Abstract
Use of allogeneic blood products to treat pediatric trauma may be challenged, particularly in relation to safety. We report successful treatment of a child with severe abdominal and pelvic injuries with preemptive fibrinogen supplementation followed by rotational thromboelastometry (ROTEM)-guided, goal-directed hemostatic therapy. Fibrinogen concentrate was administered (total dose: 2 g), while transfusion of fresh frozen plasma and platelet concentrate was avoided. Activated partial thromboplastin time was prolonged and Quick values were low but ROTEM clotting time values remained normal, therefore, no thrombin-generating drugs were considered necessary. This case shows the potential for hemostatic treatment with coagulation factor concentrates to be applied to pediatric trauma.
Keywords
Introduction
Coagulopathy is a frequent complication in pediatric patients with trauma, and it is strongly associated with mortality. 1 The management of pediatric trauma generally follows similar principles to those used for adult patients, but specific data for infants and children are scant. Hemostatic therapy is mainly based on transfusion of allogeneic blood products, such as fresh frozen plasma (FFP), platelets, and cryoprecipitate. A transfusion protocol may be advocated as a means of standardizing therapy and improving outcomes, but there are no strong data to support any particular protocol in children. 1 –3
Routine use of allogeneic blood products in pediatric trauma may be challenged, particularly in relation to safety. Red blood cell (RBC) transfusion has been associated with increased mortality among pediatric patients with trauma. 4,5 Allogeneic blood product transfusion also carries a risk of immunomodulation and infection. 6,7 Complication risks may be higher among children than adults—for example, transfusion reactions to RBCs (mostly allergic) have an incidence of 37 per 100 000 transfusions in infants, compared with 13 per 100 000 in adults. 6 The most common complications of transfusion relate to human error (eg, overtransfusion and lack of knowledge of neonatal requirements). 6 However, there are also concerns regarding potential long-term consequences of transfusion. For example, the incubation period of variant Creutzfeldt-Jakob disease (vCJD) has been suggested to exceed 50 years. 8 Possible increases in the risks of Alzheimer disease 9 and cancer such as non-Hodgkin lymphoma 10 have also been postulated among recipients of allogeneic blood transfusion. Such risks are small and/or unconfirmed, but a precautionary approach is mandatory in children: in 2006 the UK Department of Health recommended the use of imported FFP (treated with methylene blue) for all children aged <16 years, as a precaution against vCJD. 11
Coagulation factor concentrates (eg, fibrinogen concentrate and prothrombin complex concentrate) represent a possible means of increasing hemostatic capacity and reducing dependence on allogeneic blood products. For example, fibrinogen concentrate has been shown to improve fibrin polymerization and clot strength in children undergoing craniosynostosis surgery, reducing the need for FFP or platelets to control bleeding. 12 In adult trauma, fibrinogen supplementation has been proposed as first-line hemostatic therapy and experience suggests that this approach might improve outcomes. 13 –15 Pharmacosurveillance over a 22-year period reported 9 thrombotic events possibly related to administration of fibrinogen concentrate, 7 of which were in patients with congenital fibrinogen deficiency, meaning that the risk of such events is low (3.48 per 100 000 treatment episodes). 16,17 It should be acknowledged that fibrinogen concentrate is manufactured from pooled human plasma. However, viral inactivation steps during manufacture minimize the risk of pathogen transmission, 18 such that there have been no confirmed cases of pathogen transmission despite the use of more than 3 million grams since 1985.
There is increasing evidence that viscoelastic assessment (thromboelastometry or thrombelastography) is helpful for guiding coagulation therapy. 14,19 These methods provide rapid information on speed, elasticity (strength), and stability (fibrinolytic degradation) of clotting, 20,21 enabling effective tailoring of therapy to the patient’s specific needs. 19 This “theragnostic” approach also enables the effect of intervention on coagulation status to be measured quickly after administration.
Data on the use of viscoelastic assessment in pediatric patients with trauma are limited, and no previous studies have investigated fibrinogen concentrate in children with trauma. We report a severe pediatric blunt trauma case where thromboelastometry-guided treatment with fibrinogen concentrate was the predominant hemostatic therapy.
Case Presentation
A previously healthy 7-year-old caucasian boy (body weight 20 kg and height 1.2 m) was run over by a school bus at 07:30. Upon arrival of the emergency doctor (15 minutes later), the patient was conscious, with systolic blood pressure (BP) of 70 mm Hg and blood oxygen saturation (SaO2) of 98% while receiving oxygen via a mask. Clinical examination showed open unstable pelvis fracture, extensive soft tissue lesion from the pelvis to the perineum, and multiple soft tissue lesions of the upper and lower limbs. Thorax trauma and intraabdominal lesions were also suspected. Anesthesia was induced at the scene of the accident using 5 mg midazolam (Midazolam Actavis; Actavis Group PTC ehf, Hafnarfjördur, Iceland), 50 mg ketamine (Ketanest S; Pfizer Corporation Austria GmbH, Vienna, Austria), and 20 mg succinylcholine chloride (Lysthenon; Nycomed, Dübendorf, Germany) and the trachea was then intubated. Anesthesia was maintained with 5 mg midazolam, 20 mg rocuronium bromide (Esmeron; NV Organon, Oss, Holland), and a total dose of 0.1 mg fentanyl (Fentamed; DeltaSelect GmbH, Dreieich, Germany). Auscultation indicated decreased respiration in the left lung but treatment was not considered necessary. The patient was transported by helicopter to Salzburg University Hospital. The following fluid therapy was administered at the accident scene and during transport to the hospital: 300 mL crystalloid (Ringer solution; Fresenius Kabi, Graz, Austria) and 100 mL succinylated gelatine (Gelofusin; B. Braun Melsungen AG, Melsungen, Germany).
Upon arrival at the emergency room (ER) at 08:20, BP was 50/25 mm Hg and SaO2 was 100%, with a fraction of inspired oxygen (F
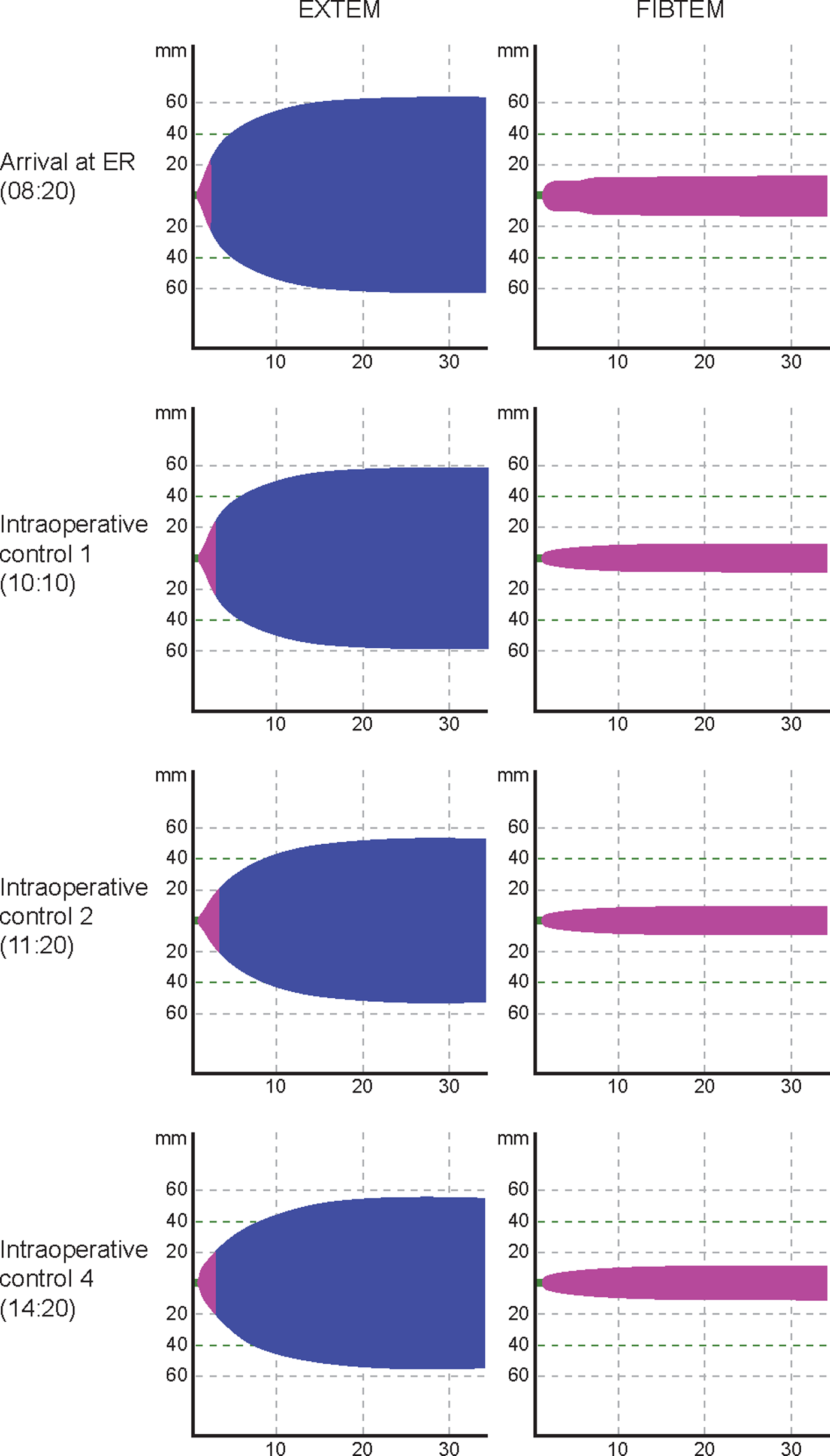
The rotational thromboelastometry (ROTEM) traces from EXTEM (extrinsically activated thromboelastometric test) and FIBTEM (extrinsically activated thromboelastometric test with platelet function inhibited) assays performed upon arrival at the emergency room (ER) and during surgery.
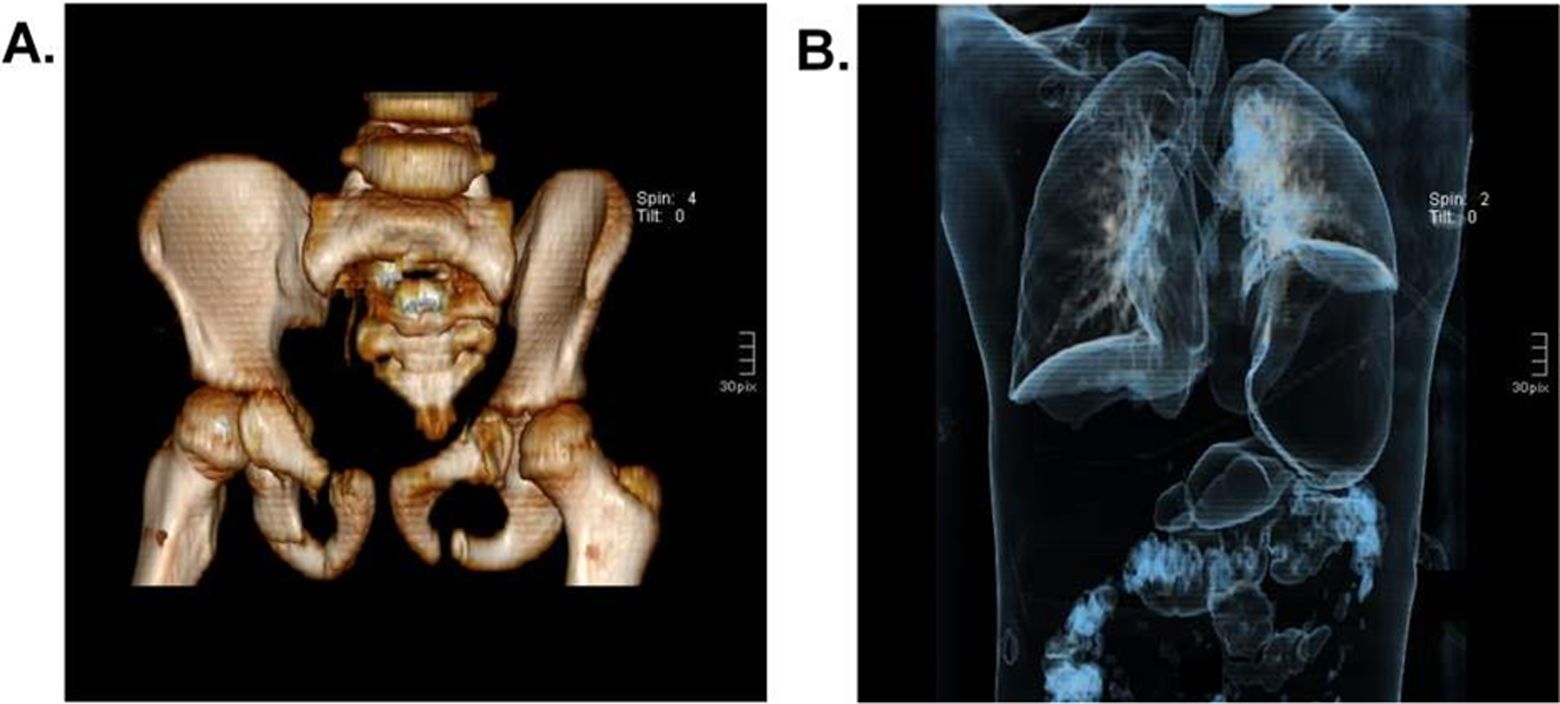
Computed tomography scan showing the patient’s injuries following admission to the emergency room. Panel A shows the pelvic area, with a bilateral fracture of the superior and inferior rami of the pubic bone, rupture of the symphysis pubis, unilateral lesion of the sacroileal joint, and bilateral fracture of massa lateralis. Panel B shows the thorax and abdomen, with rupture of the left diaphragm, herniation of stomach and spleen, splenic injury grade III, left renal trauma grade III, and hepatic contusion.
Thrombelastometry and Laboratory Results.
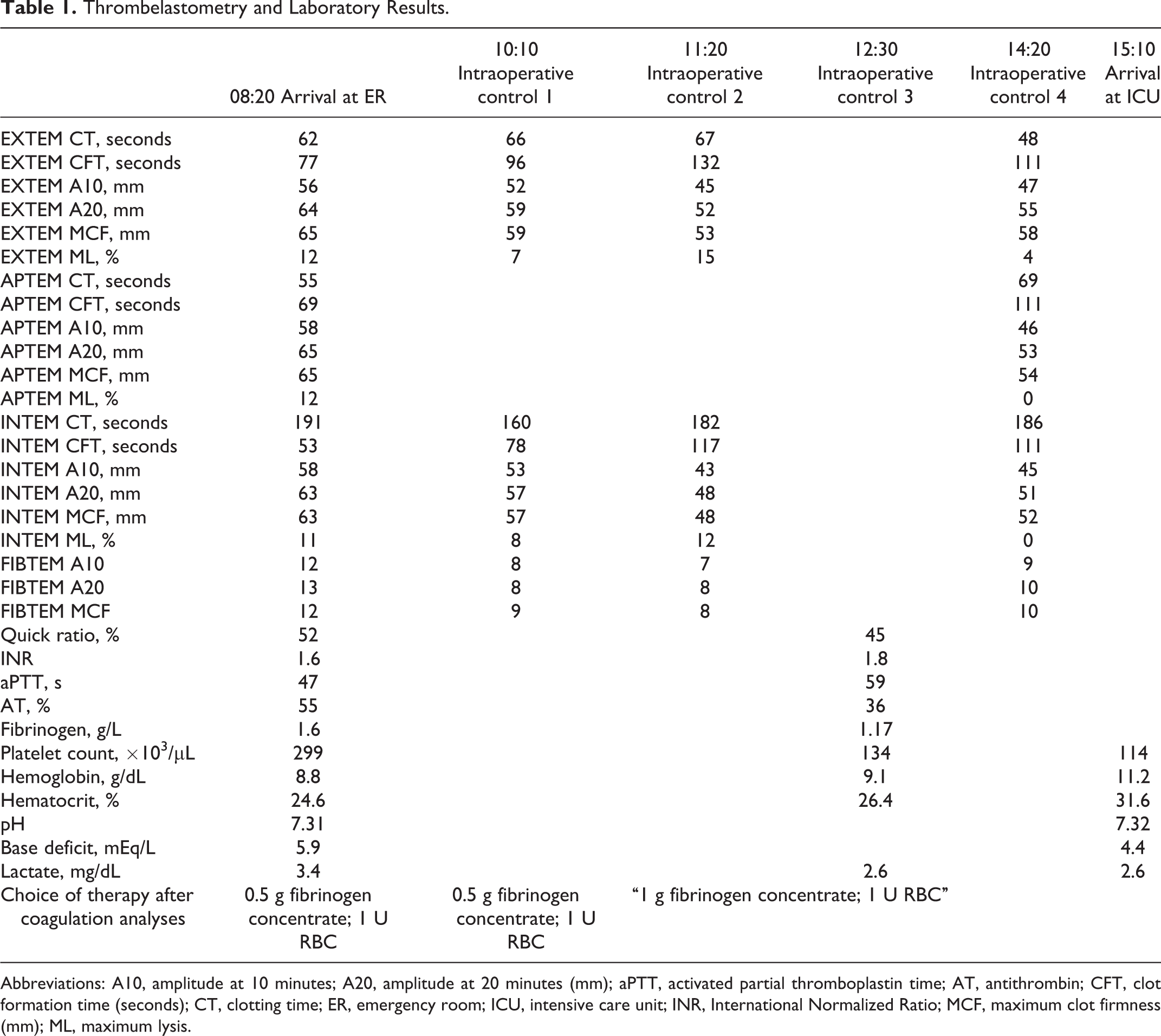
Abbreviations: A10, amplitude at 10 minutes; A20, amplitude at 20 minutes (mm); aPTT, activated partial thromboplastin time; AT, antithrombin; CFT, clot formation time (seconds); CT, clotting time; ER, emergency room; ICU, intensive care unit; INR, International Normalized Ratio; MCF, maximum clot firmness (mm); ML, maximum lysis.
One hour into surgery (10:10), diffuse, moderate bleeding was observed and point-of-care ROTEM analysis was performed using the device in the pediatric OR. Results of value in guiding further hemostatic therapy were available within minutes. In the FIBTEM assay (assessment of fibrin-based clot; extrinsically activated thromboelastometric test with platelet function inhibited), clot amplitude at 10 minutes (A10) and maximum clot firmness (MCF) had decreased to 8 and 9 mm, respectively (toward the bottom of the normal ranges for these parameters among children aged 6-10 years of 7-21 and 7-22 mm, respectively 19 ). In contrast, EXTEM (extrinsically activated thromboelastometric test) clotting time (CT) was normal (66 seconds, compared with a normal range for 6- to 10-year-olds of 43-74 seconds 19 ). In light of this information, another 0.5 g of fibrinogen concentrate was administered, but no thrombin-generating products (eg, prothrombin complex concentrate and recombinant activated factor VII) were administered. This treatment led to temporary control of bleeding. One hour later (11:20), ROTEM testing again showed low FIBTEM A10 and MCF values (Table 1). Because the surgical procedure was ongoing and bleeding had resumed, a further 1 g of fibrinogen concentrate was administered. In a sample taken at 12:30, laboratory-measured fibrinogen concentration was low and standard coagulation times were prolonged (Table 1). However, in the absence of clinically relevant bleeding, no hemostatic intervention was considered necessary.
Toward the end of surgery (14:20), the patient was clinically stable with no significant bleeding, and ROTEM analysis showed clot firmness within the normal range (INTEM and EXTEM assays; MCF normal ranges for 6- to 10-year-olds of 53-69 mm and 53-68 mm, respectively 19 ). Clotting time in both EXTEM and INTEM assays was within the normal range (EXTEM: 43-74 seconds; INTEM: 97-212 seconds 22 ), as it had been throughout the surgery.
During the 6-hour period of surgery, volume therapy comprised 1750 mL crystalloid (Elomel Isoton; Fresenius Kabi) and 500 mL gelatin. Hemoglobin levels were maintained at 7 to 9 g/dL by administering 2 units of RBC (Table 1). Blood loss during the first 3 hours after trauma was estimated at more than 1 blood volume (>70 mL/kg body weight), qualifying as massive blood loss. 23
Upon completion of surgery (15:10), the patient was transferred to the pediatric intensive care unit, with the following diagnoses: left diaphragm rupture, splenic injury grade III, left renal trauma grade III, hepatic contusion, rupture of the urethra, amputation of left testis, penetrating perineal injury with lesion of anterior rectum wall, bilateral fracture of the superior and inferior rami of the pubic bone, rupture of the symphysis pubis, unilateral lesion of the sacroileal joint, bilateral fracture of massa lateralis, fracture of the transverse processes of the lumbar vertebrae, contusion and abrasions of the left forearm, and extensive detachment of the abdominal epidermis. Herniation of the spleen, stomach, and (partly) the left colic flexure into the left thorax were also confirmed, as per the initial diagnosis. The injury severity score was 41.
On day 2 (second postoperative day), 1 unit of RBC was administered to correct a hemoglobin value of 7.5 mg/dL, but no other allogeneic blood products or hemostatic agents were needed postoperatively. Platelet count remained above 100 × 103/µL at all times. The stoma reversal operation was performed on day 40 without any need for transfusion. The patient was released from hospital 45 days after the accident, and no side-effects attributable to the treatments administered were observed.
Discussion
This is the first report of early, preemptive administration of fibrinogen concentrate followed by ROTEM-guided, goal-directed therapy in a pediatric patient with severe trauma. A favorable outcome was achieved despite severe abdominal and pelvic injuries, massive blood loss, no transfusion of FFP or platelet concentrate, and transfusion of only 3 U of RBC to supplement the 2 g fibrinogen concentrate administered intraoperatively.
High base deficit (≥6) and coagulopathy (International Normalized Ratio [INR] ≥1.5) upon arrival at the ER have recently been associated with mortality in pediatric patients with trauma. 24 The combination of coagulopathy and early shock produced an odds ratio of 3.8 for mortality. Base deficit (>5 mEq/L) and lactate concentration above 2.94 mmol/L have also been reported recently as independent predictors of mortality. 25 Our patient had a base deficit of 5.9, an INR of 1.6, and lactate above 2.94 mmol/L upon arrival at the ER. It was estimated that the patient had massive blood loss (more than one blood volume) during the first 3 hours after trauma. However, based on the clinic’s experience, no massive transfusion protocol based on administration of allogeneic blood products was implemented. Instead, RBC administration was guided by hemoglobin level, and hemostatic therapy was performed solely with fibrinogen concentrate. The dose administered (2 g or 100 mg/kg in total) is comparable with previous reports in the literature (eg, in one study, repetitive doses of 30 mg/kg body weight, up to a total median dose of 76 mg/kg body weight, were administered 12 ). Of interest, activated partial thromboplastin time (aPTT) was prolonged and Quick values were low, but antithrombin levels were very low throughout the procedure and ROTEM CT values remained normal. These data suggested that initiation of coagulation was sufficient at all times, because of a balance between levels of procoagulants and anticoagulants. As a result, no thrombin-generating drugs were considered necessary. This might not have been the case if decisions had been based on aPTT and Quick values. Lack of assessment of overall coagulation status may be considered as a significant drawback of laboratory coagulation tests. 26
The EXTEM MCF was within the normal range throughout the perioperative period of our case. This suggests sufficient contribution of platelets to the whole-blood clot, although platelet count fell slightly below the normal range of 150 000 to 450 000/µL during the latter stages of surgery. It has been suggested that fibrinogen supplementation may potentially compensate for reduced platelet count, 27,28 and this might explain our observations. Earlier, FIBTEM MCF values were at the lower end of the normal range for a child of this patient’s age, and these values were used to guide intraoperative treatment with fibrinogen concentrate. The incidence of hyperfibrinolysis in adult patients with severe trauma is high, 29 and it is reasonable to expect that tranexamic acid might also be helpful in severe pediatric trauma. In our case, the APTEM and EXTEM results showed no overt signs of hyperfibrinolysis. The EXTEM, APTEM, and INTEM maximum lysis values were 7% to 15% during the early stages of surgery and decreased to 0% to 4% before the end of surgery, indicating satisfactory stability of the whole-blood clot. However, these viscoelastic assays might not be sensitive enough to detect susceptibility to fibrinolysis/hyperfibrinolysis, and it is possible that an initial dose of tranexamic acid would have provided further improvement in clot quality and decreased transfusion.
Our treatment strategy was based on the maintenance of a consistent FIBTEM MCF at the low end of the normal range (7-22 mm). 22 Preemptive fibrinogen concentrate was administered in the ER because no ROTEM device was available nearby. The results of the ROTEM analysis, which were available later, confirmed the appropriateness of the chosen therapy. In our clinic, the first dose of fibrinogen concentrate may occasionally be administered preemptively, before the FIBTEM results are available, because fibrinogen is the first coagulation factor to reach critically low levels during bleeding. 30,31 Further testing in the OR, where the ROTEM device is available at the point of care, can be used to confirm the patient’s status and guide subsequent treatment. The ratio of intraoperative fibrinogen concentrate (g) to RBC (U) was 0.7, close to the ratio of 0.9 recently described in hemostatic therapy with coagulation factor concentrates in adult trauma. 14 The subsequent approach to hemostatic therapy was similar to our usual strategy for the management of adult trauma where, in the absence of signs of hyperfibrinolysis, fibrinogen concentrate is given as first-line hemostatic therapy when FIBTEM A10 is <7 mm—although the different FIBTEM normal ranges among children may be considered. 22 This approach to treating pediatric trauma does not require off-label use of fibrinogen concentrate in Austria, although the licensing status is different in other countries. As additional treatment, prothrombin complex concentrate may be administered later on if EXTEM CT is prolonged beyond the normal range (>80 seconds). The rationale for such goal-directed therapy is based on the evidence that it can reduce mortality compared with predicted rates 13 and reduce transfusion of allogeneic blood products compared with FFP-based therapy. 14
This case report shows the potential for preemptive fibrinogen supplementation followed by a goal-directed, theragnostic approach to hemostatic therapy to be applied to pediatric trauma. It also highlights the shortcomings of standard laboratory coagulation tests such as prothrombin time and aPTT, compared with viscoelastic clot assessment. Randomized controlled trials are needed to establish optimal treatment strategy in pediatric trauma.
Consent
Written informed consent was obtained from the patient’s parents for publication of this case report and the accompanying images. A copy of the written consent is available for review with the editor-in-chief of this journal.
Footnotes
Acknowledgments
The authors are indebted to Dr Imre Vasváry from the Department of Radiology, SALK University Hospital, Salzburg, Austria, for providing the 3D CT images. Editorial assistance with manuscript preparation was provided by Meridian HealthComms, funded by CSL Behring.
Authors– Notes
B.Z. and B.S. provided in-hospital anesthesiologic patient care. C. Schimke provided in-hospital surgical patient care. B.Z., C. Schimke, H.S., and C. Solomon collected data. B.Z., C. Schimke, H.S., and C. Solomon assisted the writing of the case report. P.M. provided emergency patient care. B.S. and P.M. reviewed the case report for scientific content.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Schöchl and Dr Solomon have received speaker honoraria and/or research support from CSL Behring (manufacturer of fibrinogen concentrate) and Tem International GmbH (manufacturer of the ROTEM device).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:Editorial assistance with manuscript preparation was provided by Meridian HealthComms, funded by CSL Behring.
