Abstract
Antithrombin III (AT) is the most important endogenous anticoagulant, and genetic variability in SERPINC1, gene encoding AT, is low. Mutations leading to AT deficiency and increased thrombotic risk are well known; however, only 2 studies have reported mutations in regulatory region of SERPINC1 gene till date. Aim of the present study was to identify genetic variations in SERPINC1 5′ untranslated region (UTR) in Indian patients with deep vein thrombosis (DVT) having AT deficiency. DNA sequencing was used to identify underlying genetic defects in SERPINC1 regulatory region. In silico tools TFBIND and PROMO were used to identify transcription factor binding sites in the promoter region. We have identified 2 novel polymorphisms, g.25G>A and g.−1A>T, and 2 known g.67G>A and rs3138521 5′ UTR polymorphisms in SERPINC1 regulatory region in Indian patients with DVT for the first time. In present study, allele frequencies of rs3138521 (S: 0.37 and F: 0.63) were similar to that reported in Western population and were not associated with low plasma AT levels (P value .5). This is the first report of regulatory region polymorphisms in SERPINC1 gene in Indian population. Our results strongly suggest that similar studies should be included when ever no mutation is detected in protein-coding region of AT gene.
Introduction
Serine proteinase inhibitors (serpins) are a superfamily of homologous proteins having both inhibitory and non-inhibitory functions. Serpin dysfunction has long been associated with diseases such as thrombosis, cirrhosis, emphysema, dementia, and hereditary angioedema. 1 Thrombosis can be classified into 2 types depending on thrombus location: arterial thrombosis (myocardial infarction and stroke) and venous thrombosis (deep vein thrombosis [DVT], portal vein thrombosis, renal vein thrombosis and Budd-Chiari syndrome). 2 Venous thromboembolism is a common cause of mortality and morbidity worldwide, with an annual incidence of 0.1%. Mutations in anticoagulants and procoagulants are known to cause pathological conditions, whereas hemostasis is balanced by procoagulant, anticoagulant, and fibrinolytic pathways under normal conditions.
Deficiency of antithrombin III (AT) has long been associated with increased risk of DVT. 3 AT, an important endogenous anticoagulant, is a member of serpin superfamily and protein product of SERPINC1 gene synthesized in the liver. SERPINC1 gene spans 13.4 kb of genomic DNA and is located on chromosome 1q23-25. AT comprises of 3 β-sheets and 7–9 α-helices. 4 AT mediates its anticoagulant action by inhibiting thrombin, factor IXa, Xa, and XIa of the blood coagulation cascade. Prevalence of AT deficiency has been estimated to be 0.07% to 0.16% 5,6 in general population, whereas this frequency is approximately 2% in patients with venous thrombosis. 7 AT deficiency can be categorized as type I where both activity and the antigen levels in plasma are reduced or as type II where normal antigen levels are associated with a reduced AT activity level. Type II AT deficiency has been further subdivided as mutations in the reactive site (type IIa), heparin-binding site (type IIb), or pleiotropic mutations (type IIc). 8 The natural variants of AT mostly identified from families with a tendency of thrombosis are collated together in the AT mutation database. 9 Different mechanisms such as RNA instability, compromised heparin-binding affinity, in vivo polymerization, impaired thrombin binding, and altered glycosylation among others have been identified for AT deficiency leading to thrombosis. 10 –13 The promoter, a regulatory region located upstream of a gene, plays a very critical role in gene expression regulation. Variations in the promoter region are a known putative cause of diseases such as β-thalassemia, hemophilia, bladder cancer, and familial hypercholesterolemia. 14,15 Although more than 250 mutations and polymorphisms spanning the introns, exons, and flanking regions in SERPINC1 gene causing DVT are known, only a single polymorphism in the SERPINC1 regulatory region (g.2085T>C) has been identified till date. 16
The present study was designed to identify gene variations in SERPINC1 regulatory region by DNA-based sequencing and their association with plasma AT activity and antigen levels in AT deficient patients. In addition, rs3138521, a known unusual DNA length polymorphism having either S (108 bp) or F (32 bp) allele in the promoter region of AT gene located −345 bp upstream of the translation initiation codon, was studied in 62 patients with DVT having low AT levels and 62 age and sex-matched healthy controls. All the gene variations were studied for potential transcriptional relevance using in silico analysis.
Materials and Methods
Patients and Blood Sampling
Total 62 Doppler-proven DVT patients with AT deficiency were recruited in Department of Hematology, All India Institute of Medical Sciences, New Delhi. In previous study, we have reported several mutations in SERPINC1 gene in AT deficient Indian population with DVT. 17 The present analysis of polymorphisms in promoter region of AT was performed on patients where no mutation was identified in protein-coding region of SERPINC1 gene. Patients with acquired factors such as pregnancy, surgery, trauma, accident, malignancy, or low anticoagulant levels other than AT were not included in this study. Sixty-two age and sex-matched healthy controls were recruited from the same geographical area with no history of arterial or venous thrombosis.
The 5 ml venous blood was collected in citrated vials. Plasma was separated from the blood by spinning at room temperature. The blood was centrifuged at 1300 Relative Centrifugal Field (RCF) for 20 minutes, supernatant was collected and centrifuged again at 2400 RCF for 30 minutes, and the supernatant plasma was immediately stored at −20°C. DNA was isolated from the remaining cell debris using kit from BIOSERVE (Hyderabad, India).
Antithrombin Assays and Other Coagulation Tests
Plasma AT activity and antigen levels were determined by amidolytic heparin cofactor assay with chromogenic substrate CBS 61.50 (STA-STACHROM ATIII; Diagnostica Stago, France) and latex immunoassay (LIATEST ATIII; Diagnostica Stago, Asnières-sur-Seine, France), respectively. Normal AT activity and antigen levels were determined to be 80% to 120%.
Thromboplastin time (TT), prothrombin time (PT), and activated partial thromboplastin time (APTT) were performed using kits from Diagnostica Stago (Parsippany, New Jersey). PT and APTT were measured as ratios where normal ratio was determined to be 1 ± 0.2 standard deviation (SD) in 20 (10 males and 10 females) age and sex-matched healthy controls. Normal TT level was determined to be 16.00 seconds. Plasma levels of protein C (PC), protein S (PS), anti-β2 glycoprotein (anti-β2 GPI) antibodies, and Activated Protein C Resistance (APCR) were determined using commercially available kits. Normal plasma levels of PC, PS, anti-β2 GPI antibodies, and APCR were estimated to be 70% to 130%, 50% to 135%, <10 U/dL, and a ratio of ≤2.1, respectively.
Genetic Analysis
Genotyping of the 7 exons comprising the SERPINC1 gene and flanking intron sequences, were amplified using the polymerase chain reaction (PCR) using the set of primers as described previously. 11 The 5′ untranslated region (UTR) of SERPINC1 gene was amplified using the primers AT3- 5′UTRF (GCCTGAAGGTAGCAGCTTGT) and AT3- 5′UTRR (AGCTCACCCCTCTTACCTTT). Thermal cycling conditions were as follows: initial denaturation of 10 minutes at 94°C followed by 30 cycles of 1 minute denaturation at 94°C, 1 minute annealing at 62.1°C, 45 seconds extension at 72°C, and final extension at 72°C for 10 minutes. Amplification was followed by commercial DNA-based sequencing using 96 capillary high-throughput sequencer with both forward and reverse primers (Applied Biosystems 3730xl DNA analyzer from Thermo Fisher Scientific). Comparison with the reference sequence (GenBank accession number NG_012462.1) was performed with MEGA 6.0 software. 18
Prediction of Transcription Factor Binding Sites
The search for sequences with potential transcriptional relevance in the present study was performed with TFBIND (tfbind.hgc.jp) and PROMO. 19 –21
Statistical Analysis
Association study between AT single-nucleotide polymorphism (SNP) rs3138521 and plasma AT activity and antigen levels was determined using Mann-Whitney U test. Statistical significance was taken as P < .05, and data have been presented as mean ± SD.
Results
Identification of SERPINC1 Gene Regulatory Region Polymorphisms
To understand the underlying basis of AT deficiency, DNA-based sequencing of promoter region was performed in AT deficient patients, where no mutation was detected in protein-coding region of SERPINC1 gene. Three patients with type I AT deficiency were found to be carrying polymorphisms in 5′ UTR of SERPINC1 gene.
Case report 1
The proband 1 was a 55-year-old Indian male with proximal DVT in popliteal vein of the left lower limb. Anticoagulant-based testing revealed normal PC (90%), PS (110%), anti-β2 GPI antibodies (2 U/dL), and APCR (2.4) levels with only type I AT deficiency (AT activity: 66% and AT antigen: 70%). Reduced APTT was observed with almost normal PT and TT levels (0.48, 0.83, and 10.00 seconds, respectively). A previously reported polymorphism, g.67G>A, present 3 bp upstream of the translation initiation codon in the 5′ UTR region, 22 was identified in the proband. The known polymorphism was confirmed by sequencing with both forward and reverse primers and was carried out 3 times with fresh template each time. Previously reported g.67G>A polymorphism was checked in 61 AT deficient patients with DVT and 62 controls and was found to be absent from above-indicated position. Figure 1A shows electropherograms of g.67G>A polymorphism in proband and control.
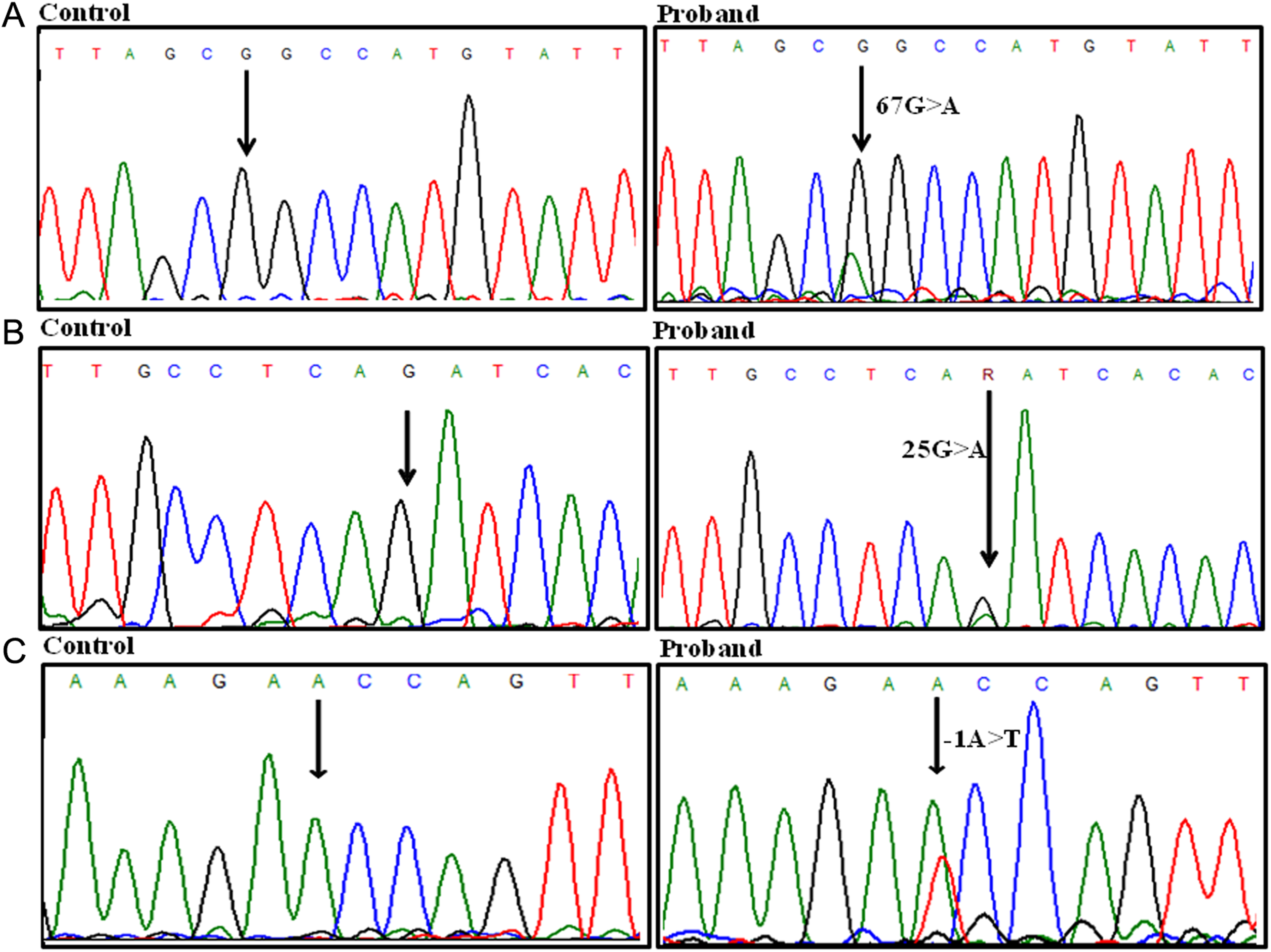
Identification of polymorphisms in 5′ UTR of SERPINC1 gene. A, Heterozygous g.67G>A in proband 1. B, Heterozygous g.25 G>A in proband 2 compared to control. C, g.−1A>T in proband 3 compared to control. UTR indicates untranslated region.
Case report 2
The proband 2 was a 46-year-old Indian male with recurrent thrombosis. First thrombotic episode occurred at the age of 36 years, second at 42 years, and the last at 45 years of age in the femoral vein of left lower limb. The patient doesn’t have an active lifestyle and is obese. Anticoagulant-based testing revealed only low AT activity and antigen levels (51% and 55%, respectively), with normal PC (120%), PS (130%), anti-β2 GPI antibodies (1 U/dL), and APCR (2.5) levels. Reduced APTT was observed with normal PT and TT levels (0.57, 0.93, and 17.00 seconds, respectively). Sequencing of entire SERPINC1 gene revealed a novel heterozygous polymorphism in 5′ UTR g.25G>A, 45 bp upstream of the initiation codon. The above-identified polymorphism was absent in controls but present in 2 other patients with DVT. One of the 2 other patients in which polymorphism was present had type II AT deficiency (AT activity and antigen levels 57% and 82%, respectively), and the other patient had type I AT deficiency (AT activity and antigen levels 62% and 65%, respectively) with other anticoagulant levels being normal in both the patients. Electropherograms showing g.25G>A polymorphism in proband and control are shown in Figure 1B.
Case report 3
The proband 3 was a 27-year-old female with swelling and DVT in popliteal vein of left lower limb. Significant reduction in anticoagulant levels was found only in AT (activity: 48% and antigen: 52%) with other anticoagulant levels being normal: PC (100%), PS (75%), anti-β2 GPI antibodies (2 U/dL), and APCR (2.4). Reduced APTT and TT levels were observed with normal PT levels (0.73, 14.00, and 0.91 seconds, respectively). A novel polymorphism, g.−1A>T, was observed in 5′ UTR of SERPINC1 gene (Figure 1C). The g.−1A>T polymorphism was present 71 bp upstream of the translation initiation codon. This polymorphism was not present in any of the control or AT deficient patients with DVT.
Effect on Transcription Factor Binding Sites Affected by Genetic Variations
The newly identified polymorphism, g.25G>A, was found to have potential transcriptional consequences as it eliminates AHRARNT, REBP-1 transcription factor binding sites and creates a binding site for MYCMAX. Similarly, g.−1A>T transversion was found to eliminate OLF1 transcription factor binding site. Previously reported g.67G>A 5′ UTR polymorphism was also tested for probable transcriptional relevance and was found to cause elimination of LMO2COM and USF and creation for catabolite activator protein-binding site.
Genetic Analysis of rs3138521 in SERPINC1 Gene
rs3138521 polymorphism was studied in 62 patients with DVT with low AT levels and 62 healthy controls (Figure 2). Table 1 shows genotypic frequencies and their association with plasma AT activity and antigen levels in patients and controls. Allele frequencies were determined to be S: 0.37, F: 0.63 and were not associated with low AT activity and antigen levels (P = .5 and .402, respectively).
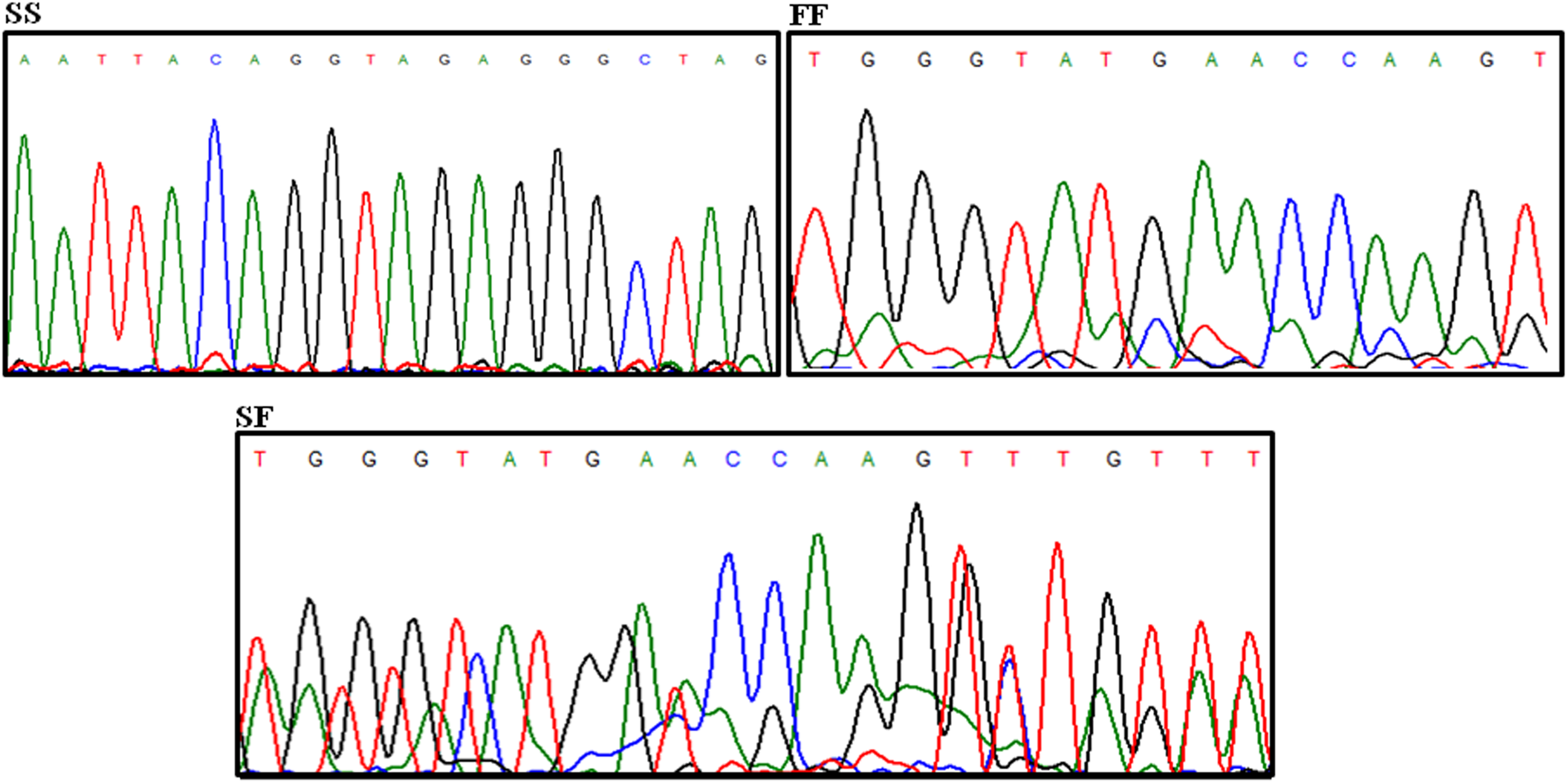
Identification of rs3138521 genotypes in Indian population with DVT. DVT indicates deep vein thrombosis.
rs3138521 Genotype Frequencies and Their Association With Plasma AT Activity and Antigen Levels in Patients and Healthy Controls.
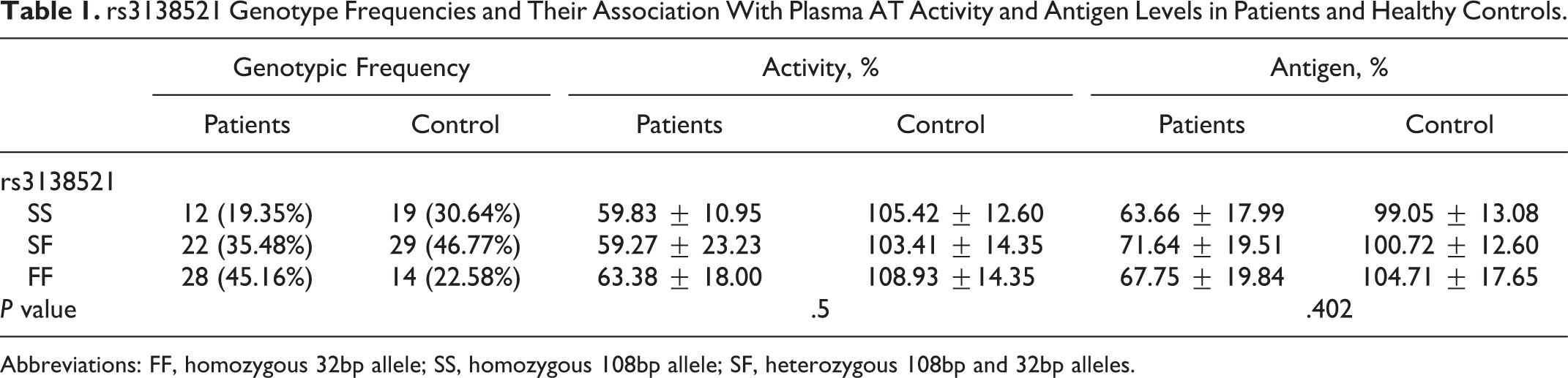
Abbreviations: FF, homozygous 32bp allele; SS, homozygous 108bp allele; SF, heterozygous 108bp and 32bp alleles.
Discussion
AT is a natural endogenous anticoagulant and is highly sensitive to minor changes in the primary structure as evident by the absence of any missense polymorphisms in the gene. 9 Transcriptional regulation is the first step in gene expression regulation, where promoter region plays a key role by binding to RNA polymerase and various transcription factors. Mutations in the promoter region have been shown to be associated with a number of human diseases. Although the first mutation linked to AT deficiency was characterized in 1983, 23 the first regulatory region mutation was characterized only in 2012. 16 In the present study, novel and known variants in AT regulatory region have been identified for the first time in Indian population with DVT. Table 2 shows the genotypic and phenotypic features of patients identified with polymorphisms in the regulatory region of AT gene.
Polymorphisms Identified in AT Regulatory Region in Indian Population With DVT.
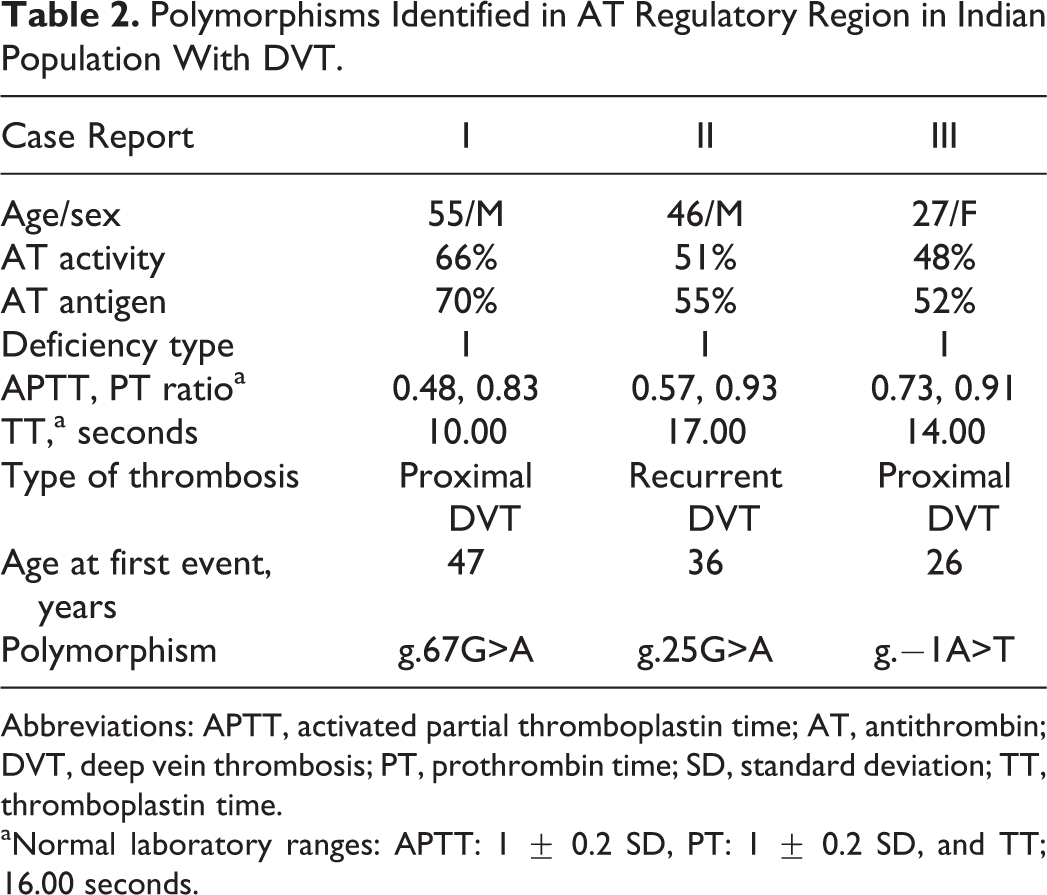
Abbreviations: APTT, activated partial thromboplastin time; AT, antithrombin; DVT, deep vein thrombosis; PT, prothrombin time; SD, standard deviation; TT, thromboplastin time.
aNormal laboratory ranges: APTT: 1 ± 0.2 SD, PT: 1 ± 0.2 SD, and TT; 16.00 seconds.
Identification of 2 novel polymorphisms in the 5′ UTR in patients with severely reduced AT activity and antigen levels indicates the genetic basis of type I AT deficiency associated with severe thrombotic risk. Both the polymorphisms affected relevant transcription factor binding sites, which might affect the rate of transcription of AT gene. A previously reported polymorphism, g.67G>A, was identified in a 55-year-old male with proximal DVT. 22 This polymorphism was previously reported with very low frequency in a family with type I AT deficiency and was absent in control. This is the first report of AT genetic variations in regulatory region identified in Indian population with DVT and till now there is only 1 report showing polymorphism in the regulatory region of AT gene in caucasian population. 16 The heterozygous polymorphism, g.2085T>C, is located 228 bp upstream of translation initiation codon and was present in a patient with high plasma AT levels. Polymorphisms identified in the present study were located closer to the translation initiation codon and might explain severity of reduced circulatory AT levels when compared to that observed for g.2085T>C polymorphism.
In addition to searching for genetic variations in AT regulatory region, we also studied rs3138521 promoter polymorphism in 62 DVT patients with low AT level and 62 healthy controls. The molecular basis of rs3138521 was first reported in 1983 when the presence of either 108 bp (S) or 32 bp (F) nonhomologous variable segments were observed 345 bp upstream of the AT gene. 24 It was later suggested that F allele derived from the longer S allele by partial deletion followed by sequence divergence during evolution, and the promoter strength of S allele was found to be 1.6 fold higher than the F allele in HepG2 cells, whereas the strength of F allele was 1.7 fold higher than S allele in Hep3B cells. 25,26 However, no difference in promoter activity for these 2 alleles was observed in case of larger constructs. 26 Also, a strong linkage disequilibrium has been observed between rs3138521 and rs2227589, a polymorphism associated with high risk of venous thrombosis. 27,28 It has been suggested that high risk of thrombosis associated with rs2227589 might be due to linkage with rs3138521 which also affects the binding of potential transcription factors, thus affecting the rate of transcription of AT gene. 27 The rs3138521 allele frequencies observed in present study are S: 0.37 and F: 0.63 and are somewhat close to previously reported caucasian populations: rs3138521 S allele—0.21, 25 0.23, 27 0.21 29 , and 0.25. 24 rs3138521 was not associated with variations in the plasma AT levels, and our results are consistent with the Western studies where no association was found between rs3138521 and low plasma AT levels.
To conclude, this is the first study of genetic variations in regulatory regions of AT gene, where novel and known variations have been identified in Indian population with DVT having reduced AT levels. Indian studies regarding deficiency and subsequent genetic analysis of AT gene leading to DVT are very limited. A similar allelic frequency of rs3138521 and lack of association with plasma AT levels in Indian population show similar trend of AT polymorphism in Indian and Western thrombotic population. All the above-identified polymorphisms should be studied in a larger sample size to understand their distribution and association with thrombotic risk in Indian population with DVT. In patients with absence of mutations in protein-coding region of AT gene and severely reduced plasma AT levels, regulatory regions should be included in the analysis for understanding the genetic basis of defect, as they are involved in regulatory functions.
Footnotes
Authors’ Note
M.A.J. was the principle investigator of the work. T. B. performed the experimental studies, compiled the data, and wrote the manuscript. A.S. contributed to data analysis and manuscript writing. M.F.A. helped in writing the manuscript. R.S. and M.M. helped in screening of patients with DVT and provided valuable clinical inputs.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work is funded by the Indian Council of Medical Research (52/2/2009-BMS). T.B. is supported by Department of Science and Technology-INSPIRE-Senior Research Fellowship, A.S. is supported by the Indian Council of Medical Research-Senior Research Fellowship, and M.F.A. is supported by Council of Scientific & Industrial Research-Senior Research Fellowship.
