Abstract
Second-generation activated protein C resistance (APC-R) assay was developed to avert interferences from lupus anticoagulant (LA) and warfarin therapy by prediluting the patient sample with factor V (FV)-depleted plasma. We investigated the effect of LA on the second generation APC-R assay in 121 LA-positive patients. Twenty-five APC-R-positive patients were tested for the mutation in FV (Leiden, Hong Kong, and Cambridge). Eleven had FV Leiden and twelve were negative for any mutation (2 were not tested). Of 12, 8 had APC-R suggestive of heterozygous and 4 had APC-R suggestive of homozygous defects. These patients had strong LA activity, compared to those with concurrent FVL. This was associated with a trend toward increased thrombosis risk compared to those with normal APC-R. These findings suggest that LA causes acquired APC-R, reflecting an in vivo pathophysiologic effect of LA rather than merely an in vitro phenomenon even with the second generation APC-R assay.
Introduction
The most common congenital and acquired causes of thrombophilia are activated protein C resistance (APC-R) and lupus anticoagulant (LA), respectively.1,2 The APC-R is a very sensitive functional assay to detect mutations in the Factor V (FV) gene, the most common being the Leiden mutation (FVL). The Cambridge and Hong Kong mutations are much less frequent. 3 The APC-R assay involves performing a partial thromboplastin time (PTT) in the presence and absence of activated protein C (APC) and calculating a ratio. Ratios of 1.7 ± 0.2 and 1.2 ± 0.2 are suggestive of heterozygous and homozygous FVL mutations, respectively. LA frequently interferes with phospholipid-based clotting tests such as the PTT. Consequently, patients with LA gave false-positive results in the first generation APC-R assay. 4 In the second generation assay, the patient’s plasma is diluted 4-fold with FV-deficient plasma to supplement all vitamin K-dependent factors for patients on warfarin. 5 This also may dilute the effect of LA. In addition, LA should prolong the PTT both in the presence and absence of APC and, thus, should not affect the APC-R ratio significantly. Current understanding is that the second-generation APC-R assay should be unaffected by both warfarin and LA.6,7 Interestingly, we observed that many patients with LA also had positive APC-R in the absence of the genetic mutation. The aim of our study was to determine the effect of LA on the second generation APC-R assay.
Materials and Methods
We analyzed data from the special coagulation laboratory at the University of Texas Southwestern Medical Center/Parkland Health and Hospital System in Dallas, TX, from January 2002 to August 2009 to identify LA-positive patients (n = 121) who were also tested for APC-R as a part of a thrombophilia work-up. The APC-R-positive patients were tested for FVL by polymerase chain reaction (PCR), and the FVL-negative patients were further tested for variant FV mutations (Hong Kong and Cambridge). The study was approved by UT Southwestern Medical Center’s institutional review board.
Lupus Anticoagulant Testing
The dilute Russell viper venom time-screen (DRVVT-S, LA1, Dade-Behring, Marburg, Germany) and partial thromboplastin time for LA (PTT-LA, Diagnostica-Stago, France) were used as screening tests for LA. 8 Prolonged DRVVT-S clotting time (>43.5 seconds) was followed with the DRVVT-confirm test (DRVVT-C, LA2, Dade-Behring), which is a DRVVT performed with excess phospholipids. Based on 40 normal healthy volunteers, we established a cut-off DRVVT-ratio (DRVVT-R = DRVVT-S/DRVVT-C) of ≥1.30 to be positive for LA. In the laboratory, the DRVVT-R also indicates the strength of LA (weak, ≤1.5; moderate, 1.5-2.0; strong, >2.0). If the DRVVT-S was normal or the DRVVT-R was not diagnostic, and if the PTT-LA clotting time was prolonged (>50 seconds), the hexagonal phase neutralization test (HPN, Diagnostica-Stago), was performed to confirm LA. Hexagonal phase neutralization test involves performing 2 PTTs on the patient’s plasma after mixing with an equal amount of normal pooled plasma: one with and one without the addition of hexagonal phase phosphatidyl ethanolamine, which neutralizes LA. An HPN test is considered positive if the clotting time corrects by >8 seconds after the addition of phospholipid.
Activated Protein C Resistance Testing
The Hemosil second-generation assay was used to detect APC-R (Instrumentation Laboratory, Lexington, Massachusetts). Patient samples are prediluted in FV-deficient plasma (4 parts FV-deficient plasma and 1 part patient plasma). The partial thromboplastin time is performed both in the presence and absence of APC, and a ratio is calculated (PTT with APC/PTT without APC). Based on 40 normal participants, we determined a ratio of ≤1.9 to be an abnormal result for APC-R. The polymerase chain reaction test is performed to confirm the FVL gene defect.
Factor V Mutation Testing
Testing for the FVL mutation was performed using the Invader assay (Third Wave Technologies, Madison, Wisconsin), while variant FV mutations (Hong Kong and Cambridge) were detected using PCR analysis. 3 Briefly, for identification of the Hong Kong and Cambridge mutations, a 228 base-pair length DNA fragment containing exon 7 of the FV gene was amplified from both control and patient DNA. Aliquots of the DNA products were digested with BstNI. Samples bearing mutations causing an arginine substitution at position 306 of the FV protein (a guanine-to-cytosine point mutation at position 1091 for Hong Kong mutations and an adenine-to-guanine point mutation at position 1090 for Cambridge mutations) would lose the restriction digestion site and remain the same length as the uncut fragments. Notably, samples containing the wild-type sequences yielded fragments of 2 lengths (66 and 162 base pairs).
Statistical Analysis
Mean, standard deviations, and t-tests were performed using Microsoft Excel 2007. Pearson correlation was performed using Sigma Plot for Windows (Ver 11.0).
Results
Of the 121 LA-positive patients (102 by DRVVT-R and 19 by HPN), 25 also tested positive for APC-R (Figure 1 ). Factor V mutation was studied in 23 of 25 APC-R-positive patients and only 11 were found to have heterozygous FVL and remaining 12 patients were homozygous for the wild type; PCR for the Hong Kong and Cambridge variants were also negative (Table 1 ). Thus, these patients (12 of 23 or 52%) had an acquired APC-R. Of these 12 patients, 8 had APC-R ratios consistent with heterozygous FVL defect (1.5-1.9) and 4 consistent with homozygous defect (1.1-1.4). The mean ratio of acquired APC-R was similar to those with APC-R due to FV mutation (1.55 and 1.65, respectively; P = .38); however, the 12 patients with acquired APC-R had significantly higher a mean DRVVT-R (2.52 vs. 1.39; P = .0022) and mean PTT-LA (147.83 vs. 61.27; P = .00081) compared to those with FVL (Table 1).
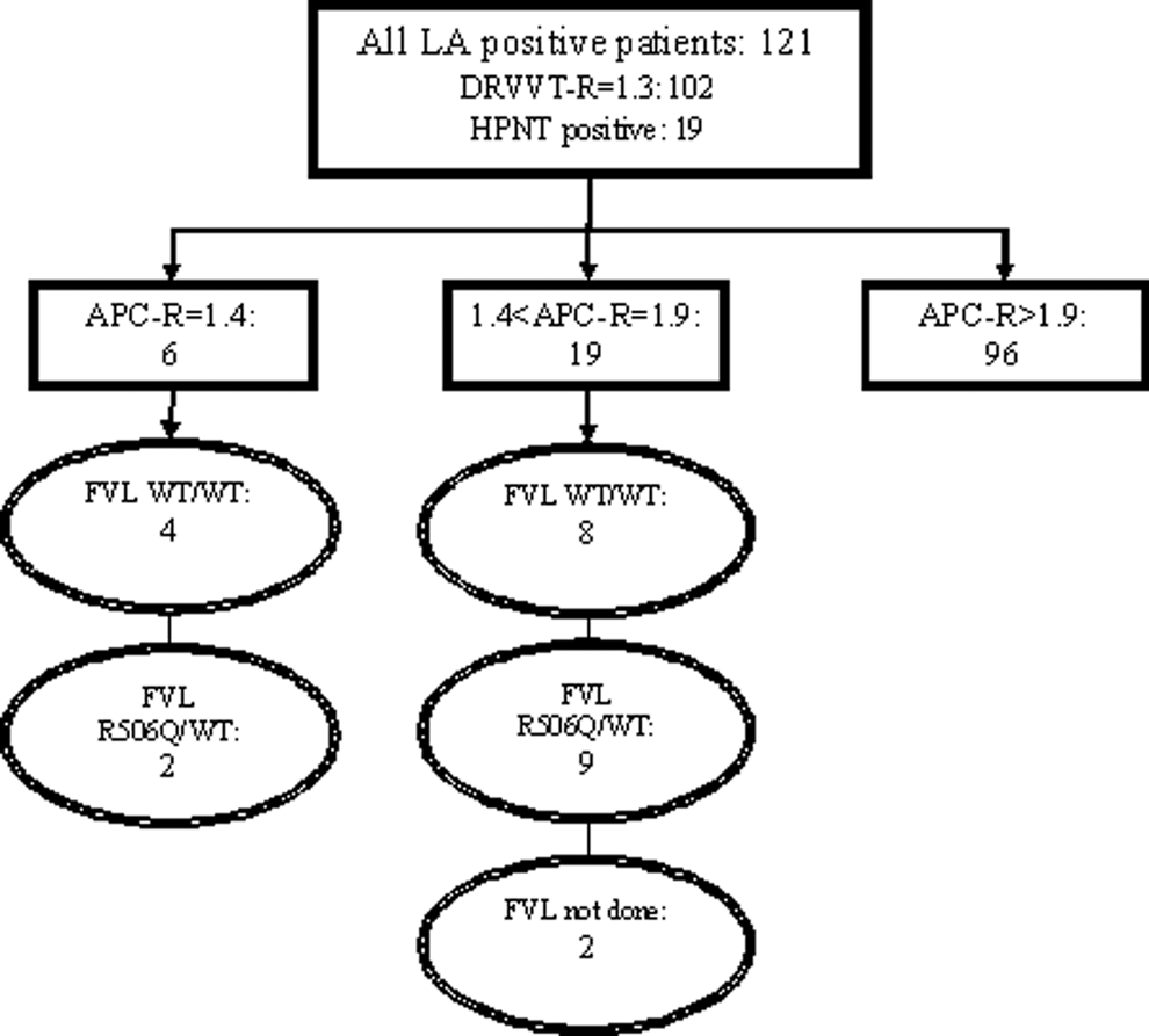
Patients With Both Lupus Anticoagulant and Activated Protein C Resistance
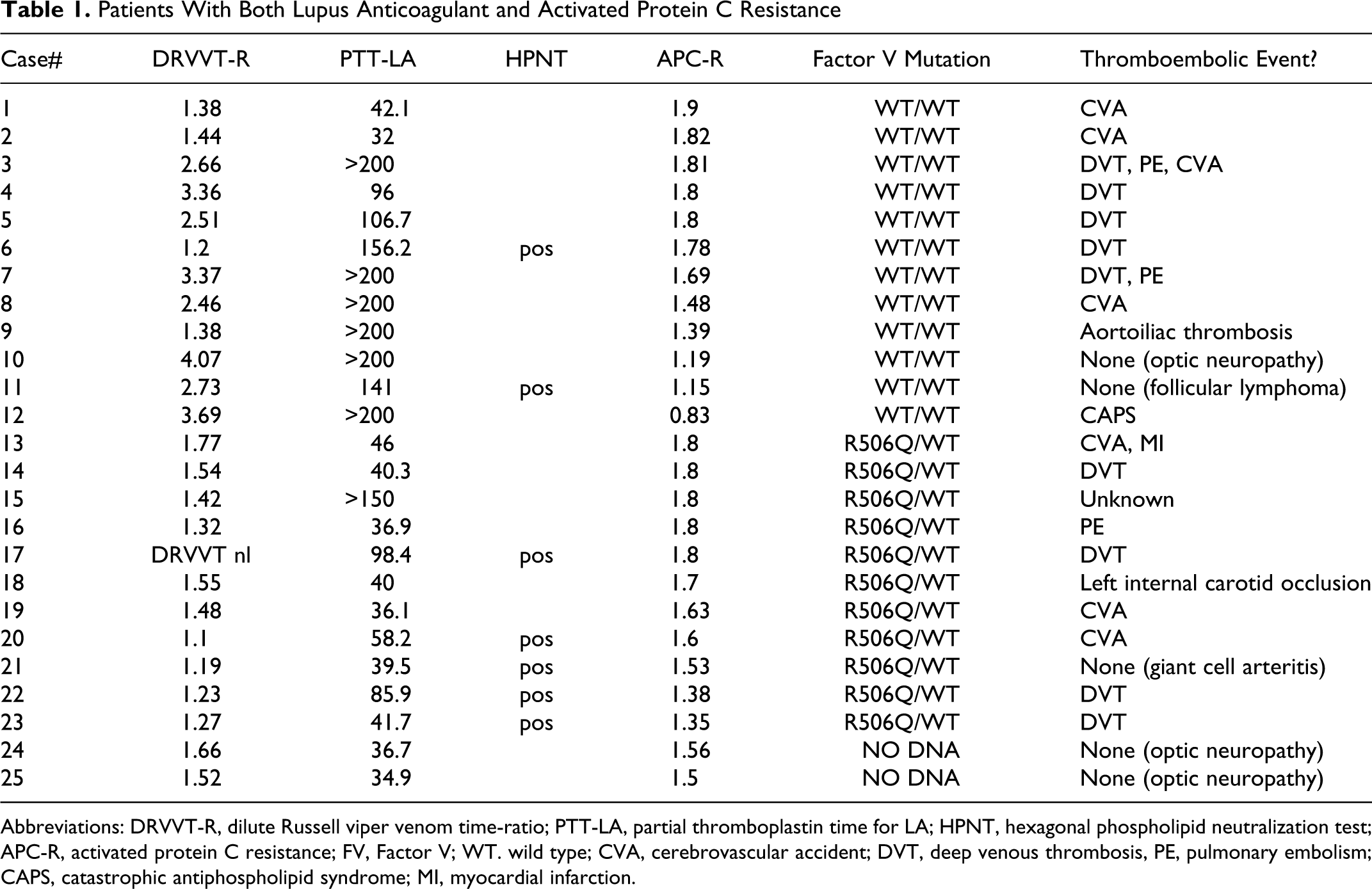
Abbreviations: DRVVT-R, dilute Russell viper venom time-ratio; PTT-LA, partial thromboplastin time for LA; HPNT, hexagonal phospholipid neutralization test; APC-R, activated protein C resistance; FV, Factor V; WT. wild type; CVA, cerebrovascular accident; DVT, deep venous thrombosis, PE, pulmonary embolism; CAPS, catastrophic antiphospholipid syndrome; MI, myocardial infarction.
A scatter plot was used to investigate correlation between DRVVT-R strength and acquired APC-R (FVL mutation and patients with no FVL testing excluded; Figure 2a). Those with DRVVT-R <2.0 had a mean APC-R of 2.30 as compared to those with DRVVT-R ≥2.0 who had a mean APC-R of 1.73 (P = .0068). Of note, 3 of 11 with DRVVT-R of ≥2.0 had APC-R ratio of >1.9 (Figure 2a top-right quadrant), indicating not all strong LA have acquired APC-R. Three patients with DRVVT-R <2.0 had acquired APC-R. Among the patients with DRVVT-R ≥1.3, there was a strong negative correlation between DRVVT-R and APC-R, with Pearson correlation coefficient of −.530 (P << .001).
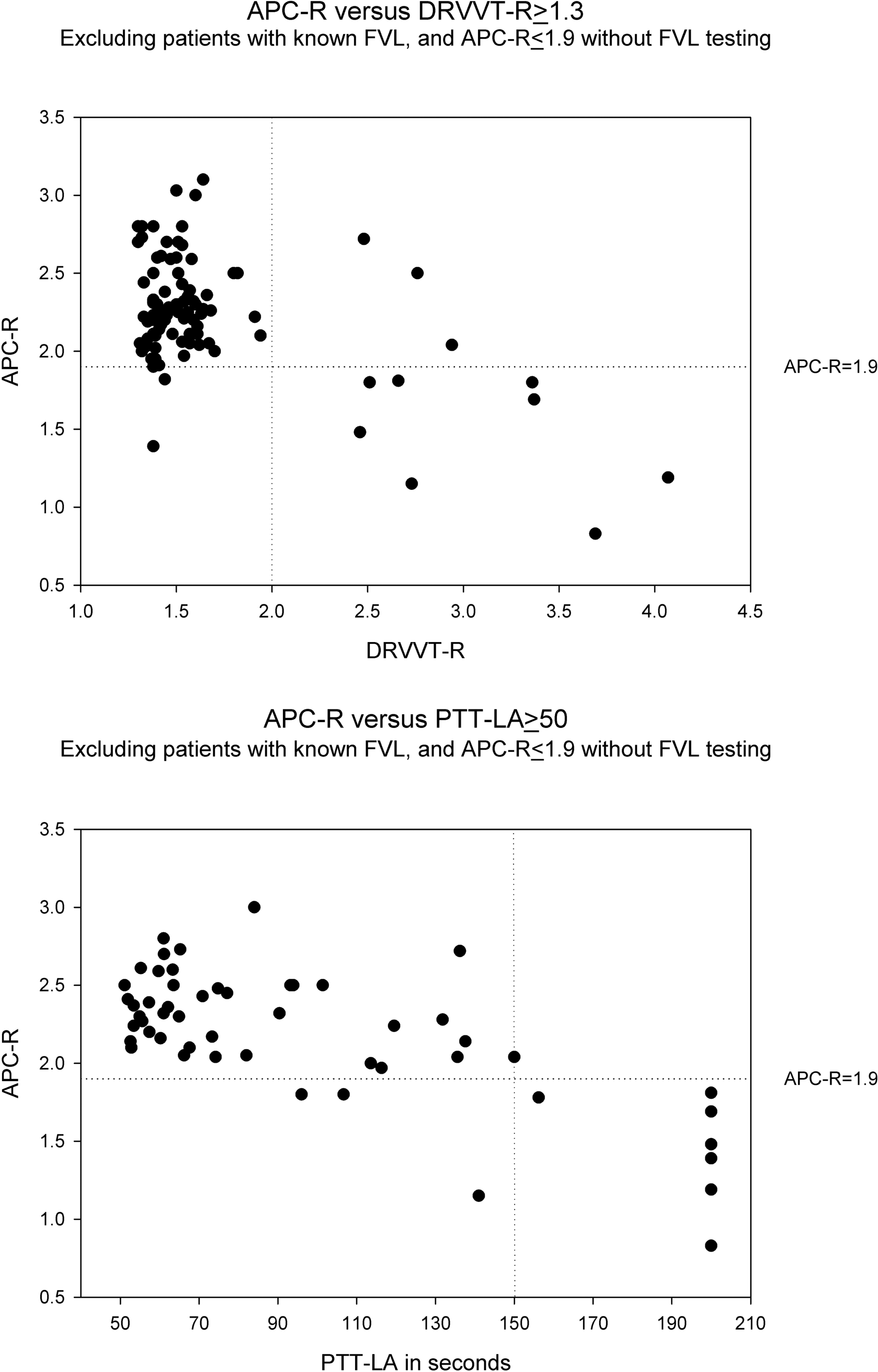
(a) Activated protein C resistance (APC-R) versus DRVVT-R ≥ 1.3. Most patients clustered in the upper left quadrant with DRVVT-R <2 and APC-R >1.9. However, in patients with DRVVT-R ≥2, most patients had APC-R <1.9, consistent with acquired APC-R. b, APC-R versus PTT-LA ≥50. Most patients clustered in the upper left quadrant with PTT-LA <150 and APC-R >1.9. However, in patients with PTT-LA ≥150, all but 1 patient had APC-R <1.9, consistent with acquired APC-R. PTT-LA reported as greater than 150 or greater than 200 were plotted as 150 and 200, respectively. DRVVT-R indicates dilute Russell viper venom time-ratio; FVL = Factor V Leiden; PTT-LA = partial thromboplastin time for LA, FVL = Factor V Leiden.
A similar analysis was done in patients with prolonged PTT-LA (≥50 seconds) and acquired APC-R (Figure 2b). Those with PTT-LA <150 seconds had a mean APC-R of 2.30, and those with PTT-LA ≥150 seconds had a mean APC-R of 1.53 (P = .00046). One of eight patients with PTT-LA ≥150 seconds had a negative APC-R, while 3 patients with PTT-LA <150 seconds had acquired APC-R. Among the patients with PTT-LA ≥50 seconds, there was a strong negative correlation between PTT-LA and APC-R, with Pearson correlation coefficient of −0.715 (P << .001).
A total of 59% (54 of 91) of the patients with positive LA and normal APC-R (>1.9) with available clinical information had a documented thrombotic event (deep venous thrombosis, pulmonary embolus, peripheral arterial occlusion, cerebrovascular accident, myocardial infarction), as compared to 83% (10 of 12) patients with positive LA and acquired APC-R (odds ratio 3.43). Patients with positive LA and documented factor V Leiden had a thrombotic rate of 90% (Table 2 ).
Thrombosis Rate: With or Without Activated Protein C Resistance
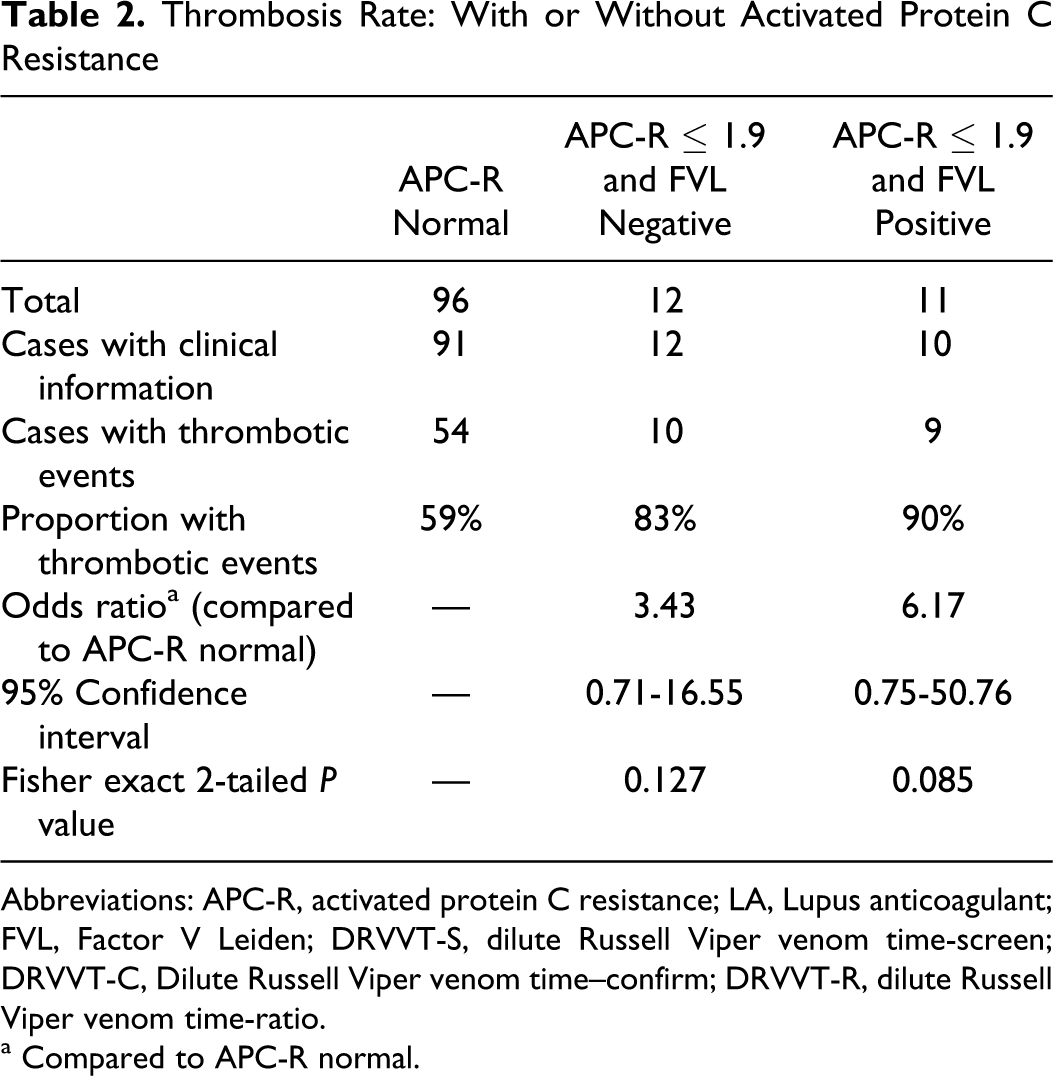
Abbreviations: APC-R, activated protein C resistance; LA, Lupus anticoagulant; FVL, Factor V Leiden; DRVVT-S, dilute Russell Viper venom time-screen; DRVVT-C, Dilute Russell Viper venom time–confirm; DRVVT-R, dilute Russell Viper venom time-ratio.
a Compared to APC-R normal.
Discussion
Our study showed that 52% of patients with LA and positive APC-R had acquired APC-R even with the second-generation APC-R assay that utilized dilution of patient plasma with FV-deficient plasma. Patients with acquired APC-R had stronger LA activity compared to those with concurrent LA activity and FVL. Furthermore, these patients had a trend toward higher thrombotic risk than those with LA alone without acquired APC-R, similar to those patients with concurrent LA activity and FVL. Since the acquired APC-R was not universally seen in patients with strong LA activity, we propose that acquired APC-R is not a false positive in vitro laboratory phenomenon, but reflects an in vivo pathophysiologic effect of LA.
In several LA-positive patients, Ragland et al observed false positive APC-R ratios that corrected with serial dilutions with FV-deficient plasma. A dilution of 1:40 appeared optimal to avoid LA interference while maintaining the capability to separate homozygous and heterozygous patients with FVL. 9 Similarly, adding platelet-derived phospholipids before the contact activation step of each PTT resulted in correction of the APC-R ratios in the first generation assays of eight LA-positive patients, but it did not in 1 patient with hereditary APC-R, likely due to neutralization of the interfering LA. 10 This suggests that phospholipid neutralization may be helpful in differentiating congenital from acquired APC-R.10,11 It has also been suggested that a dilute Russell viper venom-based APC-R assay may be resistant to LA effects. 12 Jacobsen et al proposed yet another, perhaps simpler, method for distinguishing LA from FVL as a cause of APC-R; this involves performing the APC-R assay on patient plasma in a 1:1 mix with normal pooled plasma. Normalization confirms the presence of FVL rather than LA. 13 Some investigators have hypothesized that a prolonged baseline PTT heralds false positive APC-R results in patients with LA.9,11 Nevertheless, three of our LA positive patients with very long PTT-LA (>150 sec) had normal APC-R.
The APC-R assay is simply a ratio between the two PTTs (with and without APC). Hence, both PTTs should be prolonged in the presence of LA, and the ratio should be largely unaffected. Thus, an abnormal ratio should indicate a true APC-R, either hereditary or acquired.
In vivo interference with the activated protein C pathway has been described as one possible procoagulant effect of LA. 14 Cariou et al showed that the immunoglobulin fractions from patients with LA inhibited the ability of cultured human umbilical endothelial cells to activate purified protein C. 15 Malia et al demonstrated that antiphospholipid antibodies prevented inactivation of FVa by APC and protein S by a one-stage FV clotting assay. 16 Similarly, APC-R determined by a thrombin generation assay is significantly abnormal in patients with antiphospholipid syndrome as compared to control patients. 17 De Laat et al demonstrated that all 11 plasma samples with β2-glycoprotein I-dependent LA had increased resistance to activated protein C and correlated with thrombosis, whereas only 1 of 11 samples with β2-glycoprotein I-independent LA had increased resistance to activated protein C. 18 Acquired APC-R was also detected in 31% of patients with SLE and was a risk factor for thrombosis with odds ratio of 3.36 in another recent study. 19
In our current investigation, we did not assay for antibodies with specificities against protein C or protein S, which could potentially lead to acquired APC-R. However, with 4 parts of FV-deficient plasma added to the patient plasma in the APC-R assay, the effects of any such antibody would have been diluted by the excess protein C and S. Similarly, the addition of excess FV-deficient plasma would also negate the effects of any hemostatic changes in the patient plasma due to an acute thrombotic event or the use of anticoagulant therapy. Thus, the observed acquired APC-R is thought to be a direct interference on the function of activated protein C.
In conclusion, our study showed that some patients with lupus anticoagulant have an acquired APC-R, even in the second generation assay. These findings, in light of new evidence, suggest that acquired APC-R may be an in vivo thrombotic mechanism of LA.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
