Abstract
We describe thrombosis, deep venous thrombosis (DVT) pulmonary embolism (PE; n = 9) and hip-knee osteonecrosis (n = 5) that developed after testosterone therapy (median 11 months) in 14 previously healthy patients (13 men and 1 woman; 13 Caucasian and 1 African American), with no antecedent thrombosis and previously undiagnosed thrombophilia–hypofibrinolysis. Of the 14 patients, 3 were found to be factor V Leiden heterozygotes, 3 had high factor VIII, 3 had plasminogen activator inhibitor 1 4G4G homozygosity, 2 had high factor XI, 2 had high homocysteine, 1 had low antithrombin III, 1 had the lupus anticoagulant, 1 had high anticardiolipin antibody Immunoglobulin G, and 1 had no clotting abnormalities. In 4 men with thrombophilia, DVT-PE recurred when testosterone was continued despite therapeutic international normalized ratio on warfarin. In 60 men on testosterone, 20 (33%) had high estradiol (E2 >42.6 pg/mL). When exogenous testosterone is aromatized to E2, and E2-induced thrombophilia is superimposed on thrombophilia–hypofibrinolysis, thrombosis occurs. The DVT-PE and osteonecrosis after starting testosterone are associated with previously undiagnosed thrombophilia–hypofibrinolysis. Thrombophilia should be ruled out before administration of exogenous testosterone.
Keywords
Introduction
We have recently described thrombosis, deep venous thrombosis (DVT) pulmonary embolism (PE), amaurosisfugax, and osteonecrosis of the hips after exogenous testosterone was given to 6 men with no antecedent thrombosis and previously undiagnosed familial thrombophilia. In all, 5 of these 6 men were found to be heterozygous for the factor V Leiden mutation, and 1 had high factor VIII. 1 Of 20 men having measures of total and free testosterone and estradiol (E2) before and during exogenous testosterone therapy, 2 (10%) had high pretreatment E2 (>42.6 pg/mL), but after 3 months on testosterone gel (50 mg/d), 9 (45%) had high E2. 1 In these 20 men, testosterone was positively correlated with E2 before and during testosterone therapy, and change in testosterone was correlated with change in E2. 1 The major source of E2 in men comes from the aromatization of testosterone (endogenous or exogenous) to E2. 2 We speculated 1 that when testosterone is aromatized to E2 2–5 and E2-induced thrombophilia is superimposed on thrombophilia, thrombosis occurs. 1 We suggested that men sustaining thrombotic events on testosterone therapy should be screened for thrombophilia. 1 Oral estrogen therapy in men is associated with stroke 6 and increased venous thromboembolism (VTE).7,8
Osteonecrosis may be caused, in part, by thrombophilia- and hypofibrinolysis-induced thrombus of the major efferent veins of the head of the femur, leading to increased intracortical pressure and reduced arterial inflow, with subsequent bone hypoxia and bone death. 1,9
In 13 previously healthy Caucasians and 1 African American with no antecedent DVT-PE or osteonecrosis and previously undiagnosed thrombophilia–hypofibrinolysis, our specific aim was to describe thrombosis (DVT-PE in 9 men and osteonecrosis of the hips–knees in 4 men and 1 woman) that developed after testosterone therapy (median 11 months).
Patients and Methods
Study Design
Informed consent was obtained after the nature of the procedures (blood drawing, measures of coagulation, medical history) had been fully explained. The procedures followed were in accordance with the ethical standards of the institutional review board of the Jewish Hospital, Cincinnati, Ohio, which approved the research protocol.
In the sequential order of their referral for evaluation of DVT-PE or osteonecrosis, patient information was obtained by review of history and physical examination in 7 patients seen in Cincinnati, and by review of history and physicians’ records for 7 out-of-town patients. None of the data for these 14 patients have been previously reported. 1
In all patients, after an overnight fast, blood was drawn in the seated position for assessment of thrombophilia and hypofibrinolysis. 10 Coagulation measures were also obtained in 30 men with DVT-PE or testosterone, not taking exogenous testosterone, in 104 previously reported healthy controls for the factor V Leiden mutation, 11 and in 66 previously reported healthy controls for factor VIII. 9
To assess the E2 levels in men given exogenous testosterone, we separately examined the relationships of E2 and total and free testosterone in 60 otherwise healthy men free of DVT-PE or osteonecrosis who were receiving exogenous testosterone prescribed by their referring family physicians or urologists. In 22 of these 60 men, we were able to measure total testosterone, free testosterone, and E2 before and after testosterone therapy.
We also studied 30 men with previously undiagnosed thrombophilia–hypofibrinolysis causing DVT-PE or osteonecrosis, not receiving exogenous testosterone, to allow an examination of potential relationships of endogenous sex hormone levels 12 to DVT-PE and osteonecrosis.
Laboratory Assessment of Thrombophilia and Hypofibrinolysis
Polymerase Chain Reaction Assays
Polymerase chain reaction (PCR) measures of thrombophilia–hypofibrinolysis (G1691A factor V Leiden, G20210A prothrombin, MTHFR C677T-A1298C, and 4G5G plasminogen activator inhibitor activity gene mutations) were performed in all cases using previously published methods by laboratory staff blinded to the patients’ status (diagnosis, severity of disease). 13–15
Serologic Measures of Thrombophilia
Serologic measures of thrombophilia included anticardiolipin antibodies (Immunoglobulin [Ig] G and IgM), antigenic protein C, total and free protein S, antithrombin III, resistance to activated protein C, activated partial thromboplastin time, dilute Russel Viper venom time, factors VIII and XI, and homocysteine. Established, previously, published methods were used. 13,16
Serologic Measures of Hypofibrinolysis
Serologic measures of hypofibrinolysis, lipoprotein (a), and plasminogen activator inhibitor activity (PAI-Fx) were done using established, previously published, methods. 14,17
Measurement of Total and Free Testosterone and E2
Blood samples were drawn in the morning after an overnight fast, approximately 2 hours after application of the testosterone gel, patch, or injection.
Total and free testosterone and E2 levels were measured by LabCorp in a single laboratory (Burlington, NC). Total testosterone and E2 were measured by electrochemiluminescence immunoassay 18 and free testosterone by equilibrium ultrafiltration. 19,20 Sensitivity of the E2 method was 5.0 pg/mL and intraassay coefficients of variation <7% for 25 to 50 pg/mL, <4% for 50 to 1000 pg/mL, <5% for 1000 to 3000 pg/mL, and <7% for >3000 pg/mL, respectively. As noted by Wang et al, 21 results for serum total testosterone levels vary greatly by assay technique, and it is particularly important to use a single, standardized methodology in prospective follow-up of serum testosterone levels.
Statistical Methods
Paired Wilcoxon tests of difference were used to compare E2, testosterone, and free testosterone before and on testosterone therapy. Fisher tests were used to compare categorical distributions of testosterone, free testosterone, and E2 between patient groups with and without DVT-PE or osteonecrosis. Spearman correlations were used to assess correlations between changes in testosterone and E2 on testosterone therapy as well as correlations between changes in free testosterone and E2 on testosterone therapy.
Results
Interaction of Testosterone Supplementation Therapy With Previously Undiagnosed Thrombophilia–Hypofibrinolysis, Leading to Thrombotic Events, DVT, PE, and Osteonecrosis
The temporal associations of exogenous testosterone with thrombotic events led us to evaluate for underlying thrombophilia–hypofibrinolysis in 14 patients, Table 1. Abnormal results of the PCR and serologic tests for thrombophilia and hypofibrinolysis are displayed in Table 2.
Testosterone Therapy and Subsequent Deep Venous Thrombosis (DVT), Pulmonary Embolism (PE), and Osteonecrosis (ON) in 13 men and 1 woman.
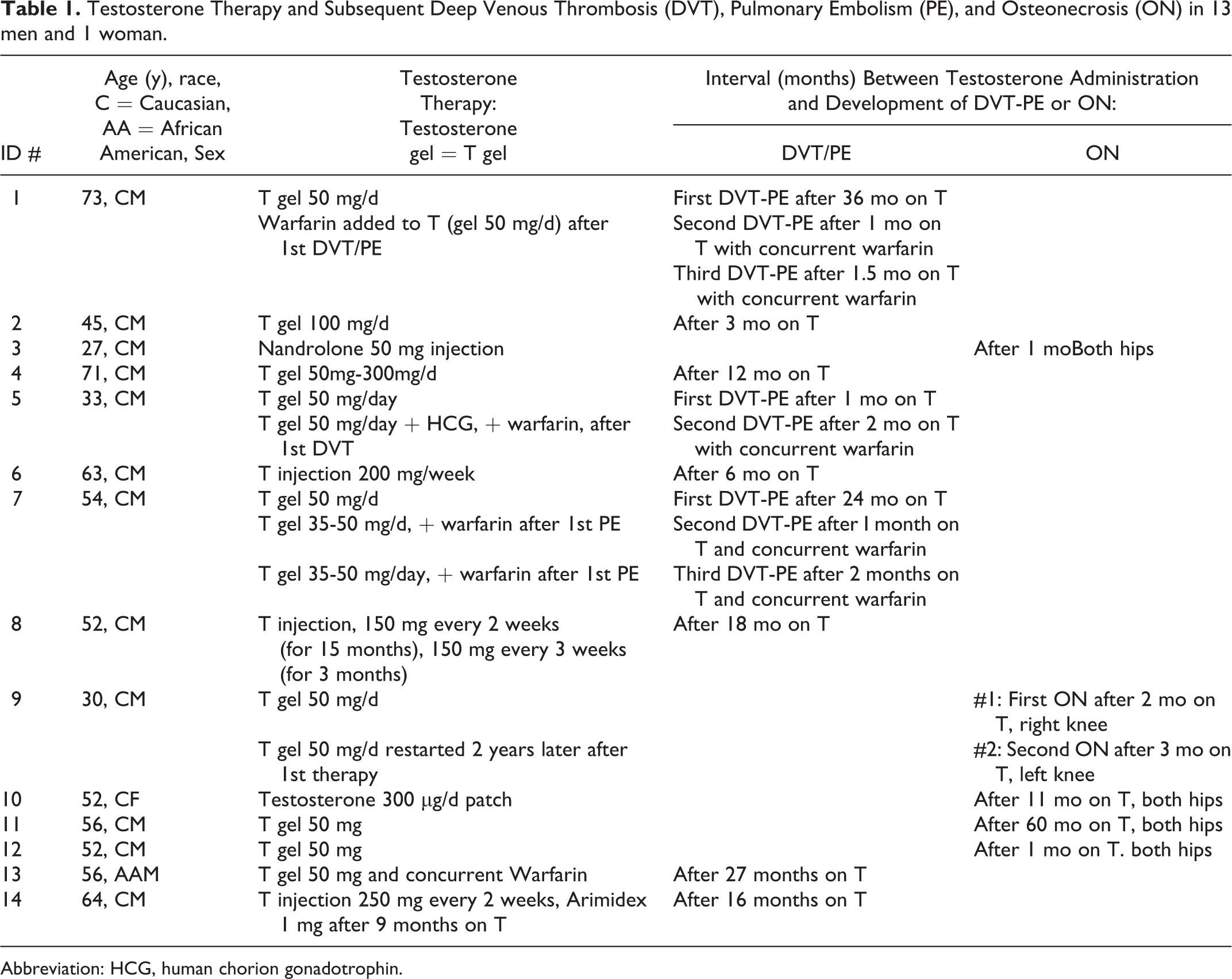
Abbreviation: HCG, human chorion gonadotrophin.
Previously Undiagnosed Thrombophilia and Hypofibrinolysis in Patients Developing Thrombotic Events After Starting Exogenous Testosterone Therapy.
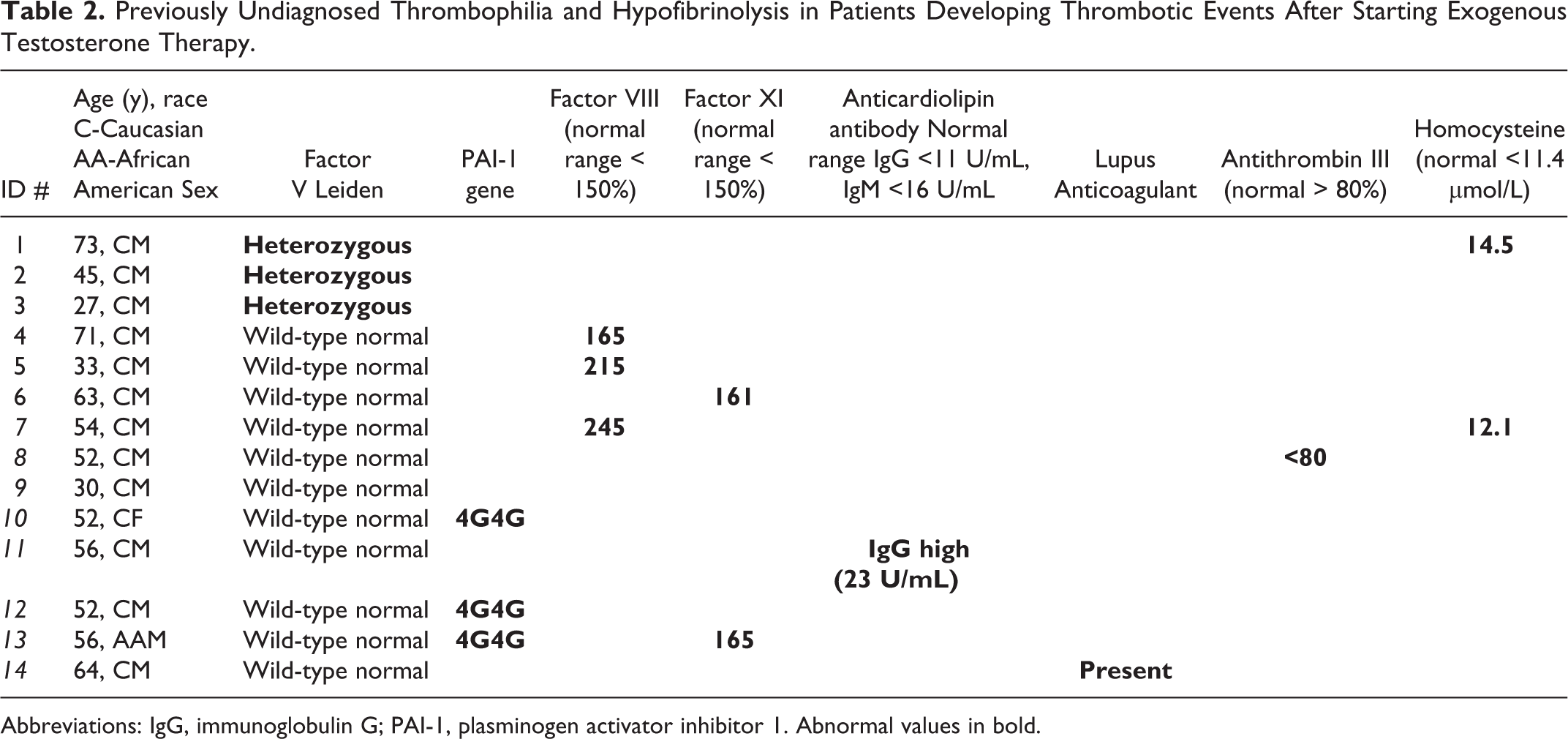
Abbreviations: IgG, immunoglobulin G; PAI-1, plasminogen activator inhibitor 1. Abnormal values in bold.
Before our evaluation that was triggered by thrombotic events, none of the previously healthy 13 men and 1 woman had a previous history of thrombosis and none had any previous testing of thrombophilia or hypofibrinolysis. They had no antecedent history of trauma, surgery, overt cancer, immobilization, or exogenous estrogen use that would predispose to thrombi and had no previously diagnosed coagulation disorders. None of the 14 patients with testosterone therapy and subsequent DVT-PE or osteonecrosis had taken long-term, high-dose corticosteroids, none were alcoholic, and 13 of the 14 were nonsmokers. None of the patients with osteonecrosis had taken biphosphonates.
Of the 14 patients, 13 were Caucasian and 1 (case #13) was African American. Of the 14 patients, 13 (93%) were found to have thrombophilia and/or hypofibrinolysis (Table 2), comparable to the findings in 23 (77%) of 30 patients with DVT-PE or osteonecrosis not receiving exogenous T (Fisher P = .40).
Of the 14 patients, 3 were found to be factor V Leiden heterozygotes, 3 had high Factor VIII, 3 had PAI-1 4G4G homozygosity, 2 had high Factor XI, 2 had high homocysteine, 1 had low antithrombin III, 1 had the Lupus anticoagulant, 1 had high ACLA IgG, and 1 had no clotting abnormalities (Table 2).
Of the 14 patients, 3 (21%) were heterozygous for the Factor V Leiden mutation (Table 2), far more common than in healthy Cincinnati controls 11 (2 of 104, 2%, chi-square = 7.26, P = .007). Three (21%) patients had high factor VIII (Table 2) versus 3 (4.5%) of 66 healthy Cincinnati controls (Fisher P =.063). 9
Case 1
This 73-year-old Caucasian male had his first DVT and pulmonary embolus 3 years after starting testosterone (Table 1). Testosterone was continued and warfarin was started with international normalized ratio (INR) held ∼ 3 to 3.5 units. After 1 month on combined warfarin and testosterone, a second DVT-PE occurred, and after 1.5 months on combined warfarin and testosterone, a third DVT-PE occurred (Table 1). Testosterone was stopped on our advice and warfarin was continued, and he has been free of DVT-PE for 8 months. He was found to be heterozygous for the factor V Leiden mutation and had high homocysteine (Table 2).
Case 2
This 45-year-old Caucasian male developed his first DVT-PE 3 months after starting testosterone and was found to be heterozygous for the factor V Leiden mutation (Tables 1 and 2). Testosterone was stopped and warfarin started, and he has been free of DVT-PE for 11 months.
Case 3
This 27-year-old Caucasian male had 2 injections of Nandrolone 15 days apart after wrist fracture and developed bilateral osteonecrosis of the hips 1 month later (Table 1). He was found to be heterozygous for the factor V Leiden mutation (Table 2). He is currently taking a 3-month course of 5000 units of Fragmin twice daily.
Case 4
This 71-year-old Caucasian male developed DVT and PE 12 months after starting testosterone (Table 1) and was found to have high factor VIII (Table 2). After discontinuing testosterone and after 6 months’ anticoagulation with warfarin, there have been no further thrombotic events for 12 months.
Case 5
This 33-year-old Caucasian male developed a DVT in his right leg 1 month after starting testosterone (Table 1). Warfarin was started and the testosterone continued, with addition of human chorion gonadotrophin (HCG; Table 1). After 2 months on warfarin (INR ∼3), testosterone gel, and HCG, he had a second DVT and a concurrent PE. He was found to have high Factor VIII (Table 2). Testosterone gel and HCG were stopped on our advice and warfarin continued, with no further DVT-PE for 16 months.
Case 6
This 63-year-old Caucasian male developed progressive shortness of breath 3 months after starting testosterone, and at 6 months sustained a massive saddle PE (Table 1). Testosterone gel was stopped, warfarin started, and was on warfarin for 7 months, he has had no further DVT-PE. Coagulation tests revealed high factor XI (Table 2).
Case 7
This 54-year-old Caucasian male developed his first PE 24 months after starting testosterone (Table 1), testosterone was continued, Warfarin was given, and INR maintained at the 3 to 3.5 therapeutic range. He sustained a second PE 1 month later, while still taking testosterone and concurrent warfarin. With testosterone and warfarin continued, despite stable INR ∼3.5, he had a third PE 1 month later (Table 1). Coagulation tests revealed high factor VIII and high homocysteine (Table 2). On our advice, testosterone gel was stopped, warfarin was continued, and he has been free of PE for 3 months.
Case 8
This 52-year-old Caucasian male with a 20 pack-year smoking history developed DVT and subsequent PE 18 months after starting testosterone (Table 1). He was found to have low antithrombin III (Table 2). After discontinuing testosterone, and after 6 months’ anticoagulation with warfarin, there have been no further thrombotic events for 24 months.
Case 9
This 30-year-old Caucasian male developed osteonecrosis in his right knee 2 months after starting testosterone (Table 1). Testosterone was stopped. Subsequently, testosterone was restarted 2 years later; and after 3 months on testosterone, he developed osteonecrosis in the left knee (Table 1). Testosterone was stopped on our advice 5 months ago, but osteonecrosis in both knees remained symptomatic. No coagulation abnormalities were found (Table 2).
Case 10
To improve libido, this 52-year-old Caucasian woman was given a testosterone patch that delivered 300 mcg testosterone/d. Bilateral hip osteonecrosis was diagnosed 11 months after starting testosterone (Table 1). Coagulation tests revealed 4G4G homozygosity of the PAI-1 gene (Table 2).
Case 11
After 5 years on testosterone, osteonecrosis of both hips was diagnosed in this 56-year-old Caucasian male (Table 1). Coagulation tests revealed elevated anticardiolipin antibody IgG (Table 2).
Case 12
This 52-year-old Caucasian male developed osteonecrosis of both hips 1 month after starting testosterone (Table 1). Coagulation tests revealed 4G4G homozygosity for the PAI-1 gene (Table 2).
Case 13
Two years before starting testosterone, this 56-year-old African American male had an unprovoked right arm radial artery thrombus; warfarin was started and continued. After 2 years on warfarin, testosterone gel was started; and after 27 months on both testosterone and warfarin, DVT-PE occurred (Table 1). High Factor XI and PAI-1 4G4G homozygosity were documented (Table 2), testosterone was stopped; and with continuation of warfin, he had no further thrombi for 4 months.
Case 14
This 64-year-old Caucasian male started testosterone because of loss of libido. Ten months after starting testosterone, serum E2 was found to be elevated (76 pg/mL, upper normal limit ≤46.2 pg/mL), and Arimidex (1 mg/d) was started and continued for 3 months. Then, 3 months after stopping the Arimidex, and after 16 months on testosterone, DVT-PE developed (Table 1). The Lupus Anticoagulant was found to be present (Table 2).
Total and Free Testosterone and E2 in Men With Exogenous Testosterone Supplementation
In 60 otherwise healthy men, without DV-PE or osteonecrosis, studied on testosterone (median testosterone dose 50 mg/d, mean ± standard deviation [SD] 61 ± 47 mg/d, minimum 7, maximum 200 mg/d), 33% had high serum E2 (>42.6 pg/mL; Table 3).
Serum Testosterone (T), Free T, and Estradiol (E2) on Testosterone Treatment (median 50 mg/d) in 60 Men.
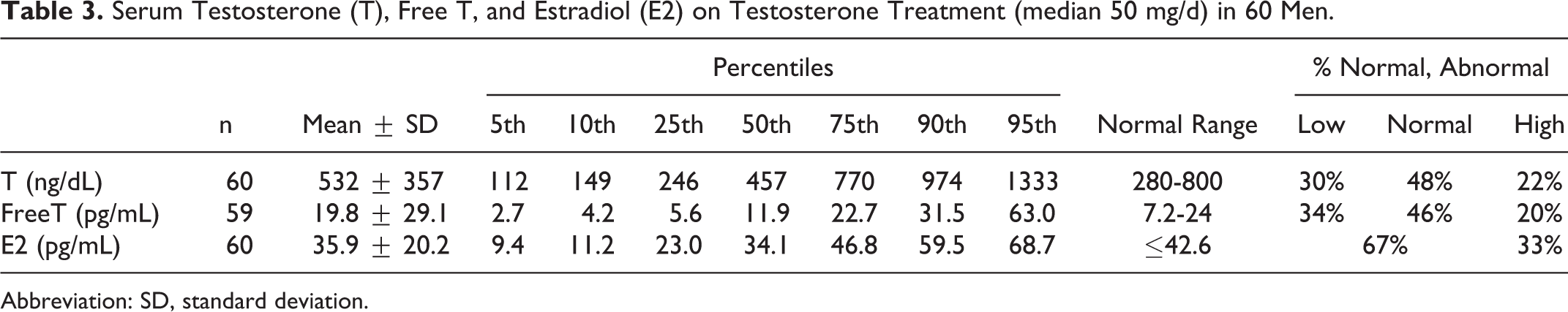
Abbreviation: SD, standard deviation.
In 22 otherwise healthy men, without DVT-PE or osteonecrosis, having measures of testosterone, free testosterone, and E2 before and during exogenous testosterone treatment (median testosterone dose 50 mg/d, mean ± SD 54 ± 46 mg/day, minimum 7, maximum 200 mg/d), 12% had high E2 before treatment and 41% had high E2 on testosterone treatment (Table 4). The E2 rose 61% on testosterone therapy (Table 4).
Testosterone (T), Free T, and Estradiol (E2) Measures in 22 Men Before and on Testosterone Treatment (median 50 mg/d), Compared With That in 30 Men With Thrombotic Events (Deep Venous Thrombosis, Pulmonary Embolus, Osteonecrosis) Not Taking Exogenous Testosterone.
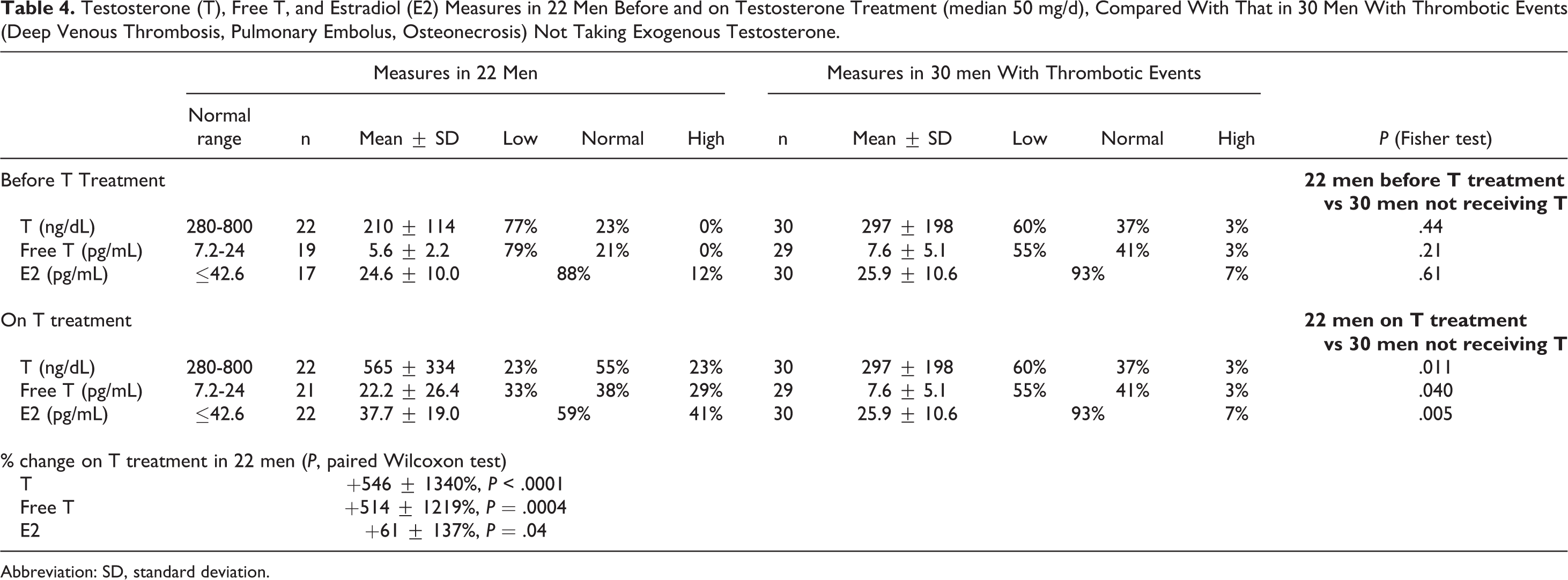
Abbreviation: SD, standard deviation.
In 30 men with previously undiagnosed thrombophilia–hypofibrinolysis causing DVT-PE or osteonecrosis, not receiving exogenous testosterone, 7% had high endogenous E2 (>42.6 pg/mL), comparable to 12% in 22 otherwise healthy men without DVT-PE or osteonecrosis studied before testosterone treatment (P = .61) but much lower (P = .005) than the high E2 in 41% of the 22 men on testosterone treatment (Table 4). Of these 30 men, 23 (77%) were found to have familial thrombophilia–hypofibrinolysis.
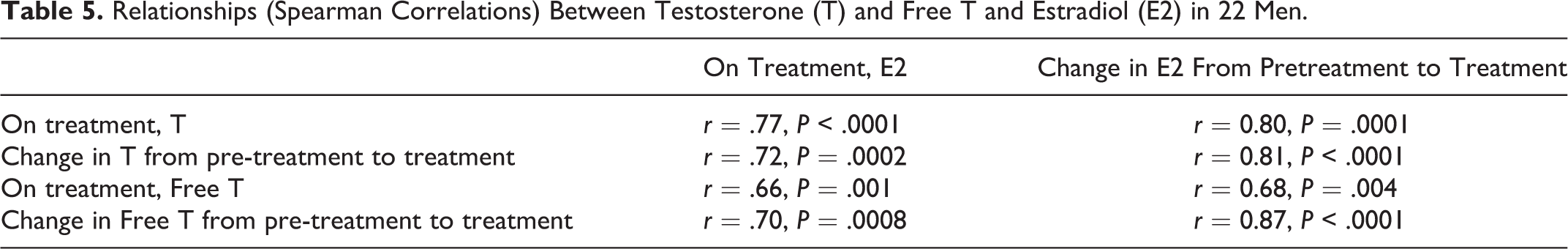
On testosterone supplementation, in the 22 otherwise healthy men, serum testosterone and free testosterone were closely correlated with serum E2, and changes in serum testosterone and free testosterone were closely correlated with changes in E2 (Table 5). In these 22 men, there was no correlation between testosterone dose and serum E2 levels (r = .11, P = .7).
Relationships (Spearman Correlations) Between Testosterone (T) and Free T and Estradiol (E2) in 22 Men.
Discussion
The novel finding of the current study, confirming and expanding our initial report of 6 patients, 1 was that in 13 previously healthy men and 1 woman without previous DVT, PE, or osteonecrosis, and without previously known thrombophilia–hypofibrinolysis, thrombotic events (9 DVT-PE, 5 osteonecrosis) after testosterone therapy were found to be associated with thrombophilia including factor V Leiden heterozygosity, high factors VIII and XI, high homocysteine, low antithrombin III, the lupus anticoagulant, high anticardiolipin antibody IgG, and the hypofibrinolytic 4G4G mutation of the PAI-1 gene. factor V Leiden heterozygosity and high factor VIII in 21% of our cases were much more common than in healthy Cincinnati controls, 2% of whom were factor V Leiden heterozygotes, 11 4.5% of whom had high factor VIII. 9 Factor V Leiden heterozygosity, 22 high factors VIII 23 and XI, 24 the lupus anticoagulant, 25 and low antithrombin III 26 are all associated with increased risk of DVT and PE. 27–29 Osteonecrosis is associated with the factor V Leiden mutation, high factors VIII and XI, and with the hypofibrinolytic 4G4G mutation of the PAI-1 gene. 9,11,30 Like other thrombophilias, risk of thrombosis is increased by E2 in patients with high factor VIII. 31
When exogenous testosterone was discontinued and on warfarin anticoagulation, 6 men, all with thrombophilia, had no further DVT-PE. However, when testosterone was continued, in 4 men with thrombophilia, despite concurrent anticoagulation with warfarin in the therapeutic INR range, DVT-PE occurred or reoccurred. Of the 4 men and 1 woman with “primary” osteonecrosis, without high-dose, long-term corticosteroids or alcoholism, 1 patient developed osteonecrosis of the right knee 2 months after starting testosterone, stopped testosterone for 2 years, and then developed osteonecrosis of the left knee 3 months after resuming testosterone.
In the current study, in 22 men whose E2 was measured before and on exogenous testosterone, 12% had high E2 (>42.6 pg/mL) before testosterone and 41% had high E2 on testosterone. In a group of 60 men studied on testosterone, 33% had high E2. Change in serum testosterone and free testosterone was closely correlated with change in E2. We speculate that as exogenous testosterone is aromatized to E2, 32 the thrombophilic increased E2 interacts with previously undiagnosed thrombophilia to produce thrombotic events, as in the current study; and in our recent report. 1 Wang et al 33 previously reported that serum E2 “… levels were significantly higher during treatment with testosterone, compared with baseline (P = .0001), and increased progressively from 6 (P = .0001) until 24 months of treatment, remaining at the upper limit of the male reference range.” In our study, 33% to 41% of the men on testosterone therapy developed high E2, congruent with reports that high E2 in testosterone-treated men is common. 33–35 Given an estimated population prevalence of heterozygosity for the factor V Leiden mutation of 5%, 36 and prevalence of thrombophilias of 18% to 28%, 37–39 there is considerable opportunity for men having high E2 on testosterone therapy to also have familial and acquired thrombophilias, with resultant increased risk of thrombosis.
Endogenous sex hormone levels in men are not associated with risk of venous thromboembolism. 12 Svartborg et al 12 obtained sex hormone measurements in 1350 men, aged 50 to 84, participating in the Tromso study in 1994 to 1995. There were 63 incident VTE events (4.5 per 1000 person years) during a mean of 10.4 years of follow-up. In age-adjusted analyses, endogenous sex hormones levels were not associated with the risk of VTE. In the current study, E2 was high in 7% of 30 men not receiving testosterone treatment but with DVT-PE caused by thrombophilia–hypofibrinolysis, not different from high E2 in 12% of 22 otherwise healthy men before testosterone supplementation, but much lower than high E2 in 41% in these 22 men on testosterone therapy (P < .005).
Although in our study and that of Svartborg et al, 12 endogenous male sex hormones were not associated with VTE, 12 there are case reports of DVT-PE in men after exogenous testosterone Nandrolone 40 and cerebral sinus thrombosis 41 after exogenous nandrolone. Exogenous androgenic hormone regimens increase the risk of breast cancer in women. 42 As in our current study, when exogenous testosterone therapy is given, the percentage of men with high E2 more than triples, speculatively facilitating interaction with underlying thrombophilia–hypofibrinoysis, leading to VTE and osteonecrosis.
Men and women who sustain thrombotic events on testosterone therapy should be screened for underlying familial and acquired thrombophilias, and any subsequent testosterone therapy is contraindicated because of the likelihood of development of second and third DVT-PE events despite adequate anticoagulation, as in our current report. Additional information on the incidence of thromboembolism in testosterone-treated men and women with positive screening tests for familial and acquired thrombophilia 43 versus those with negative screening tests is needed in order to advise consideration of systematic screening for thrombophilia and hypofibrinolysis in men and women treated with testosterone. However, based on our previous report 1 and on our current study, we suggest that thrombophilia should be ruled out before administration of exogenous testosterone.
Testosterone is increasingly given to improve libido and sexual function, 32,44–46 beyond conventional use in hypogonadotrophichypogonadism. 47 We speculate that the increasing frequency of testosterone prescription may be associated with a parallel and largely preventable increase in thrombotic events and, possibly, cardiovascular adverse events. 48
After starting testosterone, DVT-PE and osteonecrosis appear to be frequently associated with previously undiagnosed thrombophilia and hypofibrinolysis. We speculate that when exogenous testosterone is aromatized to E2, and E2-induced thrombophilia is superimposed on thrombophilia–hypofibrinolysis, thrombosis occurs. Men and women sustaining DVT-PE or osteonecrosis after testosterone therapy should be screened for familial and acquired thrombophilia and hypofibrinolysis. Continuation of testosterone therapy is contraindicated after an initial DVT-PE /osteonecrosis associated with familial or acquired thrombophilia, since the continued interaction of testosterone with the underlying thrombophilia may lead to repetitive thrombotic events, despite adequate anticoagulation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported in part by the Lipoprotein Research Fund and the Metabolic Research Fund of the Jewish Hospital of Cincinnati.
