Abstract
Background:
Factor V (FV) Leiden mutation-related activated protein C resistance (APCR) is one of the common inherited risk factors for venous thromboembolism (VTE) in caucasian population. Although APCR could be identified in some of the Chinese healthy people and patients with VTE, it was not related to FV Leiden mutation. In 2008, we have identified a novel FV mutation (FV E666D) in exon 13 in a hereditary APCR family. And we presumed that the novel mutation might be a genetic defect of APCR in the Chinese population. The aim of our study was to evaluate the prevalence of FV E666D mutation and its correlation with APCR in the Chinese population in a larger series.
Methods:
From June 2009 to January 2011, 163 consecutive patients who underwent thrombophilia tests in our hospital were recruited. The clinical data were retrospectively reviewed. Thrombophilia tests included APCR, anticoagulant proteins, and antiphospholipid antibodies. Factor V E666D mutation was detected.
Result:
Of the 163 patients, 6 (3.7%) were identified as APCR positive, 2.9% for patients without thrombosis and 5.1% for patients with thrombosis or thrombosis history. Factor V E666D mutation was not detectable in all the 163 patients including 6 APCR-positive patients.
Conclusions:
The prevalence of APCR either in the nonthrombotic patients or in the patients with thrombosis was lower than that reported in other Chinese studies. Our study couldn’t provide illustration whether FV E666D mutation is correlated with APCR in the Chinese population.
Introduction
Venous thromboembolism (VTE) is a common disease with high mortality and morbidity. 1–2 Pathophysiology of VTE is multifactorial and involves environmental, acquired, and genetic factors. 3–4 In 1993, Dahlbäck and colleagues described a new familial thrombophilia, hereditary activated protein C resistance (APCR). 5 One year later, a missense mutation at nucleotide 1691 in the factor V (FV) gene (FV Leiden mutation) was found by several groups simultaneously. It was found that the mutation was responsible for the substitution of arginine (R) to glutamine (Q) at amino acid 506, 1 of 3 sites where activated protein C (APC) normally cleaves and inactivates procoagulant-activated FV (FVa). 6 –8 Later studies showed that FV Leiden contributed to APCR phenotype and was the most common genetic defect associated with venous thrombosis. 9 –12 Other genetic abnormalities in the FV gene have been linked with APCR independently or in association with the FVL mutation. These mutations include Cambridge Arg306, Hong Kong Arg679, and the haplotype R2 polymorphism. 13
Actually, any reason that influences protein C (PC) system can cause APCR, whether hereditary or acquired. Hereditary APCR mainly focuses on FV mutation. High-risk factors such as antiphospholipid antibody, tumor, infection, pregnancy, and childbirth may disturb PC system in different ways and lead to acquired APCR in the end. The mechanisms include decreasing the degradation rate of FVa, detering the formation of complex that can hydrolyze FV rapidly, inhibiting the activation of protein C by thrombin, and so on. Protein S (PS) and thrombomodulin (TM) are important cofactors that assist APC to inactivate FV and FVIII. Protein S forms 1:1 molecular complex by uniting APC on the surface of phosphate vesicles, enhance the activation rate of FV and FVIII, and strengthen the anticoagulation. Thrombomodulin is a glycoprotein on the surface of vascular endothelial cells. It develops biological effect as thrombin receptor. When combined with thrombin, it alters the procoagulant activity of thrombin and activates PC. Any reason that disturbs PS and TM may cause APCR too. The prevalence of FV Leiden mutation is quite high in Europe, especially in the caucasian population 14–15 but it is extremely low in the Asian and African populations. 16 –18 Only 1 family with APCR and FV Leiden mutation has been reported so far in China. 19 Therefore, it is presumed that some genetic and/or acquired factors other than FV Leiden mutation might contribute to APCR in the Chinese population. In 2008, Cai identified a novel gene mutation in FV exon 13 (FV E666D) in a hereditary APCR family. 20 This mutation was detected in the proband with acute VTE and other family members with APCR but was absent in the remaining family members without APCR and in the healthy controls. This study suggested that the FV E666D mutation might be responsible for the inherent APCR and high risk of VTE in the Chinese population.
In this preliminary study, we looked for the FV E666D mutation in a larger series of Chinese patients with and without a history of thrombosis to evaluate the prevalence of FV E666D mutation and its correlation with APCR in Chinese population.
Methods
Study Population
A total of 163 consecutive patients who underwent thrombophilia tests in Peking Union Medical College Hospital from June 2009 to January 2011 were recruited. All patients gave their written informed consents according to the treatment protocol, which was approved by the Peking Union Medical College hospital institutional review board.
Activated protein C resistance assays
Activated protein C resistance was measured using the Coatest APCR V kit (Chromogenex: Orangeburg, NY, USA) in combination with the STA-R analyzer (Diagnostica Stago company, France), and the procedure was performed as described by the manufacturer. Activated protein C resistance was measured in an activated partial thromboplastin time (APTT)-based assay after dilution of the patient plasma in factor V-deficient plasma to which polybrene was added as a heparin neutralizer. This test measures the ratio of APTT in the presence and absence of exogenous APC. Activated protein C resistance was defined as the ratio less than or equal to cutoff value of 2.0, which was established in our laboratory previously. 20
Antiphospholipid Antibody Assay
Levels of anticardiolipin (aCL) antibody and anti-β2-glycoprotein I antibody (β2-GPI) were determined by enzyme-linked immunosorbent assay using separate QUANTA Lite Test Kits for isotypes (INOVA Diagnostics, Inc, San Diego, California). Lupus anticoagulant (LAC) was detected according to the guidelines of the International Society on Thrombosis and Haemostasis. Dilution Russell Viper venom time was determined by LAC Screen and confirm assay kits (Rainbow Scientific Inc, Connecticut). All the testing for aPL was performed according to instructions of the assay’s manufacturer.
Assay of Plasma Activity of Antithrombin, PC, and PS
Antithrombin, PC, and PS were measured on a STA-R Instrument (Diagnostica Stago, France) using the same batches of reagents (Diagnostica Stago). Activity of PS was assayed by a clotting method (STA-Staclot Protein S). The activities of AT and PC were assayed using a chromogenic substrate method (STA-Stachrom AT-III and STA-Stachrom PC, respectively).
DNA Extraction and Detection of FV E666D Mutation
DNA was extracted from blood samples for all patients using RelaxGene Blood DNA System by Tiangen Biotech, China. The amplification primers were synthesized corresponding to sequences upstream (5′-TGCCACAATGGATTATTGTG-3′) and downstream (5′-AGAAACGAATTCAGTGCCAT-3′) of the exon 13 sequence. For each polymerase chain reaction (PCR), a total of 100 ng DNA template was amplified at 95°C (5 minutes), then 95°C (30 seconds), 64°C (40 seconds), and 72°C (40 seconds) for 30 cycles, using 20 μmol/L primers and 2 × PCR Master Mix (Tiangen Biotech, China). A final extension step was performed at 72°C for 10 minutes. The amplification reactions were performed in a Bio-RAD cycler (PCR, Chercules, California). Finally, the amplified products were directly sequenced (ABI Prism 377 DNA Sequencer, Applied Biosystems, London, United Kingdom).
Results
Clinical Characteristics
A total of 163 participants consisting of 96 males and 67 females were enrolled in the present study. Among 163 individuals who underwent APCR testing, 59 (36.2%) presented with thrombosis whereas 104 (63.8%) did not. In 59 patients with thrombosis, the more common thrombotic episodes were VTE including deep vein thrombosis (DVT) and pulmonary embolism (PE). Other episodes were visceral or portal vein thrombosis and arterial thrombosis including cerebral and myocardial infarction (Table 1). The median age was 39 years (ranged 11-73 years). The remaining 104 individuals without thrombosis underwent APCR testing for various reasons, such as family history with thrombosis, pregnancy loss, or elevated
Demographic Data and Type of the Thrombosis in 59 Patients With Thrombosis.
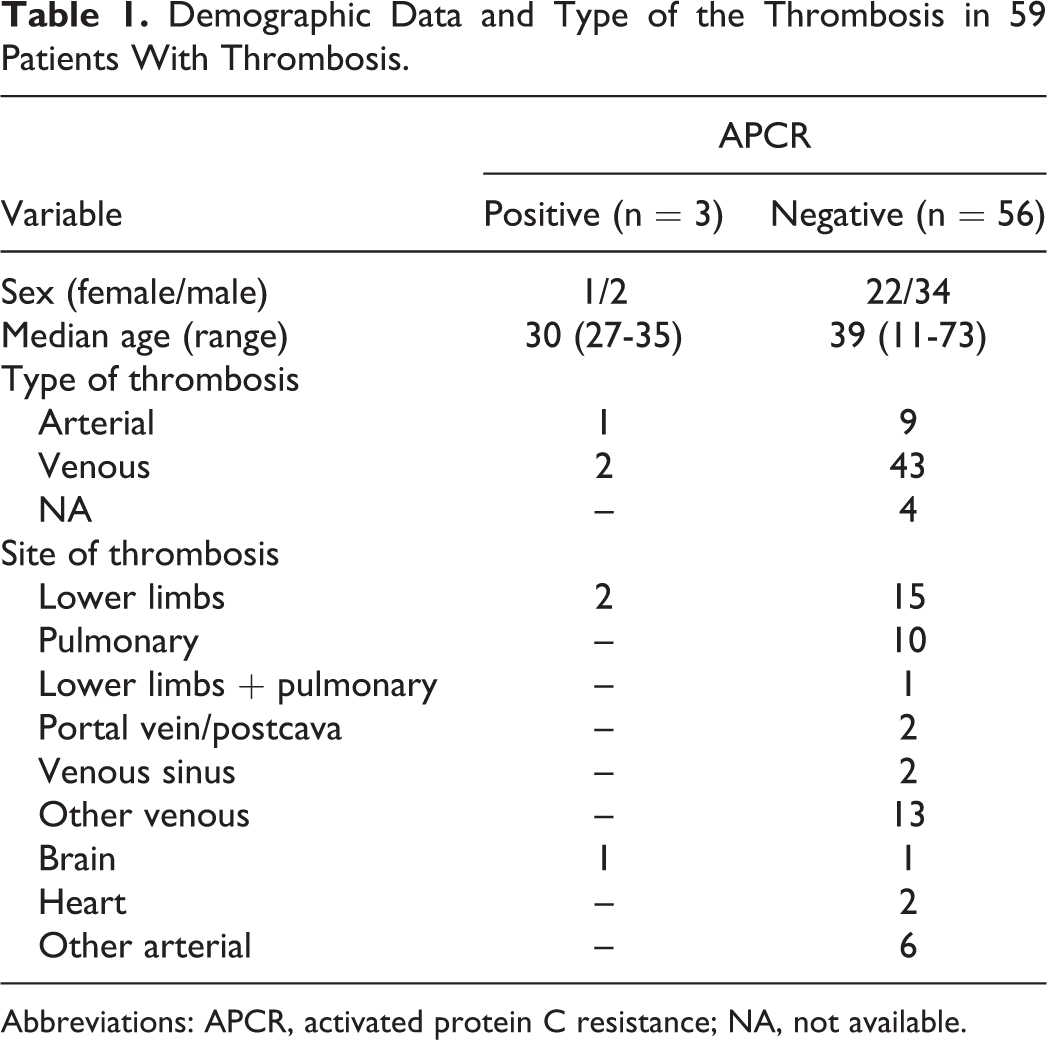
Abbreviations: APCR, activated protein C resistance; NA, not available.
Prevalence of APCR
Of the 163 patients, 6 (3.7%; 1 woman and 5 men) were APCR positive. The prevalence of APCR was higher in patients with thrombosis or thrombosis history (3/59, 5.1%) than in those without thrombosis history (3/104, 2.9%) but the difference was not statistically significant (P > .1). All the patients with positive APCR had negative results of aPL antibody tests and were excluded from the deficiencies of AT, PC, or PS.
FV E666D Mutation in Patients With APCR
All 163 individuals enrolled in this study underwent genetic analysis. No FVE666D mutation was detectable in patients with APCR and without APCR.
Discussion
Activated protein C resistance related to thrombosis closely. High-risk factors for acquired APCR give rise to thrombus through different links. For example, aPL antibodies disturb platelet membrane phospholipids, inhibit the free prostaglandin I2, activate the platelet, reduce the AT activity, weaken the anticoagulant function, interfere with the generation of kallikrein and plasminogen activator, and reduce the fibrinolytic activity. Particularly, the putative or known mechanisms for APCR are not due to FV Ledien, for example, oral contraceptives might be also responsible for the phenomenon. In Western countries, APCR has been recognized as the most frequent independent risk factor for (venous) thrombosis known to date, which is present in 2% to 5% of the general population, while 20% to 60% of patients with VTE. However, the findings of the previous Chinese studies had showed that APCR was detected in the healthy population with the frequency of 2.86% to 5.8% and in patients with VTE of 12% to 22.31%, 21 which is similar to those in the Western countries. In our study, only 6 (3.7%) patients were positive among all the patients who underwent APCR testing. Three (5.1%) patients were detected among the patients with thrombotic history, while the other 3 (2.9%) patients were detected among the patients without thrombotic history. The frequencies of APCR in these 2 groups were lower than that of the most of the previous Chinese studies. There were some possible reasons for the inconsistent results. First, the studies reported previously in China used the original assay for APCR in which the result was expressed as a ratio between a baseline APTT and the PTT after adding purified APC. A variety of factors including therapies can interfere with this test, rendering it relatively insensitive and nonspecific to diagnose APCR. 22 In the present study, new modified assays had been used. The sensitivity and specificity for the diagnosis of FV Leiden are approaching 100%. First, the modifications involve diluting the patient plasma 1:5 into FV-deficient plasma before performing the PTT or clotting time (CT) reactions, thus minimizing the effect of coagulation factor deficiency or elevation on the baseline PTT, especially FVIII. Second, adding a heparin neutralizer to eliminate the disturbance of heparin, low-molecular weight heparin, and pentose. Third, mixing FX-activator and Ca2+ to launch downstream coagulation directly, refrain from the influence of coagulation factors in the upstream coagulation cascade. Another important factor is the aPL antibody that can cause falsely abnormal (low) ratio results. In our study, aPL antibodies including aCL antibody, β2-GPI antibody, and LA were detected, but the interferences from aPL antibodies were excluded. These may explain the relative low prevalence of APCR in our study. Second, majority previous studies didn’t repeat APCR test to exclude the acquired APCR. The reasons for the acquired APCR are aPL antibodies, deficiencies of natural anticoagulants, tumor, infection, pregnancy, and hyperhomocystinemia. In our study, all the 6 patients with positive APCR were aPL antibody negative and their plasma levels of AT, PC, and PS were normal.
Although the APCR phenotype is not rare in the Chinese population, its role in thrombotic events is unclear and controversial. Moreover, in contrast to the caucasian population, it seems that the APCR phenotype in the Chinese Han nationality is not related to FV Leiden mutation. So far, only 1 Chinese family has been confirmed to have FV Leiden-related APCR. In 2008, we identified a novel mutation of FV (FV E666D) in a Chinese family with a venous thrombotic phenotype and hereditary APCR. 20 This mutation is predicted to result in the replacement of glutamate by aspartate at position 666, which is very close to the position 679. We speculate that this mutation might interfere with cleavage by APC at Arg679, which may result in reduced susceptibility of the variant FVa to APC-mediated inactivation. The mutation with a heterozygous state existed in all the APCR-positive family members but was not detectable in APCR-negative ones. Therefore, we presumed that the hereditary FV gene mutation is responsible for APCR in this family and even might be one of the genetic defects that cause APCR in the Chinese population. However, in our present preliminary study, no carrier of the FV E666D mutation was found including 6 APCR-positive patients. The limited study size and insufficient hereditary APCR samples may have contributed to this negative result.
In the present study, the age groups in the APCR+ and APCR− are quite different. The reason might be that the samples in the present study were not large enough in our epidemiological screening. If more patient samples were included, age group differences would be narrowed. In addition, the sensitivity and specificity of the assays will further improve the accuracy of the finding in the present study. However, it remains to be investigated.
In conclusion, it is still unclear whether FV E666D is responsible for APCR in the Chinese population. The well-designed and large sample studies on the prevalence of APCR should be carried out to confirm whether APCR is a risk factor for thrombosis, and FV E666D mutation is responsible for hereditary APCR in the Chinese population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from Research Fund for the Doctoral Program of Higher Education of China (20091106110007).
