Abstract
Background:
The objective of this study was to evaluate whether admission neutrophil–lymphocyte ratio (NLR) and platelet–lymphocyte ratio (PLR) might reflect amputation in patients with critical limb ischemia (CLI) who could not get surgical or radiological (percutaneous transluminal angioplasty) revascularization.
Methods:
A total of 104 patients with nonreconstructable CLI over a 5-year period were collected prospectively.
Results:
Admission NLR levels of ≥3.2 and a PLR of ≥160 were found to represent the optimal cutoff values to risk stratification of patients. If both levels were elevated, patients had a median overall limb survival of 22 months. For cases where both levels were less than the cutoff values, the median overall limb survival time was not reached but was greater than 60 months. Conclusions: Admission NLR and PLR both merit further evaluation as prognostic indices in patients with CLI.
Introduction
Patients with critical limb ischemia (CLI) comprise a subset of high-risk patients with generalized atherosclerotic disease of the arterial tree. The presence of systemic atherosclerosis is associated with a low-grade systemic inflammatory response. 1,2 Previous studies have suggested that elevated preoperative C-reactive protein (CRP) levels, 3,4 neutrophil–lymphocyte ratio (NLR), 1,5 thrombocytosis, and platelet agregation 6 may also represent potential prognostic markers in peripheral arterial disease. However, no previous studies have investigated whether platelet–lymphocyte ratio (PLR) might provide any prognostic information in CLI. Similarly, to the best of our knowledge, there is no report or study concerning NLR-amputation relation.
The objective of this study was to determine whether admission NLR levels and the PLR might represent significant prognostic indices in patients with CLI and to determine whether a combination of these parameters might provide superior prognostic information.
Methods
A total of 112 patients presented with CLI to our hospital between February 2007 and June 2012 who could not have radiological (percutaneous transluminal angioplasty) or surgical revascularization and had medical treatment are included in our study. These patients were assessed by the vascular team consisting of cardiologists, interventional radiologists, and cardiovascular surgeons and accepted as ineligible for endovascular intervention or conventional surgery thus getting medical therapy instead. All patients with CLI defined by the TASC II intersociety consensus for the management of peripheral arterial disease. The arterial anatomy of the lower extremities was assessed by digital subtraction angiography with use of either stepping techniques with a single-contrast material bolus or multiple contrast material injections. Inclusion criteria were persistent rest pain for more than 2 weeks or ischemic skin lesions, ankle pressure below 50 mm Hg, or absent palpable ankle pulses or toe pressure below 30 mm Hg in patients with diabetes and incompressible vessels. Standard treatment included cilostazol, statins, analgesics, antithrombotic drugs (aspirin), cardiovascular risk factor control, hemorrheologic agents (such as pentoxifylline), local wound care, and antibiotics, if indicated. The local ethics committee approved the study protocol.
Patients with infected ischemic wounds (n = 4), active cancer (n = 1), unstable coronary disease (n = 2), and steroid use (n = 1) were excluded from outcome analysis. Eight patients were excluded according to these criteria. Thus, 104 patients were included in the study.
Demographic characteristics, medical histories, risk factors, laboratory studies, and outcomes data were collected by the research team using a standardized database. Prior surgical or radiological revascularization was documented. Any subsequent surgical intervention was also documented as follows: major amputation (above the level of the ankle) or minor amputation (below the level of the ankle).
Primary end point was determined as amputation (limb survival) and all-cause death and separate analyses were performed. The effect of NLR and PLR on outcome was studied by constructing a receiver–operating characteristic curve. For amputation, an NLR ≥3.2 and PLR ≥160 were used to categorize patients into low- and high-NLR\PLR groups. A further stratification index was generated for those patients where both admission NLR and PLR were recorded (n = 104). If both levels were above the selected threshold values (n = 28), patients were classified as “high risk”. If either NLR or PLR were elevated individually (n = 30), patients were classified as “intermediate risk”. For the remaining cases where both levels were less than the cutoff values (n = 46), patients were classified as “low risk”.
The admission full blood count was recorded where available. Levels of hemoglobin, white blood cells, platelet, neutrophil, and lymphocyte were measured using a Siemens Advia 2120 analyzer.
After the decision was made for patients as inoperable CLI, they were called in for follow-ups at the first, third, and ninth months and then with 6-month intervals. Patients were contacted during routine check-ups at our center on a periodic, but not uniform, basis. During the follow-up, the following events were registered: (1) mortality for all causes and (2) major or minor amputation. Latest statuses of the patients were ascertained via both phone and Social Security Death Index at the end of the study.
Statistics
Continuous data were described using median, interquartile range, and 95% confidence intervals (CIs). Categorical variables were summarized as percentages. The effect of NLR on outcome was studied by constructing a receiver–operating characteristic curve with amputation as the primary variable. Potential prognostic factors were entered into univariate Cox regression models of midterm limb survival. Significant univariate correlates were then entered into a reverse stepwise multivariate Cox regression model to test for independence. The least significant variable was dropped from each iteration of this model until all remaining variables were significant. Kaplan-Meier survival curves and the log-rank test were used to compare limb survival between the low-, intermediate-, and high-risk groups. A P value <.05 was considered statistically significant and the CI was 95%. Analyses were performed using SPSS software.
Results
Of the 104 patients in the study, 83% were male and 17% were female. The median age was 69 years and median follow-up was 39 months (Table 1). In all, 71(68%) patients had nonreconstructable (radiological or surgical revascularization) arterial outflow vessels, 19(18%) was inoperable due to the comorbidities, 11(11%) did not have suitable vein grafts, and 3(3%) did not accept the revascularization (radiological or surgical revascularization). All were Rutherford classification 4 or 5 with multilevel disease. As expected in these high-risk patients, 71% were current smokers and 47% had hyperlipidemia.
Baseline Characteristics of Patients Divided According to Amputation.a
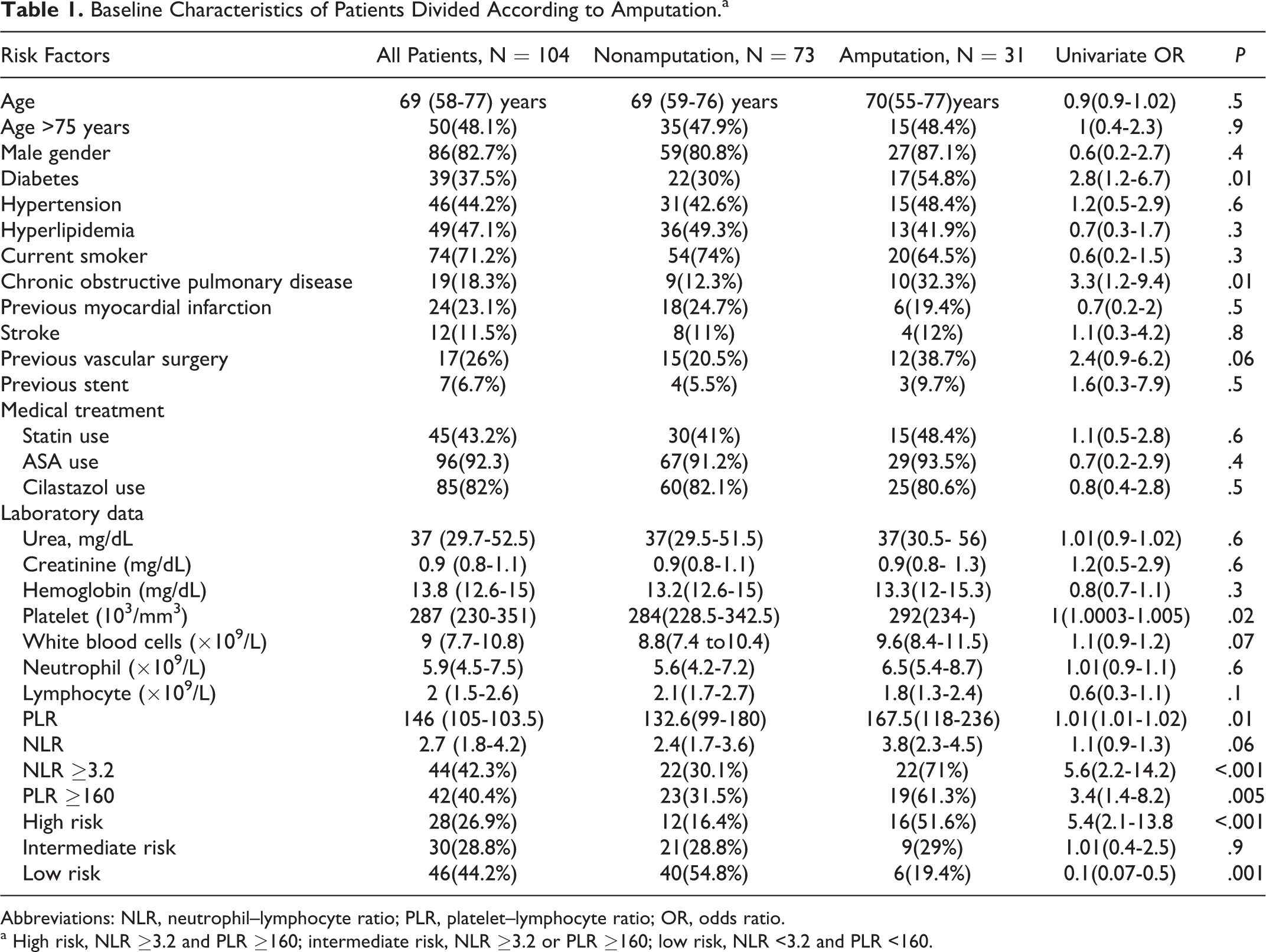
Abbreviations: NLR, neutrophil–lymphocyte ratio; PLR, platelet–lymphocyte ratio; OR, odds ratio.
a High risk, NLR ≥3.2 and PLR ≥160; intermediate risk, NLR ≥3.2 or PLR ≥160; low risk, NLR <3.2 and PLR <160.
Using an NLR cutoff of ≥3.2, the area under the receiver–operating characteristic curve was 0.71 (95% CI 0.54-0.78). Using a PLR cutoff of ≥160, the area under the receiver–operating characteristic curve was 0.66 (95% CI 0.54-0.78).
Overall, there were a total of 28 (26.9%) deaths and 31 (29.8%) amputation over a mean follow-up of 46 months. The amputations were above ankle in 24 (77%) and below ankle in 7 (23%).
Patients with a high risk showed the highest amputation (n = 16, 57%) when compared to PLR ≥160 (n = 19, 45%), NLR ≥3.2 (N = 22, 50%), low risk (n = 6, 13%), and intermediate risk (n = 9, 30%; Figure 1).
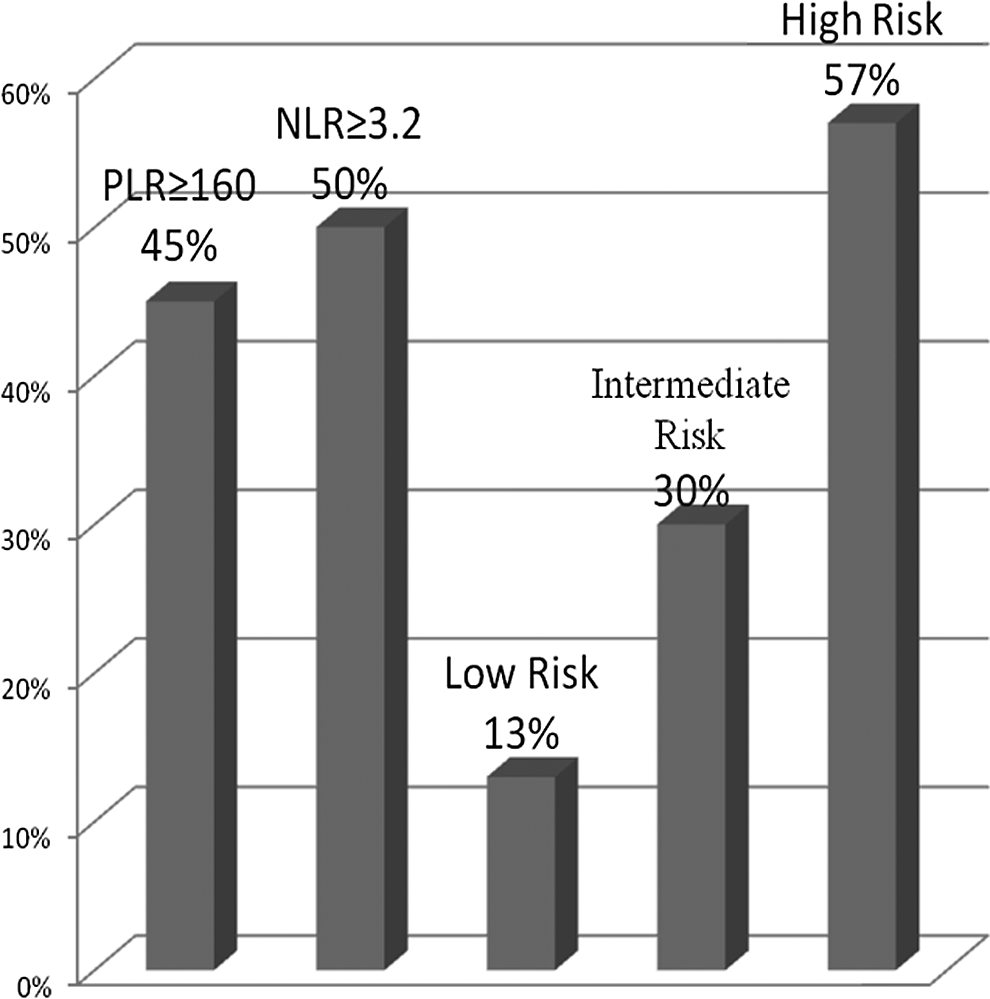
Amputations according to the neutrophil–lymphocyte ratio and platelet–lymphocyte ratio.
The median overall limb survival time of high-risk and intermediate risk-patients was 22 (95% CI = 6.2-37.8) and 36 (95% CI = 24.1-47.9) months, respectively. The median overall limb survival time for “low-risk” group was not reached but was found to be in excess of 60 months on Kaplan-Meier analysis (Figure 2). When comparing the survival between these groups a log-rank P value less than .001 was obtained.
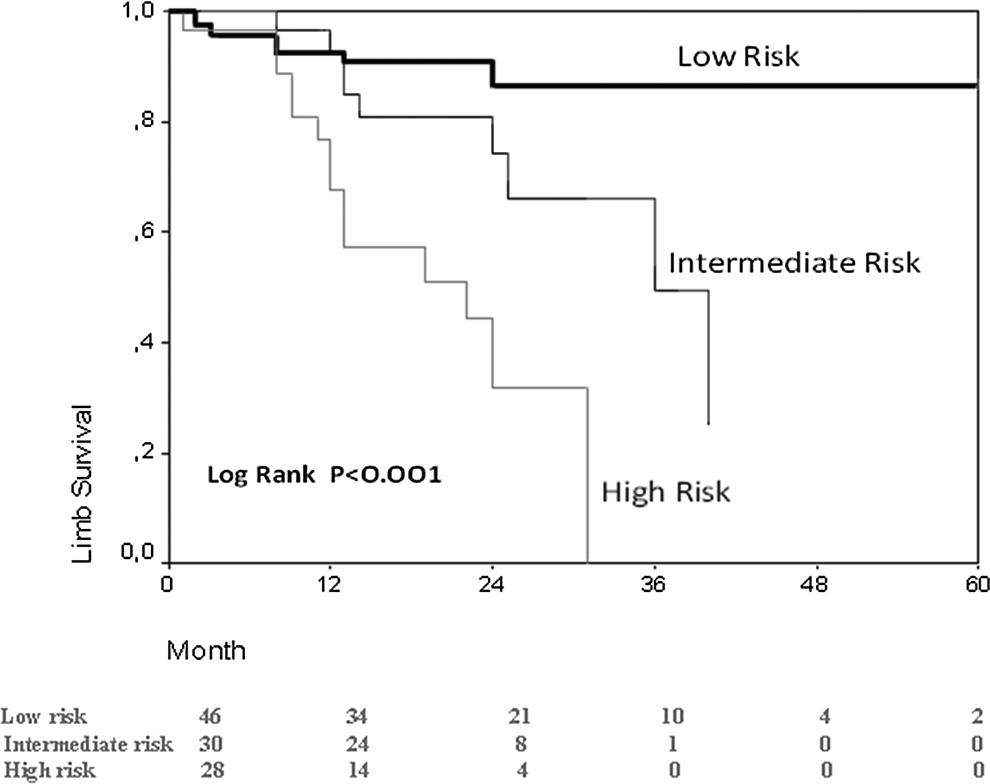
Kaplan-Meier cumulative limb survival curves for patients with critical limb ischemia according to combined neutrophil–lymphocyte ratio (NLR) and platelet–lymphocyte ratio (PLR) risk stratification.
The results of univariate limb survival analysis for each of the demographic and hematologic data are shown in Table 1. All patients were grouped in 2, those with amputation and those without amputation. Diabetes mellitus, chronic obstructive pulmonary disease, platelet, mean PLR, PLR ≥160, NLR ≥3.2, and high-risk patient (Table 1) were strong univariable predictors of amputation.
Factors associated with midterm amputation were diabetes mellitus (odds ratio [OR], 2.8; 95% CI, 1.2-6.7; P = .01), chronic obstructive pulmonary disease (OR, 3.3; 95%CI, 1.2-9.4; P = .01), and high-risk patients (OR, 5.4; 95% CI, 2.1-13.8; P < .001). When these 3 variables were included in a multivariate regression modeling, diabetes mellitus (OR, 3.1; 95% CI, 1.1-8.3, P = .02), chronic obstructive pulmonary disease (OR, 4.1; 95% CI, 1.3-13.4; P = .01), high-risk patients (OR, 4.7; 95% CI, 1.7-12.6, P = .01) remained as independent factors associated with midterm amputation (Table 2).
Multivariate (Cox Proportional Hazards) Limb Survival Analysis.
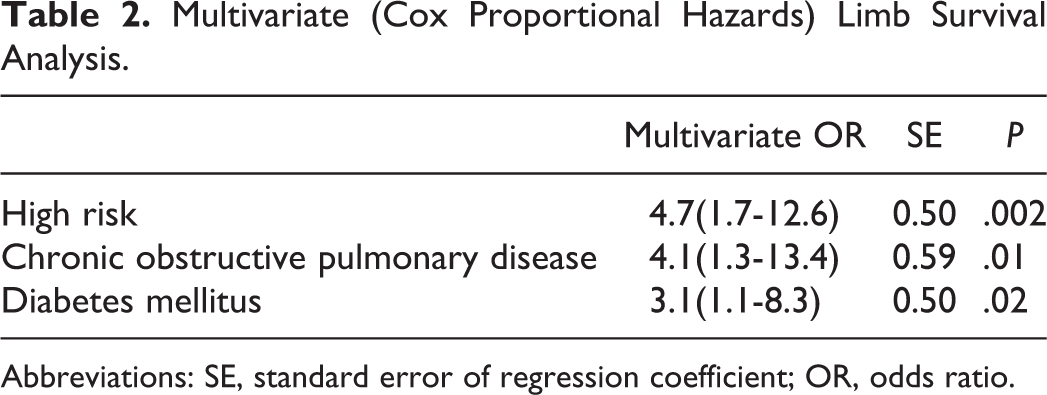
Abbreviations: SE, standard error of regression coefficient; OR, odds ratio.
Discussion
The findings of the present study demonstrate that NLR and PLR obtained from a universally available low-cost test (CBC with differential) provide relevant information regarding the risk of amputation in patients who are admitted with nonreconstructable CLI.
Chronic inflammation is both an etiological factor and a physiological consequence of peripheral arterial disease. 1,5 Experimental models provide compelling evidence for the role of inflammation in the initiation, progression, and complication of atherosclerosis, confirmed in the clinical setting. 7 High levels of high-sensitive C-reactive protein (hs-CRP) are correlated with angiographic coronary artery disease progression. 8,9 In the peripheral vasculature, both hs-CRP, 7,10,11 fibrinogen, and serum amyloid A 7 are related to the progression of atherosclerosis in peripheral arterial disease. Akinci et al 12 demonstrated that circulating levels of acute phase reactants were associated with amputation risk in diabetic foot. Similarly, Violi et al 13 reported that elevated levels of CRP were associated with poor long-term prognosis in patients with peripheral artery disease. Previous studies have shown that progression of atherosclerosis is associated with increased vascular events. 1,14 Progression of atherosclerosis and additional vascular events increase limb ischemia. Based on the findings of the present study, we also think that amputation may increase in patients with peripheral vascular disease with high-inflammatory markers.
Unlike many other inflammatory markers and bioassay, NLR is an inexpensive and readily available marker that provides an additional level of risk scores in predicting inhospital and long-term mortality. Recently, NLR has also been used as an indicator of poor prognosis in patients with acute coronary syndrome. 15 The NLR has been shown to predict cardiovascular events and mortality. 16 Moreover, multiple studies have suggested that NLR is a strong predictor of outcomes in percutaneous coronary intervention and coronary artery bypass grafting. 17,18 In patients undergoing vascular surgery, NLR provides useful information about postoperative mortality. 1,5
Thrombocytosis is commonly associated with a peripheral arterial disease and has been widely reported as an adverse prognostic marker. 6 The proinflammatory phase associated with systemic inflammation results in release of various immunological mediators such as interleukin (IL)-1, IL-3, and IL-6, which increase circulating platelet counts as a result of megakaryocyte proliferation. 19,20 It is well documented that patients with pathologically increased platelet counts, that is, thrombocytosis, have an enhanced risk of thrombotic complications. 6,21,22 Experimental animal studies indicate that blood platelets both initiate atherogenesis and trigger its complications. 23 It is shown that, in healthy individuals with platelet count lower than normal, platelet aggregation increases when number of platelets increase (>250 × 109/L, >150 × 109/L). 6,21,22 Activated platelets have been demonstrated in a wide variety of clinical settings including peripheral arterial disease despite the use of aspirin. 24 –28 Robless et al found that whole blood platelet aggregation was significantly enhanced in patients with peripheral arterial disease. 6 Also, previous studies have indicated that lymphopenia is associated with the progression of atherosclerosis. Lymphopenia may be caused by lymphocyte apoptosis in atherosclerotic lesions, which gradually increases with atherosclerotic burden. Likewise, a reduced lymphocyte count has also been associated with poorer prognosis. 18,29,30
Thrombocytosis and lymphocytopenia both correlate with the degree of host systemic inflammation, and the PLR reflects a novel marker incorporating both hematologic indices. Several studies has shown a relationship between the PLR and the prognosis in colorectal and pancreatic cancer. 31 –35 No previous studies to our knowledge have investigated this parameter in peripheral arterial disease.
Although PLR is shown to be an independent risk factor for high inflammatory process, with respect to NLR, PLR is more sensitive. 31,35 We tried to emphasize the likeliness of chronic systemic inflammation and its related complications when both high NLR and high PLR is present; thus, we have classified the patients as having high risk when both NLR and PLR are high and having low risk when both values are low, respectively. At the end of our study, we have found that high-risk patients had an independent risk factor for amputation and highest rate of amputation is present. Although these findings were interesting, the clinical relevance is not yet completely understood and requires further research.
The limitations of our study include the possibility of selection bias, the relatively small number of patients with a highly prevalent disease, and the possibility of missing some outcomes using the Social Security Death Index. Only one measurement of admission full blood count and calculation of NLR and PLR was included in the analysis, and it was not possible to determine whether an acute and brief inflammation was responsible for the correlation observed. Additionally, inflammatory markers, such as CRP, myeloperoxidase, and IL-6, were not analyzed and compared to NLR and PLR.
The results of the present study suggest that significant prognostic information can be obtained from routine blood results in patients with CLI. Stratification of patients with CLI according to both admission NLR and PLR should be considered in the limb survival analyses of future adjuvant and neoadjuvant trials to validate these findings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
