Abstract
Although animals housed for research purposes are strictly monitored for lighting, temperature, and humidity, the acoustic environment receives less attention. In a retrospective study, we investigated the effect of construction-induced noise on coagulation using thromboelastography in a group of healthy control rats. Animals were unintentionally exposed to noise due to construction in the vicinity of the animal care facility where these rats were housed. We compared the results to those of age-, gender-matched nonexposed rats. There was a significant shortening of the reaction (R) time (P = .009) and a trend toward an increase in coagulation index (CI; P = .09), indicating hypercoagulability. The short R time and increase in CI were correlated with an elevated plasma cortisol and corticosterone, indicating that the hypercoagulability seen in these rats is stress induced. Noise is a stress factor for which animals need to be monitored, particularly if those animals are selected as controls for hemostasis studies.
Introduction
The use of animals for laboratory experiments should always follow not only ethical rules but also careful biological and medical standards. Mice and rats are 2 of the most widely used animal models in biomedical research. When housed under standard laboratory conditions, these rodents are sedentary and have virtually no environmental stimulation. Animals maintained under these conditions are widely used as “standard” controls not only in basic but also in translational biomedical research studies, including preclinical drug testing. 1
Monitoring the animal environment in relation to lighting, temperature, and humidity is usually strictly performed and recorded in animal care facilities. However, little attention is given to the acoustic environment. Noise causes stress and can significantly impact laboratory animals and disturb cardiovascular, biochemical and behavior, and reproductive biology. 2
The effect of stress, due to a variety of reasons and levels, on the coagulation profile in animals has been studied, and data showed considerable controversies. 3–5 In some of these studies, stress induced a prolonged activated partial thromboplastin and thrombin time and shortened euglobulin clot lysis time attributed to increased fibrinolytic activity. Interestingly, there was no change in prothrombin time as well as a prolonged decrease in plasma fibrinogen levels. In other studies, platelet dysfunction as well as thrombocytopenia was observed. 3,6,7 Other studies reported prolonged bleeding time, elevated plasma fibrinogen content, and progressively shorter activated partial thromboplastin time. 8
Glucocorticoids are known to have fundamental role in carbohydrates, protein, and lipid metabolism and energy regulation. 9 They also play a critical role in reducing inflammation and immune responses and are commonly referred to as stress hormones. Corticosterone (C21H30O, Kendall’s Compound B) is a glucocorticoid secreted by the cortex of the adrenal gland in response to the stimulation by adrenocorticotropic hormone (ACTH) and is known to be a major indicator of stress in nonhuman mammals. 9 Cortisol on the other hand, also known as hydrocortisone, is the most important glucocorticoid in human. The relationship between cortisol and stress in human has been established, and the effect of cortisol (hydrocortisone) on coagulation in human was studied since 1950. 10,11 However, such relationship has not been clearly defined in rats.
In this study, we retrospectively investigated the effect of construction-induced noise on coagulation in a group of rats. The study was initiated by an unexpected observation of increased blood coagulability in a group of healthy control rats that were exposed to noise due to construction in a building in the vicinity of the animal care facility where these rats were housed. We show that the nearby construction induced stress and resulted in hypercoagulability that limited the utilization of this control rat group in further coagulation-based studies.
Materials and Methods
Animals
The Animal Care Committee of Queen’s University approved the protocol, which followed the Guidelines of the Canadian Council on Animal Care. 14 Sprague-Dawley rats, weighing between 380 and 500 g, were studied retrospectively by dividing into 2 groups according to their exposure to construction noise. Group A (6 rats; 3 males and 3 females) was housed under standard conditions with no exposure to noise or any type of stress, and group B (8 rats; 4 males and 4 females) was unintentionally exposed to noise for approximately 6 hours daily due to construction taking place at a separate building in the vicinity of the animal care facility where these rats were housed. The possibility of construction-induced stress was retrospectively noticed and was unintended.
Blood Sampling
Citrated blood samples (sodium citrate 3.2% 1:9 ratio) were collected from the jugular vein in all the studied rats, consistently in the morning between 8:00 and 9:00
Thromboelastography
Thrombelastography was performed using the Thrombelastograph Analyzer 5000 (Hemoscope Corporation, Skokie, Illinios) immediately after sample collection, using disposable standard cuvettes and pins from Medtel (for Hemoscope Corp) and according to manufacturer’s instructions. Briefly, 340 µL of whole blood was added to 20 µL of calcium chloride 0.2 mol/L in the cuvette, and the procedure was started without a delay. Four TEG parameters were measured as a representation for hemostasis: reaction (R) time, time for initiation to initial fibrin clot formation; alpha angle (α), rate of fibrin formation; maximum amplitude (MA), the maximum strength of the clot and a reflection of the structural interactions and fibrinogen interlaced with fibrin polymers as well as platelet function; and coagulation index (CI), an indicator of the overall coagulation activity, measured using a formula that is derived from the R, MA, and α. All the TEGs were carried out using the same analyzer within 30 minutes after sampling, and generation of full trace lasted for at least 1 hour. Quality control was maintained per manufacturer’s instructions.
Plasma Cortisol and Corticosterone Levels
Stored plasma samples were used to analyze the levels of stress hormones in the 2 groups. This analysis was performed at Dr Wynne-Edwards laboratory, Faculty of Veterinary medicine, University of Calgary. Protein precipitation was first performed followed by solid phase extraction. Cortisol and corticosterone were quantified using liquid chromatography–tandom mass spectrometry.
Statistical Analysis
Analysis was performed using Prism 5 for Windows, Graph Pad version 5.00 (Graph Pad software Inc). A paired 2-tailed Student t test was used to compare the TEG parameters as well as the plasma cortisol and corticosterone levels in both the groups. Spearman rank correlation was used to study the correlation between the changes in TEG values and the plasma cortisol and corticosterone levels.
Results
Construction Induces Hypercoagulability in Rats as Evidenced by TEG
The TEG showed hypercoagulability in group B (stressed rats) when compared to group A (no stress). As shown in Figure 1, there was a significant shortening of the R time (P = .009) and a trend toward an increase in CI (P = .09), indicating hypercoagulability. Although the average values for α and MA in group B (52.14 ± 13.14 and 68.20 ± 5.89 mm, respectively) were considerably higher than that in group A (35.55 ± 20.76 and 60.62 ± 10.86 mm, respectively), these changes were not statistically significant (P = .13 and P = .24, respectively). Representative TEG traces are shown in Figure 2.
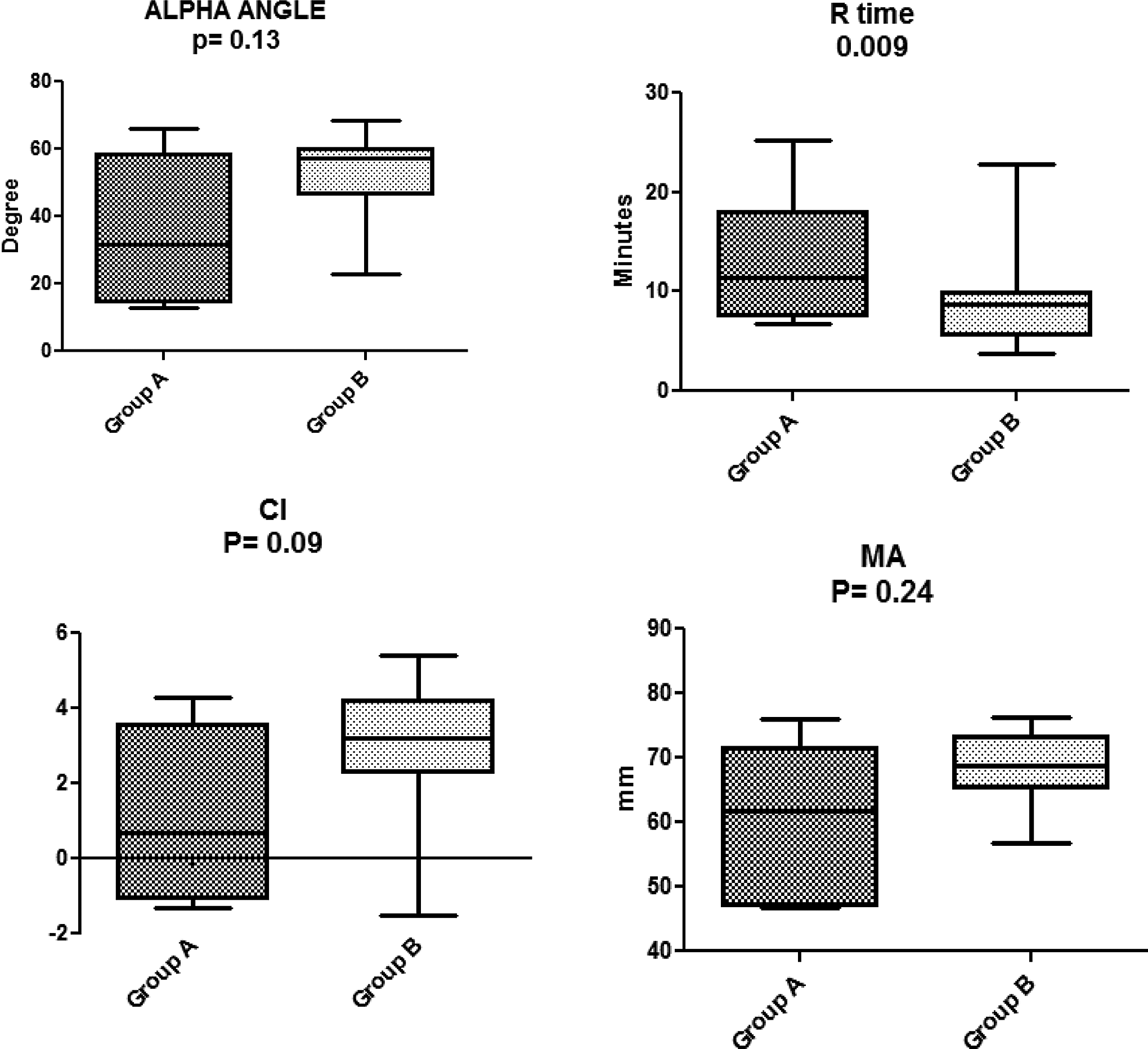
Major thromboelastography (TEG) parameters in group B (exposed to construction noise) in comparison to group A (nonexposed animals). There is significant shortening of R time and significant elevation in overall clotting index (CI). The means for α angle and maximum amplitude (MA) are higher in group B compared to group A. However, this was not statistically significant.
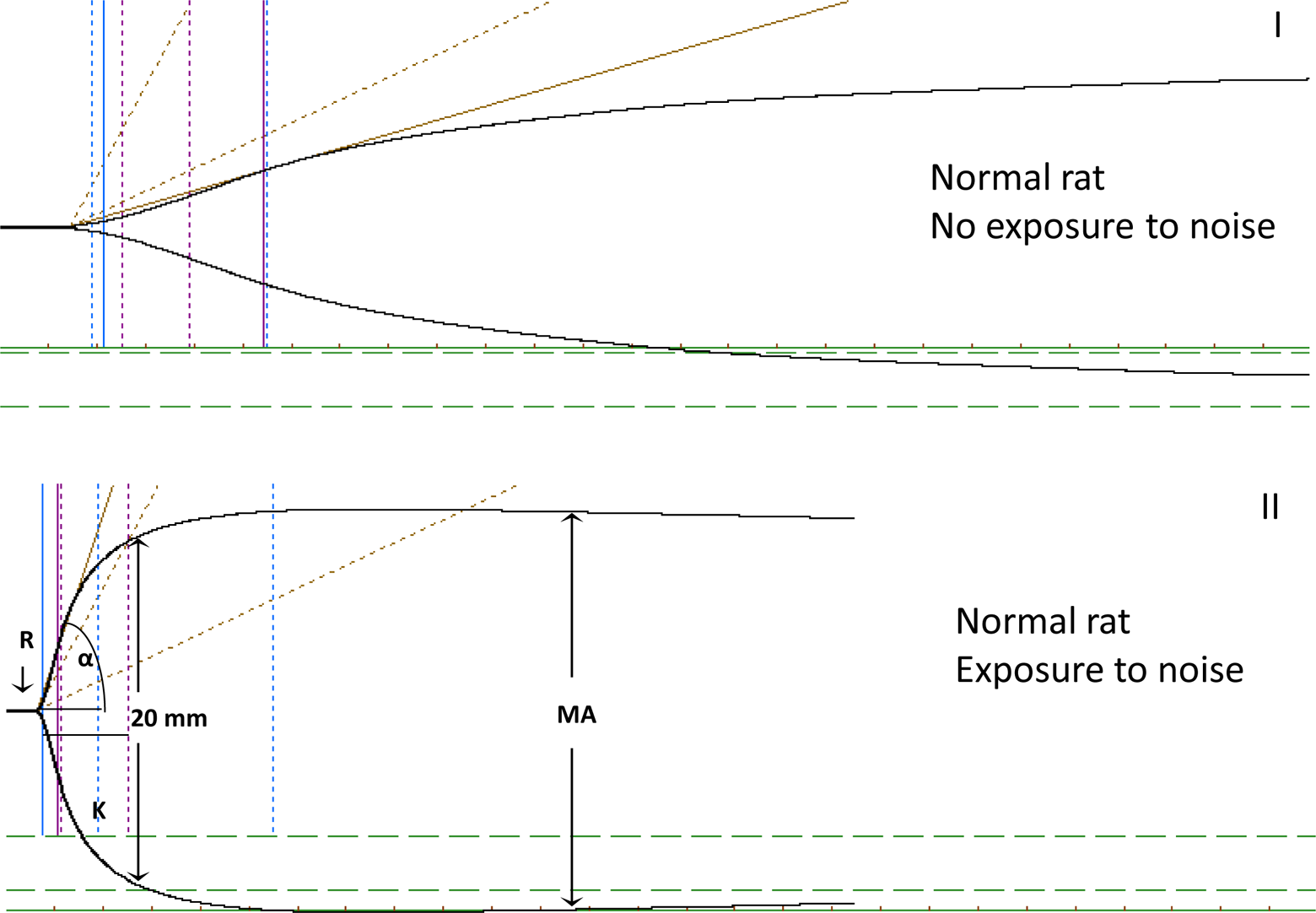
Representative thromboelastography (TEG) traces. A, Normal TEG trace obtained from a rat in group A. B, Hypercoagulable trace obtained from a rat in group B.
Construction Causes Elevation in Plasma Cortisol and Corticosterone Levels
In order to confirm that rats exposed to noise during construction period (group B) had sufficient stress factor to stimulate coagulation, we measured their plasma glucocorticoid levels. We compared these levels to group A, the control rats that were not exposed to construction. We found that the plasma cortisol and corticosterone were elevated in group B when compared to group A (0.66 and 562.2 ng/mL for cortisol and corticosterone compared to 0.52 and 225.7 ng/mL, respectively). Based on student t test, this difference was statistically significant (P = .039 for cortisol and P = .003 for corticosterone; Figure 3).
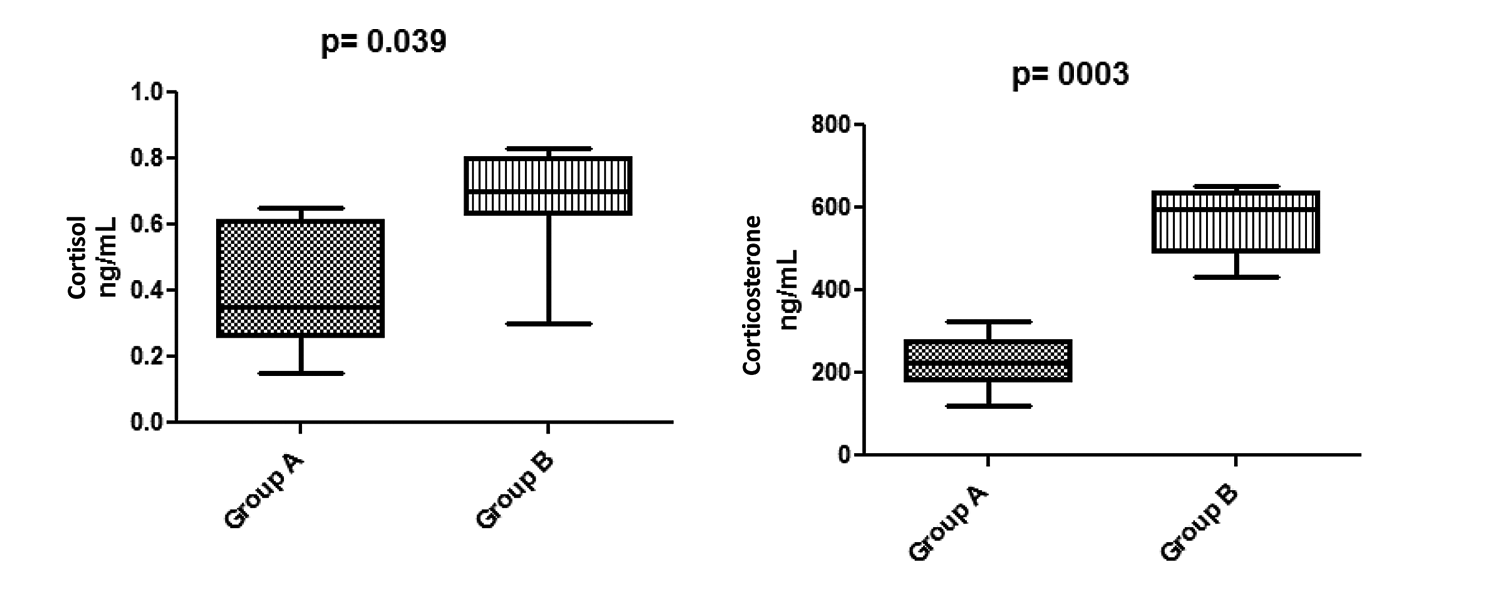
Plasma cortisol and corticosterone levels in rats of group B (exposed to construction noise) in comparison to group A (nonexposed animals). Both stress hormones are significantly elevated in rats exposed to construction noise.
Construction-Induced Hypercoagulability is Correlated With Plasma Glucocorticoid Levels in Rats
We were interested in investigating whether the change in coagulation was correlated with plasma stress hormone levels. Spearman correlation (Table 1) confirmed a significant positive correlation between plasma corticosterone and plasma cortisol. In addition, both R and α correlated with the increase in corticosterone level, while R, α, and CI were correlated with the increase in cortisol levels. There was a reversed correlation with R time and a direct relation to each of α, MA, and CI, indicating that hypercoagulability seen in these rats can be stress induced.
Correlation of Each of the Major TEG Parameters With Plasma Cortisol and Corticosterone.a
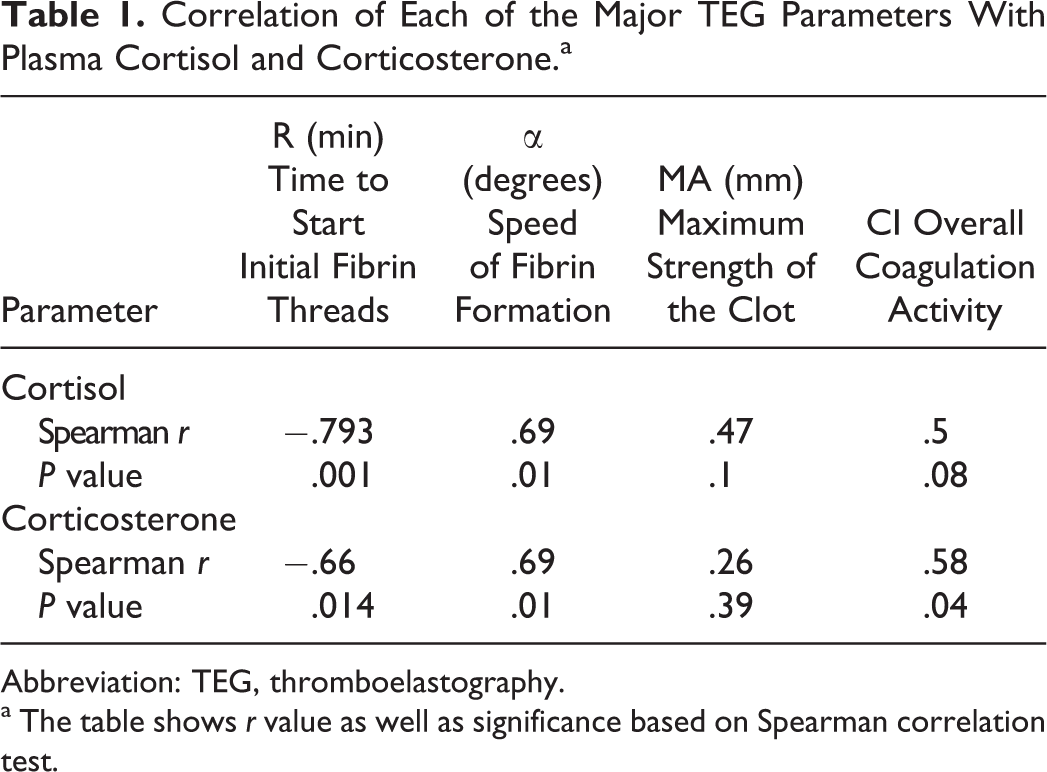
Abbreviation: TEG, thromboelastography.
a The table shows r value as well as significance based on Spearman correlation test.
Discussion
This study shows hypercoagulability in 8 healthy Sprague-Dawley rats that were exposed to intermittent construction noise for approximately 6 hours daily for more than 15 days (retrospectively observed and unplanned), as a result of construction taking place in the vicinity of the animal care facility where these rats were housed. The TEG documented this hypercoagulability in comparison to age-, sex-, and weight-matched 6 other rats tested under complete standard nonstressful conditions. Hypercoagulability significantly correlated with high levels of the 2 stress hormones, cotrisol and corticosterone, in these rats, indicating that this hypercoagulability was stress induced.
There is evidence in the literature about the relation between elevated cortisol and coagulopathy. A study on 302 human blood samples showed that higher morning serum cortisol level predicts increased fibrinogen but not shortened activated partial thromboplastin time. 12 In many species, such as amphibians, reptiles, rodents, and birds, corticosterone is a main glucocorticoid involved in the regulation of metabolism, immune reactions, and stress responses, 11 and in rats, stress-induced elevation of corticosterone was shown to cause subsequent thrombotic complications. 13 Adrenaline, the stress hormone, is a known prothrombogenic substance. 14 In rats, the sympathoadrenal and hypothalamo–pituitary–adrenocortical/thyroid systems are interrelated, and the continuous exposure to cold stress lead to temporary increase in both ACTH and corticosterone serum levels. 15 The levels of these hormones are significantly higher in association with acute intermittent compared to continuous stress. These studies indicate that the relationship between coagulation, stress, and endocrine functions is well established.
The TEG has been used in veterinary medicine since the early 1960s, with numerous publications reporting its first use and was reviewed recently. 16 Furthermore, the standardization of TEG use and application has been a focus of international studies 17 but to our knowledge has not been used to evaluate stress-induced hypercoagulability. The mechanism of hypercoagulability in these rats needs to be further studied. However, based on the correlation between both corticosterone and TEG values, a more pronounced effect with respect to α and R time and less pronounced effect with respect to MA indicate that the stress-induced coagulability and subsequent glucocorticoid release may be related to an increase in coagulation factor levels as well as fibrinogen buildup. However, platelet activation remains a possible factor involved in these mechanisms.
Although the effect of noise on animal biology has been proven in relation to blood pressure, 18 breeding, elevation of plasma catecholamine and steroids, 19 sleep, 20 and behavioral functions, 21 monitoring and recording the change in the acoustic environment is not routinely performed. Although sterilizing the animal environment from noise related to daily routine in animal care facilities including cage movements and washing machines can be difficult, recognizing the effect of noise and the induced stress of animal biology would be important among researchers. Caution has to be particularly exercised when selecting animals for control groups, as failure to do so can result in false data interpretation and considerable waste in animal care research. For details on the effect of noise in animal care facilities and the lack of attention in monitoring acoustic environment, readers are advised to refer to Turner et al. 22
The hypercoagulability documented by TEG in these stressed rats in association with elevated glucocorticoid levels suggests the role of these hormones in thrombotic tendencies. This stress was most probably induced by noise or vibration generated by the construction that took place in the vicinity of animal care facility where the animals were housed. However, other factors such as dust or machinery fumes could not be excluded. To our knowledge, this is the first study using TEG to evaluate stress-induced coagulation in rats and correlate TEG values with plasma stress hormones.
We conclude that noise has to be considered as a stress factor in rats, and caution has to be exercised when selecting animals as controls for hemostasis studies. A larger detailed study to investigate the mechanisms of hypercoagulability in stressed rats and to validate the role of TEG as a tool to identify stress-induced coagulopathy following exposure to unfavorable conditions would be recommended.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by funds from the Canadian Haemophilia Society.
