Abstract
Background:
Older patients with cancer are at high risk of developing venous thromboembolism (VTE) and bleeding. Aspirin may decrease VTE in the general population without significant bleeding. Here, we examined whether aspirin is associated with reduced rates of VTE in older patients with cancer.
Methods and Results:
Using the National Inpatient Sample 2016, we retrospectively identified a cohort of patients with cancer ≥65 years old who received aspirin and a similar cohort who did not receive aspirin (n = 31 654, each). The cohorts were matched for age, sex, race, patient demographics, insurance, hospital demographics, and 9 comorbidities (smoking, obesity, hypertension, hyperlipidemia, diabetes, chronic kidney disease, chronic obstructive pulmonary disease, congestive heart failure, and history of deep venous thrombosis [DVT]/pulmonary embolism [PE]). Primary outcomes were discharge diagnosis of acute PE or acute DVT. Secondary outcomes were inhospital mortality, bleeding, length of hospital stay (LOS), and total hospitalization cost. The aspirin group, compared with the nonaspirin group, had a significantly lower incidence of acute PE (matched, 2.1% vs 2.6%, P < .001), acute DVT (matched, 2.3% vs 3.2%, P < .001), and inhospital mortality (matched 4.0% vs 6.5%, P < .001); shorter LOS (matched, 5.29 ± 5.01 vs 6.20 ± 6.56 days, P < .001); and lower total costs (matched, US$14 700 ± 15 031 vs US$16 363 ± 20 219, P < .001). The primary and secondary outcomes were similar before and after propensity matching. We found no increase in bleeding in the aspirin group compared to the nonaspirin group: gastrointestinal bleeding (matched, 3.8% vs 4.0%, P= .168), hematuria (matched, 3.5% vs 3.7%, P = .102), hemoptysis (matched, 0.9% vs 0.9%, P = .532), and hemorrhagic stroke (matched, 0.8% vs 0.8%, P = .443). In subgroup analyses, aspirin was associated with decreased inhospital mortality, mostly in patients with lung, colon, pancreatic, prostate, breast cancer, lymphoma, and leukemia.
Conclusions:
Among older patients with cancer, aspirin was associated with lower VTE incidence and overall inhospital mortality without significantly increased bleeding.
Introduction
The incidence of venous thromboembolism (VTE) is high among patients with cancer because of their hypercoagulable state due to increased procoagulant factors, platelet activation, and the endothelium damage associated with cancer. 1 -6 Venous thromboembolism, particularly pulmonary embolism (PE), is a common complication in patients with cancer and usually requires hospitalization. 7,8 Advanced age is also an independent risk factor for VTE. Epidemiologic data have shown that the incidence of VTE increases exponentially with age. 9,10 Patients with cancer who develop VTE are at high risk of bleeding when treated with anticoagulants. 11 -14 Studies have shown that being older than 65 years is a risk factor for bleeding in response to anticoagulation. 15 -17 The risk of major bleeding increases about 2-fold in patients over 65 years. Moreover, older age is also associated with higher mortality. These factors make decisions about prophylactic anticoagulant use challenging when treating older patients with cancer. 15 -17
The 2 leading causes of death worldwide—cancer and cardiovascular disease (CVD)—share many risk factors, including chronic inflammation, age, and smoking. 18 Aspirin, which reduces arterial thrombosis, is used to help prevent CVD. In addition, aspirin is effective in preventing VTE. 19,20 In 2 multicenter randomized trials, WARFASA and ASPIRE, with a combined total enrollment of 1224 patients, aspirin was better than placebo in reducing VTE recurrence in patients with unprovoked VTE after anticoagulant discontinuation and did not significantly increase the risk of bleeding. 21 -23 Both studies were performed in the general population and focused on unprovoked VTE; neither was conducted in patients with cancer who usually have provoked VTE due to cancer or cancer therapy–related hypercoagulability. Although guidelines have considered the value of aspirin in thromboprophylaxis in patients with multiple myeloma on chemotherapy with thalidomide- or lenalidomide-based regimens and/or dexamethasone, the efficacy of aspirin remains unclear in patients with other types of cancers. Moreover, no guidelines have focused on aspirin use in elderly patients with cancer. 24,25
In the present study, we examined whether aspirin was associated with lower rates of VTE and mortality in older patients with cancer. We used data from the latest available National Inpatient Sample (NIS), which is the largest inpatient discharge database in the United States and stores data on more than 210 000 cancer patients who are ≥65 years old.
Materials and Methods
Study Design and Data Source
The NIS is part of a family of databases developed for the Healthcare Cost and Utilization Project (HCUP). It is a database of hospital inpatient stays derived from billing data submitted by hospitals to statewide data organizations across the United States. The inpatient data include clinical and resource use information typically available from discharge abstracts. The NIS approximates a 20% stratified sample of discharged patients from the US community hospitals, excluding rehabilitation and long-term acute care hospitals. Beginning with the data year 2016, the NIS includes discharge diagnoses coded using only International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) from the discharge records from all HCUP participating hospitals. In this retrospective cohort study, we retrieved data from the NIS 2016 database on the following variables: primary and secondary diagnoses, discharge status, length of hospital stay (LOS), patient demographic characteristics, hospital characteristics, total charges, expected payer, and severity and comorbidity measures. This database conforms to the definition of a limited data set, which is defined as health care data in which direct identifiers (eg, name, address, birth date, telephone number, email address, social security number, and medical record number) have been removed. Thus, review by an institutional review board was not required under the Health Insurance Portability and Accountability Act.
We used the ICD-10-CM to identify or eliminate diseases, comorbidities, and outcomes. Specifically, we used discharge diagnosis ICD-10 Z7982, which captures long-term (current) use of aspirin, to identify patients who were receiving aspirin. Based on the definition of ICD-10 Z7982 and the data source, the patient was receiving long-term (current) aspirin treatment at the time of discharge. However, no information is available regarding when the aspirin regimen was started.
Patient Selection
We included all patients with cancer aged ≥65 years who were admitted to the hospital from January 1, 2016, to December 31, 2016. We used ICD-10-CM codes to identify all types of cancers and their locations (Supplemental Table 1).
Patients were excluded if they were taking anticoagulant or antiplatelet drugs other than aspirin or if the information on discharge status was unavailable. Eligible patients were stratified on the basis of aspirin use: 31 654 patients used aspirin (aspirin group) and 234 421 patients did not use aspirin (Figure 1).

Flow chart of the selection process for the final patient sample used in this study. Inclusion criteria were applied to the National Inpatient Sample 2016 database. All eligible patients were matched 1:1 based on propensity scoring to generate the aspirin versus nonaspirin comparison cohorts. ICD-10-CM indicates International Classification of Diseases, Tenth Revision, Clinical Modification.
Variables
We extracted data directly from the NIS on baseline demographic information, including age, sex, race, household income (based on ZIP code), primary payer, patient location, and hospital characteristics including hospital type and region. Nine comorbidities were identified by ICD-10-CM codes (Supplemental Table 1): smoking, obesity, hypertension, hyperlipidemia, diabetes, chronic kidney disease (CKD), chronic obstructive pulmonary disease (COPD), congestive heart failure (CHF), and history of deep vein thrombosis (DVT)/PE.
Outcomes
The primary outcomes were a discharge diagnosis of acute PE or acute DVT, based on ICD-10-CM codes (Supplemental Table 1). The secondary outcomes identified by ICD-10-CM codes (Supplemental Table 1) were inhospital mortality, LOS, total hospitalization costs, and 4 types of bleeding: gastrointestinal (GI) bleeding, hematuria, hemoptysis, and hemorrhagic stroke.
Statistical Analyses
Categorical variables are expressed as numbers and proportions and were compared by using the χ2 test. Continuous variables are expressed as the mean and standard deviation. The 95% CI for the mean difference was also provided.
To reduce selection bias in an unmatched cohort, we performed a propensity score-matched analysis of the aspirin versus nonaspirin group. To measure propensity scores, we constructed a multivariate logistic regression model and adjusted it for age, sex, race, mean household income, hospital type, hospital region, and the 9 comorbidities described above. The subsequent propensity scores were used for nearest-neighbor matching between the 2 groups, with a caliper match tolerance of 0.1.
We compared primary and secondary outcomes between the aspirin and nonaspirin groups in both the unmatched and the propensity score–matched cohorts. Because different cancer types may carry differing risks of DVT, 6 we divided patients into subgroups based on the type of cancer and performed a propensity score-matched analysis of aspirin versus nonaspirin effects in the subgroups. We evaluated the primary (acute PE and acute DVT) and secondary (inhospital mortality, LOS, and total cost) outcomes in the subgroups.
We used R statistics software (version 3.6.1, R Development Core Team) to perform all statistical analyses. The matching process was conducted using the MatchIt package in R software. This package has built-in features that allow the process to be performed easily and therefore is widely used for propensity score matching and other types of matching. P values <.05 were considered significant.
Results
Baseline Characteristics
Baseline characteristics before and after propensity score matching are presented in Table 1. The matched cohorts comprised 31 654 patients with cancer in both the aspirin and nonaspirin groups. In the matched cohorts, the 2 groups were similar in age (76.46 ± 7.39 years vs 76.47 ± 7.41 years; P = .907), sex (men, 58.7% vs 58.3%; P = .692), and hospital type (P = .494). These 2 groups were also matched with regard to the 9 comorbidities: smoking (51.6% vs 51.6%, P = .886), obesity (11.2% vs 11.0%, P = .631), hypertension (56.3% vs 56.7%, P = .378), hyperlipidemia (54.4% vs 54.1%, P = .444), diabetes (34.1% vs 34.0%, P = .693), CKD (24.2% vs 24.3%, P = .718), COPD (25.4% vs 25.9%, P = .145), CHF (19.0% vs 19.0%, P = .879), and history of DVT/PE (2.0% vs 2.2%, P = .318). All standardized differences were less than 10%.
Baseline Characteristics.
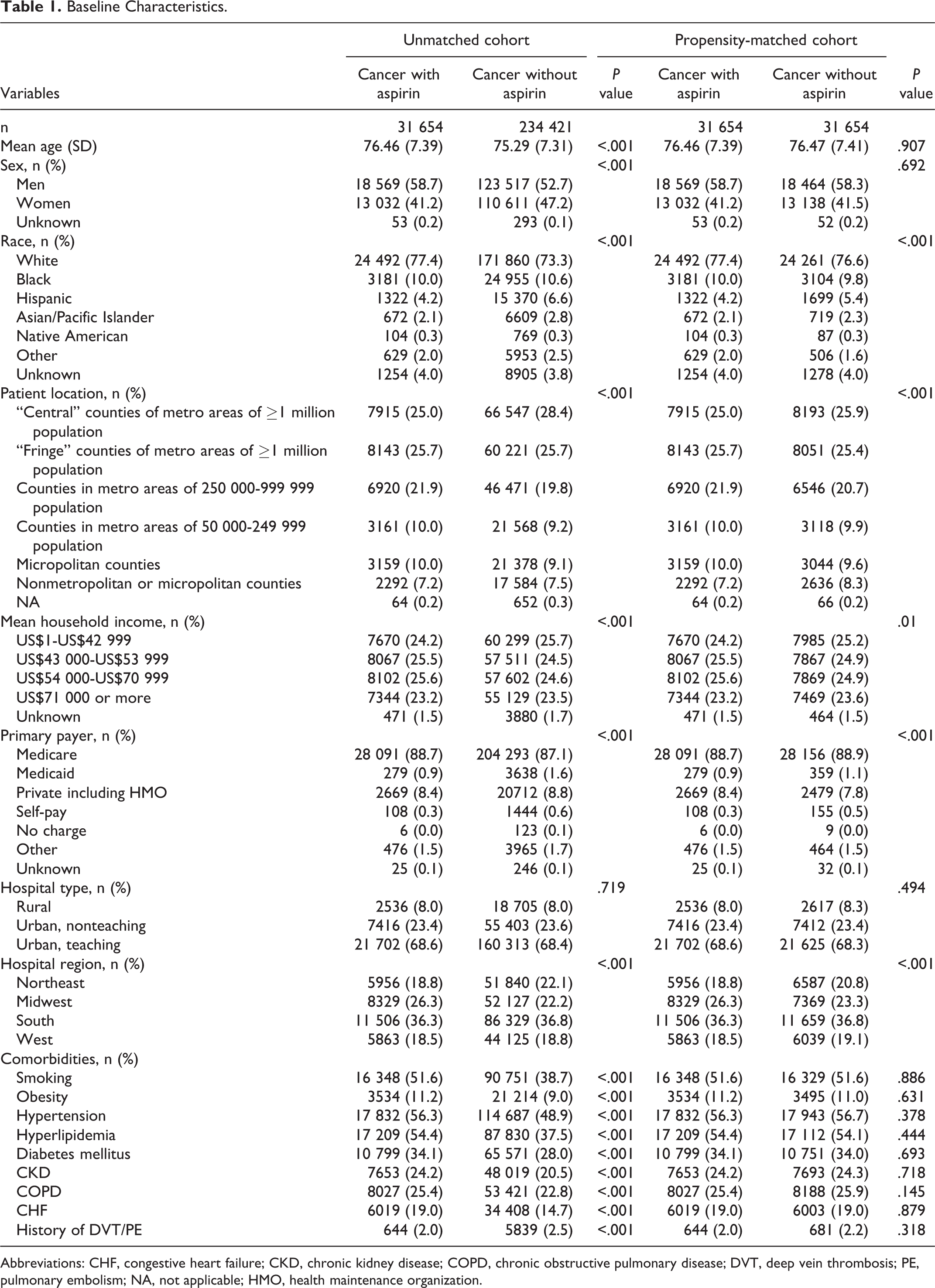
Abbreviations: CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; DVT, deep vein thrombosis; PE, pulmonary embolism; NA, not applicable; HMO, health maintenance organization.
Acute PE and DVT
We compared primary and secondary outcomes in the aspirin and nonaspirin groups, both before and after propensity score matching (Table 2). The aspirin group, compared with the nonaspirin group, had significantly lower incidences of acute PE (unmatched, 2.1% vs 2.6%, P < .001; matched, 2.1% vs 2.6%, 95% CI for the mean difference [−0.7%, −0.3%], P < .001) and acute DVT (unmatched, 2.3% vs 3.3%, P < .001; matched, 2.3% vs 3.2%, 95% CI for the mean difference [−1.1% to −0.6%], P < .001), respectively.
Primary and Secondary Outcomes.

Abbreviations: DVT, deep vein thrombosis; GI, gastrointestinal; PE, pulmonary embolism.
Inhospital Mortality, Length of Stay, and Total Cost
In both the unmatched and matched cohorts, the aspirin group, compared to the nonaspirin group, had significantly lower inhospital mortality (unmatched, 4.0% vs 6.8%, P < .001; matched, 4.0% vs 6.5%, 95% CI for the mean difference [−2.9% to −2.2%], P < .001), a shorter LOS (unmatched, 5.29 ± 5.01 days vs 6.26 ± 6.96 days, P < .001; matched, 5.29 ± 5.01 days vs 6.20 ± 6.56 days, P < .001), and a lower total cost (unmatched, US$14700 ± US$15 031 vs US$16 545 ± US$21 125, P < .001; matched, US$14 700 ± US$15 031 vs US$16 363 ± US$20 219, P < .001), respectively (Table 2).
Bleeding Events
In unmatched cohorts, compared with the aspirin group, the nonaspirin group had an increased risk of GI bleeding (unmatched, 3.8% vs 4.0%, P = .04) and hemorrhagic stroke (0.8% vs 0.9%, P = .039). However, in the matched cohorts, all 4 types of major bleeding were comparable in the aspirin and nonaspirin groups: GI bleeding (matched, 3.8% vs 4.0%, P = .168), hematuria (matched, 3.5% vs 3.7%, P = .102), hemoptysis (matched, 0.9% vs 0.9%, P = .532), and hemorrhagic stroke (matched, 0.8% vs 0.8%, P = .443; Table 2).
Aspirin Effects in Different Cancer Types
The results of the cancer subgroup analyses are shown in Table 3. After propensity score matching, the incidence of acute PE/DVT was not significantly different in the aspirin and nonaspirin groups except for patients with lymphoma, but the inhospital mortality was lower in the aspirin group than in the nonaspirin group in lung, colon, pancreatic, prostate, kidney, breast cancer, lymphoma, and leukemia.
Subgroup Cancer.

Abbreviations: DVT, deep vein thrombosis; LOS, length of stay; PE, pulmonary embolism.
Discussion
In this study, we found that aspirin use was associated with reductions in acute PE and DVT in both matched and unmatched cohorts. Furthermore, in older patients with cancer, aspirin use was associated with a decreased inhospital mortality. Our cancer subgroup analyses showed an association between aspirin use and greater reductions in mortality in patients with certain cancer types. Most importantly, aspirin achieved these effects without evidence of increased bleeding. Thus, our study suggests that aspirin may be considered in select patient groups who have a high risk of bleeding or contraindications to anticoagulation for VTE prophylaxis.
Our finding that VTE was reduced in older patients with cancer supports results reported in previous studies in the general population with unprovoked VTE. 19 -23 The WARFASA trial showed that aspirin reduced the risk of VTE recurrence by about 40% in patients with unprovoked VTE after anticoagulant discontinuation. 21 In the ASPIRE trial, the annual recurrence of VTE was reduced by 26% with aspirin use, but the effect was not statistically significant. 22 Both studies found that aspirin did not significantly increase the risk of bleeding, a finding similar to ours.
Platelet activation is part of the thrombus formation process, and platelets are a component of venous thrombi. 26 -28 Tumor cells can directly induce tumor-platelet aggregates, trigger platelet granule release, and enhance thrombopoiesis; these activities contribute to hypercoagulability in patients with cancer. 29,30 Aspirin prevents platelet activation and VTE formation by inhibiting cyclooxygenase-1 (COX-1), which is constitutively expressed in platelets. 31
In our study, the aspirin group had a decreased incidence of VTE and, even more importantly, a significant reduction in inpatient mortality. Our finding differs from that reported in the ASPREE trial, which was conducted in 19 114 men and women ≥70 years old without CVD. The ASPREE investigators gave patients 100-mg aspirin daily and followed them for a median of 4.7 years; they found an increase in all-cause mortality, which was attributed primarily to cancer-related deaths. 32 Several factors may have contributed to these disparate results. First, the ASPREE trial was conducted in relatively healthy older patients without CVD, whereas we included older patients with cancer who could also have had CVD because CVD and cancer have similar risk factors, and aspirin is beneficial in the secondary prevention and reduction of myocardial infarction, stroke, and cardiovascular death. 18,22,33,34 Second, the ASPREE trial evaluated all-cause mortality during a median of 4.7 years of follow-up, and we evaluated only inpatient all-cause mortality. Third, VTE is associated with increased morbidity and mortality in individuals with cancer 4,33,35 ; thus by reducing the VTE incidence in our study, aspirin may have indirectly reduced mortality among hospitalized older patients with cancer.
In our cancer subgroup analysis, aspirin had significant effects in reducing inpatient mortality in lung, colon, pancreatic, prostate, kidney, and breast cancers and in leukemia and lymphoma. In a previous study, investigators combined data from 8 large randomized trials of 25 570 patients with cancer and found that aspirin reduced the number of deaths due to several common cancers, including GI (colorectal and esophageal), pancreatic, lung, prostate, and brain cancers. Moreover, aspirin decreased the risk of distant metastasis in adenocarcinoma, which might account for a reduction in cancer deaths. 36,37 In those studies, low-dose and high-dose aspirin had similar effects. These findings may be explained by multiple factors. First, platelet activation has been linked to tumorigenesis and metastasis by reinforcing tumor growth with proliferation signals, antiapoptotic effects, and angiogenic factors. 31,38,39 Second, aspirin inhibits COX-2, which is not expressed in most normal cells but is overexpressed in certain cancer cells, including colon, pancreatic, and breast cancers; COX-2 catalyzes the synthesis of prostaglandin E2, which promotes tumor proliferation and angiogenesis and inhibits immune surveillance and apoptosis. 40 -42
We were able to calculate 4 major types of bleeding in this study: GI bleeding, hematuria, hemoptysis, and hemorrhagic stroke. Our analysis found that aspirin did not increase the risk of these types of bleeding. Of note, the bleeding risk calculated from this study is total bleeding risk instead of major bleeding risk in other studies. In the ASPREE study (n = 19 114 healthy older patients), the major bleeding rate was 8.6 and 6.2 events per 1000 person-years in the aspirin and placebo groups, respectively. Aspirin-induced bleeding occurred most commonly in the GI tract, particularly in the upper tract. The rate of fatal bleeding was less than 1 per 1000 person-years in both groups (0.7 and 0.6 in the aspirin and placebo groups, respectively). 43 In comparison, a large meta-analysis of 9 randomized trials was performed to study bleeding in patients with cancer who were treated with anticoagulants for VTE. The calculated major bleeding rate within 1 year was 3.6% (95% CI: 2.3%-4.9%; P < .001) with warfarin 4.5% (95% CI: 2.9% to 6.2%; P < .001) with low molecular weight heparin, and 3.6% (95% CI: 1.3%-6.0%; P < .001) with direct oral anticoagulants. However, the investigators did not report the bleeding rates in older patients, which presumably would have been even higher. 44 In the more recent SELECT-D trial, VTE in patients with cancer was treated either with rivaroxaban or dalteparin; the 6-month cumulative rate of major bleeding was 6% and 4%, respectively. 45 Even if aspirin is associated with increased bleeding risk, these data show the risk is not nearly as high as that associated with anticoagulants.
Our study had several major strengths. First, we used the NIS database, which included more than 210 000 older patients with cancer. Second, our findings are applicable clinically. Although oral anticoagulant therapy and low molecular weight heparin are both effective in treating and preventing VTE, they carry a significantly high risk of bleeding, particularly in older patients, which limits their long-term use. 11,12,15 -17,46 In contrast, for older patients with cancer who need aspirin for secondary prophylaxis of CVD or stroke, aspirin may provide additional benefits because of its association with a lower incidence of provoked VTE and inpatient mortality, with no increased risk of bleeding. Our findings suggest that the use of aspirin may require more consideration; aspirin is inexpensive (in contrast to novel oral anticoagulants), convenient (in contrast to low molecular weight heparin), does not require monitoring (in contrast to warfarin), and does not accumulate in patients with renal insufficiency (in contrast to rivaroxaban).
Our study has limitations. First, due to its retrospective design, our study allows to establish associations but not causality. Additional findings from a multicenter, randomized controlled study should be used to confirm our conclusions. Second, the dose and duration of aspirin administration were not specified in the NIS data set. We used the discharge ICD-10 code Z7982, which indicates long-term (current) use of aspirin by definition, to identify aspirin use. However, we were unable to obtain the medication compliance data of the population; therefore, we could not define the minimal effective dose. Third, the absence of cancer therapy data in the data set prevented us from examining therapy-related effects, and the lack of prechemotherapy hemoglobin level and platelet and leukocyte counts prevented calculation of the Khorana risk score for identifying those patients with cancer at highest risk of VTE. 47,48 Fourth, the validity of ICD-10 in identifying VTE is unknown in this database. Fifth, patients who are at high risk of bleeding and who are sicker may be less likely to receive aspirin, which may contribute to the reduction of mortality seen in the aspirin group. Finally, although the strength of the study relies on the use of a large national database, it can also be a weakness because of the lack of precise information, such as hospice transfer and accurate household income, and the problems inherent with using administrative data.
In conclusion, we have shown that the use of aspirin was associated with a reduced incidence of provoked VTE and inpatient mortality in older patients with cancer.
Supplemental Material
Supplemental Material, 4-15-Supplement_Table - Aspirin Is Associated With Reduced Rates of Venous Thromboembolism in Older Patients With Cancer
Supplemental Material, 4-15-Supplement_Table for Aspirin Is Associated With Reduced Rates of Venous Thromboembolism in Older Patients With Cancer by Pengyang Li, Ying Ning, Mu Li, Peng Cai, Ahmad Daniyal Siddiqui, Eric Yang Liu, Michelle Hadley, Fangcheng Wu, Su Pan, Richard A. F. Dixon and Qi Liu in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgment
The authors thank Rebecca Bartow, PhD, at the Texas Heart Institute for providing editorial support.
Author Contribution
Pengyang Li, MD, and Ying Ning, MD, contributed equally to the writing of this article and share primary authorship.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
