Abstract
Introduction
Off-label, under-, and overdosed direct oral anticoagulants (DOACs) are commonly prescribed to patients with atrial fibrillation (AF), but real-world evidence on their effectiveness and safety is limited.
Methods
MEDLINE, Embase, and Cochrane Library databases were systematically searched from 01 July 2020 to 28 February 2022 to update a previous systematic review with the same search strategy from the inception to 30 June 2020. Eligible studies were those that reported effectiveness (stroke/systemic embolism and myocardial infarction) or safety (gastrointestinal or major bleeding and death) outcomes of off-label doses of DOACs compared to on-label doses in AF patients. A random-effects meta-analysis was performed to estimate the pooled hazard ratio (HR) and 95% confidence interval (CI). Subgroup analyses were performed by specific DOACs and geographic regions.
Results
Twenty-two studies were included. Off-label, underdosed DOACs, compared to on-label doses, were not associated with an increased risk of stroke (HR 1.03, 95%CI: 0.88-1.17) but were associated with an increased risk of death (HR 1.26, 95%CI: 1.09-1.43). However, risk varied depending on the active ingredient. No other safety outcomes were associated with underdosed DOACs. No significant differences were observed by geographic regions. Compared to on-label DOACs, overdosing increased the risk of stroke (HR 1.17, 95%CI: 1.04-1.31), major bleeding (HR 1.18, 95%CI: 1.05-1.31), and death (HR 1.19, 95%CI: 1.03-1.35). Risk varied between geographical regions.
Conclusions
Off-label underdoses, compared to on-label dosing of DOACs, did not increase the risk of stroke but did increase overall mortality. Overdosed DOACs, compared to on-label doses, were associated with an increased risk of stroke, major bleeding, and death. Future studies must examine these associations, focusing on specific active ingredients and geographic settings.
Introduction
Atrial fibrillation (AF) is the most common type of heart arrhythmia, and its prevalence increases with age and is associated with a significantly higher risk of stroke. 1 Direct oral anticoagulants (DOACs) are crucial for managing patients with AF, and their use has been continuously increasing. Randomized controlled trials have confirmed the noninferiority of DOACs in stroke prevention compared to vitamin K antagonists (VKAs), with a lower bleeding risk.2–5 Dabigatran, rivaroxaban, apixaban, and edoxaban are the main active ingredients used worldwide to treat non-valvular AF. 6 The US Food and Drug Administration states that dose adjustment should be performed based on patient clinical characteristics, including age, body weight, renal function, and concomitant medications7–11 However, in clinical practice, off-label doses of DOACs are commonly prescribed instead of approved doses (on-label doses) to treat patients with AF, with a prevalence of 26.2%–39.6%.12,13
The clinical outcomes associated with off-label under- and overdose of DOACs remain unclear, with previous studies reporting inconsistent results.8,14 High-quality evidence on the effectiveness and safety of off-label DOAC dosing in patients with AF remains unclear. Three recent meta-analyses have been conducted to fill this gap15–17 However, inconsistencies persist in the results. These could be attributed to small sample sizes, population characteristics (ie, mean age and ethnicity), different definitions of under and overdosing, and other factors. Therefore, an updated systematic review is warranted based on the newly available evidence from real-world studies.
Methods
This systematic review and meta-analysis were performed based on the PRISMA reporting guidelines and Cochrane Collaboration.18,19 The protocol was prospectively registered with the International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY, registration number INPLASY202290073 and DOI number 10.37766/inplasy2022.9.0073).
Data Sources and Searches
As an update to a previous systematic review, 15 the same databases and search strings were used to identify newly published real-world evidence studies on the safety and effectiveness of off-label over and under-dosing of DOACs, compared to on-label dosing. The previous systematic review performed searches from MEDLINE, Embase, and Cochrane CENTRAL from inception (the first literature) to June 30, 2020. 15 In this systematic review, we expanded the search from July 1, 2020, to February 28, 2022. Studies cited by the retrieved articles were also screened. The detailed search strategy is presented in Supplemental Table S1. All studies (n = 10) included in the first systematic review were included in the present study.
Study Selection
The study selection was performed by the same researchers that selected the studies in the first systematic review. Previous inclusion criteria were adopted: 1) prospective or retrospective cohort studies of patients with AF, and 2) studies that compared the effectiveness or safety of off-label DOAC dosing (dabigatran, rivaroxaban, apixaban, and edoxaban) versus on-label DOAC dosing. We excluded studies on patients who received short-term DOACs after catheter ablation and studies without adjusted data. If the same data source or overlapping data were reported in more than one study, the most comprehensive data with the longest follow-up were included. The on-label prescribing referred patients who received DOAC doses based on drug instructions. Off-label underdosing was defined as a dose reduction, although the patient did not meet the dose reduction criteria. In contrast, off-label overdosing was defined as patients who received standard doses, although patients met the criteria for dose reduction. The definition of the off-label dose criteria for DOACs varies according to the geographic area of the included studies and is reported in Supplemental Table S2.
Two authors (Nan-Nan Shen and Eliana Ferroni) reviewed study titles and abstracts independently. The studies were assessed based on inclusion criteria. Disagreements or uncertainties between the reviewers were resolved by consensus or discussion with a third reviewer (Zhi-Chun Gu).
Outcomes
The effectiveness outcomes were stroke (ischemic and hemorrhagic stroke)/systemic embolism (SE) and acute myocardial infarction (MI). The safety outcomes included major bleeding, intracranial hemorrhage (ICH), gastrointestinal (GI) bleeding, and all-cause mortality.
Data Extraction
The following data from eligible articles were independently extracted by two investigators (Nan-Nan Shen and Eliana Ferroni): study characteristics (the first author, publication year, country or region, data source, study design, follow-up period, the proportion of each DOAC in the study, the total number of patients, and definition of DOAC non-standard dosing), baseline characteristics (mean age, sex ratio, comorbidities, concomitant medications, CHA2DS2-VASc score, and HAS-BLED score), effectiveness and safety outcomes. The included studies were also stratified into main geographic areas based on the country where the study was conducted: Asia, North America, and Europe.
Quality Assessment
The modified Newcastle-Ottawa Scale (NOS) was used to evaluate the methodological quality of all included studies. NOS considers five domains: sample population, sample size, participation rate, outcome assessment, and analytical methods to control bias (Cota et al., 2013). Each item was assigned from 0 to 2 points. The study was considered high quality when it scored > 6 points (Supplemental Table S3).
Data Synthesis and Statistical Analysis
A random-effects model meta-analysis was used to compare clinical outcomes of AF patients treated with off-label doses versus on-label doses of DOACs by calculating the pooled hazard ratio (HR) and the 95% confidence interval (CI), regardless of the presence of heterogeneity. Heterogeneity was assessed between studies using the I2 value, with I2 > 50% considered a high degree of heterogeneity. Subgroup analyses were performed by stratifying individual DOACs (dabigatran, rivaroxaban, apixaban, and edoxaban) and main geographical areas (Asia, North America, and Europe). Interaction analyses (P for interaction) were conducted to assess comparability in each subgroup. A sensitivity analysis was performed by removing one study at a time from the pooled analysis to explore the influence of the study on the overall outcomes of non-standard DOAC dosing. A meta-regression analysis was conducted to assess factors influencing the results. Publication bias was evaluated qualitatively using funnel plots and quantitatively using the Begg and Egger tests (Liberati et al., 2009). All statistical analyses were performed with STATA version 13.0 (StataCorp, College Station, Texas, TX, and the United States).
Results
Study Selection and Characteristics
Figure 1 describes the study selection process. Overall, 3427 articles were identified (368 duplicate studies), and 2960 were excluded after screening titles and abstracts. Subsequently, 77 studies were removed from the full-text review process for the reasons outlined in Supplemental Table S4. Finally, 22 studies (10 in the first systematic review), with data from 223,057 patients, were included.8,13,14,20–38 Among these studies, DOACs were evaluated as a class in five studies, dabigatran in 5, rivaroxaban in 7, and apixaban in 6. The characteristics of the studies are summarized in Table 1. Among the 22 studies, five were conducted in North America (all in the United States), 14 in Asia (3 in Taiwan, 5 in Japan, 4 in Korea, and 2 in Israel), and 3 in Europe (Portugal, Spain, and the UK).
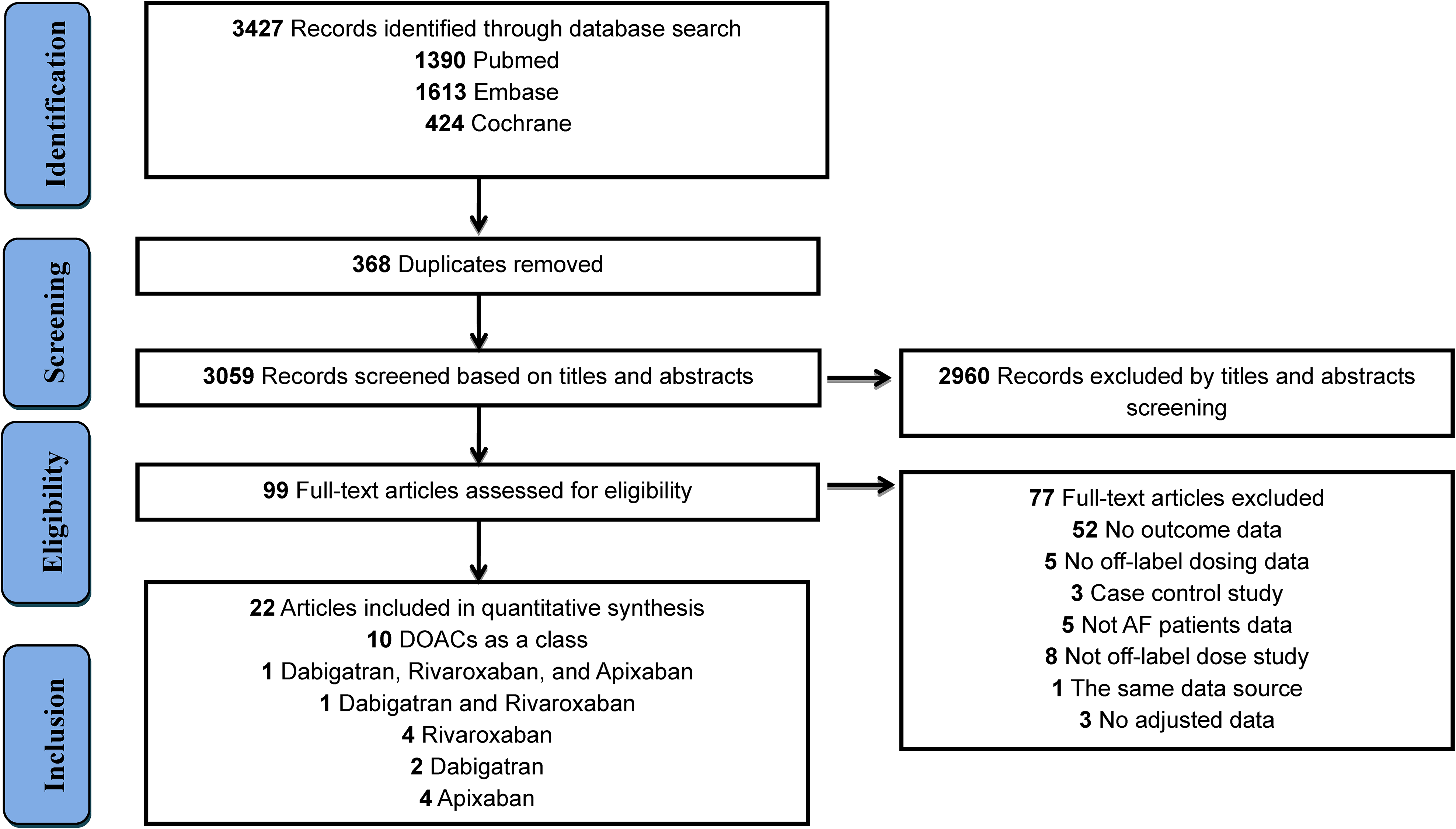
Flow diagram for the selection of eligible studies.
Detailed Characteristics of the Included Studies.
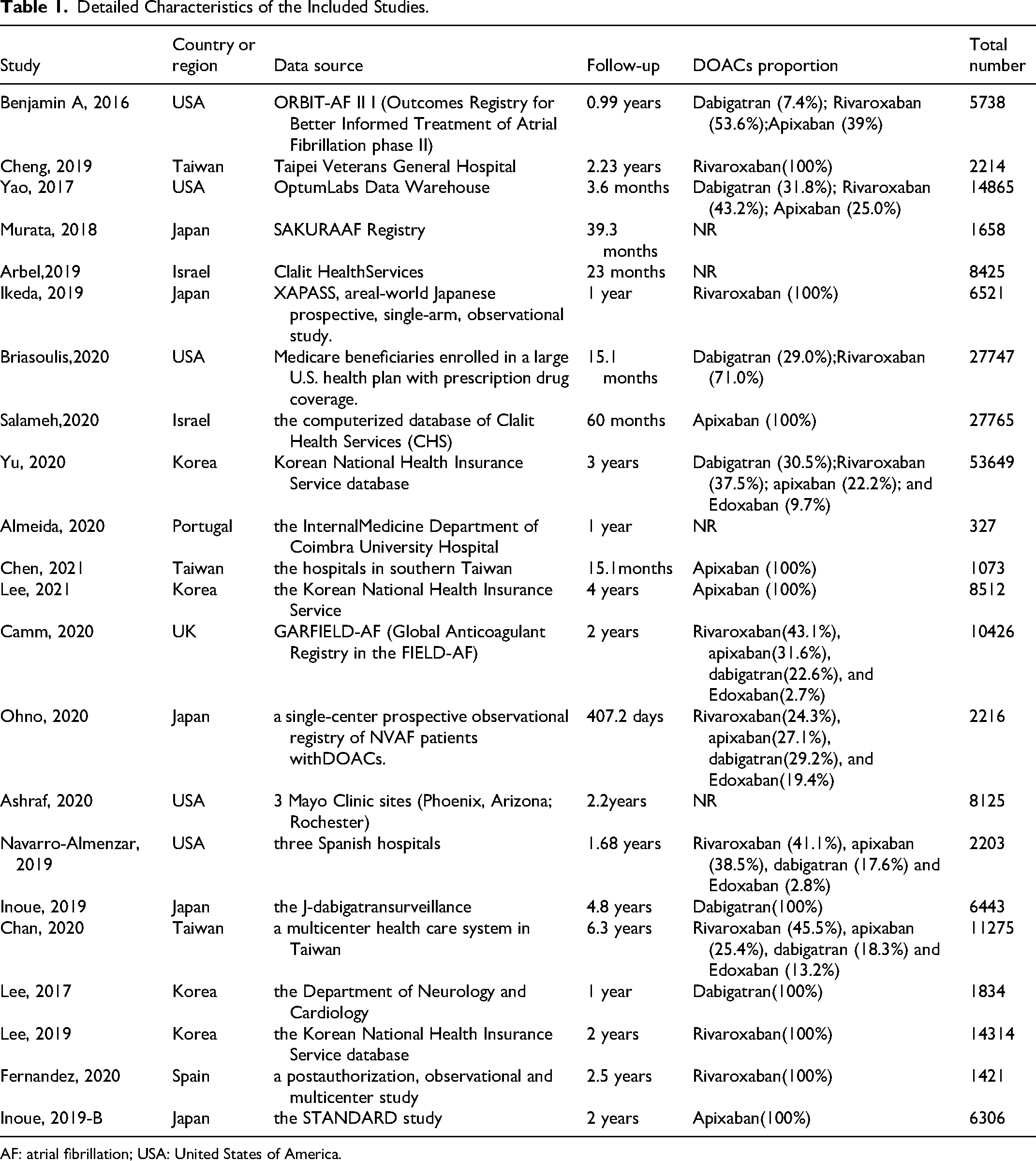
AF: atrial fibrillation; USA: United States of America.
Patient Characteristics and Quality Assessment
Detailed demographic and clinical characteristics of patients included in each study are outlined in Supplemental Table S5. The age of the patients ranged from 51 to 84 years (about 73 years on average), and most were men (about 56%). The mean CHA2DS2-VASc and HAS-BLED scores were 3.8 and 2.1, respectively. The mean body mass index was 26.3 kg/m2, and 23.4% of the patients received concomitant antiplatelet agents. The most common comorbidities were hypertension (75.5%), heart failure (29.8%), diabetes (30.6%), and transient ischemic attack (22.9%). All studies had the following risk biases: sample population, sample size, and participation rate. The overall quality of the included studies was high, with NOS scores ranging from 8 to 9 (Supplemental Table S6).
Off-Label Underdosing of DOACs
Compared to on-label doses of DOACs, off-label underdosing was not associated with an increased risk of stroke (HR: 1.03, 95%CI: 0.88–1.17, I2: 57.2%) and MI (HR:1.07, 95%CI: 0.89–1.25, I2: 0.0%), but was associated with a higher death rate (HR: 1.26, 95%CI: 1.09–1.43, I2: 73.2%). No significant association was observed for major bleeding (HR: 0.93, 95%CI: 0.80–1.07, I2: 56.8%), ICH (HR: 0.93, 95%CI: 0.66–1.20, I2: 40.6%) and GI bleeding (HR: 1.24, 95%CI: 0.95–1.54, I2: 80.3%) (Figure 2; Supplemental Figures 1–6).

Effectiveness and safety of under-dose DOACs and individual DOAC
Analyses of the individual DOACs (dabigatran, rivaroxaban, apixaban, and edoxaban) are shown in Figure 2 and Supplemental Figures 7–12. The underdosing of each DOAC presented similar results regarding effectiveness and safety outcomes. Interestingly, off-label dabigatran underdosing (HR: 0.76, 95%CI: 0.59–0.93, I2: 9.6%) was associated with a reduced mortality risk compared to on-label dosing, while there was an excess risk for rivaroxaban (HR: 1.31, 95%CI: 1.08–1.54, I2: 32.8%) and apixaban (HR: 1.15, 95%CI: 1.00–1.31, I2: 0.0%). Only one study examined death associated with the underdosing of edoxaban. Only one study examined the association of MI with the underdosing of dabigatran, apixaban, and edoxaban.
When stratifying for geographic regions, no excess risk could be observed for most of the outcomes, except for the risk of death, which was significantly increased in North America (HR: 1.41, 95%CI: 1.26–1.57, I2: 0.0%). However, this result was based on only two studies, including one conducted in Europe (HR: 1.51, 95%CI: 1.13–1.89, I2: 51.4%), with a pooled HR from four studies (Figure 3; Supplemental Figures 13–18). Subgroup analyses did not detect statistical heterogeneity of the effectiveness and safety outcomes (P for interaction > 0.05 for each outcome).

Effectiveness and safety of under-dose DOACs by regions
Off-Label Overdosing of DOACs
Compared to on-label dosing, the off-label overdose of DOACs was significantly associated with an increased risk of stroke (HR: 1.17, 95%CI: 1.04–1.31, I2:0.0%), major bleeding (HR: 1.18, 95%CI: 1.05–1.31, I2:0.0%), and all-cause mortality (HR: 1.19, 95%CI: 1.03–1.35, I2:0.0%) (Figure 4). No significant differences were observed in the risk of ICH (HR: 1.16, 95%CI: 0.79–1.53, I2:0.0%), GI bleeding (HR: 1.13, 95%CI: 0.98–1.28, I2:0.0%), and MI (HR: 0.81, 95%CI: 0.20–1.43, I2:47.2%).
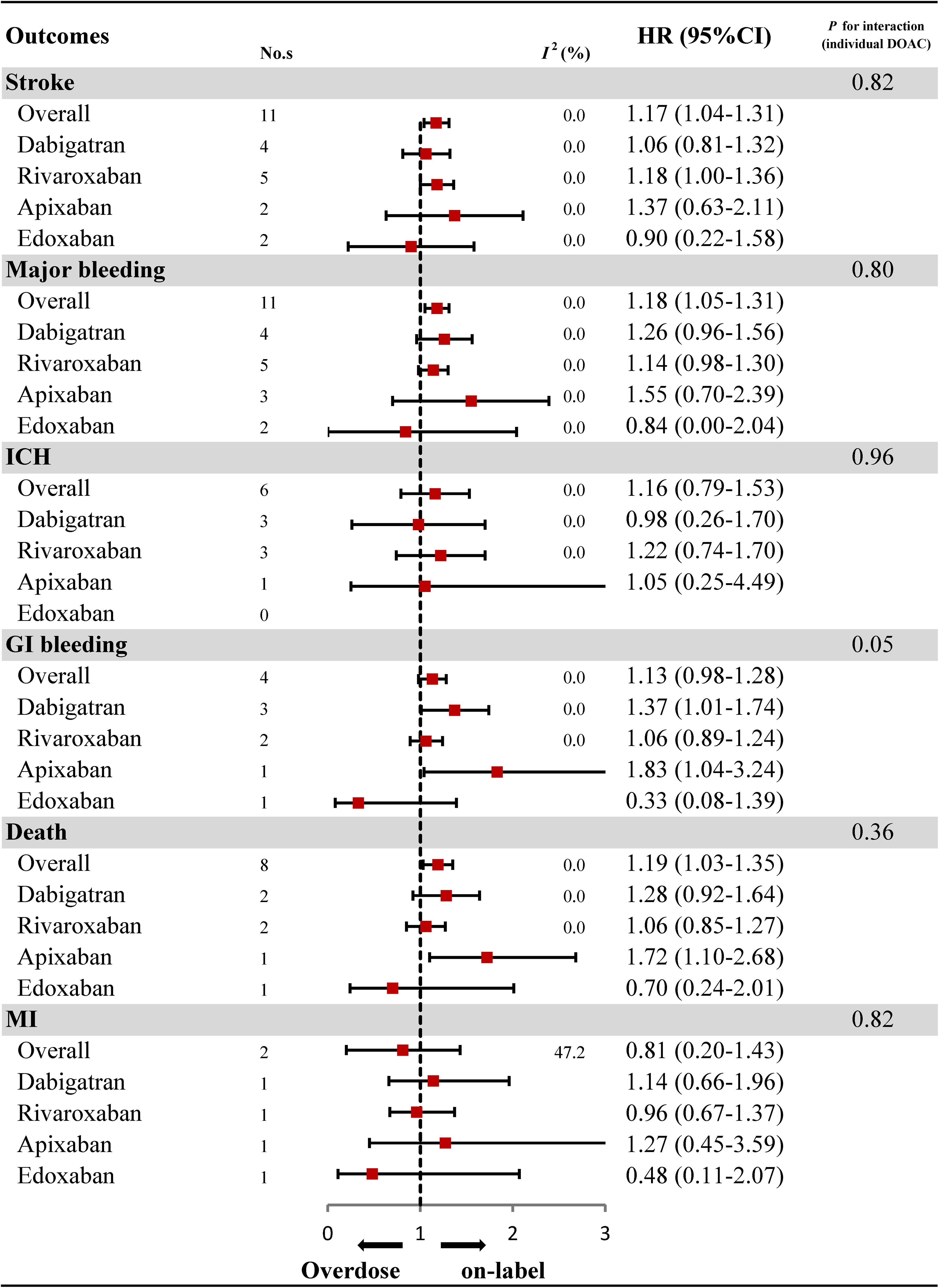
Effectiveness and safety of over-dose DOACs and individual DOAC
When stratifying for specific DOAC, the results were consistent with overall analyses, except for off-label overdosed apixaban which was associated with an increased risk of GI bleeding (HR: 1.83, 95%CI: 1.04–3.24) and an increased risk of death (HR: 1.72, 95%CI: 1.10-2.68). However, the results were based on only one study (Figure 4; Supplemental Figures S19–S29).
In different geographic regions, compared to on-label dosing, increased risks of major bleeding (HR: 1.21, 95%CI: 1.03–1.39, I2:0.0%) and death (HR: 1.18, 95%CI 1.00–1.36, I2:0.0%) were observed among patients in Asia exposed to overdosed DOACs, as well as an increased risk of all-cause mortality among patients in North America (HR: 1.91, 95%CI: 1.02–3.60) (Figure 5; Supplemental Figures 30–34). No apparent heterogeneity was found in the subgroup analyses (P for interaction > 0.05 for each outcome).

Effectiveness and safety of over-dose DOACs by regions
Sensitivity Analyses and Meta-Regression
Sensitivity analyses were repeated by sequentially removing each study, and the pooled results were consistent with the results of the main analyses (Supplemental Tables 7 and 8). Meta-regression analyses did not reveal any potential influencing factors associated with any outcomes considered for off-label over- or under-dosed DOACs (P > 0.05 for each variable; Supplemental Tables 9 and 10).
Publication Bias
A visual inspection of the funnel plot showed relative symmetry, suggesting that publication bias was absent among the selected studies (Supplemental Figures S35–S42).
Discussion
The need to update a previous systematic review on the same topic, conducted with the same methodology, was driven by the significant increase in published evidence in the past two years. This review aimed to evaluate the clinical outcomes of off-label DOAC dosing compared to on-label dosing in patients with non-valvular AF in a real-world setting. Of the included studies, most of the effectiveness and safety outcomes of off-label under-dose DOACs did not suggest any increased risk compared to on-label dosing. However, counterintuitively, mortality risks were significantly increased. Off-label over-dosing of DOACs compared to on-label dosed DOACs was associated with a substantially higher risk of stroke/SE and an increased risk of major bleeding and death. When comparing studies conducted in different geographic regions, findings were similar between non-standard (underdose and overdose) and on-label DOAC dosing, except for underdosed DOACs which were associated with increased risks of death in North America and Europe. In contrast, overdosed DOACs were associated with increased risks of death in North America and Asia.
Previous studies reported that low off-label doses are mainly prescribed to frail patients. Compared to those treated with standard doses, patients treated with underdoses of DOACs were older, more likely to be women, with a low body weight, or a high CHA2DS2-VASc score.20,36 Patients with a history of renal dysfunction, stroke, and bleeding were also found to be treated more often with low off-label doses of these drugs.20,36 Previous meta-analyses have demonstrated the effect of off-label doses of DOACs, but contrasting results persist. Kong et al reported an increased risk of all-cause mortality among patients treated with off-label, underdosed DOACs compared to those with on-label dosing. However, they also observed increased risks of stroke and systemic embolism, 39 similar to what Liu et al. and Zhang et al. observed.17,40 Kong et al. reported that off-label overdosed DOACs were associated with an increased risk of stroke and systemic embolism, similar to what we observed, which is surprising considering an opposite effect might have been expected. A similar result was observed by Liu et al. in 2021, 17 with increased risks of systemic embolism, major bleeding, and all-cause mortality. Zhang et al. also reported an increased risk of ischemic stroke and systemic embolism. 40
We also observed some contrasting results among the clinical outcomes of non-standard DOAC doses reported in the observational studies included in this systematic review. While an increase in the risk of stroke in a study conducted in Denmark on patients treated with underdosed DOACs was reported, 41 a prospective registry study from the US found higher mortality in patients treated with overdosed DOACs, compared to on-label doses. 8 A Japanese study reported similar risks of stroke/SE and death among patients administered both standard and underdosed DOACs. In contrast, higher composite events (stroke/SE, major bleeding, or death) have been observed in the overdose group compared to the standard dose group. 14 There was no increased risk of stroke in underdosed DOACs compared to on-label dosing in this meta-analysis. These discrepancies could be partially attributed to the patient's characteristics, especially body weight. Body weight is an essential factor influencing stroke risk for patients on DOAC therapy. 14
Patients with a high BMI had a higher stroke and death rate than patients with a low BMI.8,41 The stroke rate was similar between the underdoses and standard doses of the DOAC group in patients with low BMI levels. 24 There was a relatively low BMI level of less than 25 kg/m2 in the included studies of this meta-analysis, and it may not result in significant differences in stroke risk. Furthermore, the missing BMI data in some observational studies could lead to uncertainty. Additionally, we found that the mortality risk varied depending on the active ingredient. The mortality risk increased for rivaroxaban and apixaban, but the risk was reduced for dabigatran. Underdoses of DOACs are more likely to be prescribed to frail patients, older individuals, women, and patients with a low body weight or a high CHA2DS2-VASc score than those treated with standard doses. Therefore, this group's overall risk of death is increased in observational studies. Compared to other DOACs, the kidney metabolizes almost 80% of ingested dabigatran. Therefore, the clinical application of dabigatran is relatively strict. Patients who receive dabigatran do not have renal disease, indicating a relatively healthy physical state. This may be a reason for the relatively low mortality risk in patients treated with dabigatran.
Possible inconsistencies between observed results may also be related to difficulty adhering to DOAC therapy. The impact of adherence to DOAC treatment among AF patients on the outcomes evaluated is well known. 12 The patient's body weight may partly explain the observed differences, as it strongly influences a critical pharmacokinetic parameter, the volume of distribution. 14 Patients who may require a higher dose of DOAC are those with higher body weights. Some observational studies included patients whose BMI data were unavailable or could not be thoroughly examined. These studies may have reported unadjusted results with significant confounding or effects with residual confounding from incomplete weight adjustment. Ethnic differences, only partially accounted for in the analyses stratified by geographical region, also entail substantial differences in average body weight and pharmacokinetic and pharmacodynamic properties. The standard dose of rivaroxaban in Japan is 15 mg instead of 20 mg as in other countries, according to guidelines tailored to national pharmacokinetic studies conducted among Japanese adults, 14 who have a relatively lower BMI than most Europeans and Americans. When comparing clinical outcomes in different geographic regions (Asia and North America), no significant differences were observed between underdosing and on-label dosing for most outcomes.
Nevertheless, geographical differences were associated with different risks for all-cause mortality. Underdosed DOACs were associated with increased risks of death in North America and Europe. A possible explanation may be the discrepancy of patient populations in different regions. It is worth noting that the patient demographics and clinical characteristics affected death risk. A relatively high BMI in North Americans and Europeans may be an increased risk factor for death. Regretfully, the baseline characteristics were not compared between these regions due to some observational studies’ lack of patient baseline characteristics. Additionally, the pooled analysis was based on limited studies, so the result should be interpreted cautiously. Therefore, more studies are needed to better understand this problem. Lower doses of DOACs could be safe and effective in clinical practice, especially for Asian patients, but not for Europeans and North Americans. On the other hand, overdosed DOACs seem less safe in Asia, while they do not pose a serious threat in Europe. The increased risk of overall mortality associated with DOAC overdoses in North America should be considered cautiously, as it derives from one study only.
Overall, DOAC overdosing was less frequent than underdosing, ranging from 3.4% to 8.4%.8,13,14 According to this study, compared to the standard DOAC dosing, a significant association was found between DOAC overdose and increased risks of stroke/SE, major bleeding, and death. These results follow those of the ORBIT-AF II study, in which higher all-cause mortality was observed with an overdosing of DOACs. 8 Physicians may adjust the dose of DOACs according to the underlying risks, regardless of the label recommendation and the favorable risk-benefit profiles across risk strata demonstrated by a large clinical trial. 8 In this analysis, overdosing on DOACs was associated with significantly higher risks of stroke/SE, major bleeding, and death than on-label DOAC dosing in Asian patients. However, there were no significant differences in patients with other ethnicities. The results agreed with a Korean database study8,13,14 and a Japanese SAKURA AF study,36,42 suggesting that Asian patients might be more at risk of adverse events in the case of DOACs overdosed. Nevertheless, the results were difficult to explain, as it is generally accepted that a higher dose of anticoagulants would be associated with a lower risk of stroke/SE. Therefore, more studies are needed to better understand the study findings.
Study Strengths and Limitations
The main strength of this study was to evaluate the effectiveness and safety of nonstandard dosing of DOACs in AF patients in a real-world setting by a systematic and rigorous approach. The quality of the included studies was assessed using the revised NOS tool. In addition, we performed subgroup analyses, considering individual DOACs and geographic regions where the studies were conducted. Sensitivity analyses were performed to test the robustness of the results. Due to the observational nature of the included studies, a possible selection bias could limit the generalizability of the reported results and their implications for clinical practice. Future large-scale studies are needed to confirm our findings. Deliberate under or over-dosing may not be appropriate in many clinical practice scenarios. Given the limited number of included studies, the results should be taken cautiously. However, the low degree of heterogeneity in the overall and subgroup analyses supports the robustness of our results.
Conclusion
Similar risks related to most outcomes of interest were observed when comparing patients with nonvalvular AF treated with off-label doses of DOACs to those with on-label doses. The risk of stroke and SE among patients treated with off-label under-dosed DOACs compared to on-label dosing had similar risks but an increased risk of all-cause mortality. A significantly increased risk of stroke/SE, major bleeding, and death was observed when comparing patients with overdosed DOACs with those with standard doses. Underdosed DOACs were associated with increased mortality risks in North America and Europe, while overdosed DOACs were associated with increased mortality risks in Asia. A reduced dose of DOACs may be appropriate, especially for Asian patients, whereas high-dose DOACs may not be well tolerated. More research is needed to confirm these findings, especially concerning the increased risk of stroke/SE observed with DOAC overdose and differences between patient ethnicity based on the geographic region in which the studies were conducted.
Supplemental Material
sj-docx-1-cat-10.1177_10760296231179439 - Supplemental material for An Updated Pooled Analysis of Off-Label Under and Over-Dosed Direct Oral Anticoagulants in Patients with Atrial Fibrillation
Supplemental material, sj-docx-1-cat-10.1177_10760296231179439 for An Updated Pooled Analysis of Off-Label Under and Over-Dosed Direct Oral Anticoagulants in Patients with Atrial Fibrillation by Nan-Nan Shen, Eliana Ferroni, Claudio Barbiellini Amidei, Cristina Canova, Viviana Peron, Jia-Liang Wang, Hou-Wen Lin and Zhi-Chun Gu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions
Zhi-Chun Gu and Hou-Wen Lin are guarantors of the entire manuscript. Nan-Nan Shen, Eliana Ferroni, Cristina Canova, Viviana Peron, and Claudio Barbiellini Amidei contributed to the study conception, design, and critical revision of the manuscript for important intellectual content. Zhi-Chun Gu and Nan-Nan Shen contributed to the data acquisition, analysis, and interpretation. All authors contributed to the study design, critically reviewed the first draft, approved the final version, and agreed to be accountable for the work.
Data Availability Statement
The data that support the findings of this study are available at reasonable request from the corresponding authors.
Ethical Statement
Ethical approval is not necessary for this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Research Project of Grassroots health science of Zhejiang Province (2022ZD09), Clinical Medical Research Special Fund Project of Zhejiang Medical Association (2022ZYC-Z37), the Program of General Scientific Project of Zhejiang Education Department (Y202249053), the Research Project of Drug Clinical Comprehensive Evaluation and Drug Treatment Pathway (SHYXH-ZP-2021-001), Clinical Research Innovation and Cultivation Fund of Ren Ji hospital (RJPY-LX-008), and Ren Ji Boost Project of National Natural Science Foundation of China (RJTJ-JX-001).
Sources of Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
