Abstract
FVA4070G (R2 polymorphism) influences plasma factor V (FV) concentration and was associated with mild activated protein C resistance. This polymorphism was reported to have a trans inheritance with FV Leiden (FVL) mutation. The aim of this study is to investigate the inheritance of R2 polymorphism in the homozygous FVL carriers. In this study, 99 patients with thrombosis and 7 individuals without a history of thrombosis all of which homozygous for FVL were included. Of 99 patients, 1 was heterozygous for FV A4070G. Additionally, 6 polymorphisms in the FV gene were analyzed for the heterozygous R2 patient and her family. When the allelic distribution was classified, 8 different haplotypes were obtained. In contrast to the literature, it was shown that R2 polymorphism could be inherited in cis position with FVL and also the family members could have co-inheritance of the FVL and R2 on the same chromosome as proband.
Keywords
Introduction
Factor V 1691 G→A (FV Leiden [FVL]) is the most common genetic risk factor for inherited thrombophilia resulting from G→A substitution at nucleotide 1691, leading to Arg→Glu replacement at amino acid 506 (R506Q) and causing activated protein C resistance (APCR) commonly among Caucasians. 1,2 In addition to FVL mutation, an A to G change at nucleotide 4070 results in His→Arg amino acid substitution at position 1299 in exon 13 of the factor V (FV) gene was described and called as R2 polymorphism. 3 Afterward, R2 polymorphism has been reported to be in tight association with more than 12 polymorphisms of the FV gene which is collectively known as HR2. 4 The R2 variation has been shown to influence plasma FV concentration and its association with mild APCR. 3 –5 It also has been associated with an increased risk of thrombosis either by itself or in combination as a compound heterozygote with FVL. 6,7 However, there are studies that were opposed to these associations. 8,9
Due to genetic variations in different ethnic populations, the presence of the FV 4070 A→G alteration has been reported in different populations 3 –5,10,11 and the frequency of FV4070G allele was found to be 8.5% in Turkish population. 12 –14 Additionally, we reported that FV R2 polymorphism is not an independent risk factor in thrombosis, but it can be important when associated with FVL in VTE. 12 On the other hand, it was shown that 4070G allele was frequently co-inherited in symptomatic FV1691A carriers in our previous study. 15 In the literature, it was reported that HR2 haplotype and R506Q mutation did not reside on the same chromosome 4,16 as well as in another study analyzing familial transmission of the FV R506Q mutation and the 4070 G allele in the participants who carry both these alleles. 17 So, we aimed to investigate FV 4070 A→G polymorphism in the homozygous FVL mutation carriers and to detect allelic distribution of R2.
Materials and Methods
The DNA samples that were determined as carrying FVL mutation in homozygous state by real-time polymerase chain reaction ([PCR]; LightCycler; Roche Diagnostics, Roche Molecular Biochemicals, Mannheim, Germany) 18 were included in the study from 99 patients with the diagnosis of thromboembolism and 7 healthy unrelated individuals without any familial history of thrombosis and stroke from our DNA bank. All patients and controls gave a written informed consent for the study.
The FV A4070G polymorphism was detected by the previously described method using the following primers: 5′-CAAGTCCTTCCCCACAGATATA-3′ and 5′-AGATCTGCAA AGAGGGGCAT-3′ for PCR of exon 13 of the FV gene. Amplification was performed for 35 cycles with annealing temperature of 57°C (Biometra, Germany). The amplified 703 bp PCR fragment was digested with RsaI enzyme (Fermentas, Lithuania) at 37
Results
The presence of the FV A4070G was investigated in 99 patients with thrombosis and 7 healthy individuals who were homozygous for FVL mutation. Of the 99 patients, 98 were homozygous for the wild-type FV A4070G polymorphism. Only 1 patient with recurrent venous thromboembolism was found in heterozygous state (R1R2). Among the healthy individuals, none of them had R2 allele. So, we analyzed the family members of the patient with R2 haplotype.
As shown in Figure 1 , there was 3-generation vertical and horizontal transmission of the FVL and FV1299. The index patient with the homozygous FVL was a 41-year-old woman (II-1) who developed pulmonary embolism and was under warfarin treatment. Family members, her 35-year-old brother (II-2) and 34-year-old sister (II-3) were compound heterozygotes for FVL—FV1299 as well as the proband’s 56-year-old mother (I-1), and none of them had a thrombotic event. The FVL was observed in 3 children (aged between 13 and 20) and none of them had thrombosis. Although the 2 children (III-2 and III-3) were heterozygous for R2 polymorphism, only 1 was found to carry R1 allele in homozygous state (III-1; Figure 2 ). None of the family members had the same mutation profile as the index patient.
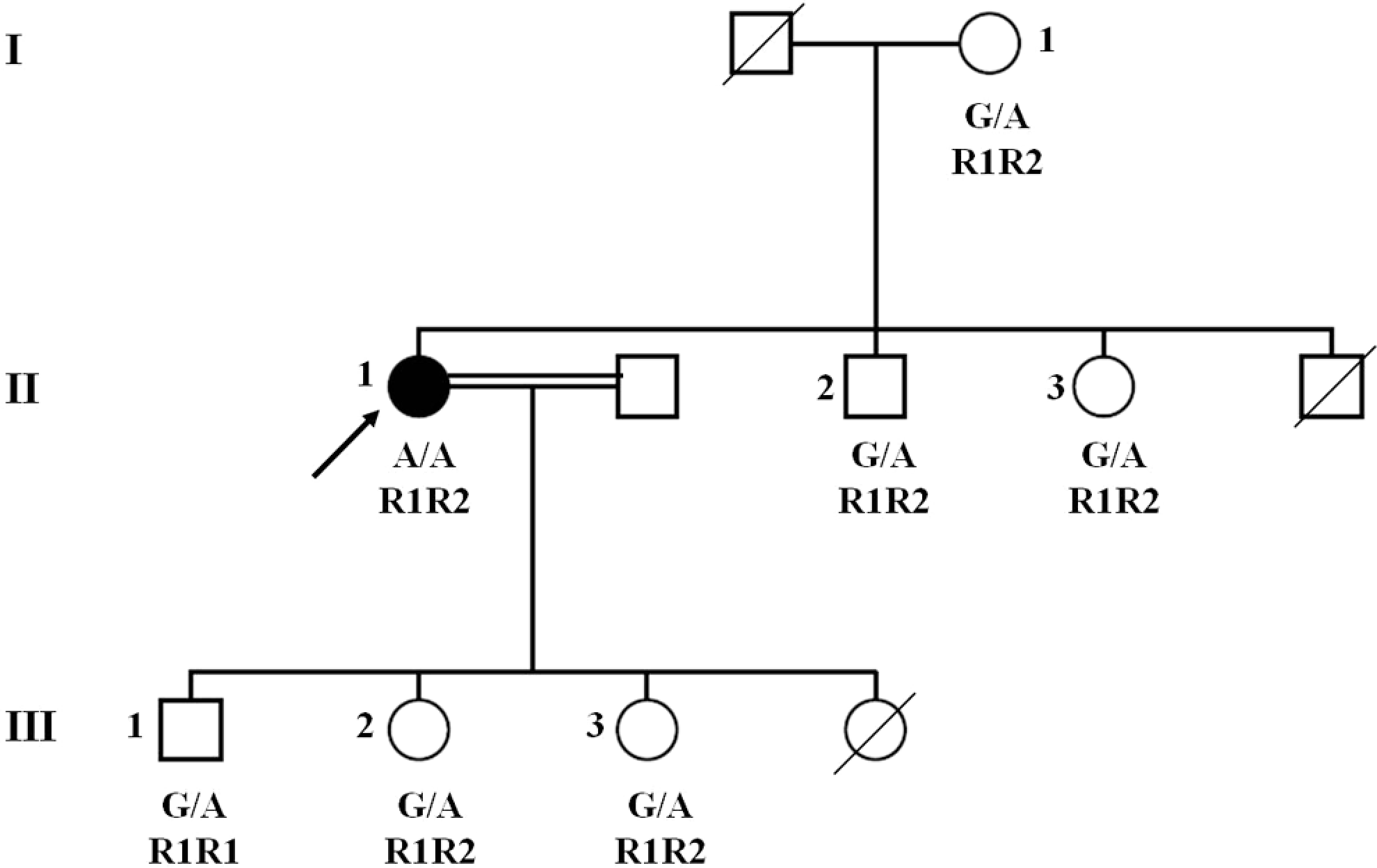
The familial pedigree of the patient carrying FV1691AA/R1R2.
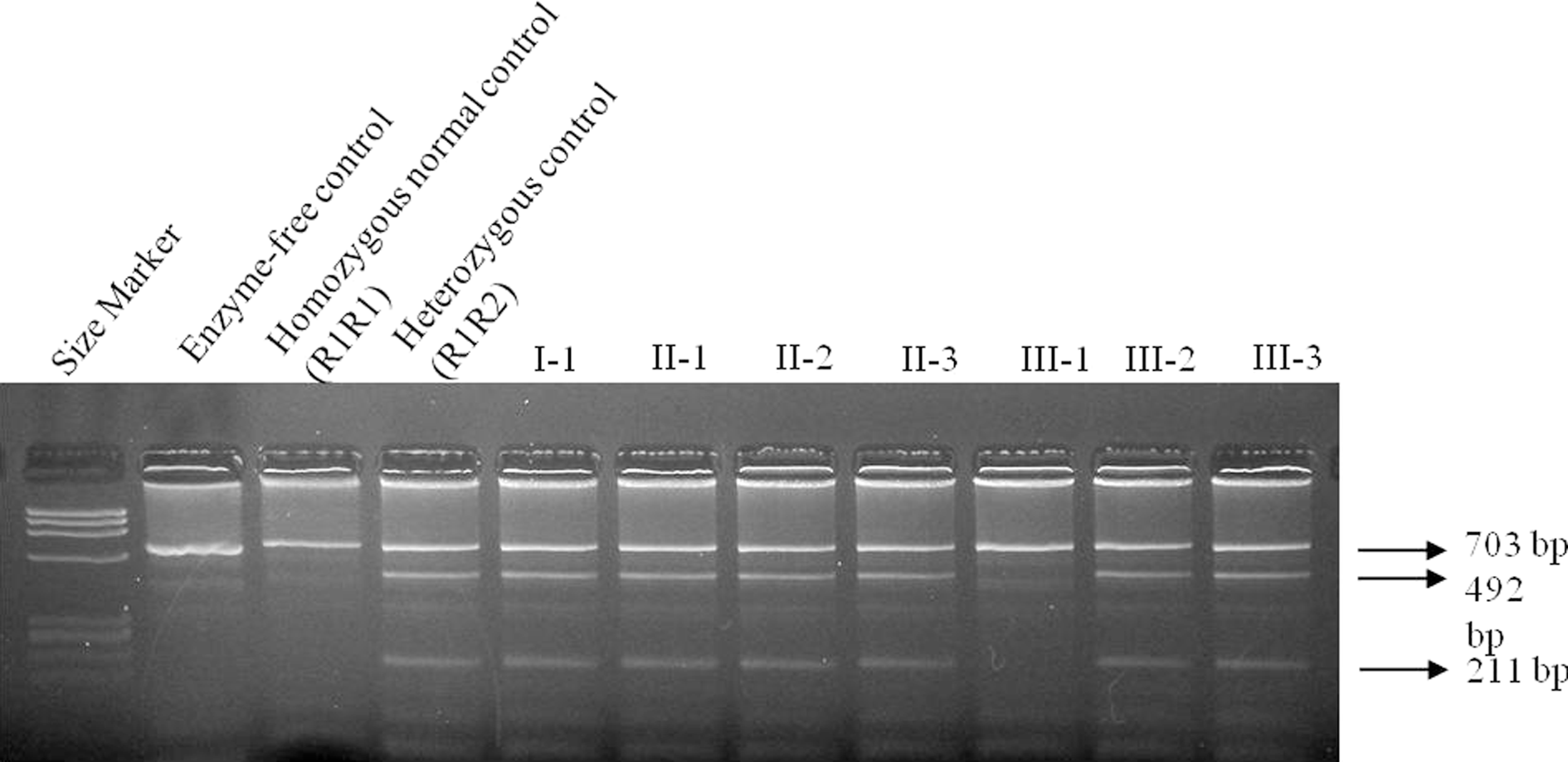
Agarose gel electrophoresis of FV A4070G polymorphism in the family members. Polymerase chain reaction products of 703 bp were digested by RsaI restriction endonuclease and band profiles were examined in 2% agarose gel. After digestion, 703-, 492-, and 211-bp fragments were observed in heterozygosity (R1R2), whereas 703-bp fragment in wild-type (R1R1). M: ØX174 DNA-HaeIII Marker. I-1, II-1, II-2, II-3, III-2, III-3: R1R2, III-1:R1R1.
Additionally, 6 single-nucleotide polymorphisms of FV gene were analyzed in all family members. All the participants were found to carry −426G (promoter), 495G (exon 4), 1470C (exon 9), 1806G (exon 11), and 2298C (exon 13) alleles identical to the index patient. Interestingly, A327G (exon 2) polymorphism was detected to be heterozygous in 6 individuals from the family, whereas the proband was homozygous for this variant. When the allelic distribution was classified, 8 different haplotypes were obtained. In this way, their allelic linkage could be shown. We also detected a haplotype that FVL and R2 were not present on the same chromosome. Proband had haplotypes 7 and 8 as shown in Table.1.
Factor V Haplotype Distribution for the Family Members Using Polymorphisms in Promoter and Exons 2, 4, 9, 11, 13.a
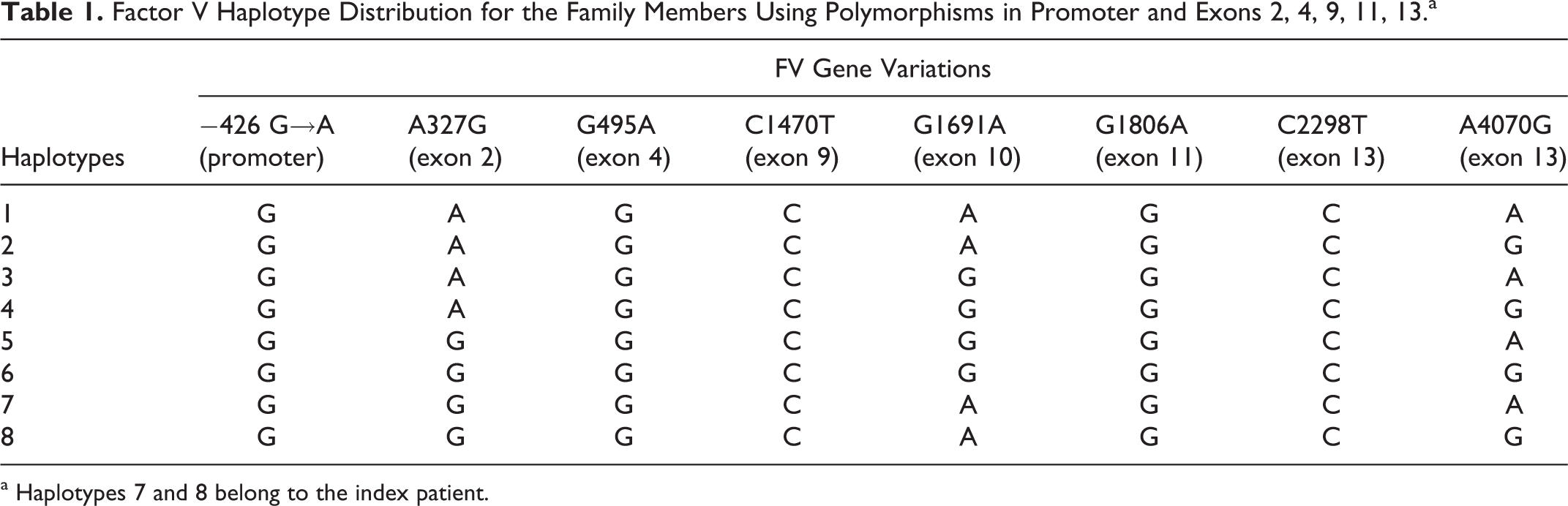
a Haplotypes 7 and 8 belong to the index patient.
Discussion
The pathogenesis of thromboembolism is multifactorial and involves acquired and genetic factors. Inherited gene defects related to the coagulation system have been reported as risk factors for thrombosis. The common inherited gene defect is a single-point mutation in exon 10 of the FV gene; FVL (G1691A and R506Q) causes a resistant to activated protein C. This mutation represents the most common genetic risk factor for venous thrombosis in caucasian population. 1 Recently, the HR2 haplotype consisting of a group of polymorphisms in different exons of the FV gene has been described and marked as R2 polymorphism, an A→G alteration at position 4070 in exon 13 that predicts the His1299Arg substitutions. The FV A4070G polymorphism was shown to affect plasma FV levels and increase the resistance to activated protein C. 3 –5 The frequency of the FV 1691A and FV 4070G was found to be 10.4% and 8.5%, respectively, in the Turkish population. 12,21,22
Several gene defects often coexist in thrombophilia, and these genes show an attenuated effect in individuals for the occurrence of thrombosis. Although the effect of A4070G on the pathogenesis of thrombosis was controversial, 10,12 compound heterozygosity of FV1691A and FV 4070G produced a 3- to 4-fold increase in venous thromboembolism risk when compared with FVL alone. 7 Other studies did not find such an association or risk. 4,8,9 We previously reported that Turkish patients with thromboembolism showed a co-inheritance for the 4070G allele, frequently in symptomatic FV1691A carriers. 15
Bernardi et al 4 showed the gene frequencies of the HR2 haplotype and of the R506Q mutation in the Italian population and have reported that HR2 haplotype and the R506Q mutation did not reside on the same chromosome. In a French study, an analysis of familial transmission of the FV R506Q mutation and the 4070G allele in the 18 participants who carry both these alleles demonstrated that none had these 2 mutations on the same chromosome. 17 Additionally, it was pointed out that FVL and FV HR2 never resided on the same allele and thus individuals with both defects did not have any normal FV. 16 So, in this study, we investigated the question of the inheritance of the R2 allele in FV 1691AA carriers. Although all the FV 1691AA carriers were found in 1691A/4070G haplotype, only one of these carriers was found heterozygous for FV A4070G polymorphism. Based on these results, R2 allele was found neither in the patient group with thrombosis (n: 99) nor in the healthy control group (n = 7). Based on these results, we also investigated the family of the R1R2 patient. The family members were heterozygous for FVL and were without thrombosis. Of the FVL heterozygotes, only 1 individual carried the R1R1 genotype, although 5 of them were found as R1R2 (Figure 1).
Additionally, 6 polymorphic sites in the promoter region and exons 2, 4, 9, 11, and 13 of FV gene were chosen for haplotype analysis and examined in the family members. Zivelin et al 20 also suggested that crossover could be obtained in this polymorphic region and haplotypes were created from participants carrying homozygous state for FVL mutation. In this study, haplotype analysis also based on multiple polymorphisms in alleles from individuals of the family was limited to only participants homozygous for FV 1691A to minimize the number of uninformative alleles. The use of only homozygotes maximized the number of informative alleles for generation of probable haplotypes. Eight different haplotypes were detected but, in this respect, 2 different haplotypes could be more informative. The FV 1691A and R2 alleles could be obtained by crossover events in the proband within the range of exon 2 to between exons 10 and 13. In this manner, the family members could have co-inheritance of the FVL and R2 polymorphism on the same chromosome as proband. Analyzing the FV levels of the family members would be more informative; however, it was not possible as the family lived in another town.
In conclusion, an interesting observation of this study is that R2 polymorphism was found to be on the same chromosome with FV 1691A mutation, in contrast to earlier results.
Footnotes
Acknowledgments
We are grateful to Muzaffer Demir, Hakan Gürkan, and Hilmi Tozkır from Trakya University, Reyhan Diz Küçükkaya from İstanbul Bilim University, Ender Altıok from Acıbadem Hospital, Ferit Avcu from GATA, Solaf M. Elsayed from Medical Genetics Center, Cairo, Egypt, and Selçuk Öztürk who assisted for the collection of homozygous FVL samples.
Declaration of Conflicting Interests
The author(s) declared a potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by the Ankara University Research Fund (Project no: 08B3330008) and partially by the Turkish Society of Hematology Research Projects Support Program (Project no: 2010-1).
