Abstract
A 4-factor prothrombin complex concentrate (4F-PCC, Kcentra®) was recently approved in the United States for the reversal of vitamin K antagonist-associated major bleeding, but it is often used to reverse coagulopathy in patients with liver disease (LD). This single-center, retrospective study analyzed the efficacy and safety of 4F-PCC administered in patients with and without LD. Prothrombin time/International Normalized Ratio (PT/INR) reversal with 4F-PCC was attempted in 85 patients; LD was documented in 31 patients. Coagulopathy reversal and hemostasis with 4F-PCC were inferior in patients with LD compared to patients without LD. Coagulopathy reversal, defined as INR = 1.5 after 4F-PCC administration, was achieved in 6 (19.4%) LD patients, compared to 44 (81.5%) non-LD patients (p < 0.01). Hemostasis was achieved in 6 LD patients (19.4%) compared to 23 non-LD patients (42.6%) (p = 0.03). Thromboembolic events occurred in 1 LD patient (3.2%) and 8 non-LD patients (14.8%) (p = 0.15). Mortality was 51.6% in LD patients and 18.5% in non-LD patients (p < 0.01). These observations suggest that the efficacy of 4F-PCC is suboptimal to correct coagulopathy and hemostasis in patients with LD, who have high rates of in-hospital mortality due to sequelae of LD. The incidence of thromboembolic events appeared comparable, suggesting that 4F-PCC does not cause undue thromboembolism in LD patients. In conclusion, 4F-PCC appears to be safe in LD patients when administered judiciously; however, further studies are necessary to optimize its use and elucidate its hemostatic potential in this patient population.
Introduction
Four-factor prothrombin complex concentrate (4F-PCC) contains nonactivated vitamin K-dependent coagulation factors and anticoagulant proteins C and S. The first 4F-PCC product in the United States, Kcentra (CSL Behring, Marburg, Germany), was approved by the Food and Drug Administration in April 2013, for “urgent reversal of acquired coagulation factor deficiency induced by vitamin K antagonists (VKAs) therapy in patients with acute major bleeding.” 1 This approval was based on a phase IIIb trial that compared 4F-PCC, Beriplex (CSL Behring, Marburg, Germany) with concurrent vitamin K (phytonadione) administration to fresh frozen plasma (FFP) with vitamin K. 2 The 4F-PCC product was noninferior to plasma for hemostasis but was superior in rapid reduction of the prothrombin time (PT), expressed as International normalized ratio (INR). 2 Kcentra contains the same 4 vitamin K-dependent coagulation factors, factors II, VII, IX, and X, as Beriplex, with minor differences in the amount of sodium citrate, sodium chloride, albumin, antithrombin III, and heparin. 3 –5 The use of 4F-PCC in combination with vitamin K for VKA-associated major bleeding is recommended as an alternative to FFP in the 2012 American College of Chest Physicians guidelines because of less potential for allergic reactions and transmission of infection, short preparation time, and low infusion volume. The 4F-PCC has, therefore, been adopted by our institution for this indication. 6
Since vitamin K-dependent clotting factors are also decreased in liver disease (LD), these patients are often perceived to have an increased risk of bleeding and are treated with PCC in an attempt to correct clotting factor deficiencies. In LD, the production of fibrinogen and procoagulant factors, such as factors II, VII, IX, and X, is reduced due to hepatic synthetic dysfunction. 7,8 Therefore, PT and INR are often prolonged, similar to what is observed in patients treated with VKAs. 9,10 Because of the recent availability of 4F-PCC in the United States and the difficulty in correcting coagulopathies with FFP, 4F-PCC is increasingly used to treat major bleeding due to LD-associated coagulopathy or to correct abnormal clotting tests in patients with LD prior to invasive procedures. 11 Due to its significantly smaller infusion volume and shorter infusion time, 4F-PCC is gaining popularity over FFP for bleeding reversal, especially in patients with LD who often present volume overloaded. However, in addition to reduced procoagulant factors, patients with LD have simultaneously reduced production of natural anticoagulants, such as proteins C/S and antithrombin, and exhibit disturbance of the fibrinolytic pathway, changes which together may result in a new hemostatic “balance.” 7,8,12 In clinical practice, these changes are often not taken into account when assessing bleeding risk, and providers may perceive that patients with LD are at risk for bleeding based on a prolongation of the PT/INR alone. Unlike in patients treated with VKAs, where the correction of PT/INR can be used as an end point to guide reversal strategies with plasma products or hemostatic agents, 2 –4 this is less certain in patients with LD where alterations in hemostasis parameters are fluctuating, complex, and less amenable to assessment with routine hemostatic assays. As a result, the safety and efficacy of using procoagulant products, such as 4F-PCC, to correct PT/INR in patients with LD is unknown. Therefore, we compared the efficacy and safety of 4F-PCC in patients with and without LD when used for the treatment or prophylaxis of significant bleeding.
Methods
Data Collection
This was a single-center, retrospective, observational study of patients who received 4F-PCC (Kcentra; CSL Behring, Kankakee, Illinois) between July 1, 2013 and April 20, 2014, performed at the University of California San Diego Health System, a level 1 trauma center and a tertiary referral center for patients with LD and liver transplantation. The study was approved by the institutional review board and conducted in accordance with the ethical standards of the institutional Human Research Protections Program. Patients were included if they received at least 1 dose of 4F-PCC, had at least 1 INR prior to 4F-PCC administration (pre-INR), and at least 1 INR after 4F-PCC administration (post-INR). Both pre- and post-INR values must have been obtained within 48 hours of 4F-PCC administration. Exclusion criteria included documented congenital factor deficiency, pregnancy, and age <18 years. Demographic data, including age, sex, height, weight, body mass index, fibrinogen, anticoagulant, or antiplatelet use prior to admission, documented LD based on International Classification of Diseases (ICD-9) codes, type of bleeding, and indication of 4F-PCC administration were collected from the electronic medical record. The dose of 4F-PCC was selected based on pre-INR, actual body weight, and indication per institutional guidelines (Supplemental Appendix 1). For warfarin reversal, 4F-PCC dose (25-50 units/kg) was selected per package insert based on INR and actual body weight. A fixed low-dose 4F-PCC (approximately 500 units) for INR reversal in nonbleeding coagulopathic patients prior to procedures was recommended to minimize thromboembolic risk. Concurrent use of blood products including FFP, packed red blood cells (PRBCs), platelets, and cryoprecipitate received within 48 hours of 4F-PCC administration was also collected. Other treatments affecting INR or hemostasis including vitamin K, recombinant factor VIIa, tranexamic acid, aminocaproic acid, or desmopressin received within 48 hours of 4F-PCC administration were also recorded.
Outcomes
The primary outcome was coagulopathy reversal, defined as post-INR ≤1.5, which was the INR collected at least 30 minutes after 4F-PCC administration. Secondary outcomes included 24-hour post-INR, 48-hour post-INR, hemostasis at 48 hours, intensive care unit (ICU) length of stay (LOS), hospital LOS, and all-cause in-hospital mortality. The 24- and 48-hour post-INR were defined as the latest INR obtained within 24 hours and 48 hours after 4F-PCC administration, respectively. Hemostasis was defined as a composite end point having met all 3 of the followings—achieved and maintained hemoglobin ≥ 7 g/dL for 48 hours after 4F-PCC administration, discontinuation of all blood products, and physician documented assessment of hemostasis in the electronic medical record within 48 hours of 4F-PCC administration. Safety outcomes included thromboembolic events (deep vein thrombosis [DVT], pulmonary embolism, myocardial infarction, cerebral vascular accident) within 30 days, as recorded in the electronic medical record, or new thrombus observed on ultrasound, computed tomography, or high probability on ventilation–perfusion scan.
Statistical Analyses
Categorical variables were analyzed using the χ2 or the Fisher exact test. Ordinal and nonnormally distributed continuous variables were analyzed by Wilcoxon rank sum test. A P-value < .05 was considered as statistical significance. Subgroup analysis was performed to determine if the differences in numbers of patients in each group receiving low fixed dose of 4F-PCC would explain the differences in outcomes. Only patients who received reversal doses of 4F-PCC (25-50 units/kg) were included in the subgroup analysis. Patients who received approximately 500 units of 4F-PCC prior to invasive procedure were excluded in the subgroup analysis. We performed analyses with SAS version 9.4 (SAS Institute, Cary, North Carolina).
Results
Patient Characteristics
Ninety-five patients received at least 1 dose of 4F-PCC during the study period; 85 patients were eligible for the analysis, including 31 patients with LD and 54 patients without LD (non-LD; Figure 1). Ten patients were excluded for the following reasons—4 patients did not have a post-INR, 3 patients did not have a pre-INR, 1 patient was less than 18 years, 1 patient has documented congenital protein C deficiency, and 1 patient had inconclusive documentation regarding the administration of 4F-PCC. The most common etiology of LD was hepatitis C cirrhosis (32.3%), followed by alcoholic cirrhosis (25.8%). Other etiologies of LD are detailed in Table 1. The majority (77.4%) of patients with LD had severe LD (Child-Pugh C score). The median Model for End-Stage Liver Disease score was 29 (interquartile range [IQR]: 21-40). Baseline patient characteristics are described in Table 1 and were similar between groups, except patients in the LD group were younger (58 vs 70 years; P < .01) and had lower fibrinogen level (median: 109 [IQR: 83-105] vs 176 [IQR: 140-257]; P < .01). The pre-INR was similar between groups (median: 2.8 [IQR: 2.1-4.1] in LD group vs 2.6 [IQR: 1.8-4.9] in non-LD group). Bleeding patterns differed among patients with and without LD. The most common cause of bleeding in the LD group was gastrointestinal hemorrhage (32.3%), whereas the most common cause in the non-LD group was intracranial hemorrhage (42.6%). Bleeding sites in both groups are detailed in Table 1. Oral anticoagulation use prior to admission was more prevalent in the non-LD group. More patients in the non-LD group were taking aspirin (33.3% vs 9.7%; P < .05) or warfarin (74% vs 12.9%; P < .05) prior to admission. Details regarding antiplatelet agent and anticoagulants use prior to admission are summarized in Table 2.
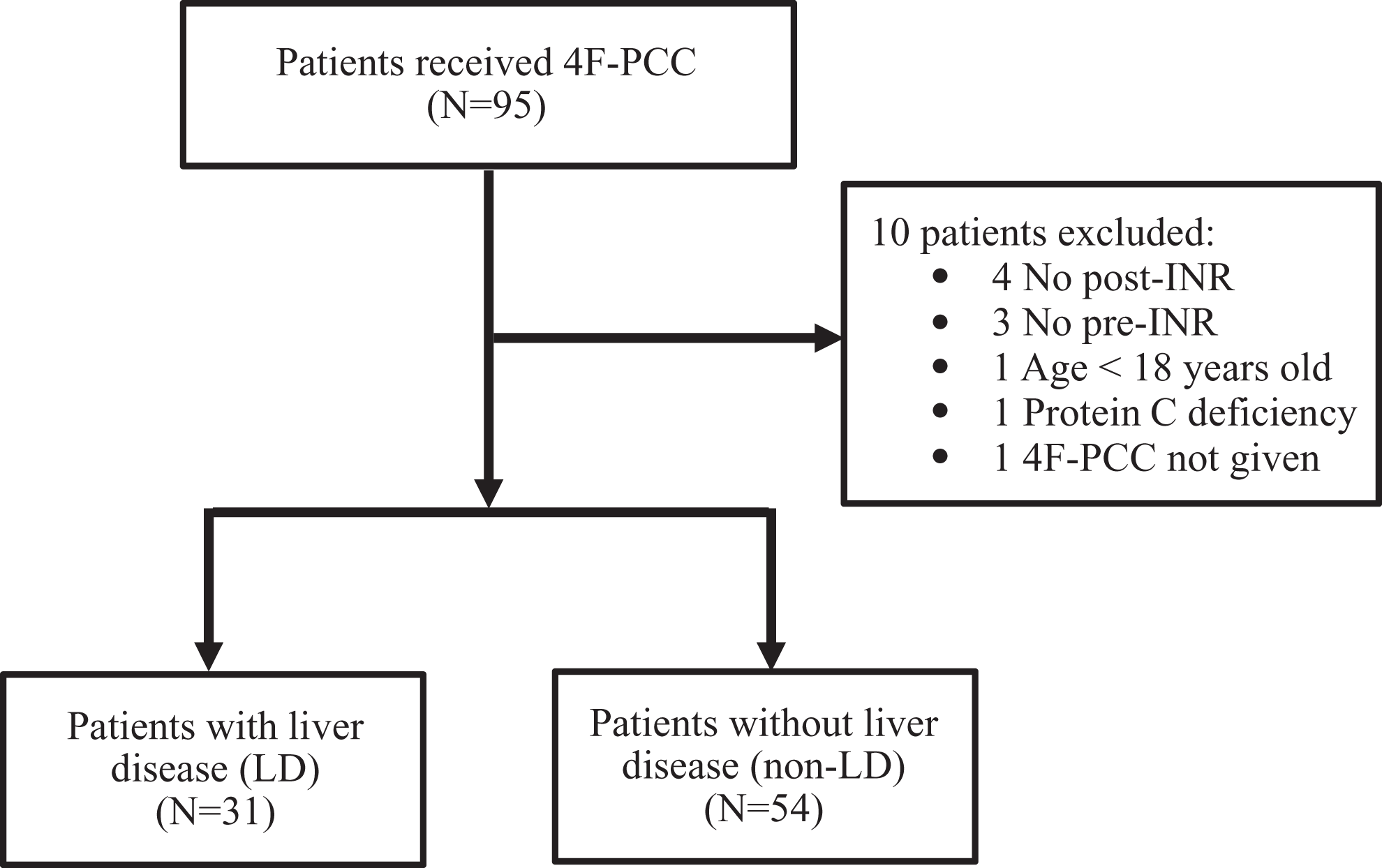
Patient distribution. 4F-PCC indicates 4-factor prothrombin complex; INR, international normalized ratio.
Demographics and Baseline Patient Characteristics.
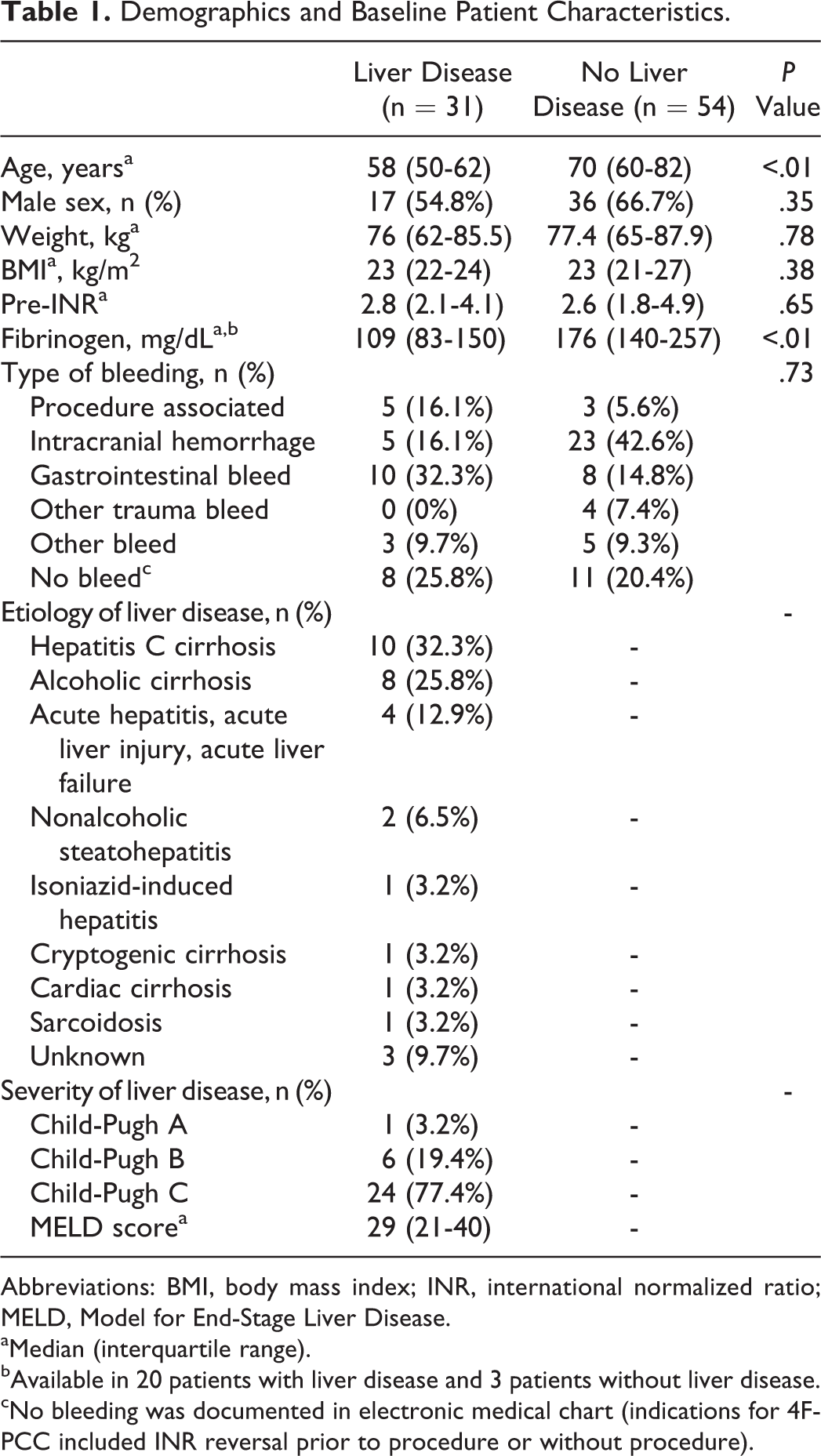
Abbreviations: BMI, body mass index; INR, international normalized ratio; MELD, Model for End-Stage Liver Disease.
aMedian (interquartile range).
bAvailable in 20 patients with liver disease and 3 patients without liver disease.
cNo bleeding was documented in electronic medical chart (indications for 4F-PCC included INR reversal prior to procedure or without procedure).
Antiplatelet and Anticoagulant Agent Use Prior to Administration of 4F-PCC.
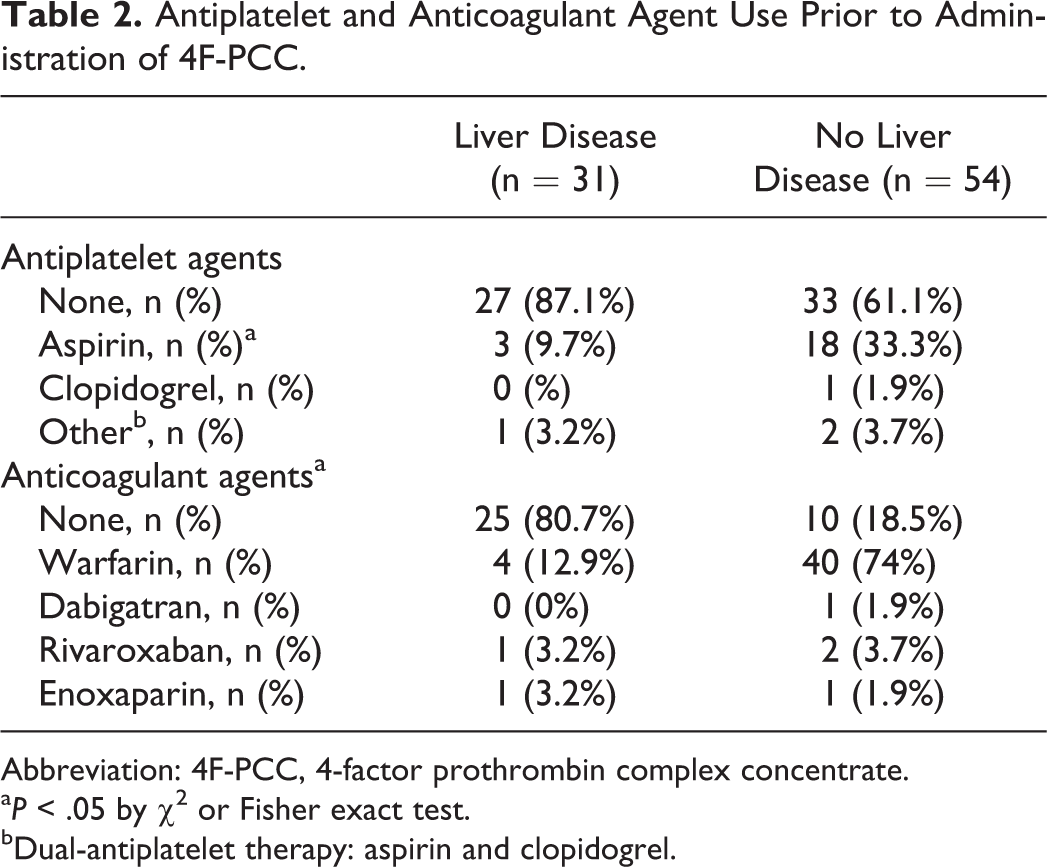
Abbreviation: 4F-PCC, 4-factor prothrombin complex concentrate.
aP < .05 by χ2 or Fisher exact test.
bDual-antiplatelet therapy: aspirin and clopidogrel.
Use of 4F-PCC, Hemostatic Agent, and Blood Products
The median dose of 4F-PCC administered in the LD group was 1638 units (23 units/kg) and 2146 units (27 units/kg) in the non-LD group (P = .01). Compared to patients in the non-LD group, more patients in the LD group received low-dose 4F-PCC at a fixed dose of approximately 500 units (32.2% vs 9.3%; P = .01) for urgent invasive procedures per our institutional guidelines. There was no significant difference in vitamin K administration between the 2 groups. No patients in either group received factor VIIa, tranexamic acid, aminocaproic acid, or desmopressin. Blood product use was significantly higher in the LD group compared to the non-LD group, including PRBC, FFP, platelets, and cryoprecipitate. Patients in the LD group received approximately 2 to 3 times more blood products than patients in the non-LD group. The use of blood products and hemostatic agents is summarized in Table 3.
Use of Blood Products and Hemostatic Agents Within 48 Hours of 4F-PCC Administration.
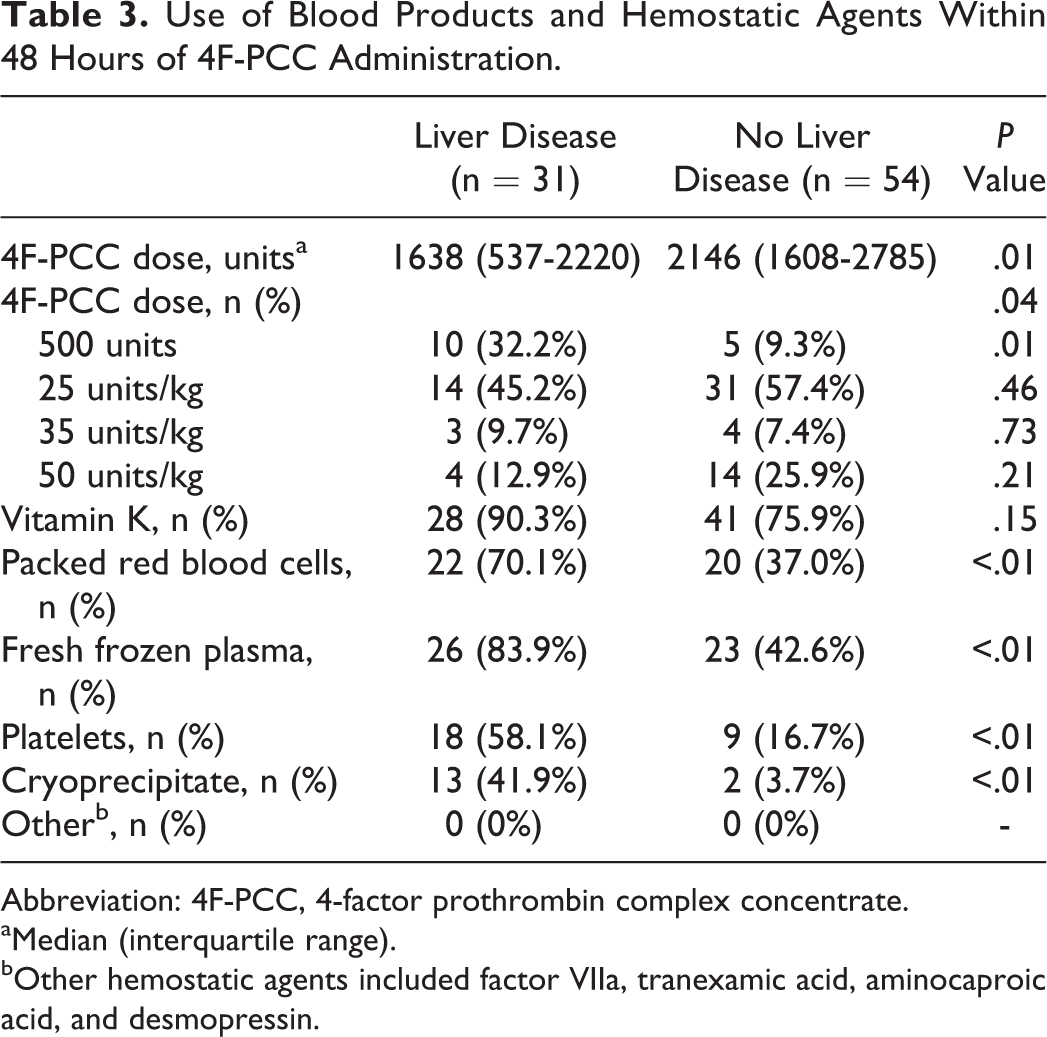
Abbreviation: 4F-PCC, 4-factor prothrombin complex concentrate.
aMedian (interquartile range).
bOther hemostatic agents included factor VIIa, tranexamic acid, aminocaproic acid, and desmopressin.
Correction of Coagulopathy and Hemostasis
Six (19.4%) patients in the LD group achieved coagulopathy reversal compared to 44 (81.5%) patients in the non-LD group (P < .01). Furthermore, the median post-INR after 4F-PCC administration was significantly higher in patients with LD than patients with non-LD (median post-INR: 2 vs 1.3; P < .05). The change of median INR at various time points is represented in Figure 2. Similar results were observed for the 24-hour post-INR and for the 48-hour post-INR. At 24 hours, 7 (22.6%) patients with LD achieved an INR ≤1.5 compared to 47 (87.0%) patients with non-LD (P < .01). At 48 hours, 6 (19.4%) patients with LD achieved an INR ≤1.5 compared to 44 (81.5%) patients with non-LD (P < 0.01). At 48 hours after 4F-PCC administration, 6 (19.4%) patients in the LD group achieved hemostasis compared to 23 (42.6%) patients in the non-LD (P = .03). Patients in the LD group had a significantly longer ICU stay (11 days [5-22] vs 3 days [1-9]; P < .01) and hospital stay (20 days [11-53] vs 8 days [3-19]; P < .01) compared to patients without LD. Primary and secondary outcomes are summarized in Table 4.
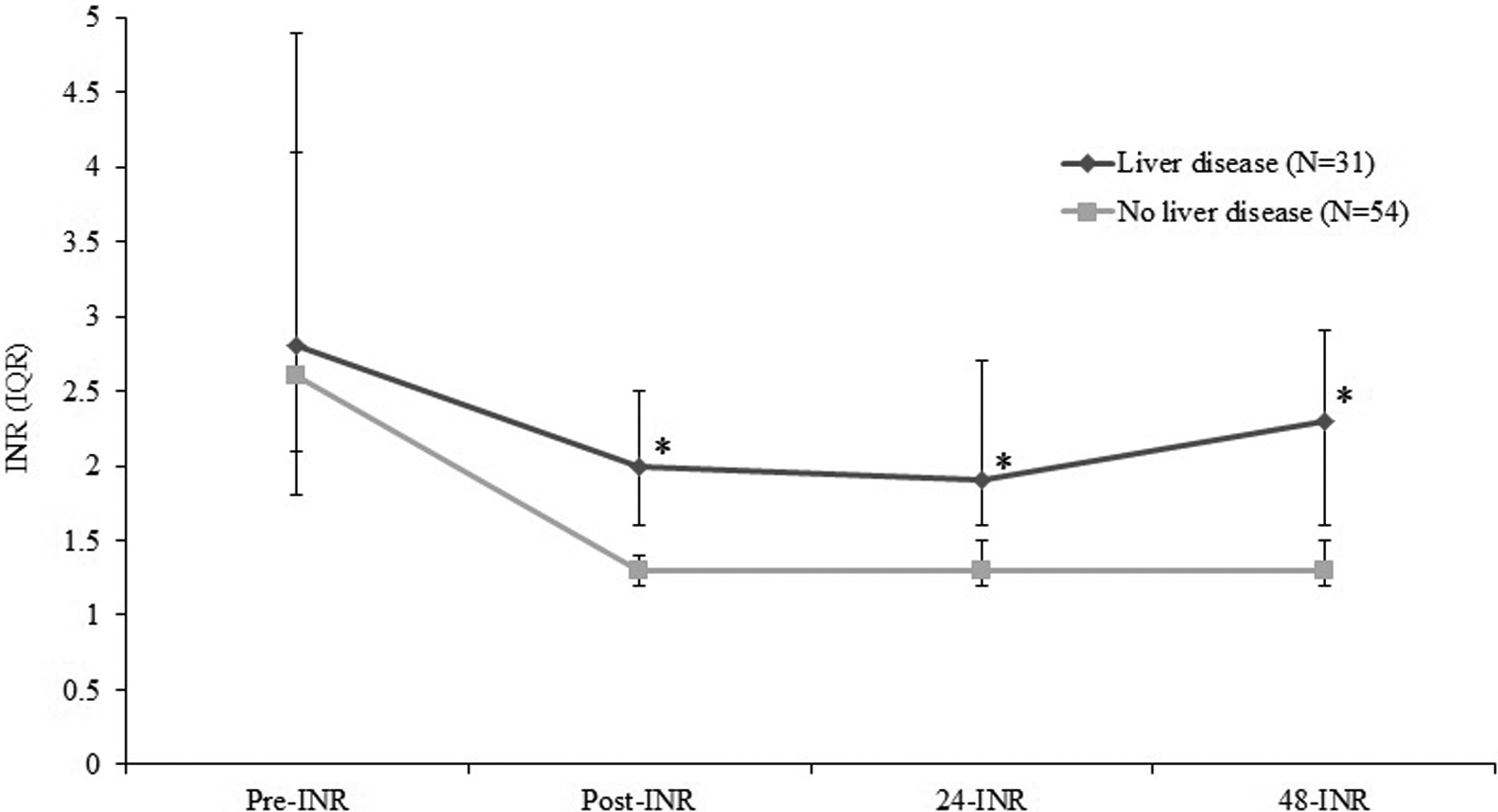
International normalized ratio (INR) in relationship to 4-factor prothrombin complex (4F-PCC) administration. International normalized ratio correction after 4F-PCC administration at various time periods. Median INR is represented along with interquartile range (IQR) in error bars. *P < .05 by Wilcoxon rank sum test.
Outcomes: INR Correction, Hemostasis, and Mortality.
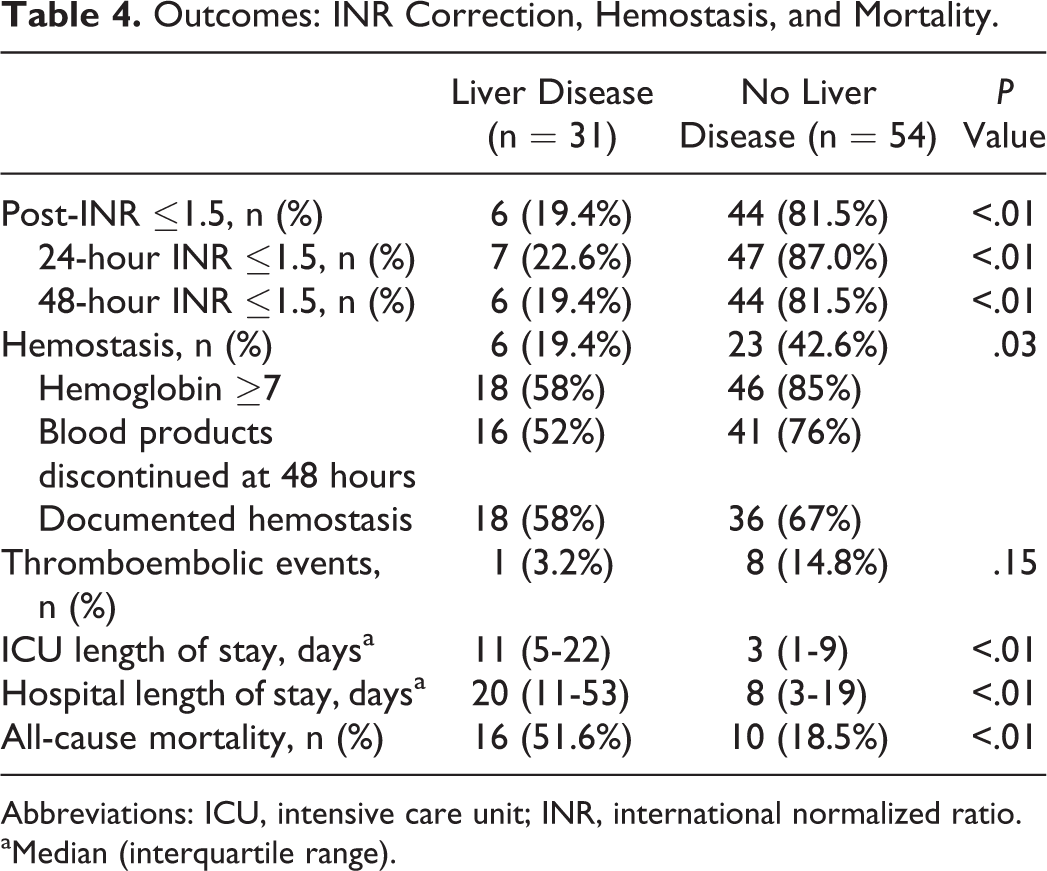
Abbreviations: ICU, intensive care unit; INR, international normalized ratio.
aMedian (interquartile range).
Thromboembolic Events and Mortality
One thromboembolic event was documented in a patient with LD within 30 days of 4F-PCC administration compared to 8 patients without LD (P = .146). The patient in the LD group had a DVT, which was discovered within 1 day of 4F-PCC administration. In the non-LD group, median time to diagnosis of a thromboembolic event (6 DVTs, 1 left ventricular thrombus, 1 pulmonary embolism) was 8 days after 4F-PCC administration (range: 3-17 days). All-cause in-hospital mortality was significantly higher in patients with LD (51.6%) compared to patients without LD (18.5%; P < .01).
Subgroup Analysis Excluding Patients Who Received Nonweight-Based Fixed Doses of 4F-PCC
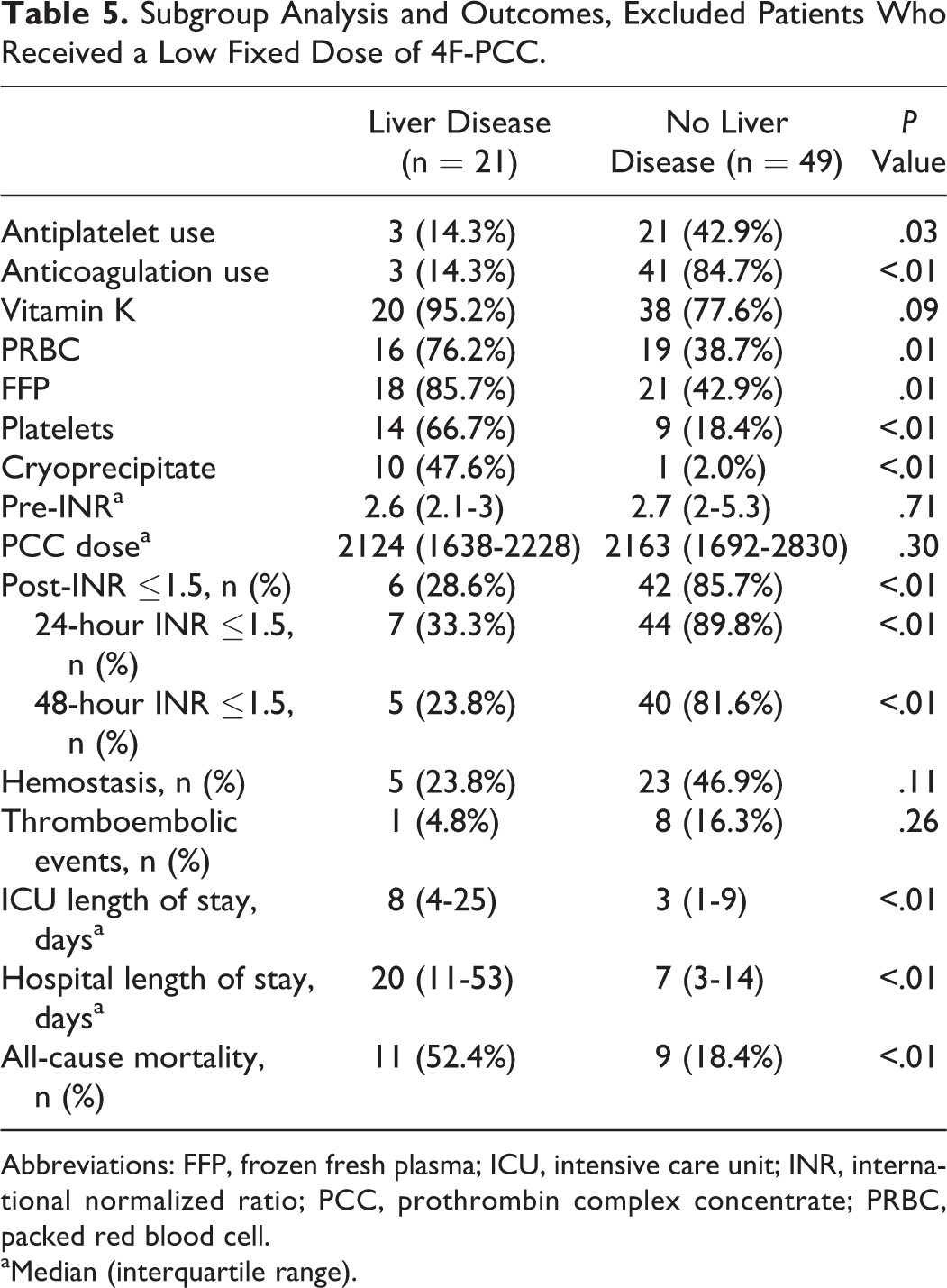
A subgroup analysis, which excluded patients who received the low fixed dose of approximately 500 units of 4F-PCC prior to urgent invasive procedure, was performed. For the subgroup analysis, 10 patients in the LD group and 5 patients in non-LD group were excluded. After excluding these patients, the overall findings indicating inferior coagulopathy reversal and hemostasis, as well as longer hospitalization and higher mortality in patients with LD compared to patients without LD, did not change. The median doses of 4F-PCC administered in both groups were similar in the subgroup analysis. The median dose of 4F-PCC administered in the LD group was 2124 units [IQR: 1638-2228], which was comparable to 2163 units [IQR: 1692-2830] in the non-LD group (P = .30). The pre-INR was similar between the 2 groups (median: 2.6 [2.1-3] in LD and 2.7 [2-5.3] in non-LD; P = .71). 6 (28.6%) patients in the LD group achieved post-INR ≤1.5, which is significantly less than 42 (85.7%) patients in the non-LD group (P < .01). Similar results were observed at 24- and 48-hour INR. Hemostasis was achieved in 5 (23.8%) patients in the LD group, compared to 23 (46.9%) patients in the non-LD group (P = .11). Patients in the LD group had longer ICU and hospital LOS. All-cause mortality was higher in LD group (52.4% vs 18.4%; P < .01). Subgroup analysis results are summarized in Table 5.
Subgroup Analysis and Outcomes, Excluded Patients Who Received a Low Fixed Dose of 4F-PCC.
Abbreviations: FFP, frozen fresh plasma; ICU, intensive care unit; INR, international normalized ratio; PCC, prothrombin complex concentrate; PRBC, packed red blood cell.
aMedian (interquartile range).
Discussion
Limited options exist to control major bleeding events in patients with LD. Historically, a variety of hemostatic agents, such as plasma products, fibrinogen concentrates, factor VIIa, antifibrinolytics, and PCCs have been used for this indication. 13 Data supporting the efficacy of these agents for LD-associated coagulopathy are scant and limited to case series and expert opinions. 13 Despite limited efficacy and safety data, 4F-PCC is now frequently used to correct prolongation of the PT/INR in patients with LD. This practice perpetuates because currently no readily available or validated tools exist to assess bleeding risk in patients with LD. Therefore, PT/INR is used as a surrogate marker by clinicians to guide bleeding reversal strategies.
Similar to the reversal of VKAs, the smaller infusion volume and shorter infusion time compared to FFP have led prescribers to favor 4F-PCC over FFP in this patient population. In addition, many institutions, including ours, experienced a remarkable increase in the use of 4F-PCC in patients with LD to reserve coagulopathy following its addition to formulary. Nevertheless, there is little published “real-world,” postmarketing experience with this factor concentrate, specifically in patients with LD. To create first knowledge regarding the effectiveness and safety of 4F-PCC for coagulopathic patients with LD in relative comparison to the approved indication of VKA reversal, we evaluated both the efficacy and safety of this approach by analyzing real-world data of all patients treated with 4F-PCC since its introduction on our formulary. Our findings demonstrated that the majority of patients with LD did not reverse their coagulopathy, defined as an INR ≤1.5, after 4F-PCC administration, and that hemostasis was far less optimal compared to patients with non-LD. The determination of hemostasis was based on chart extracted data, which may therefore not be entirely accurate. However, we believe that it allows a realistic estimate and a rough comparison between patient populations. Reversal of the PT/INR was neither achieved with fixed low-dose administration nor weight-based dosing in relation to the INR per package insert. Little comparative data are available for review, but preliminary findings expressed in 2 abstracts align with our observations and suggest that 4F-PCC administration has only a small impact on INR correction in patients with LD. 14,15 It was also suggested that the probability of INR correction may correlate with the severity of LD, but this appears controversial based on results from an open-label, prospective study on the use of 4F-PCC (Beriplex P/N) in 22 patients with severe LD and major bleeding or in need of urgent surgical or invasive intervention. 14 –16 This study did not find a dose responsiveness to 4F-PCC in relationship to LD but demonstrated wide ranges of 4F-PCC doses (1000-4000 units) and clotting assay parameter responses. 16 Taken together, these observations highlight substantial variability of 4F-PCC dosing requirements to attempt correction of clotting assay parameters and achievement of clinical hemostasis in patients with LD, which appears different from the more uniform and successful response in the approved indication of 4F-PCC for VKA reversal. 2,3
Several explanations for the relative lack of INR correction and achievement of hemostasis with 4F-PCC in patients with LD are proposed. First, the effective dose of 4F-PCC for INR reversal in patients with LD has not been established. Dosing is performed empirically based on study results using 4F-PCC for VKA reversal, which may result in underdosing for patients with LD. For instance, at our institution, we recommend using 25 units/kg, up to a maximum of 100 kg, for patients with LD and life-threatening bleeding with no known history of VKA consumption. If the patient does not respond adequately, a second repeat dose of 25 units/kg may be used. For patients who are not taking VKA and only require a diagnostic or therapeutic invasive procedure to prevent major bleeding, we recommend a low fixed dose of 4F-PCC (approximately 500 units) weighing the risk of procedure-associated bleeding against thromboembolic complications with prophylactic use of 4F-PCC. 17 Other institutions may choose different algorithms, but none has been established to be efficacious and/or safe. Second, PT/INR is not a reliable indicator for overall coagulation status in patients with LD. The PT/INR was developed to guide VKA therapy by capturing the attenuation of vitamin K-dependent procoagulant factors, especially FVII, within the extrinsic pathway. In this context, without abnormalities of other hemostatic parameters, PT/INR provides a reasonable tool to guide anticoagulation and estimate the risk of bleeding outside the therapeutic range. 6,18 In LD, abnormal platelet counts, perturbed fibrinolysis, low natural anticoagulants, low fibrinogen, factor V, and the compensatory increase of FVIII/von Willebrand factor render the PT/INR insensitive to the determination of bleeding risk. 7,8,10,19 Therefore, coagulation tests such as the PT/INR may be abnormal in LD, but they are poor predictors of bleeding risk in this patient population. 20 –22 Global assays of hemostasis, such as thrombin generation and thromboelastography (TEG), have been applied to determine the coagulation status in patients with LD. 23 –25 Studies have shown normal to increased thrombin generation capability in patients with LD despite abnormal PT/INR, which suggested the limitation of using PT/INR to determine bleeding risk in this patient population. 26 –28 However, thrombin generation is not routinely available in clinical laboratories since it is not yet standardized and approved for clinical use. On the other hand, TEG is a commercially available point-of-care assay and has been used to assess hemostasis in patients with LD. 29 It has the potential to guide 4F-PCC therapy in patients with LD by better identifying imbalanced hemostasis; however, this method has not been validated in clinical setting and warrants further investigations. Therefore, PT/INR was selected to be the marker in guiding 4F-PCC therapy despite its limitation in patients with LD. Finally, in addition to low vitamin K-dependent clotting factors, low fibrinogen levels often present in advanced LD not only impede the PT/INR correction but also impact clot formation and stability, further contributing to the bleeding diathesis. 7,30,31 In our study, patients with LD had significantly lower fibrinogen levels compared to patients without LD and received significantly more fibrinogen containing blood products such as FFP and cryoprecipitate. These observations suggest that low fibrinogen was a major contributor to bleeding in patients with LD, and probably contributed to the lesser ability to correct the PT/INR with 4F-PCC.
We also observed a high mortality rate in patients with LD, which may be more inherent to multiple comorbidities associated with LD rather than the use of 4F-PCC. Of interest, only one of patients with LD experienced thromboembolic events within 30 days of 4F-PCC administration, whereas the incidence of thromboembolic risk in patients without LD was comparable to previous observations in the pivotal trials for VKA reversal. 2,3 This suggests that the thromboembolic risk associated with 4F-PCC administration in patients with LD is low, or at least not increased, barring the limitation that there were no guidelines on when or who to screen for thromboembolic events. Most diagnostic tests were performed when a patient presented with signs or symptoms of thromboembolism and included 1 or more different imaging studies such as ultrasound, computed tomography, or ventilation–perfusion scan.
Further limitations of our study were the relatively small sample size and significant heterogeneity between groups. We had broad inclusion criteria to reflect clinical practice at our institution; therefore, more patients without LD than patients with LD were available for analysis since 4F-PCC was used more frequently in that population. Bleeding sites and urgency of reversal differed between patients with and without LD, which explains in part why the doses of 4F-PCC were slightly different between groups. For instance, intracranial bleeds, which required urgent reversal with 4F-PCC, were more frequent in patients without LD than with LD. There was no established time interval for measuring INR after 4F-PCC administration during the study period. This led to variability in the frequency and timing of INR obtained after 4F-PCC administration.
Despite the limitations, we believe that this study represents a real-world experience and permits the conclusion that 4F-PCC, at the doses utilized here, resulted in poor coagulopathy reversal and hemostasis correction in patients with LD having major bleeding or in need of coagulopathy reversal prior to procedures. Based on our findings, and against common current belief, PT/INR reversal and hemostasis correction with 4F-PCC in patients with LD cannot be expected to occur to the same extent as in patients treated with VKAs. Studies to determine whether 4F-PCC in patients with LD increases hemostasis efficacy above and beyond that achieved with the concomitant use of plasma products are necessary. These studies will require a look-back analysis of bleeding patients with LD before and after the introduction of 4F-PCC or a prospective randomized clinical trial. In addition, cost effectiveness of such therapy has to be assessed. In the interim, the use of 4F-PCC cannot be routinely recommended for coagulopathy reversal in patients with LD but may be an option for selected patients with excessive, life-threatening bleeding, after carefully weighing risks and benefits of such therapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Annette von Drygalski has received honoraria for participating in scientific advisory board panels, consulting, and speaking engagements for Baxalta, Pfizer, Biogen, CSL-Behring, Novo Nordisk, and Grifols. She is a cofounder and member of the board of Hematherix LLC, a biotech company that is developing superFVa therapy for bleeding complications.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
