Abstract
Objective: The aim of this study was to evaluate mean platelet volume (MPV), thrombopoietin (TPO), and platelet levels in patients with ischemic stroke and compare this with healthy controls. Methods: We prospectively studied 50 patients with ischemic stroke and compared them with 37 control participants who have evaluated in internal medicine polyclinic and had no history of cerebrovascular events. All patients were within 24 hours after stroke; MPV and TPO were measured on admission. Results: Both TPO and MPV values were significantly higher in patients with stroke (P = .00; P = .001). Conclusion: Increased TPO levels may increase both platelet count and platelet size, resulting in more hemostatic tendency, which may contribute to the progression of ischemic stroke.
Introduction
Platelets have essential role in the consistence of ischemic stroke by developing intravascular thrombosis after the rupture of an atherosclerotic plaque. 1 Megakaryocytopoiesis is a process that depends on early and late acting hematopoietic growth factors. Thrombopoietin is the most critical cytokine regulator of megakaryopoiesis.2,3 Platelet proliferation and maturation is highly dependent on thrombopoietin (TPO) function.
Circulating platelets are heterogeneous with respect to their size, density of granules, and reactivity. Increased mean platelet volume (MPV) is considered as a marker of platelet function. Large platelets aggregate in vitro more rapidly and effectively to agonists like collagen and adenosine diphosphate than smaller ones; release more thromboxane and serotonin; and contain more dense granules. 4 Previous studies have shown higher MPV levels among patients with stroke, myocardial infarction, and accompanying diseases which cause vascular risk factors such as diabetes mellitus, hypertension, and hyperlipidemia4,5; however, some studies did not observe such effect.6,7 There is no available data assessing the TPO level in patients with acute ischemic stroke.
The aim of the present study was to investigate whether MPV, platelet count, and TPO are associated with acute ischemic stroke. We also studied other laboratory parameters such as fibrinogen, leukocyte, hemoglobin, and bleeding parameters and compared them.
Participants and Methods
We included consecutive patients with ischemic stroke within 24 hours after ictus, admitted to our neurology department between January 2010 and February 2011. The diagnosis was made according to World Health Organization criteria. Stroke is defined as rapidly developing clinical symptoms/signs of cerebral dysfunction lasting more than 24 hours without any cause other than a vascular abnormality. 8 Stroke subtype among all admitted patients was classified according to trial of Org 10172 in acute stroke treatment (TOAST) classification. 9 Control group consisted of 37 participants evaluated in internal medicine polyclinic for routine control.
Exclusion criteria included transient ischemic attack (TIA), hemorrhagic stroke, intracerebral hemorrhage, cranial trauma, late admission (after 24 hours of ictus), subarachnoid hemorrhage, bleeding diathesis, primary thrombosis, and medical treatment with the anticoagulant/antiaggregant drugs. In all, 25 patients with TIA and 15 patients with intracerebral hemorrhage were excluded.
Informed consent was obtained from each patient and the study protocol conforms to the ethical guidelines of the Declaration of Helsinki as reflected in a prior approval by the institution’s human research committee. The local ethic committee approved the study.
Peripheral venous blood samples of patients were drawn with 21 G multiple drawing blood collecting needles to EDTA-anticoagulated tubes rapidly on admission to our clinic. Venous samples were taken from controls without regarding the fasting level. The blood samples were centrifugated immediately at 3000 g for 15 minutes and the plasma was stored at 70 C until assayed. Coulter Gen S analyzer device was used to yield platelet parameters (Beckman Inc., Miami, Florida). Thrombopoietin (Quantikine, R&D Systems, Minneapolis, Minnesota) was assayed by sandwich-type enzyme-linked immunosorbent assays.
The Student t test was used for comparison of the platelets, MPV, and TPO levels between patients and control participants. The Spearman test was used for correlation of TPO, MPV, and platelet counts. A P value of <.05 was considered to indicate the statistical significance.
Results
In all, 50 patients (mean age of 69.5 ± 1.6 years) who had inclusion criteria and 37 control participants (mean age of 64.8 ± 1.86 years) were prospectively documented in this study. Demographic characteristics and risk factors of study group are summarized in Table 1 . There was no significant difference about age and gender. Diabetes mellitus and hypertension; but not coronary artery disease, smoking, and hyperlipidemia, were significantly different in patients when compared to controls (P = .001; P = .019).
Comparison of Demographic and Clinical Features Between Patients and Control Group
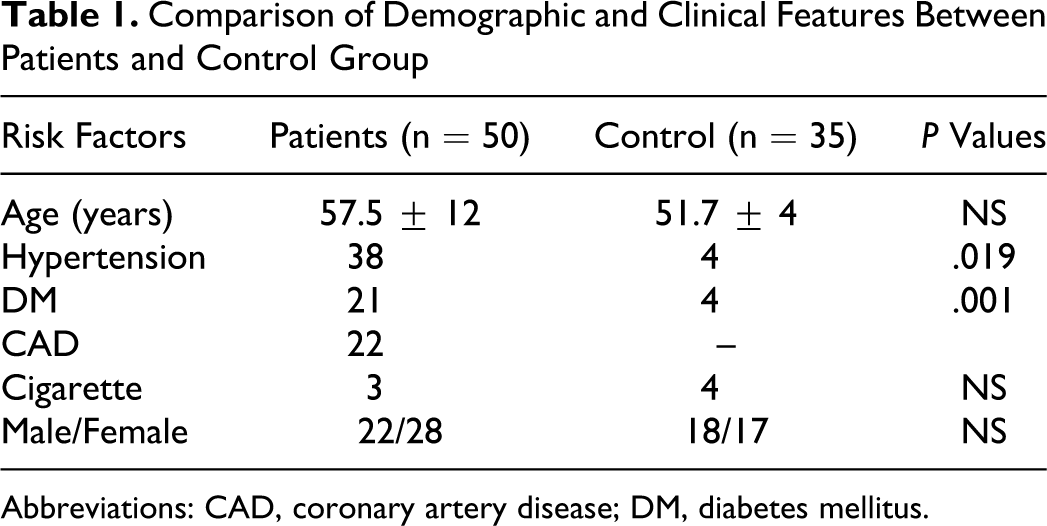
Abbreviations: CAD, coronary artery disease; DM, diabetes mellitus.
Thrombopoietin was significantly higher in stroke group when compared to control participants (P = .00 and P = .001, respectively). Platelet count was inversely associated with MPV. Also, MPV values were significantly higher in stroke group (P = .00; 8.6 vs 7.7 fL; P = .001). Figure 1 emphasizes MPV and TPO differences between the groups. Comparison of other laboratory parameters is summarized in Table 2 .
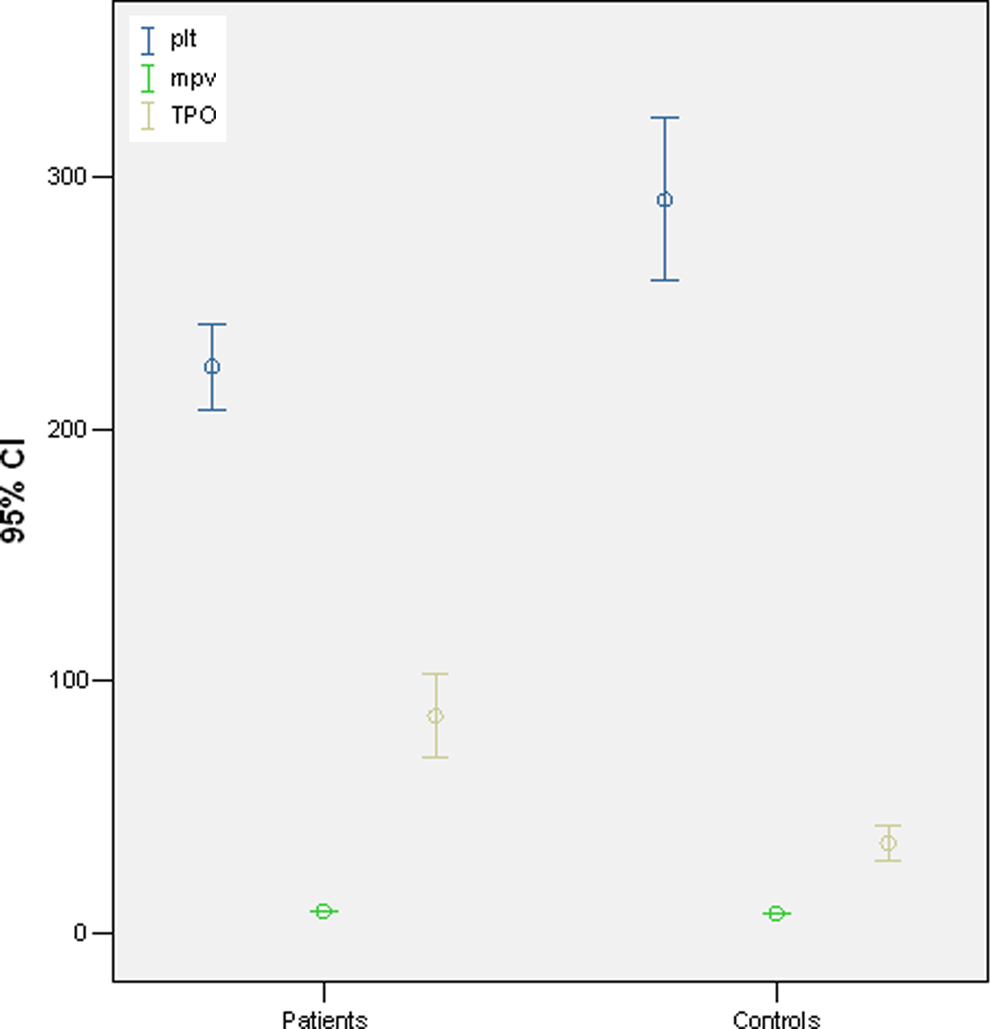
Comparison of platelet count, mean platelet volume and thrombopoietin levels between ischemic stroke and control groups.
Comparison of Laboratory Parameters Between Patient and Control Groups
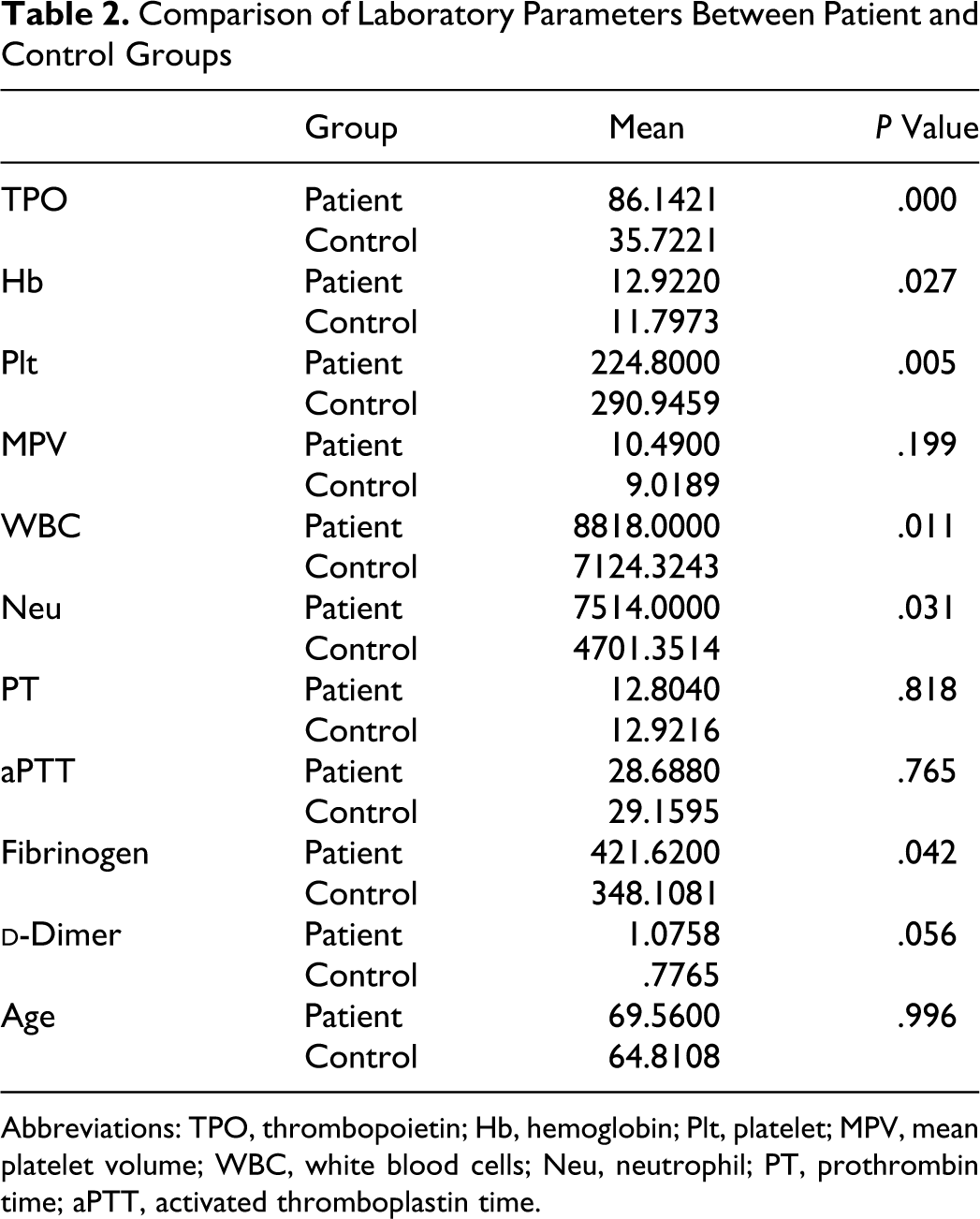
Abbreviations: TPO, thrombopoietin; Hb, hemoglobin; Plt, platelet; MPV, mean platelet volume; WBC, white blood cells; Neu, neutrophil; PT, prothrombin time; aPTT, activated thromboplastin time.
Discussion
Mean platelet volume and platelet count can be used in the evaluation of hemostatic index. Mean platelet volume is increased in disorders affecting platelet turnover or increased megakaryocytes like Bernard-Soluier syndrome. 10 So, MPV value indicates accelerated platelet production. Larger platelets contain more dense granules, express higher levels of prothrombotic substances, and decrease the effectiveness of prostaglandin on platelet aggregation. 11 The platelet size is determined in progenitor cell level and not changed in circulation; so it may be concerned that before ischemic stroke, prothrombotic condition may be the reason of higher platelet count. 12 There are many large trial studies emphasizing importance of higher MPV values in ischemic stroke.13–16 Our results were similar to those studies that show a significant relationship between MPV and ischemic stroke. But there are some reports affirming that MPV value has nonsignificant relationship with ischemic stroke17,18; the other reports showed decreased MPV values in patients with acute ischemic stroke.19,20
So the mean life span of platelets is 10 days; elevated MPV values in thrombotic diseases indicate macrothrombocytosis. It may contribute to the disease process; this condition may result from a prior activation of megakaryocytes to release larger and more active platelets. 11 It is suggested that increased hemostatic activity of large platelets is in part due to an increased number of glycoprotein IIb-IIIa receptors on each platelet in acute myocardial infarction. 21 The relationship between circulating platelets and megakaryocytes is not yet fully described. Thrombopoietin is the major stimulant molecule of megakaryocytopoiesis. Also a large number of cytokines including interleukin 3 (IL-3), IL-6, IL-1 I, erythropoietin, stem cell factor, and granulocyte–macrophage–CSF play a role in megakaryocytopoiesis.22–24 Furthermore, in addition to these numerous positive effectors, megakaryocytopoiesis might be physiologically regulated by inhibitory proteins.
Since TPO is considered the primary physiological regulator of megakaryopoiesis, its level is very essential for platelet production. Injection of recombinant TPO protein selectively induced thrombocytosis in vivo 25 and forced expression of TPO in mice via retroviral vectors, adenoviral vectors, or transgenes resulted in thrombocytosis correlating with the TPO expression. 26 Some studies affirmed that feedback mechanism regulating TPO production is directly related to its binding to platelets and/or megakaryocytes. So that, in normal hemostasis, TPO concentration remains within basal limits. During thrombocytopenia, degradation of TPO by c-mpl-positive cell is reduced; so the level of free TPO is increased.27,28 But TPO production remains constant. Its concentration is regulated by total mass of platelet and megakaryocytes capable of binding and degrading this protein. 29 Some studies about this subject expressed that c-mpl-deficient mice shows markedly reduced platelet and megakaryocyte numbers, but increased TPO concentration. 3 Platelet count and TPO levels were significantly higher in patients with ischemic stroke. There is no explanation for this result which is expected as the inverse relationship between platelet count and TPO level according to our knowledge. We speculate that TPO level may be affected due to various factors apart from platelets and megakaryocytes. It is also reported that the life span of platelets may be shortened because of mechanical stress when passing through from narrowed atherosclerotic vessels. 30 Because of shortened life span and accelerated thrombopoiesis, TPO level may remain elevated in these patients. This concern is also denoted in a study in which increased TPO levels were observed in patients with acute myocardial infarction and unstable angina pectoris. 31 In our study, we found that MPV and TPO levels were elevated significantly in patients with stroke when compared to healthy ones. The authors encountered no previous studies on the relationship between TPO levels and ischemic stroke. Platelet count was also higher in our patient group.
In conclusion, patients with ischemic stroke had increased platelet count, TPO and MPV values. Follow-up studies to ascertain the prognostic value of TPO should be conducted. The pathophysiologic mechanism is not clear and need to be determined in larger series.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
