time ratio was markedly elevated in the group with DVT (13.65 ± 7.96%) compared with those in the group with non-DVT (9.77 ± 4.91%, P = .048). The level of TFPI anticoagulant rate in DVT patients (32.61 ± 15.43%) also tended to be increased compared with that in non-DVT patients (25.87 ± 10.32%, P = .034).
For free-TFPI antigen concentration, the level of free-TFPI antigen showed a similar significant increasing trend in trauma patients with DVT (33.43 [16.15] ng/ml) compared with those without DVT (29.43 ± 9.93ng/ml, P = .035).
For the routine clinical coagulation parameters, a significant difference in the level of PT was observed between the DVT (12.63 ± .89 s) and non-DVT groups (12.23 ± .76 s, P = .030). The level of DD was also increased in the DVT group (7.30 [5.85] mg/L) compared with the non-DVT group (3.31 [10.68] mg/L, P = .039).
Factors significant in the univariate analyses were included in the multivariate logistic regression analysis (Table 5).
Independent Risk Factors for DVT

Independent Risk Factors for DVT

Abbreviation: CI, confidence interval; OR, odds ratio.
In trauma patients, the TFPI initial anticoagulant time ratio (OR = .902, 95% CI 0.814-0.998, P = .046), free-TFPI antigen (OR = 0.941, 95% CI 0.890-0.995, P = .033), and PT (OR = 0.364, 95% CI 0.174-0.764, P = .008) were protective factors for DVT, while RBC (OR = 2.748, 95% CI 1.085-6.957, P = .033) was a risk factor of DVT.
We then created a formula for the TFPI correlation coefficient that fitted the model and the new indicator as: (−0.104) × TFPI initial anticoagulant time ratio (%) + (−0.061) × free-TFPI antigen (ng/ml)+(−1.010) × PT (s)+(1.001) × RBC ( × 10 9 /L)+(7.239).
To investigate the usefulness of the parameters (TFPI initial anticoagulant time ratio, TFPI whole anticoagulant time ratio, TFPI anticoagulant rate, free-TFPI antigen, PT, DD, and TFPI correlation coefficient) for diagnosing DVT in trauma patients, a ROC curve analysis was conducted for the DVT and non-DVT groups (Table 6 and Figure 4).

Receiver operating characteristic curves for indicators to predict DVT in trauma patients. The TFPI initial anticoagulant time ratio is represented by a dark blue line with an area under the curve of .646(P < .016); free-TFPI antigen is represented by a light blue line with an area under the curve of 0.627(P = .035); prothrombin time is represented by a purple line with an area under the curve of 0.617(P = .048); D-dimer is represented by a green line with an area under the curve of .624(P = .039); and TFPI correlation coefficient is represented by a red line with an area under the curve of 0.821(P < .001).
Diagnostic Values of the Identified Indicators

All cut-off values were determined by the Youden index.
Abbreviation: AUC, area under the curve; CI, confidence interval.
For the TFPI initial anticoagulant time ratio, the optimal cut-off value was 33.53% with a sensitivity of 71.11% and specificity of 55.32% (AUC 0.646, 95% CI 0.533-0.759, P = .016). For the free-TFPI antigen, the optimal cut-off value was 37.65 ng/mL with a sensitivity of 40.43% and specificity of 82.61% (AUC 0.627, 95% CI 0.513-0.740, P = .035). For the PT, the optimal cut-off value was 12.05s with a sensitivity of 72.92% and specificity of 47.92% (AUC 0.617, 95% CI 0.505-0.730, P = .048). For the DD, the optimal cut-off value was 3.825 mg/L with a sensitivity of 85.42% and specificity of 51.11% (AUC 0.624, 95% CI 0.505-0.744, P = .039). For the TFPI correlation coefficient, the optimal cut-off value was 0.370 with a sensitivity of 64.44% and specificity of 89.36% (AUC 0.821, 95% CI 0.734-0.907, P <.001).
In this study, we developed a method to quantify and evaluate the anticoagulant activity of TFPI in plasma based on the dPT experiment that extends the clotting time. This method uses plasma turbidimetry to monitor and graph, in real time, changes in plasma absorbance over time. The graphs were used to compare differences between the plasma coagulation curve with and without TFPI antibody and calculate the relevant parameters of the first derivative curve to evaluate TFPI anticoagulation ability and quantify the anticoagulant effect produced by TFPI in plasma. We also established the reference range for this method in healthy people and found that the parameters were not significantly different in age and gender groups.
Research findings in recent years on the anticoagulant effect of TFPI can be divided into two parts.8,9 The first part is inhibition of FXa and TF-FVIIa which is considered to occur in two steps. The first step is the combination of TFPI- Kunitz domain 2 and FXa, leading to inhibition of FXa's coagulation function. The second step is to combine TFPI- Kunitz domain 1 in the tissue factor pathway inhibitor-activated coagulation factor X (TFPI-FXa) complex with FVIIa in the TF-FVIIa complex to form the TF-FVIIa-TFPI-FXa quadruplex complex. The binding of TFPI and FXa is the rate limiting step. Due to the presence of the C-terminus in full-length TFPI, the rate at which it binds and inhibits FXa increases. Therefore, compared with truncated TFPI, full-length TFPI can form a TFPI-FXa complex faster, resulting in a more rapid and effective anticoagulation effect, thereby prolonging the clotting time. 18 The second part is the inhibitory effect of TFPI on prothrombinase, which was discovered in recent years and represents the unique anticoagulant function of full-length TFPI. 19 Studies have shown that the anticoagulant activity of TFPI depends mainly on full-length TFPI,20,21 and therefore we used full-length TFPI antibody in the test. Because the TFPI anticoagulant activity measured is due mainly to the effect of full-length TFPI, the test may not be sensitive to truncated TFPI. The anticoagulant activity of TFPI can therefore be regarded as a measurement of the anticoagulant activity of free full-length TFPI.
At present, the commonly used methods for TFPI are the double antigen sandwich method for measuring the antigen level of TFPI and the substrate color method for assessing the ability of TFPI to inhibit the TF-FVIIa complex.22,23 The free-TFPI antigen detection method does not evaluate the true anticoagulant effect of TFPI in the body, while the TFPI substrate color method does not distinguish between full-length and truncated TFPI. Therefore what is detected is only part of rather than the entire anticoagulant effect of TFPI. 24 However, the detection method has the advantage of measuring the anticoagulant effect in the whole body, which has greater biological relevance.
We then applied this test to trauma patients and compared the anticoagulant activity of TFPI between trauma patients and healthy people. We found that the anticoagulant activity of TFPI in trauma patients was significantly higher than that in healthy controls, a finding consistent with the results of Keyloun's study. 13 The reason for the increased anticoagulant activity of TFPI in patients with trauma may be related to extensive injury of vascular endothelial cells and inflammation caused by traumatic fractures. There is no doubt that traumatic fracture as an external force leads to endothelial cell activation and injury. 25 TFPI is expressed and released mainly by vascular endothelial cells, 26 with its main pool related to the surface of endothelial cells, 27 and is speculated to play a major role in regulating the function of vascular wall cells. Previous studies have shown that during the clinical course of DIC, the level of plasma free-TFPI antigen increases in conjunction with the increase in the plasma TF antigen level.28,29 This indicates that plasma TFPI is released from the inside or surface of vascular endothelial cells. Kato et al. 27 found an inverse relationship between the level of high density lipoprotein cholesterol and free TFPI before and after administration of heparin, a finding that reflects the compensatory enhancement of vascular wall antithrombotic properties to maintain normal endothelial function. The inverse relationship between lipoprotein lipase and free TFPI after heparin indicates competitive sharing of the binding sites of heparan sulfate proteoglycans on the endothelial surface. These results suggest that full-length TFPI in plasma may be a marker of endothelial cell dysfunction. Basic experiments carried out by Zhao et al. 30 found that TFPI can reduce endothelial cell injury by inhibiting endothelial autophagy and the class III PI3K/Beclin-1 pathway, while in clinical studies, Keyloun et al. 13 used TFPI as a biomarker of endothelial injury and showed that the high level of TFPI in burn patients was related to a nine-fold increase in mortality (OR:9.59), and provided a predictive index of burn mortality. On the other hand, the inflammation caused by trauma has always been the focus of research attention, with studies showing that the levels of pro-inflammatory factors in plasma of post-traumatic patients were increased significantly. 31 In recent years, the relationship between TFPI and inflammation has also been studied extensively.32,33 Ameri et al. 34 found in endothelial cell experiments that the inflammatory mediators (phorbol myristate acetate, endotoxin, interleukin-1, or tumor necrosis factor-α) stimulated endothelial cells to express high levels of TFPI mRNA and release high levels of TFPI into the supernatant. This indicated that unlike thrombomodulin and plasmin synthesized by endothelial cells, TFPI synthesis was not down-regulated, or even slightly up-regulated during inflammation. Cibor et al. 35 then reported that the level of free-TFPI antigen in patients with inflammatory enteritis and acute pancreatitis was significantly higher than that in healthy controls, with the concentration of plasma TFPI related to the severity of disease and prognosis. These results suggest that the increase in the anticoagulant activity of TFPI in patients with trauma may be related to the release of inflammatory mediators after trauma. However, further investigations are needed to determine the reasons for the increase in TFPI levels in trauma patients.
DVT is one of the most common complications of traumatic lower extremity fractures. 1 At present, the diagnosis of post-traumatic DVT relies of invasive angiography, 36 and there is a lack of new biomarkers for accurate assessment of coagulation status and diagnosis of DVT. We therefore divided the trauma patients into DVT and non-DVT groups. A case-control study by Dham et al. 12 showed that a decrease in the TFPI concentration was a risk factor for DVT. Interestingly, in contrast with the results of Dham et al. we observed that the anticoagulant activity and antigen level of TFPI in the DVT group were significantly higher than those in the non DVT group, consistent with the results of Sidelmann et al.. 14 This variability in results may be due to the different pathophysiological mechanism of DVT caused by different diseases. For example, Dham et al. 12 studied the relationship between the occurrence of DVT and TFPI, Sidelmann et al. 14 studied acute DVT patients, while our study focused on the occurrence of DVT in trauma patients. As expected after trauma, TFPI was not depleted due to hypercoagulability, although the anticoagulant activity and antigen level of TFPI increased. We made the following assumptions to elucidate the cause of the increase of TFPI level in patients with DVT after trauma. First, we speculated whether the relationship between the occurrence of DVT and the increase of TFPI concentration after trauma was related to the injury of vascular endothelial cells. In previous studies, our team observed that endothelial dysfunction in trauma patients played an important role in the occurrence and development of DVT. Thrombomodulin (TM), as an endothelial cell membrane protein, was increased significantly in trauma patients with DVT. 37 In addition, the content of endothelial tissue factor microparticles was increased significantly in post-traumatic DVT patients (results not published). On one hand, the increase of TFPI level may be a stress reaction to the hypercoagulable state in trauma patients with DVT, and on the other hand, may reflect the degree of endothelial cell damage, one of three major factors of thrombosis. We subsequently speculated on the potential impact of inflammation on the relationship between DVT and TFPI after trauma, because TFPI can be produced by over-reaction to inflammatory stimuli and it has been reported that inflammation is related to the formation of DVT after trauma. The above discussion needs to be verified by follow-up studies.
Although the current research study achieved its objectives, it had certain limitations. Because of the low DVT positive rate in trauma patients and the difficulty of sample collection, the sample size and the number of detection indicators were relatively small. In order to prevent interference from other factors such as surgery and drugs, no samples were collected at consecutive time points. Therefore, in order to verify our results, a large-sample, multicenter cohort study needs to be conducted. At present, only the potential relationship between the occurrence of DVT in trauma patients and changes of TFPI have been studied. In order to clarify the relationship between TFPI and trauma and the complication of DVT, in-depth basic research is needed in the future, such as cell experiments and study of post-traumatic mouse models of DVT. The results of the current study lay the foundation for future research directions.
Conclusions
In conclusion, trauma patients have a risk to suffer DVT, in which TFPI plays an important role. We consider that the risk of posttraumatic DVT may be correlated with increased anticoagulant activity of TFPI and endothelial injury. We propose the anticoagulant activity of TFPI can be used as a potential biomarker for diagnosing DVT in trauma patients.
Supplemental Material
sj-docx-1-cath-10.1177_10760296211063877 - Supplemental material for The Impact of Anticoagulant Activity of Tissue Factor Pathway Inhibitor Measured by a Novel Functional Assay for Predicting Deep Venous Thrombosis in Trauma Patients: A Prospective Nested Case-Control Study
Supplemental material, sj-docx-1-cath-10.1177_10760296211063877 for The Impact of Anticoagulant Activity of Tissue Factor Pathway Inhibitor Measured by a Novel Functional Assay for Predicting Deep Venous Thrombosis in Trauma Patients: A Prospective Nested Case-Control Study by Xiangyu Cao, Yu Su, Wenjie Zhang, Huiru Zhao, Meng Wen, Shan Lu, Yujing Zhao, Yuying Chen, Lei Liu, Xinwei Zang and Jun Wu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Note
Xiangyu Cao and Jun Wu designed and performed the research. Xiangyu cao, Yu Su, Wenjie Zhang, Huiru Zhao, Yujing Zhao, Meng Wen, Shan Lu, Lei Liu and Xinwei Zang acquired the data. Yu Su, Huiru Zhao, Yujing Zhao, Meng Wen, Shan Lu, Wenjie Zang, Yuying Chen and Lei Liu provided valuable technical assistance. Xiangyu Cao and Jun Wu analyzed and interpreted the data and wrote the manuscript. All authors reviewed and made critical revisions and approved the final version of the manuscript. The study was conducted with approval of the Institutional Ethics Committee of Beijing Jishuitan Hospital. (NO.202004-89)
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
