Abstract
Background:
Atrial fibrillation (AF) is the most common cardiac rhythm disorder. Atrial fibrillation causes a 5-fold increased risk for thromboembolic stroke. It is known that eosinophils play an important role in thrombosis. We aimed to compare the number of eosinophil counts of the patients with and without thrombi in the left atrium (LA) or in the left atrial appendage (LAA) and to ascertain the association of eosinophil counts with the presence of thrombi.
Method:
The study included 89 patients diagnosed with persistent AF who underwent transesophageal echocardiography and designated to undergo cardioversion. The patients were divided into 2 groups: group 1 consisted of 40 patients (18 male; average age 63.27 ± 1.4) who had thrombus formation in the LA or LAA, and group 2 consisted of 49 patients (23 male; average age 66.53 ± 1.56) who did not have any thrombus in the LA or LAA. These patients underwent concurrent routine biochemical tests and eosinophil count on whole blood count was also performed.
Results:
Baseline characteristics of the study groups were comparable. Group 1 patients had higher eosinophil and mean platelet volume values than group 2 (233.0 ± 30.7 vs 118.9 ± 11.8 and 9.77 ± 0.20 vs 8.27 ± 0.12 fL,
Conclusion:
As a result, our study revealed a relationship between eosinophil count and LA thrombus in patients with nonvalvular AF.
Introduction
Atrial fibrillation (AF) is the most common cardiac rhythm disorder. The estimated prevalence of AF ranges from 0.4% to 1% in the general population and increases by age. Atrial fibrillation is an important public health issue, which increases the risk of mortality, stroke, and cardiac failure. 1 –3
There are evidences regarding that AF is associated with abnormal hemostasis, fibrinolysis, thrombocyte, and endothelial functions as well as occurrence of prothrombotic and hypercoagulable states. 4 The thromboembolic event that is the most common cause of mortality and morbidity in AF results from the embolus of thrombus material formed in the atria. The pathogenesis of thrombus formation in AF has not been disclosed thoroughly.
The traditional thromboembolic risk classification methods concentrate on the hemodynamics of the left atrium (LA) but do not focus sufficiently on the components forming the thrombus. It is known that eosinophils play an important role in endothelial dysfunction, inflammation, and thrombosis. 5,6 Eosinophils stimulate the activation and aggregation of platelets. Moreover, they ease the formation of thrombosis via inhibition of thrombomodulin. 7 –9
In this trial with patients having nonvalvular persistent AF, we aimed to compare the number of eosinophils in concurrent blood counts of the patients with and without thrombi in the LA or in the left atrial appendage (LAA) and to ascertain the association of eosinophil counts with the presence of thrombi.
Materials and Methods
Selection of the Patients
The study included 89 consecutive patients diagnosed with persistent AF who were 40 to 80 years old. These patients underwent transesophageal echocardiography (TEE) and were designated to undergo cardioversion. The patients were divided into 2 groups; group 1 consisted of 40 patients (18 male; average age 63.27 ± 1.4) who had thrombus formation in the LA or LAA, and group 2 consisted of 49 patients (23 male; average age 66.53 ± 1.56) who did not have any thrombus in the LA or LAA. These patients underwent concurrent routine biochemical tests and the eosinophil count on whole blood count was also performed. According to the study protocol that was approved by the Ethics Committee of Bursa Şevket Yılmaz Education and Research Hospital, the patients were asked to sign the informed written consent.
A careful record of the underlying disorders of the patients which might lead to AF and the accompanying diseases and the medicines used was made. The blood pressure of the patients was measured. The patients having a systolic blood pressure ≥140 mm Hg and/or a diastolic blood pressure ≥90 mm Hg and those taking antihypertensive drugs were considered hypertensive. The patients using oral antidiabetic drugs or insulin or those having 2 measurements of fasting blood glucose level ≥126 mg/dL were accepted to be diabetic.
Exclusion criteria included the presence of the following: known coronary artery disease, chronic renal failure, chronic liver disorders, moderate or severe valvular disease, congenital heart disease, left ventricular (LV) systolic dysfunction on echocardiography (EF<50%), marked LA dilatation (>55 mm), recent acute coronary syndrome, anemia, hyperthyroidism, pregnancy, obstructive sleep apnea, chronic obstructive pulmonary disease, stroke, secondary AF (due to a temporary cause), hematological disorders, known malignancy, and drug history including antigout agent, anti-inflammatory agent (steroid or nonsteroid), antiaggregant or anticoagulant agents, and antihistaminic agents. Patients who had a recent history of an acute infection and or high body temperature >38°C, and inflammatory or allergic disease are also excluded from the study. The study did not include male and female patients younger than 40 years or older than 80 years.
Laboratory Tests
Biochemical parameters were analyzed spectrophotometrically on ArchitectC16000 autoanalyzer (Abbott, Abbott Laboratories, Abbott Park, IL, USA) using enzymatic–colorimetric assay.
For whole blood count (eosinophil count, hematocrit, hemoglobin, mean corpuscular volume [MCV], mean platelet volume [MPV], leukocytes, platelets), the blood samples were collected in tubes with EDTA and analyzed on CELL-DYN 3700 (Abbott. USA) device using impedance and optic scatter method.
Echocardiography Protocol
Echocardiographic evaluation was done on a Vivid 7 Pro TTE with 3.5 MHz transducer. All echocardiographic examinations were performed according to the criteria of the American Society of Echocardiography and European Association of Echocardiography guidelines. 10 All echocardiographic images were recorded while the patient was lying in the lateral decubitus position. On echocardiographic evaluation, dimensions of the left ventricle chamber, wall thickness, left ventricle ejection fraction (with Teichholz method), diameter of the LA, abnormal blood flows due to valve insufficiency, if present, were determined. The degree of valvular stenosis was evaluated with 2-dimensional, M-mode, Doppler, and tissue Doppler studies. Parasternal long axis, apical 2-, 4-, and 5-chamber views were taken.
Thicknesses of the LV septal and posterior free walls and LV end-diastolic and end-systolic diameters were measured in parasternal long-axis view with M-mode. Systolic pulmonary artery pressure was calculated by adding the estimated right atrial pressure to the right ventricle systolic pressure obtained from the tricuspid insufficiency peak velocity.
On completion of the TTE investigations, all patients underwent conscious TEE using a 5-MHz multiplane probe. Pharyngeal anesthesia was applied using a topical spray of lidocaine (10%); neither sedation nor atropin was employed, and probe insertion and image acquisition were carried out using conventional techniques.
Statistical Analysis
Statistical analyses were done using SPSS (Statistical Package for the Social Sciences version 10; SPSS Inc, Chicago, Illinois) software and Epi info pack program. Numeric variables were presented as median ± standard deviation; categorical variables were presented as percentage values. The equality of the data to the normal distribution was assessed with the Shapiro-Wilk test. Since the data were not normally distributed, the Mann-Whitney
Results
By evaluating the basic characteristics, no statistically significant difference between the 2 groups in terms of medications, age, gender distribution, and body mass index (Table 1) was found.
Comparison of Clinical Features of Patients With AF.
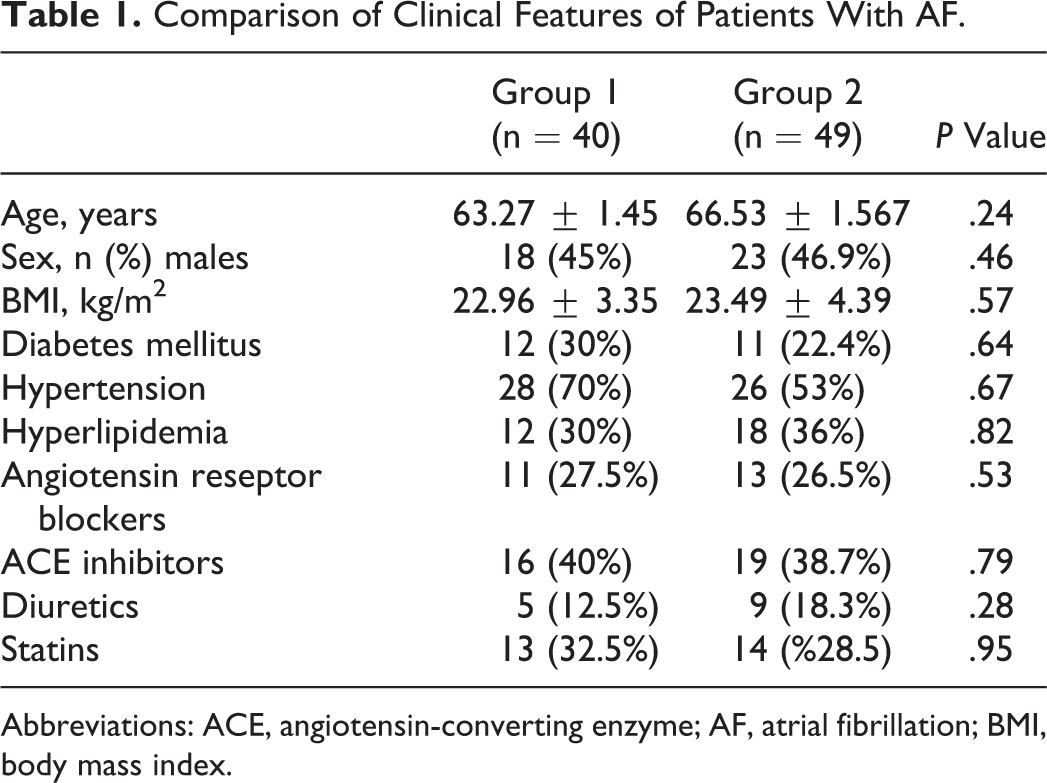
Abbreviations: ACE, angiotensin-converting enzyme; AF, atrial fibrillation; BMI, body mass index.
Blood leukocyte count, eosinophil count, and MPV ratio, were significantly higher in patients with LA/LAA thrombus than that of nonthrombus patients (P < .001). There was no statistically significant difference between the 2 groups with regard to hemoglobin level, MCV, hematocrit, and platelet count (Table 2).
Comparison of Biochemical and Hematological Features of Patients With AF.
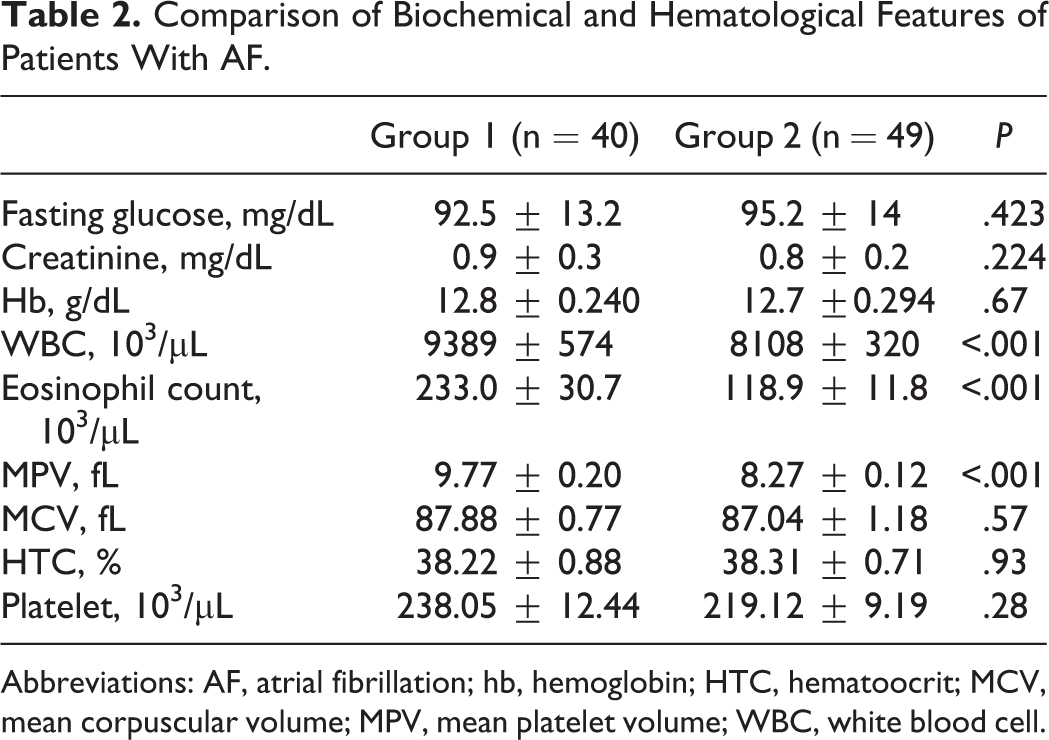
Abbreviations: AF, atrial fibrillation; hb, hemoglobin; HTC, hematoocrit; MCV, mean corpuscular volume; MPV, mean platelet volume; WBC, white blood cell.
When the initial conventional echocardiographic parameters of the patients were evaluated, the average LA diameter of the group of the patients with LAA thrombus (group 1) was significantly larger than that of the group 2 patients (
Comparison of Conventional Echocardiographic Features of Patients With AF.
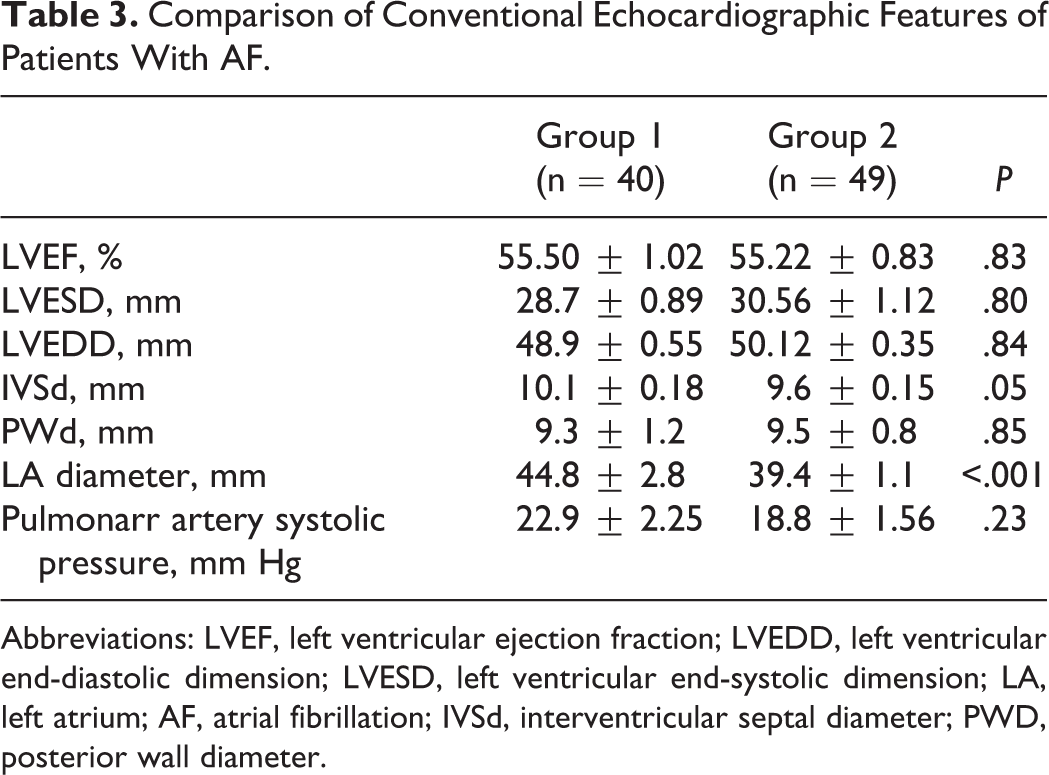
Abbreviations: LVEF, left ventricular ejection fraction; LVEDD, left ventricular end-diastolic dimension; LVESD, left ventricular end-systolic dimension; LA, left atrium; AF, atrial fibrillation; IVSd, interventricular septal diameter; PWD, posterior wall diameter.
The parameters that were determined in previous studies as predictors of thrombus formation within LAA among patients with nonvalvular AF and eosinophil count were assessed by univariate and multivariate logistic regression analyses. Using univariate logistic regression analysis, it was found that, with regard to thrombus formation, eosinophil count, MPV, white blood cell, and diameter of LA were found to be significant parameters. When these 4 parameters were evaluated with multivariate logistic regression analysis, it was found that eosinophil count and MPV ratio were significant to predict thrombus formation within LA/LAA in patients with nonvalvular AF (Table 4).
Logistic Regression Analysis for the Independent Risk Factors for Atrial Thrombus.

Abbreviations: HR, hazard ratio; MPV, mean platelet volume; LA, left atrium; MCV, mean corpuscular volume; LVEF, left ventricular ejection fraction.
Discussion
Atrial fibrillation is a commonly encountered arrhythmia, which reduces the quality of life of the patients and negatively affects the prognoses of the present diseases. Therefore, impeding the development of AF and protective treatments become more popular today. The AF causes a 5-fold increased risk of stroke, and one-fifth of all strokes attributed to this arrhythmia.
The mechanism of thrombus formation in AF has not yet been clearly explained. The trilogy of stasis, endothelial dysfunction, and hypercoagulability is already the effective mechanism for the thrombus that occurs within the atrium. In AF, the most common location of thrombus is LAA, since it is structurally favorable for stasis.
There are many biochemical abnormalities increasing the risk of thrombus formation in AF. During AF, significantly decreased levels of nitric oxide and increased levels of prothrombotic plasminogen activator inhibitor 1 (PAI-1) within LAA were found. 11 Moreover, increases in plasma β thromboglobulin, platelet factor 4, von Willebrand factor (vWF), thrombomodulin, and fibrinogen levels in the patients with AF were demonstrated. 12 In SPAF III study, it was found that in patients with AF increased vWF levels were correlated with the predictors of risk of stroke. 13
Eosinophils cause activation of coagulation via release of tissue factor, inhibition of protein C, and activation of factors VII and X. 14 –17
In our study, since the association between eosinophil count and thrombus formation was investigated, the patients at high risk for thrombus formation and those having serious structural or congenital heart diseases resulting in stasis were excluded. The traditional thromboembolic risk classification methods concentrate on the LA hemodynamics but do not focus sufficiently on the components forming the thrombus.
Eosinophils are equipped with several granule-associated molecules that play a role in the occurrence of thrombosis and vascular injury. Eosinophils generate an increased tendency to thrombosis through leukocyte, platelet stimulation, and release of tissue factor. 14,15,18,19 All these effects contribute to procoagulation through preventing the activation of thrombin and endorsing fibrin formation. Eosinophils store and release tissue factor as well as other cationic proteins. Major basic protein, eosinophilic cationic protein, activates platelets and promotes thrombus formation by inhibiting thrombomodulin in hypereosinophilic syndromes and allergic diseases. In addition, antineutrophil cytoplasmic antibodies may shift the endothelial lining to proadhesive and prothrombotic surface. 20,21
Sakai et al demonstrated that large thrombus has greater eosinophil counts both in thrombi and in peripheral blood and also speculated that thrombus growth might be facilitated in patients with higher eosinophil counts in the peripheral blood. 22
Recent studies showed that eosinophils were associated with stent thrombosis, stent restenosis, and acute coronary syndromes. Furthermore, it was reported that elevated serum eosinophil concentration might be responsible for cardiac mural thrombus and embolic events. 23 –25
The powerful procoagulant effects of eosinophils made us hypothesize that there might be a correlation between eosinophil concentration and thrombus formation in the LA and LAA in patients with AF. In the literature, there is no study investigating the association between thrombus formation in LA and LAA in patients with AF and eosinophils. Our study is of importance with regard to this matter, and we investigated the effect of eosinophil concentration on thrombus formation in LA/LAA among patients with nonvalvular persistent AF.
In our study, the eosinophil count was significantly higher in the group of patients with thrombus in LA/LAA in comparison with the nonthrombus group (
In a recent study, reduced LAA outflow velocity, LA enlargement, and statin use were determined as independent predictors of the development of spontaneous echo contrast that can be accepted as precursor of thrombus formation in LAA. 26 In the literature, it was reported that in approximately 30% to 80% of serial TEEs performed after appropriate therapy, thrombi were treated. The most important factors for insufficient treatment response were found to be large and dense thrombi and enlarged LA diameter.
In our study, in consistent with the literature data, the mean LA diameter of the group of patients with thrombus in LAA was significantly greater than that of the control group (
Recently, it was demonstrated that the MPV was greater in the patients with mechanic valve thrombosis in comparison with the control group, and it was reported that MPV could be used as a marker for following up thrombosis in the patients with mechanical valve thrombosis. 27
High MPV indicates the presence of more reactive and larger thrombocytes that can be a risk factor for thromboembolic states. In a study investigating the association of MPV and platelet count with thrombus formation in LAA in patients with AF, it was reported that there was no significant difference between the 2 groups.
28
However, in our study, MPV value of the group of patients with thrombus in LA/LAA was significantly higher in comparison with the control group (
In conclusion, the correlation between the loss of atrial mechanic contractility and consequently reduced blood flow in the LA and thrombus formation has been exactly demonstrated before. Our current data demonstrate that eosinophil concentration independent of hemodynamic risk factors has procoagulant effects in predicting thrombus formation, and MPV value can be predictive of thrombus formation in LA/LAA in patients with AF. The most important restriction of our study is the limited number of patients. There is a need for large-scale research into this issue.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
