Abstract
Factor V (FV) plays a crucial role in both procoagulant and anticoagulant pathways. It was indicated that -426G/A change in the promoter of the FV gene may have an effect on the occurrence of thrombosis. We aimed to investigate the effect of -426G/A gene variation on thrombus formation in the thrombosis patients of age 0 and 18 years and 70 years and older. The study included 179 patients with the diagnosis of thromboembolism and also 221 healthy individuals as controls. The Polymerase chain reaction–restriction fragment length polymorphism method was used to detect -426G/A. It was observed that carrying AA genotype does not have a significant risk of thrombosis between the patients and controls (for 0-18 age group P = .96, odds ratio [OR]: 1.13 [0.38-3.30], for ≥70 age P = .94, OR: 0.69 [0.11−4.28]). The same results were obtained when FV Leiden mutation carriers were excluded (for 0-18 age group P = .95, OR: 1.15 [0.35−3.78], for ≥70 age P = .89, OR: 0.73 [0.11−4.56]). In this study, an effect on thrombosis of -426G/A polymorphism was not determined.
Introduction
Coagulation factor V (FV) plays an important role in the coagulation cascade. Its active form, factor Va (FVa), participates in the conversion of prothrombin into thrombin. Factor Va is inactivated by activated protein C (APC), an anticoagulant protein that acts in conjunction with protein S. Structure and function of factor V have been investigated following the description of APC resistance as an important clinical phenotype. 1,2 The most common genotype that reveals APC resistance is a Arg→Gln variation at 506 (c.1691G>A/p.R506Q) in FV gene. This APC-resistant form of FV is now generally known as FV Leiden (FVL). 3
Although there are several studies on the nature and biology of FVL, there are still gaps on FVL in order to understand the effect and the role of FV gene polymorphisms on the pathogenesis of thrombosis caused by the single nucleotide polymorphisms (SNPs) existing at the gene. 4,5
Lunghi and colleagues reported that -426 G>A change may help in decreasing the FV levels in case of linked Asp79His mutation in the study group of 50 and over. 6 Akar have pointed out possible importance of thrombophilic FVL mutation frequency differences between adults and newborns. 7 Based on these, we aimed to study the possible role of FV gene promoter variation -426G/A in the occurrence of thrombosis in thrombotic patients with and without FVL, in age group between 0 and 18 and 70 and over.
Materials and Methods
Four groups of samples were included in the study, that is, thrombosis group of age 0 to 18 and 70 and over and control groups, respectively. The groups consist of 179 patients of age 0 to 18 years, 110 patients of age 70 and older with the diagnosis of thromboembolism, and also in same age groups 148 and 118 healthy individuals as controls, respectively. All patients with thrombotic event were selected. Patients of age over 70 with arterial thrombosis and patients of age 0 to 18 with venous thrombosis were excluded.
Factor VL mutation was determined by real-time polymerase chain reaction ([PCR] LightCycler, Roche Diagnostics, Roche Molecular Biochemicals, Mannheim, Germany). 8 Patients included in the study are patients with the diagnosis of thromboembolism and healthy unrelated individuals without any familial history of thrombosis and stroke from our DNA bank. All patients/when necessary parents and controls gave a written informed consent for the study. We collected these samples over a period of 2 years.
Factor V promoter -426 G/A polymorphism was detected by the previously described method using the following primers: 5′-CTATGCTGCAGCTTAGCTGG-3′ and 5′-GCTGCAAT GAGCTCTAGAGG-3′ of the FV gene. 9 Amplification was performed for 35 cycles with an annealing temperature of 55°C (Biometra, Germany). Variation in -426G/A gene was detected after amplification of 597 base pair (bp) fragments by PCR of FV gene promoter region using the appropriate primers, digested with MvaI (Fermentas, Lithuania) restriction endonuclease enzyme by restriction fragment length polymorphism method, and subjected to 3% agarose gel electrophoresis. Presence of the mutant allele is identified by digestion of the 597-bp fragment to 351, 131, and 115 bp fragments in size.
Statistical analysis was performed using SPSS software (SPSS Inc, Chicago, Illinois). The groups were compared using chi-square test.
Results
Distribution of -426 G/A genotype is shown in Table 1. It was observed that carrying AA genotype does not have a significant risk for thrombosis between the patients and controls (for 0-18 age group P = .96, odds ratio [OR]: 1.13 [0.38−3.30], for 70 years and older P = .94 OR: 0.69 [0.11−4.28]; Table 1). In 2 groups, the FVL A allele frequencies are 0.03 and 0.04, respectively. The same results were obtained when FVL mutation carriers excluded (for 0−18 age group P = .95, OR: 1.15 [0.35-3.78], for 70 years and older P = .89, OR: 0.73 [0.11−4.56]; Table 2).
Distribution of FV-426 G/A Polymorphism.
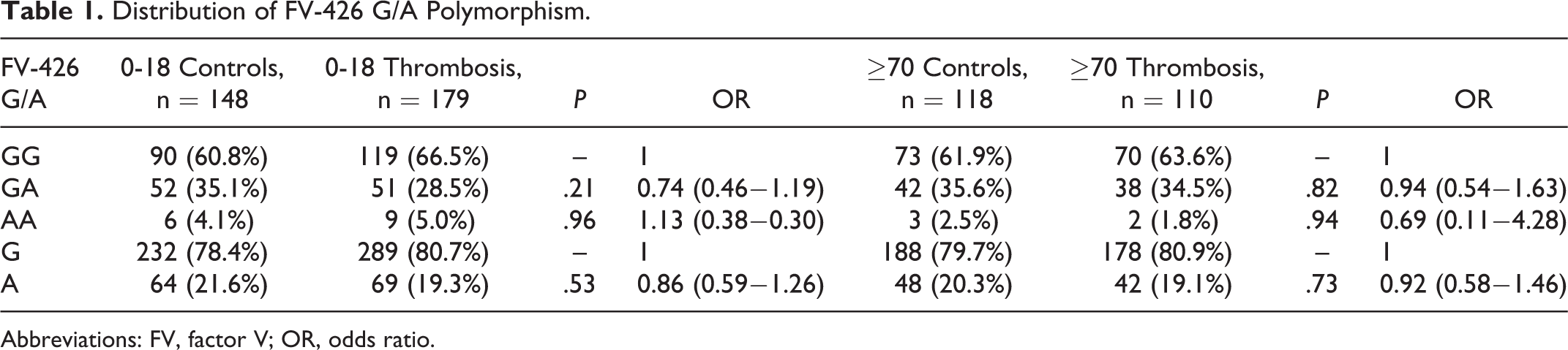
Abbreviations: FV, factor V; OR, odds ratio.
Distribution of FV-426 G/A Polymorphism After Exclusion of Factor V Leiden Carriers.
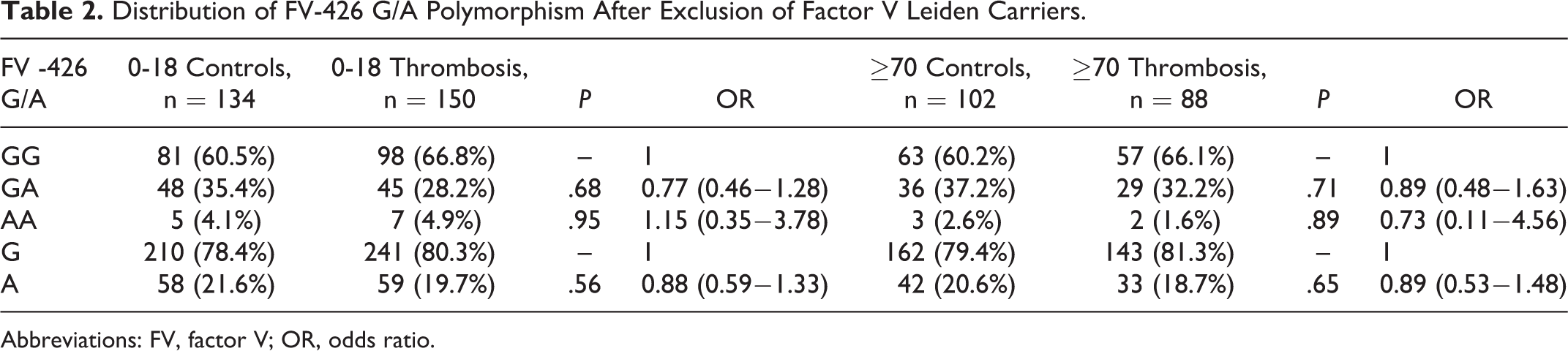
Abbreviations: FV, factor V; OR, odds ratio.
Discussion
The pathogenesis of thromboembolism is multifactorial and involves acquired and genetic factors. Inherited gene defects related to the coagulation system have been reported as risk factors for thrombosis. The common inherited gene defect is a single point mutation in exon 10 of the FV gene, FVL (G1691A and R506Q) causes a resistant to APC. This mutation represents the most common genetic risk factor for venous thrombosis in Caucasian population. 1 –3 Several gene defects either within or other than the FV gene often coexist in thrombophilia, and they have an attenuated effect on individuals in the occurrence of thrombosis. Furthermore, it is interesting that some FVL carriers do not experience thrombosis. 10
Promoter mutations affect the expression levels of the genes. Since -426G/A promoter mutation of the FV gene was published previously, 9 we decided to analyze the possible effect of a promoter mutation -426 of FV gene. Lunghi et al 6 showed that the 79His allele is linked with the A allele of the 426G/A polymorphism. Most carriers of the Asp79His change were also carriers of the 426G/A polymorphism (98%). Among Asp79His carriers, the heterozygote for the 426G/A polymorphism had very similar levels of FV compared to the general population, whereas the 426A homozygote had low levels. This observation suggested that the homozygous condition for the 426G/A change may help in decreasing FV levels. However, they did not find any effect of -426 on FVL in patients with DVT. 4,6 –9 Further Scanavini et al 11 reported that this SNP does not have any effect on FV levels.
Our study consisted of 4 groups with and without thrombosis in pediatric and elderly individuals. We were not able to find any association of -426A in the occurrence of thrombosis, even after the exclusion of the carriers of FVL.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Ankara University Research Fund (Project no: 08B3330008) and partially by the Turkish Society of Hematology Research Projects Support Program (Project no: 2010-1).
