Abstract
Platelets are crucial for maintaining physiological equilibrium, thrombosis formation, inflammation, bacterial defense, wound repair, angiogenesis, and tumorigenesis. In the Pediatric Intensive Care Unit (PICU), children frequently exhibit platelet reductions or functional alterations due to diverse pathological conditions, which significantly influence disease progression and therapeutic approaches. We analyzed the association between platelets count and its derived parameters and all-cause mortality. Adjusted smoothing spline plots, subgroup analysis and segmented multivariate logistic regression analysis were conducted to estimate the relative risk between proportional risk between platelets and all-cause mortality. Of the 11625 children, 677 (5.82%) died. After adjusting for confounders, there was a negative association between platelets and the risk of all-cause mortality in PICU. For every 100 × 10^9/L increase in platelets, the risk of death was reduced by 17% (adjusted OR = 0.83, 95% CI: 0.78, 0.89). The results of sensitivity analysis showed that in different stratified analyses (age, ICU category,WBC Count), the effect of platelets count on all-cause mortality remained stable. After adjusting for inflammation, nutrition, and liver function factors, platelets reduction is still an independent risk factor for PICU all-cause mortality.
Keywords
Introduction
Platelets play an important role in homeostasis and thrombosis in the body, in addition to their involvement in a variety of physiological and pathological processes, including inflammation, antimicrobial defense, wound healing, angiogenesis, and tumor growth. 1
Measurement of platelets count (PC) is the basis for assessing platelet function. Other platelet-derived parameters include mean platelet cell volume (MPV), platelet distribution width (PDW), plateletcrit (PCT), and mean platelet volume/platelet count ratio (MPR). Platelet count and MPV are simple and cost-effective methods that can be obtained from routine complete blood count (CBC) tests in clinical practice.In critically ill patients, platelet counts and platelet morphology are altered and platelet-related parameters change simultaneously. 2 Mean platelet volume (MPV), which reflects platelet size a simple and accurate marker of platelet function and activation status. MPV serves as an indicator of the size of platelets, which diminishes with the extension of the platelet lifespan. An elevation in MPV signifies a greater proportion of freshly formed platelets within the circulation, suggesting increased platelet production and/or destruction, indicating enhanced platelet activation. The platelet distribution width (PDW), which is a marker of platelet activation, is a parameter of the heterogeneity of the volume of platelets. PDW increases with cell size in the circulation, suggesting the presence of both mature and nascent platelets in the circulation. PCT is the arithmetic product of platelet count and platelet volume.2,3 In cases of thrombocytopenia and mortality, there is a decrease in platelet count, platelet distribution width (PDW), and plateletcrit (PCT). However, the mean platelet cell volume (MPV) and the mean platelet volume to platelet count ratio (MPR) may either remain normal or become elevated.
Platelet activation and depletion predicts a poorer prognosis, 4 and thrombocytopenia is associated with increased mortality in patients in the intensive care unit. 3 Platelet parameters have been commonly studied in critically ill patients in adults, and in the prediction of neonatal sepsis.5,6 However, only few studies have focused on the relationship between platelet parameters and Pediatric Intensive Care Unit (PICU) mortality, and those that have existed have limitations, with study populations limited to preterm infants or with small sample sizes.
In the present study, we investigated platelet-related parameters at admission in a large cohort of unselected ICU children from a public database of PICU and assessed their association with all-cause mortality in PICU patients. To our knowledge, this is the first study to date to link all-cause mortality in unselected ICU children with admission platelet-related parameters.
Methods
Subjects
We performed a retrospective analysis using the Pediatric Specialty Intensive Care (pediatric ICU) database, selecting 11625 critically ill children with comprehensive laboratory test results (Figure 1). PIC (Pediatric Intensive Care) is a large pediatric-specific, single-center, bilingual database containing information related to children in the intensive care unit at the Children's Hospital of ZheJiang University School of Medicine, China, from 2010 to 2018. The pediatric ICU database includes vital sign measurements, medications, laboratory measurements, fluid balance, diagnosis codes, length of stay, survival data, and more. 7 The laboratory indicators we included are the results of the first blood work after admission to the ICU. Exclusion criteria: missing platelets results, thrombocytopenia, pancytopenia, leukemia, anemia, K-M syndrome, post-chemotherapy bone marrow transplantation, phagocytic syndrome. The first nursing unit includes the Neonatal Intensive Care Unit (NICU), Surgical Intensive Care Unit (SICU), Pediatric Intensive Care Unit (PICU), and Cardiac Intensive Care Unit (CICU). The thrombopoietic drugs include recombinant human thrombopoietin injections and recombinant human interleukin-11 injections.
PIC database is a public database. The requirement for individual patient consent was waived because the project did not impact clinical care, and all protected health information was deidentify. 7 We formally applied for access through the procedures recorded on the PIC website and PhysioNet, and we have signed a data usage agreement. We handle data responsibly and adhere to the principle of cooperative research.
Statistical Analysis
Data is expressed as mean (SD) or median (Q1-Q3) for continuous variables and percentage (%) for dichotomous variables. Subgroup analyses examined the relationship between platelets and the risk of all-cause mortality according to age, ICU category, inflammation, nutrition, liver function and other indicators. Interaction tests in the logistic regression model were used to compare odd ratios between the subgroups analyzed.
Logistic regression models were used to examine the effect of platelets and other variables on the occurrence of all-cause mortality. Multivariate regression models included other variables, including sex, age in months, ICU category, creatinine; albumin; total cholesterol (TC); hemoglobin; alanine aminotransfease (ALT); WBC Count; oxygen saturation (sO2); platelet-raising drugs. The risk associated with all-cause mortality is reported per 100 × 10^9/L of continuous platelets. Data was analyzed with the use of the statistical packages R (The R Foundation; http://www.r-project.org; version 3.4.3). All P values for statistics were 2-tailed, and P < 0.05 was regarded as statistically significant.
Results
Our study included 11625 children, including 6638 boys and 4987 girls. Median age at ICU admission was 7.82 months (Q1-Q3: 1.08–38.20), and 5.82% percent (677 patients) died. The mean platelets count is 308.73 ± 141.34 × 10^9/L.
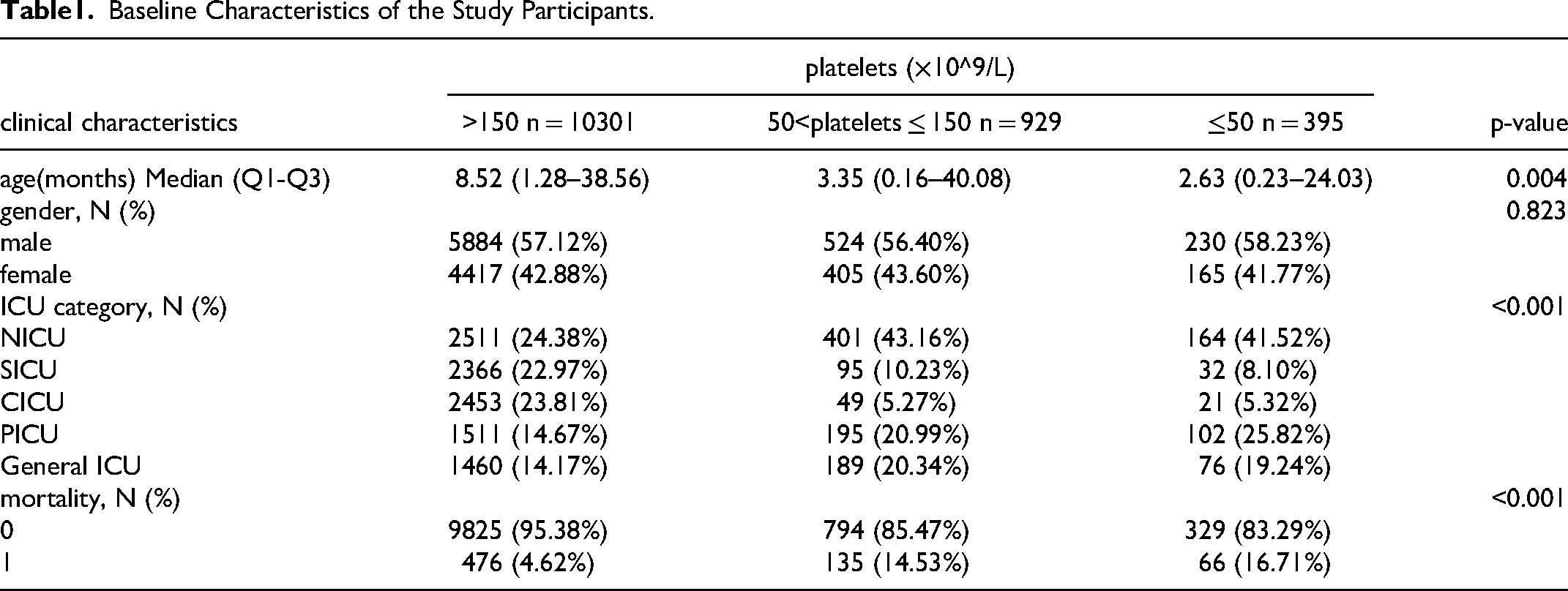
Tables 1 and 2 describes the baseline characteristics of the subjects, including demographic characteristics and some laboratory test results that may be related to the occurrence of mortality.

Flowchart of participants.
Baseline Characteristics of the Study Participants.
Characteristics of Clinical Laboratory Results of Study Participants.

The effect of age on platelets count was observed in Table 1, so we adjusted for age as a confounding factor when studying the effect of platelets count on all-cause mortality. Sensitivity analyses were performed to verify the effect of platelets count on all-cause mortality in patients of different ages. The results in Table 3 show that, after stratification by age, platelets count was a protective factor for all-cause mortality across age groups without interaction (p-value for interaction = 0.3985). Other subgroup analyses are shown in Table 3. We found that according to ICU type, WBC Count, the effect of platelets on all-cause mortality remained consistent between the subgroups analyzed. In the different subgroups, the incidence of all-cause death in the SICU decreased significantly with increasing platelets count.
The Sensitivity Analysis Between Platelets and Mortality.
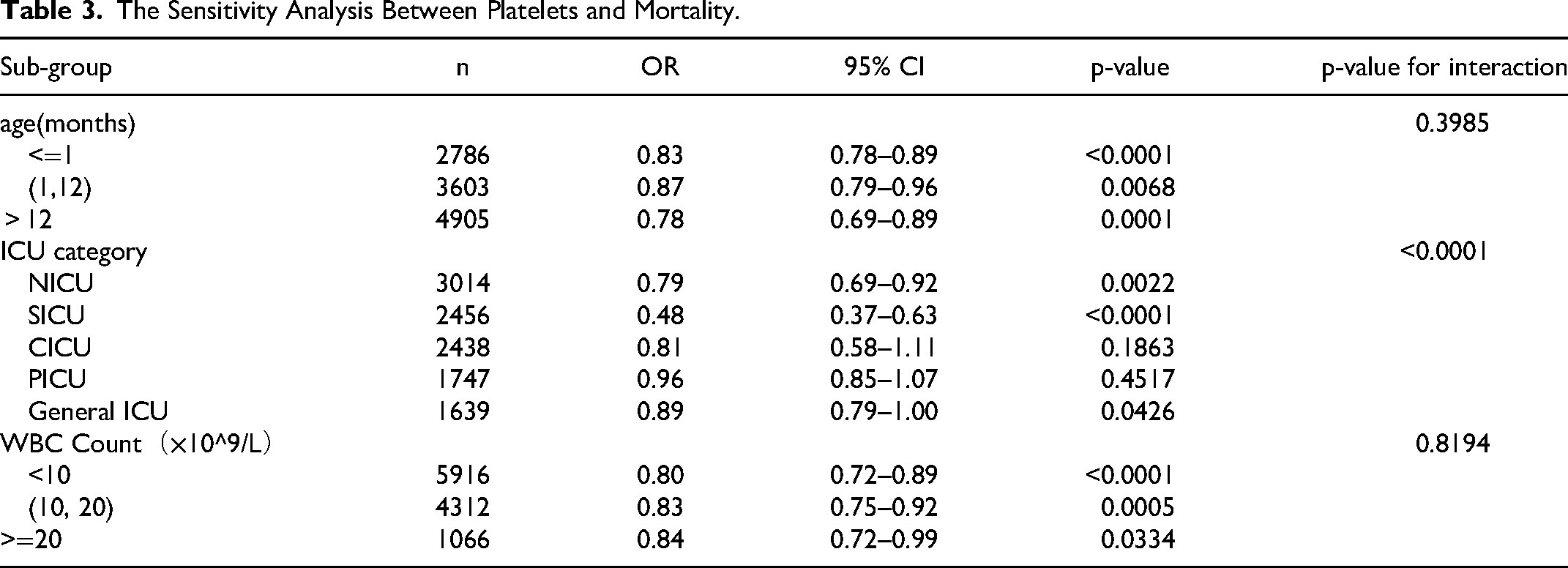
Table 4 shows the results of the multiple regression of platelets count on the risk of death. Without adjustment for confounders, for every 100 × 10^9/L increase in platelets, the risk of death was reduced by 33% (adjusted OR = 0.83, 95% CI: 0.63, 0.73) ; after full adjustment for confounders, the risk of death was reduced by 17% (adjusted OR = 0.83, 95% CI: 0.78, 0.89). platelets count was grouped according to cutoff values of 150 and 50, and after full adjustment for confounding, compared with patients with platelets count >150 × 10^9/L, the risk of death increased statistically significantly by 1.71-fold at 50–150 × 10^9/L and 1.93-fold at platelets ≤ 50 × 10^9/L.
Individual Effect of Platelets on all-Cause Mortality.
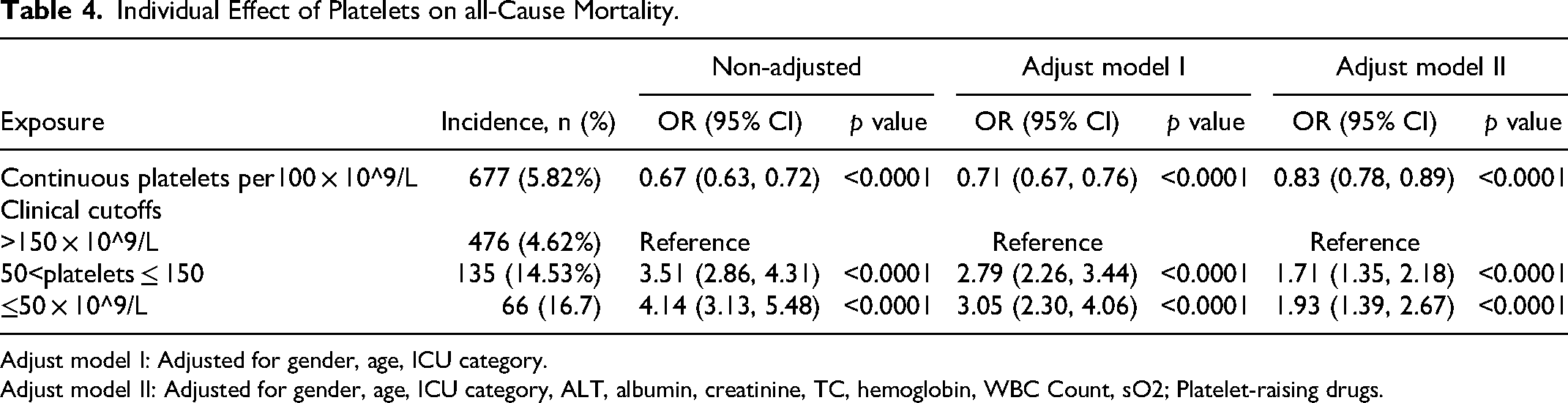
Adjust model I: Adjusted for gender, age, ICU category.
Adjust model II: Adjusted for gender, age, ICU category, ALT, albumin, creatinine, TC, hemoglobin, WBC Count, sO2; Platelet-raising drugs.
Table 5 shows the results of the multiple regression of platelets-related derived parameters on the risk of death. After full adjustment for confounders, each 0.1 increase in PCT was associated with an 18% reduction in the risk of death (adjusted OR = 0.82, 95% CI: 0.76, 0.88); each 1 increase in PDW was associated with an 11% increase in the risk of death (adjusted OR = 1.11, 95% CI: 1.07, 1.55).
Individual Effect of Platelets-Derived Parameters on all-Cause Mortality.
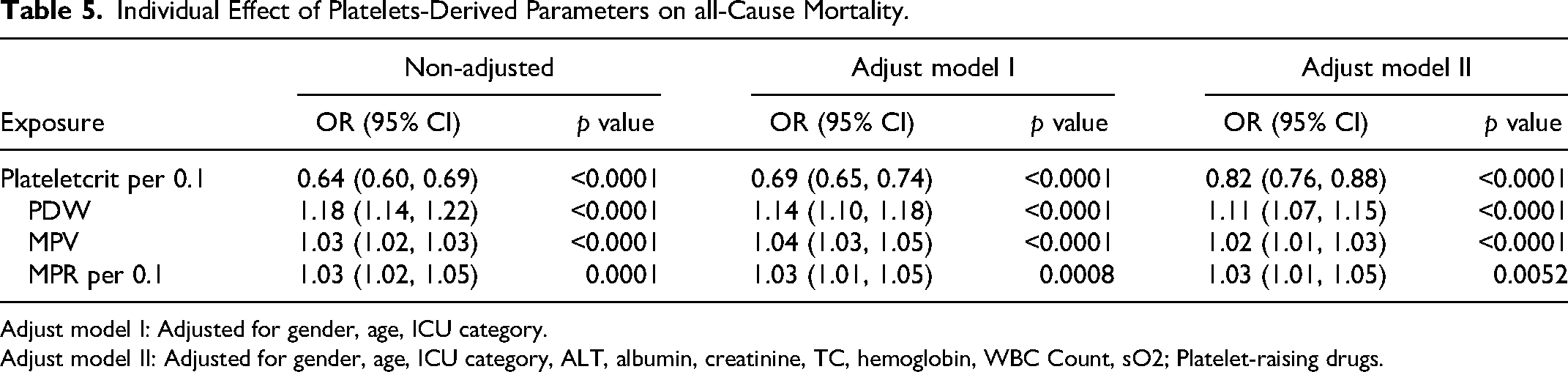
Adjust model I: Adjusted for gender, age, ICU category.
Adjust model II: Adjusted for gender, age, ICU category, ALT, albumin, creatinine, TC, hemoglobin, WBC Count, sO2; Platelet-raising drugs.
Discussion
Identifying risk factors associated with PICU mortality can help identify high-risk children who may benefit from early intervention and is also very challenging. Our study is the largest analysis of the prognostic value of platelet parameters on admission to the PICU population, including 11,625 children in unselected multiple ICU types. Our study identified lower platelet counts on admission, lower PCT and elevated PDW as independent risk factors for all-cause mortality in the PICU and as important prognostic bio-markers for optimal patient care and management. In a heterogeneous ICU population, the risk of all-cause mortality was reduced by 17% for every 100 × 10^9/L increase in platelet count on admission, and by 18% for every 0.1 increase in plateletcrit, and by 11% for every 1 increase in PDW, after full adjustment for confounders. After sensitivity analysis stratified by age, ICU type, and white blood cell count, platelet count had a consistent effect across subgroups, and after adjustment for parameters related to oxygen saturation, hemoglobin, albumin, and liver and kidney function, reduced platelet count remained an independent risk factor for all-cause mortality risk.
Platelet-related parameters can be obtained from routine complete blood count tests at low cost and are readily available in critical practices. Our study found platelet count, PCT, and PDW to be simple and effective independent bio-markers associated with mortality in the PICU.
In adults, the vast majority of studies were consistent with the effect of platelets on mortality that we observed in the PICU, but there were differences in the magnitude of the effect. Low platelet count is an independent risk factor for death in ICU trauma patients. 8 In septic shock, thrombocytopenia is associated with mortality. 9 The severity of sepsis was associated with a decrease in platelet count, and ICU mortality was significantly higher in thrombocytopenic patients. 4 Another study reported thrombocytopenia in sepsis-3 patients with worsening disease and increased 28-day mortality in patients with platelet counts <50 × 10^9/L. However, the mortality rate in the mild thrombocytopenia group (platelet count: 51–100 × 10^9/L) was not significantly different from the non-thrombocytopenia group. 10 However, one study also reported that mean admission platelets count levels did not differ significantly between the surviving and dying groups and that platelet was not considered a prognostic indicator at admission. 3
In adults, patients with sepsis combined with thrombocytopenia had a higher 30-day mortality rate (HR 2.00 (95% CI 1.32–3.05)). 11 Another study found that early thrombocytopenia increased the risk of death in critically ill patients by 60% to 110%. 12 In our study, we observed that PICU patients with thrombocytopenia (0–150 × 10^9/L) exhibited a significant mortality rate in children. This finding persisted even after adjusting for various factors such as gender, age, ICU category, ALT, albumin, creatinine, TC,WBC, and oxygen saturation (sO2). Notably, the risk associated with platelet-raising drugs was found to be 1.71-fold higher (95% CI: 1.35, 2.18) and 1.93-fold higher (95% CI: 1.39, 2.67) for children with severe thrombocytopenia (<50 × 10^9/L).
Similar to our study, Sayed et al showed in PICU patients that both the platelet count and PCT were significantly lower in non-survivors compared to survivors (OR = 0.989;95% CI: 0.982,0.996;P< 0.001) . The low platelet count in non-survivors may be attributed to the depletion of coagulation factors and platelet consumption during the septic process. 13
In a subgroup analysis of different ICU categories, we found that platelets had the greatest impact on the risk of death in the surgical ICU, with a 52% reduction in the risk of death for every 100 × 10^9/L increase in platelets, (OR = 0.48, 95% CI: 0.37 - 0.63). A previous study also found an association between thrombocytopenia and poor prognosis in adult surgical ICU patients. 14 This study further confirmed the pivotal role of platelets in severely ill patients and provides strong support for our findings.Thrombocytopenia is a common clinical phenomenon that may be caused by various factors, such as infection, drug response, myelosuppression, etc In surgical ICU patients, thrombocytopenia is often accompanied by a higher risk of infection, a longer hospital stay, and higher mortality rates. Therefore, by monitoring and regulating platelet levels, patients can be identified for early interventions, effectively reducing the risk of infection, shortening hospital stay and reducing mortality.
Our study also found that in addition to platelet count, PCT and PDW can be risk factors for PICU mortality. The findings of previous studies on platelet parameters and PICU mortality are not exactly the same as our results, and there were statistically significant differences in platelet parameters in the results of different studies. PCT was found to be an independent risk factor for intravascular hemorrhage in preterm infants in a study of 305 preterm infants. 15 In 209 children in the pediatric intensive care unit, a first platelet distribution width (PDW)/platelet count ratio above 0.07 after admission served as an independent predictor of mortality with a sensitivity and specificity of 77.1% and 77.5%, respectively. 2 In 40 children aged 1 month to 14 years in the study population and 40 children in the control group, MPV/PCT, PDW/platelet count and MPV/platelet count in the first sample after hospital admission were predictors of death and accurately predicted 65% to 67% of deaths. 16 Our study concluded that the parameters platelets count, PDW and PCT abnormalities given directly in the routine blood report can serve as a good warning.
Our findings suggest limitations of MPV as a continuous variable as a risk factor, with a median MPV of 9.50 (Q1-Q3: 8.70–10.40) in our study population and a narrow range of variation in MPV, with each 1 increase in MPV increasing the risk of death in PICU children by only 2%, with insufficient variation in risk values to attract clinical attention. In previous studies, the presence of MPV and PDW during hospitalization in critically ill children receiving mechanical ventilation was considered to be associated with mortality, but MPV at initial admission was not associated with mortality in these children. 17
MPV levels are positively correlated with platelet activation, and there have been many studies examining the relationship between mortality and MPV in adult intensive care units. However, the results are inconsistent. Some studies have concluded that MPV does not correlate with mortality,18,19 and others have concluded that mean platelet volume MPV is a very good independent predictor of mortality outcome in critically ill and injured patients in the surgical ICU.20,21 The MPV/platelet count ratio is not superior to MPV or platelet count alone. 21 In patients admitted to the intensive care unit with normal platelet counts, increased MPV values and decreased platelet counts may alert intensivists to a worse course of disease. 22 The mean MPV levels in patients who die are higher than the initial MPV levels, while MPV levels in patients who are alive are reduced. 3 High MPV is a risk factor for overall cardiovascular mortality. In addition, high MPV is also associated with poor prognosis in critically ill patients with non-cardiovascular disease. 23
Our findings suggest limitations of MPR as a continuous variable as a risk factor, with a median MPR of 0.03 (Q1-Q3:0.02–0.04) in our study population and a narrow range of variation in MPR, with each 0.1 increase in MPR associated with a 3% increase in the risk of death in PICU children, with insufficient variation in risk values to warrant clinical attention. In contrast to our findings, the MPR was considered to independently predict adverse outcomes in critically ill patients in previous adult studies. It was found that patients in the MPR > 0.099 group were more severely ill and that MPR was predictive of in-hospital mortality in patients with Acute Kidney Injury (AKI). 23 In 120 patients with sepsis, MPV or platelet count alone did not predict mortality; whereas MPR was an independent predictor of 28-day mortality. 21
Decreased platelet activation and count can identify patients with worsening conditions in the ICU. The development of thrombocytopenia is important in assessing the prognosis of ICU patients. These patients should be reassessed for adequacy of their diagnosis and treatment and should be monitored for possible complications of their disease and/or treatment.
Conclusion
In conclusion, reduced platelet count is an independent risk factor for all-cause mortality risk in the PICU, and platelet count can be used to identify high-risk patients at the time of PICU admission, allowing us to implement early and appropriate interventions in daily practice, leading to a better prognosis for PICU patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
