Abstract
Left ventricular (LV) systolic dysfunction and chronic systolic heart failure (HF) predispose to intraventricular thrombus formation and embolization resulting in stroke. Current guideline recommends the use of oral anticoagulants in patients with atrial fibrillation and history of previous thromboembolism. However, anticoagulant treatment in patients with LV systolic dysfunction with sinus rhythm and without history of previous thromboembolism is still on debate. Recent epidemiologic date has reported increased stroke rate in patients with systolic HF shortly after diagnosis. This review focuses on the possible causes of increased stroke rate shortly after the diagnosis of HF and subsequently suggests a rationale for the use of oral anticoagulant in these patient groups.
Keywords
Introduction
Congestive heart failure (HF) due to severe left ventricular (LV) dysfunction is the major public health problem. Beyond its high risk of mortality, systemic thromboembolism is also a major contributing factor of mortality and morbidity. Current AHA/ACC/ESC guidelines recommend anticoagulation in patients with coexistent atrial fibrillation or history of thromboembolic event. 1,2 However, there is no definite guideline recommendation in patients with LV systolic dysfunction but without atrial fibrillation and history of previous embolic events.
Left Ventricular Thrombi and Stroke
Blood stasis, endothelial dysfunction, and hypercoagulable state, which are known as triads of Wirchov, exist in patients with advanced HF (Figure 1). Stasis of blood in a poorly functioning left ventricle is thought to predispose to ventricular thrombus formation, and patients with chronic HF may have hemostatic abnormalities. Stagnant blood can be visualized by echocardiography appearing as spontaneous echo contrast or smoke in dilated atria or ventricles. Although, endothelial dysfunction, arising as a consequence of coronary atherosclerosis, is often present in patients with HF who have LV systolic dysfunction due to myocardial infarction or ischemic cardiomyopathy, local endothelial damage in the ventricular cavity may also take part to result in thrombus formation. Evidence that a hypercoagulable state exists in patients with HF is supported by the fact that elevated levels of von Willebrand factor, heightened platelet activation, and increased markers of thrombin production have all been described in patients with cardiomyopathy and in patients with LV systolic dysfunction in sinus rhythm. 3,4 Ikeda et al 5 measured the levels of molecular markers of hemostasis in 17 patients with idiopathic cardiomyopathy and compared these values with those of normal individuals. All study patients were in sinus rhythm and had no past history of systemic emboli. Plasma levels of fibrinopeptide A and thrombin–antithrombin III complex significantly exceeded levels found in normal participants. Interestingly, fibrinopeptide A and thrombin–antithrombin III complex levels correlated positively with LV end-diastolic volume and negatively with the fractional shortening of the left ventricle. 5 Erbay et al 6 have shown increased coagulation and platelet activation in patients with dilated cardiomyopathy (Figure 2A). They have also reported that this activation did not differ between patients with and without LV thrombus. Additionally, our group has also shown that mean platelet activation is significantly increased in patients with HF. Concerning to mean platelet volume, there was no significant difference between patients with and without LV thrombus. These results underline that platelet activation exists in patients with systolic LV dysfunction irrespective of echocardiographically visible thrombus. 7
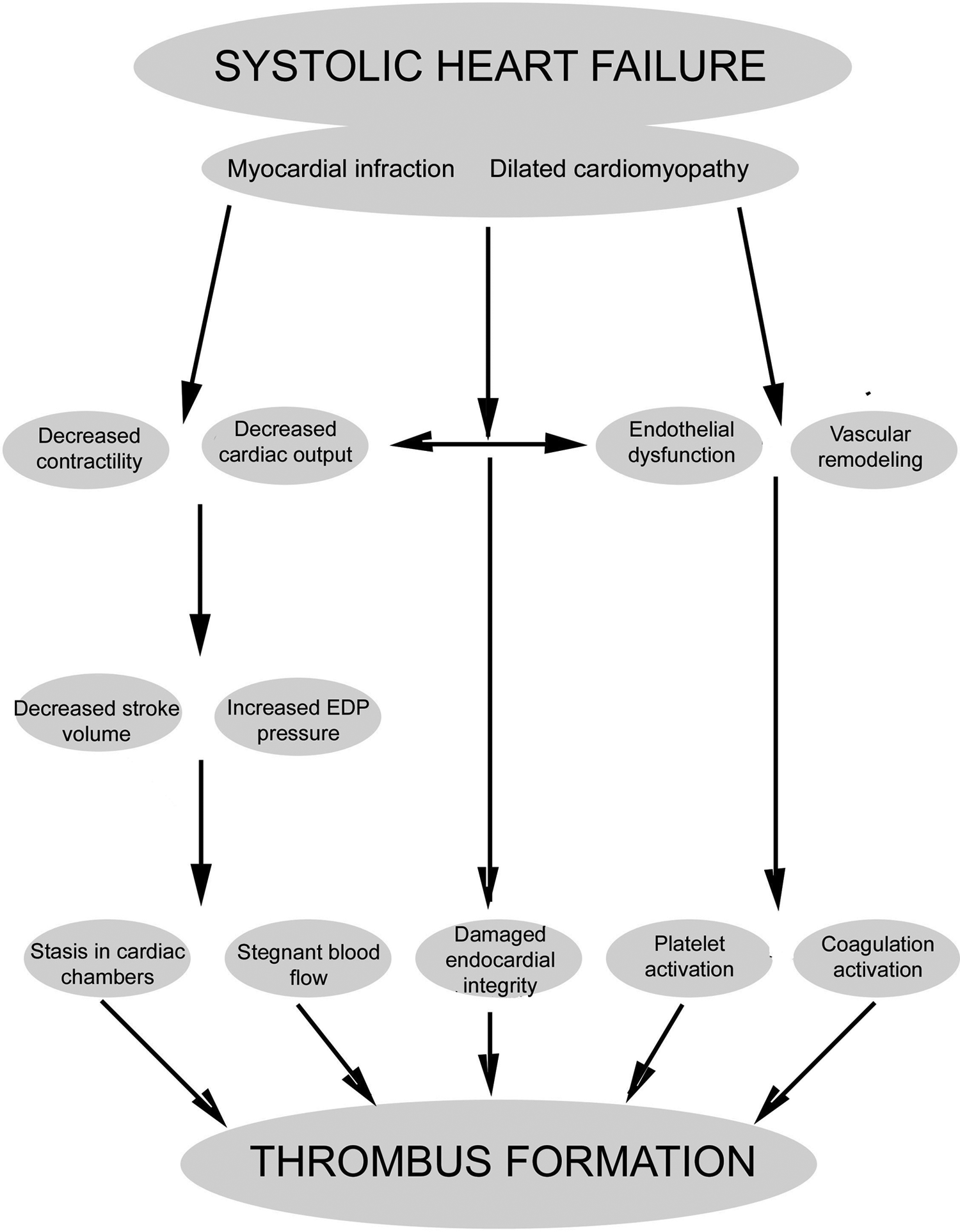
Cascade of pathophysiology in systolic left ventricular dysfunction leading to thrombus formation.
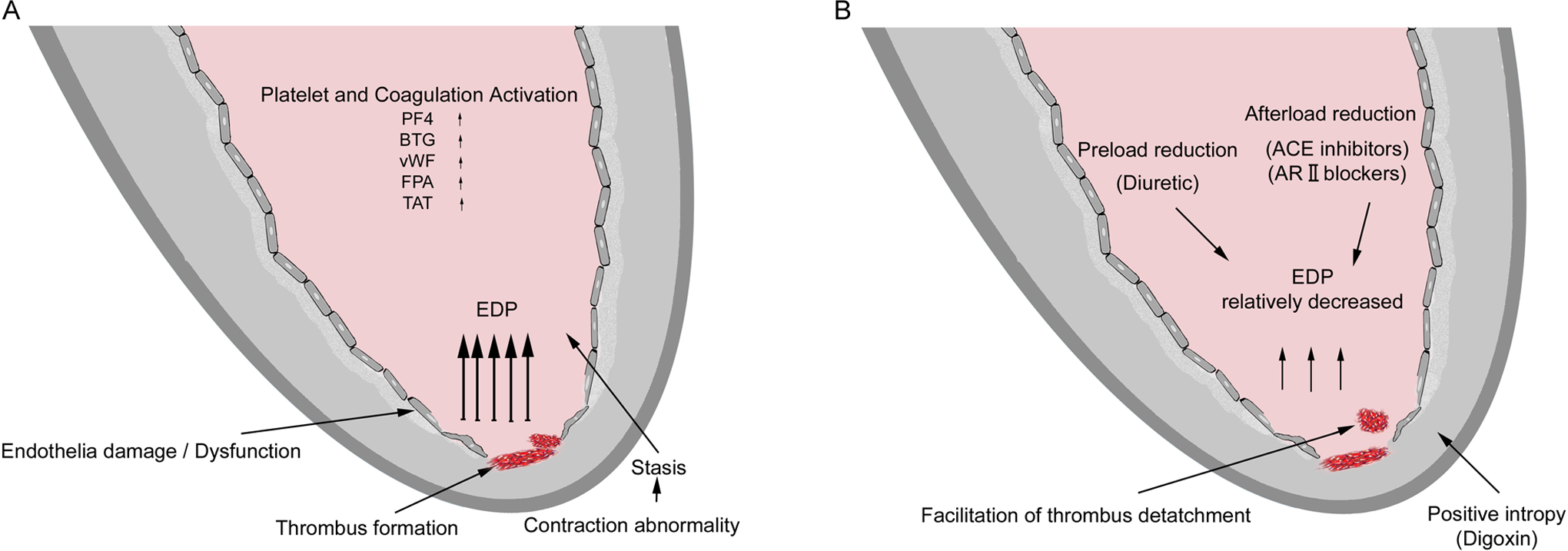
A and B, Proposed mechanism of thrombus formation and embolization in patients with systolic heart failure after diagnosis and treatment. EDP indicates end-diastolic pressure; PF4, platelet factor 4; BTG, β-thromboglobulin; vWF, von Willebrand factor; FPA, fibrinopeptide A; TAT, thrombin–antithrombin III complex; ACE, angiotensin converting enzyme; AR, angiotensin receptor.
The incidence of LV thrombus in patients with idiopathic dilated cardiomyopathy and LV systolic dysfunction ranges from 11% to 44% in the literature. 8,9 However, autopsy studies have reported relatively high rate of LV thrombus ranging from 37% to 50%. 10,11 The annual risk of systemic embolization in patients with dilated cardiomyopathy is 1.4% to 12%. 12,13 Mobile or protruding thrombi have shown to carry high risk of thromboembolism. 14 Fuster et al 8 have reported an 18% frequency of thromboembolic events in patients not taking warfarin. In the vasodilators in heart failure trial, the incidence of thromboembolism has been reported to be 2.5/100 patient years and did not differ between patients receiving and not receiving anticoagulation. 15 Furthermore, Ciaccheri et al has reported 11% prevalence of LV thrombus in patients with nonischemic-dilated cardiomyopathy, with no relation between intracavitary thrombus and systemic embolization. 13 In the survival and ventricular enlargement trial, the incidence of stroke was 1.5/100 patient years, and the risk was higher in older patients with low ejection fraction. 16,17 Interestingly, women were at greater risk of thromboembolism than men (2.4 vs 1.8 events/100 patients years) in the studies of left ventricular dysfunction trial. 18 Sharma et al have found that ischemic etiology and lower ejection fraction were both independent predictors of systemic embolism in patients with and without LV thrombi. 19 In patients with nonischemic-dilated cardiomyopathy, the rate of stroke appears similar to the rate in those with cardiomyopathy from ischemic heart disease.
Time-Dependent Changes in Stroke Rate
A few epidemiological studies have reported time-dependent risk of stroke in patients with HF. Alberts et al 20 have found a 4-fold increased risk of stroke during the month following the diagnosis of HF in a population-based cohort (7546 patients, of whom 233 had HF at baseline) that was followed since 1990. Prevalent HF at baseline was assessed using a validated score that was based on the HF definition of the European Society of Cardiology. 21 Interestingly, this risk has returned to normal by the end of 6 months follow-up in patients who had survived. Previous studies have also reported increased risk of stroke shortly after the diagnosis of HF. 22 –25 However, these studies did not have long-term follow-up and did not adjust for confounders. Witt et al have also reported 17.4-fold increased risk of stroke in the first 30 days of HF diagnosis, which decreased to 2.9-fold after 5 years in a random sample of patients with HF from Rochester followed 630 persons with incident HF for a longer period (median follow-up 4.3 years; Figure 3). Although the methodology and follow-up periods show some differences between studies, the common and clear finding is the increased risk of stroke after the first month of HF diagnosis. However, we believe that there is a misnaming or hidden part in terms of “first month after HF diagnosis.” Treatment of HF after the diagnosis has never been mentioned and thought in the epidemiologic studies. Any patients who have the diagnosis of HF with reduced LV ejection fraction concurrently would receive treatment, such as diuretics, angiotensin-converting enzyme inhibitor, angiotensin receptor antagonists, and digitalis, on the basis of clinical symptoms according to New York Heart Association functional capacity. Recently, we have reported 2 consecutive sporadic cases in whom cerebrovascular event occurred shortly after diagnosing and initiating HF treatment. Both patients were in sinus rhythm, and there was no echocardiographically visible LV thrombus formation. 26
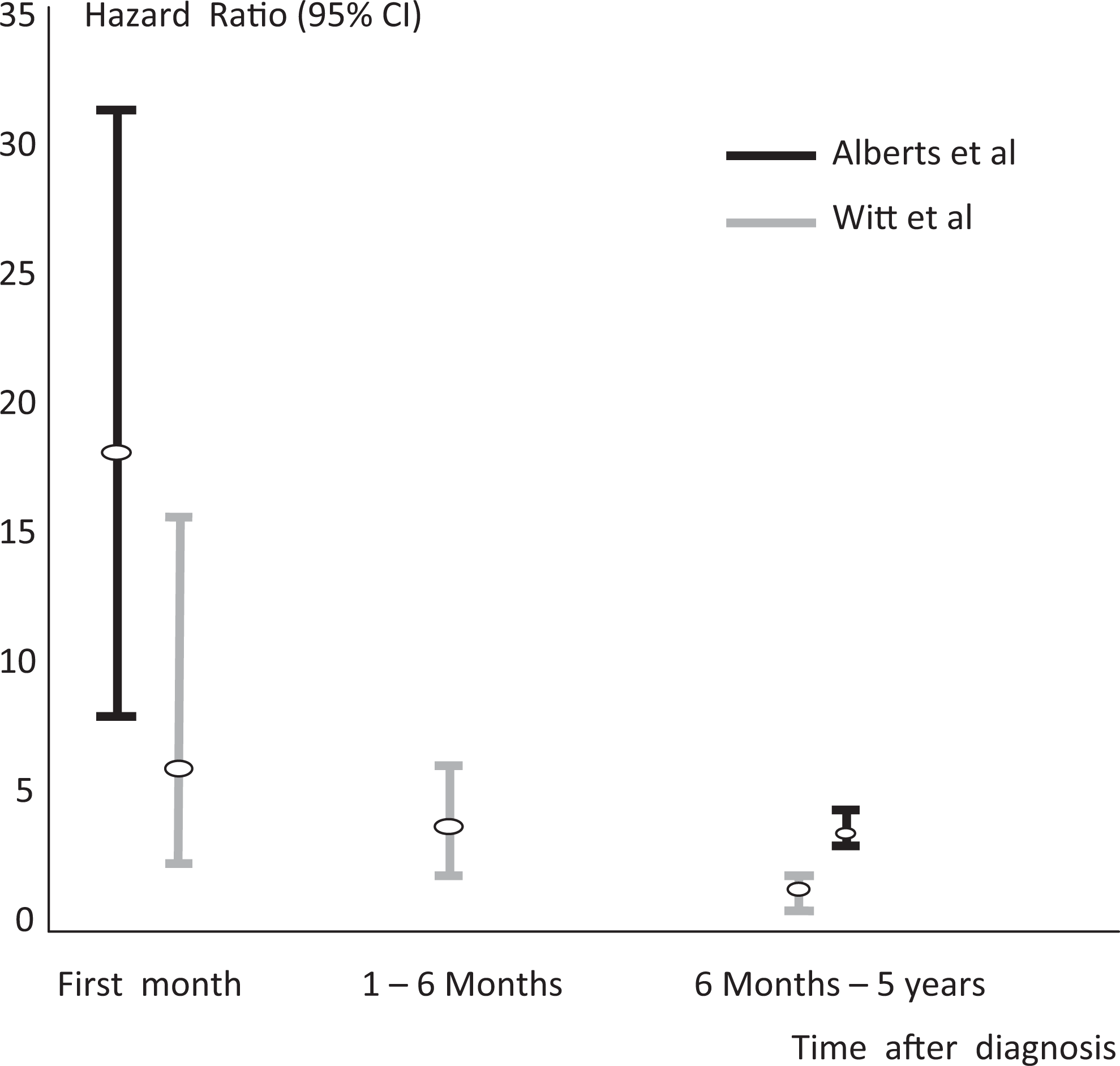
Time-dependent change in stroke risk in patients with heart failure.
Even without gross thrombus formation, these regional blood flow disturbances could lead to an increase in platelet adhesion due to prolonged residence time of platelets in the cardiac chambers. 27 At least 11% of the patients with dilated cardiomyopathy have 1 or more embolic events during the course of their illness. 12 However, echocardiographically invisible thrombus formation may also occur in the LV cavity. Roberts et al 10 have demonstrated that even in the absence of LV thrombi by echocardiography, it is possible that mural endocardial plaques, most likely resulting from organized thrombi in 25% of the patients with dilated cardiomyopathy, may exist in left ventricle. Additionally, comparably increased platelet and coagulation activation have been demonstrated in patients with and without LV thrombus compared to normal participants, indicating patients without LV thrombus carry risk of thromboembolism in terms of platelet and coagulation activation. 7 In spite of the hemodynamical and clinical improvement in patients with severe LV dysfunction, manifestation of cerebrovascular event is an unexpected and unpleasant complication. Besides the coincidental embolization risk of echocardiographically invisible or visible LV thrombus, relative improvement in LV contraction, preload, and afterload reduction, and relative decrease in end-diastolic LV pressure might have facilitated the release of freshly formed small thrombi from the LV or less likely from the left atrial cavity (Figure 2B). These local hemodynamic changes in LV cavity may lead to freshly formed microthrombus to detach easily from the endocardium, resulting in thromboembolic events. 28 Recent epidemiological data have taken the attention of possible role of HF treatment paradox in the increased risk of stroke shortly after the diagnosis. Several weeks or months shortly after the diagnosis and concurrently treatment of patients with systolic HF might be a critical time interval for patients to be anticoagulated to decrease thromboembolic events.
Warfarin Therapy in HF With Sinus Rhythm
The role of antithrombotic therapy in patients with chronic HF has been a subject of debate for many years. Anticoagulation has been shown to reduce the rates of thromboembolic events, particularly strokes in initial controlled clinical trials. However, those studies had included all spectrum of patients with HF such as atrial fibrillation and valvular heart disease and relatively low number of patients with sinus rhythm. 29 –31 Mujip et al 32 have shown in 1642 patients with systolic HF and sinus rhythm that warfarin treatment has no benefits in terms of all-cause hospitalizations and cardiovascular mortality. The last randomized study included patients who had already been diagnosed and receiving oral-anticoagulant treatment. Likewise, warfarin and antiplatelet therapy in chronic HF study results suggest that for the primary composite end point and all-cause mortality, major differences between anticoagulation with warfarin and antiplatelet therapy with either aspirin or clopidogrel are unlikely. 33 However, subgroup analysis has shown that warfarin is superior to aspirin or clopidogrel for preventing fatal and nonfatal stroke. Since the stroke rate is increased shortly after diagnosis and initiation of HF, it is reasonable to include anticoagulant in the HF treatment modalities after diagnosis for several months. However, randomized clinical studies are needed to prove the benefits of this approach.
Open Questions
Anticoagulation therapy itself is not without risk. Therefore, clinicians have to balance the benefit of risk reduction against the risk of hemorrhage with warfarin therapy in patients with systolic HF and sinus rhythm and without LV thrombi and previous ischemic stroke. Beyond the possible temporary use of warfarin in certain time interval, there are also other options that are needed to be questioned. The major examples of these newer anticoagulants are the direct thrombin inhibitors (ximelagatran and dabigatran) and the factor Xa inhibitors (apixaban and rivaroxaban). 34,35
However, on the contrary of their promising effect on ischemic event rates, safety concerns in terms of intracranial bleeding still exist. Since aspirin as a single agent has been suggested for patients with congestive HF, because many have coronary disease as the basis of their systolic HF, dual antiplatelet therapy by adding clopidogrel or prasugrel to aspirin is another option needed to be discussed as an alternate to warfarin for limited time interval shortly after the diagnosis.
Conclusion
Inconsistency continues on the use of anticoagulation in patients with systolic dysfunction, normal sinus rhythm, and without previous thromboembolic event. However, review of epidemilogic data, which show increased risk of embolic events shortly after diagnosis of HF, and case reports support the hypothesis that initiation of systolic LV dysfunction may lead to transient increase in thromboembolic events. Possible mechanisms cover the decrease in LV-filling pressure and positive inotropic effects of afterload reducing drugs and positive inotropic drug namely digoxin. This treatment might facilitate detaching of fresh thrombi from the LV cavity. Accordingly, patients with newly diagnosed LV systolic dysfunction in sinus rhythm and without previous thromboembolic events should be closely followed up for several months, and temporary anticoagulant treatment should be considered to prevent thromboembolic events.
Footnotes
Acknowledgment
The authors would like to thank Denise Newman for her creative and skillful help on the figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
